Transport of Gametes and Fertilization
Chapter 1 describes the origins and maturation of male and female gametes and the hormonal conditions that make such maturation possible. It also describes the cyclic, hormonally controlled changes in the female reproductive tract that ready it for fertilization and the support of embryonic development. This chapter first explains the way the egg and sperm cells come together in the female reproductive tract so that fertilization can occur. It then outlines the complex set of interactions involved in fertilization of the egg by a sperm.
Ovulation and Egg and Sperm Transport
Ovulation
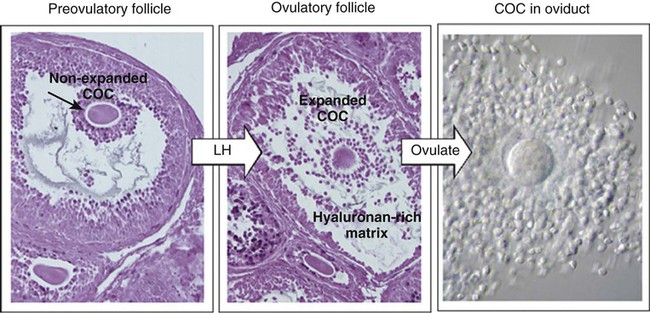
The stimulus for ovulation is the surge of LH secreted by the anterior pituitary at the midpoint of the menstrual cycle (see Fig. 1.16). Within hours of exposure to the LH surge, the follicle reorganizes its program of gene expression from one directed toward development of the follicle to one producing molecules that set into gear the processes of follicular rupture and ovulation. Shortly after the LH peak, local blood flow increases in the outer layers of the follicular wall. Along with the increased blood flow, plasma proteins leak into the tissues through the postcapillary venules, with resulting local edema. The edema and the release of certain pharmacologically active compounds, such as prostaglandins, histamine, vasopressin, and plasminogen activator, provide the starting point for a series of reactions that result in the local production of matrix metalloproteinases—a family of lytic enzymes that degrade components of the extracellular matrix. At the same time, the secretion of hyaluronic acid by cells of the cumulus results in a loosening of the cells surrounding the egg. The lytic action of the matrix metalloproteinases produces an inflammatorylike reaction that ultimately results in rupture of the outer follicular wall about 28 to 36 hours after the LH surge (Fig. 2.1). Within minutes after rupture of the follicular wall, the cumulus oophorus detaches from the granulosa, and the egg is released from the ovary.
Egg Transport
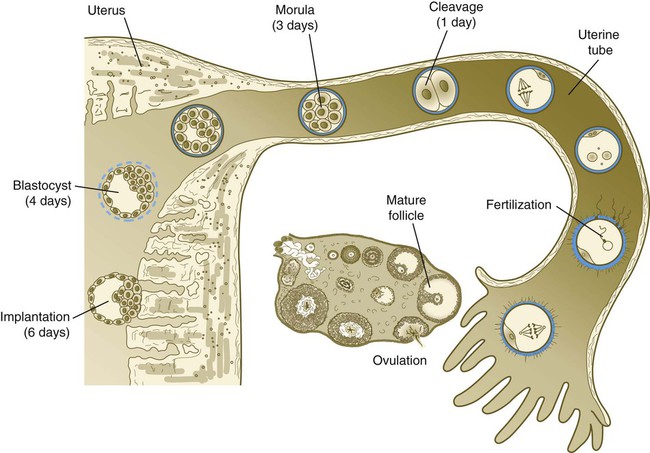
Tubal transport of the egg usually takes 3 to 4 days, whether or not fertilization occurs (Fig. 2.2). Egg transport typically occurs in two phases: slow transport in the ampulla (approximately 72 hours) and a more rapid phase (8 hours) during which the egg or embryo passes through the isthmus and into the uterus (see p. 51). By a poorly understood mechanism, possibly local edema or reduced muscular activity, the egg is temporarily prevented from entering the isthmic portion of the tube, but under the influence of progesterone, the uterotubal junction relaxes and permits entry of the ovum.
By roughly 80 hours after ovulation, the ovulated egg or embryo has passed from the uterine tube into the uterus. If fertilization has not occurred, the egg degenerates and is phagocytized. (Implantation of the embryo is discussed in Chapter 3.)
Sperm Transport
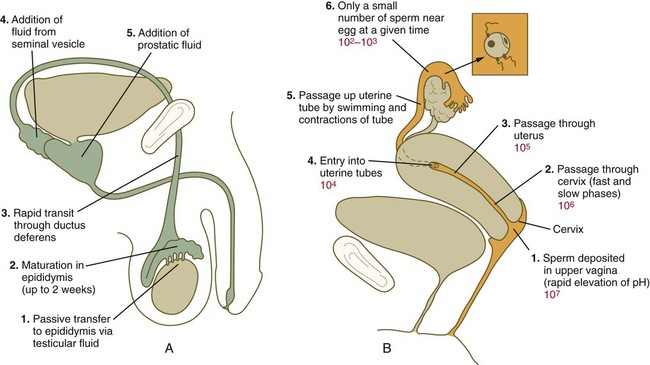
After spermiogenesis in the seminiferous tubules, the spermatozoa are morphologically mature but are nonmotile and incapable of fertilizing an egg (Fig. 2.3). Spermatozoa are passively transported via testicular fluid from the seminiferous tubules to the caput (head) of the epididymis through the rete testis and the efferent ductules. They are propelled by fluid pressure generated in the seminiferous tubules and are assisted by smooth muscle contractions and ciliary currents in the efferent ductules. Spermatozoa spend about 12 days in the highly convoluted duct of the epididymis, which measures 6 m in the human, during which time they undergo biochemical maturation. This period of maturation is associated with changes in the glycoproteins in the plasma membrane of the sperm head. By the time the spermatozoa have reached the cauda (tail) of the epididymis, they are capable of fertilizing an egg.
In the female reproductive tract, sperm transport begins in the upper vagina and ends in the ampulla of the uterine tube, where the spermatozoa make contact with the ovulated egg. During copulation, the seminal fluid is normally deposited in the upper vagina (see Fig. 2.3), where its composition and buffering capacity immediately protect the spermatozoa from the acid fluid found in the upper vaginal area. The acidic vaginal fluid normally serves a bactericidal function in protecting the cervical canal from pathogenic organisms. Within about 10 seconds, the pH of the upper vagina is increased from 4.3 to as much as 7.2. The buffering effect lasts only a few minutes in humans, but it provides enough time for the spermatozoa to approach the cervix in an environment (pH 6.0 to 6.5) optimal for sperm motility.
Once inside the uterine tube, the spermatozoa collect in the isthmus and bind to the epithelium for about 24 hours. During this time, they are influenced by secretions of the tube to undergo the capacitation reaction. One phase of capacitation is removal of cholesterol from the surface of the sperm. Cholesterol is a component of semen and acts to inhibit premature capacitation. The next phase of capacitation consists of removal of many of the glycoproteins that were deposited on the surface of the spermatozoa during their tenure in the epididymis. Capacitation is required for spermatozoa to be able to fertilize an egg (specifically, to undergo the acrosome reaction; see p. 29). After the capacitation reaction, the spermatozoa undergo a period of hyperactivity and detach from the tubal epithelium. Hyperactivation helps the spermatozoa to break free of the bonds that held them to the tubal epithelium. It also assists the sperm in penetrating isthmic mucus, as well as the corona radiata and the zona pellucida, which surround the ovum. Only small numbers of sperm are released at a given time. This may reduce the chances of polyspermy (see p. 31).
Formation and Function of the Corpus Luteum of Ovulation and Pregnancy
While the ovulated egg is passing through the uterine tubes, the ruptured follicle from which it arose undergoes a series of striking changes that are essential for the progression of events leading to and supporting pregnancy (see Fig. 1.8). Soon after ovulation, the basement membrane that separates the granulosa cells from the theca interna breaks down, thus allowing thecal blood vessels to grow into the cavity of the ruptured follicle. The granulosa cells simultaneously undergo a series of major changes in form and function (luteinization). Within 30 to 40 hours of the LH surge, these cells, now called granulosa lutein cells, begin secreting increasing amounts of progesterone along with some estrogen. This pattern of secretion provides the hormonal basis for the changes in the female reproductive tissues during the last half of the menstrual cycle. During this period, the follicle continues to enlarge. Because of its yellow color, it is known as the corpus luteum. The granulosa lutein cells are terminally differentiated. They have stopped dividing, but they continue to secrete progesterone for 10 days.
Fertilization
Penetration of the Corona Radiata
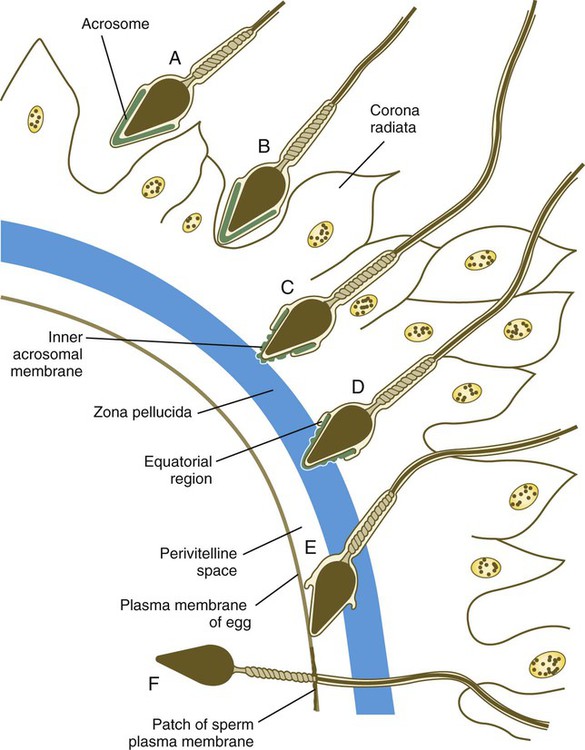
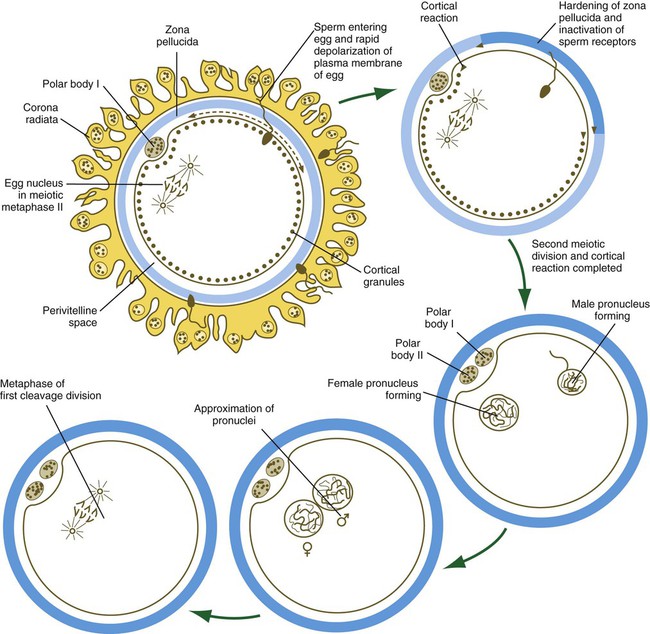
When the spermatozoa first encounter the ovulated egg in the ampullary part of the uterine tube, they are confronted by the corona radiata and some remnants of the cumulus oophorus, which represents the outer layer of the egg complex (Fig. 2.4). The corona radiata is a highly cellular layer with an intercellular matrix consisting of proteins and a high concentration of carbohydrates, especially hyaluronic acid. It is widely believed that hyaluronidase emanating from the sperm head plays a major role in penetration of the corona radiata, but the active swimming movements of the spermatozoa are also important.
Attachment to and Penetration of the Zona Pellucida
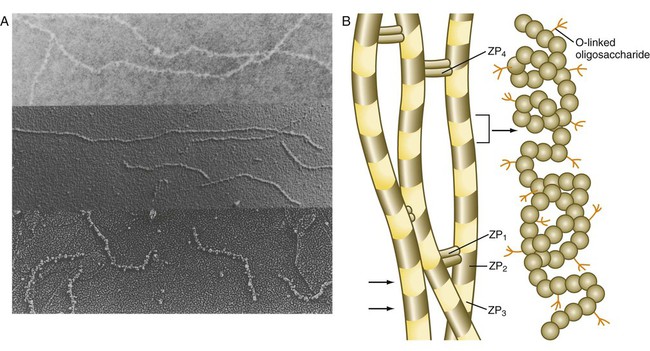
The zona pellucida, which is 13 µm thick in humans, consists principally of four glycoproteins—ZP1 to ZP4. ZP2 and ZP3 combine to form basic units that polymerize into long filaments. These filaments are periodically linked by cross-bridges of ZP1 and ZP4 molecules (Fig. 2.5). The zona pellucida of an unfertilized mouse egg is estimated to contain more than 1 billion copies of the ZP3 protein.
After they have penetrated the corona radiata, spermatozoa bind tightly to the zona pellucida by means of the plasma membrane of the sperm head (see Fig. 2.4). Spermatozoa bind specifically to a sialic acid molecule, which is the terminal part of a sequence of four sugars at the end of O-linked oligosaccharides that are attached to the polypeptide core of the ZP3 molecule. Molecules on the surface of the sperm head are specific binding sites for the ZP3 sperm receptors on the zona pellucida. More than 24 molecules have been proposed, but the identity of the zona-binding molecules remains unknown. Interspecies molecular differences in the sperm-binding regions of the ZP3 molecule may serve as the basis for the inability of spermatozoa of one species to fertilize an egg of another species. In mammals, there is less species variation in the composition of ZP3; this may explain why penetration of the zona pellucida by spermatozoa of closely related mammalian species is sometimes possible, whereas it is rare among lower animals.
On binding to the zona pellucida, mammalian spermatozoa undergo the acrosomal reaction. The essence of the acrosomal reaction is the fusion of parts of the outer acrosomal membrane with the overlying plasma membrane and the pinching off of fused parts as small vesicles. This results in the liberation of the multitude of enzymes that are stored in the acrosome (Box 2.1).
After the acrosomal reaction, the inner acrosomal membrane forms the outer surface covering of most of the sperm head (see Fig. 2.4D). Toward the base of the sperm head (in the equatorial region), the inner acrosomal membrane fuses with the remaining postacrosomal plasma membrane to maintain membrane continuity around the sperm head.
Binding and Fusion of Spermatozoon and Egg
After initial fusion, the contents of the spermatozoon (the head, the midpiece, and usually the tail) sink into the egg (Fig. 2.6), whereas the sperm’s plasma membrane, which is antigenically distinct from that of the egg, becomes incorporated into the egg’s plasma membrane and remains recognizable at least until the start of cleavage. Although mitochondria located in the sperm neck enter the egg, they do not contribute to the functional mitochondrial complement of the zygote. In humans, the sperm contributes the centrosome, which is required for cell cleavage.
Completion of Meiosis and the Development of Pronuclei in the Egg
After penetration of the egg by the spermatozoon, the nucleus of the egg, which had been arrested in metaphase of the second meiotic division, completes the last division and releases a second polar body into the perivitelline space (see Fig. 2.6).
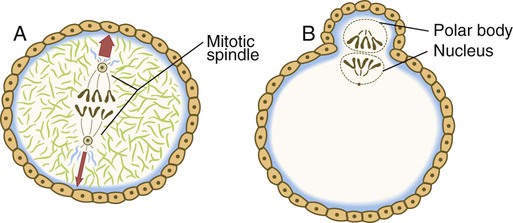
The nucleus of the oocyte moves toward the cortex as the result of the action of myosin molecules acting on a network of actin filaments that connect one pole of the mitotic spindle to the cortex. The resulting contraction draws the entire mitotic apparatus toward the surface of the cell (Fig. 2.7). This determines the location at which both the first and second polar bodies are extruded.
What is Accomplished by Fertilization?
The process of fertilization ties together many biological loose ends, as follows:
1. It stimulates the egg to complete the second meiotic division.
2. It restores to the zygote the normal diploid number of chromosomes (46 in humans).
3. The genetic sex of the future embryo is determined by the chromosomal complement of the spermatozoon. (If the sperm contains 22 autosomes and an X chromosome, the embryo is a genetic female, and if it contains 22 autosomes and a Y chromosome, the embryo is a male. See Chapter 16 for further details.)
4. Through the mingling of maternal and paternal chromosomes, the zygote is a genetically unique product of chromosomal reassortment, which is important for the viability of any species.
5. The process of fertilization causes metabolic activation of the egg, which is necessary for cleavage and subsequent embryonic development to occur.
Summary
 Ovulation is stimulated by a surge of LH and FSH in the blood. Expulsion of the ovum from the graafian follicle involves local edema, ischemia, and collagen breakdown, with a possible contribution by fluid pressure and smooth muscle activity in rupturing the follicular wall.
Ovulation is stimulated by a surge of LH and FSH in the blood. Expulsion of the ovum from the graafian follicle involves local edema, ischemia, and collagen breakdown, with a possible contribution by fluid pressure and smooth muscle activity in rupturing the follicular wall.
 The ovulated egg is swept into the uterine tube and transported through it by ciliary action and smooth muscle contractions as it awaits fertilization by a sperm cell.
The ovulated egg is swept into the uterine tube and transported through it by ciliary action and smooth muscle contractions as it awaits fertilization by a sperm cell.
 Sperm transport in the male reproductive tract involves a slow exit from the seminiferous tubules, maturation in the epididymis, and rapid expulsion at ejaculation, where the spermatozoa are joined by secretions from the prostate and seminal vesicles to form semen.
Sperm transport in the male reproductive tract involves a slow exit from the seminiferous tubules, maturation in the epididymis, and rapid expulsion at ejaculation, where the spermatozoa are joined by secretions from the prostate and seminal vesicles to form semen.
 In the female reproductive tract, sperm transport involves entry into the cervical canal from the vagina, passage through the cervical mucus, and transport through the uterus into the uterine tubes, where capacitation occurs. The meeting of egg and sperm typically occurs in the upper third of the uterine tube.
In the female reproductive tract, sperm transport involves entry into the cervical canal from the vagina, passage through the cervical mucus, and transport through the uterus into the uterine tubes, where capacitation occurs. The meeting of egg and sperm typically occurs in the upper third of the uterine tube.
 The fertilization process consists of several sequential events:
The fertilization process consists of several sequential events:
1. Penetration of the corona radiata
2. Attachment to the zona pellucida
3. Acrosomal reaction and penetration of the zona pellucida
4. Binding and fusion of sperm and egg
6. Metabolic activation of the egg
7. Decondensation of the sperm nucleus
 Attachment of the spermatozoon to the zona pellucida is mediated by the ZP3 protein, which also stimulates the acrosomal reaction.
Attachment of the spermatozoon to the zona pellucida is mediated by the ZP3 protein, which also stimulates the acrosomal reaction.
 The acrosomal reaction involves fusion of the outer acrosomal membrane with the plasma membrane of the sperm cell and the fragmentation of the fused membranes, thus leading to the release of the acrosomal enzymes. One of the acrosomal enzymes, acrosin, is a serine proteinase, which digests components of the zona pellucida and assists the penetration of the swimming spermatozoa through the zona.
The acrosomal reaction involves fusion of the outer acrosomal membrane with the plasma membrane of the sperm cell and the fragmentation of the fused membranes, thus leading to the release of the acrosomal enzymes. One of the acrosomal enzymes, acrosin, is a serine proteinase, which digests components of the zona pellucida and assists the penetration of the swimming spermatozoa through the zona.
 After fusion of the spermatozoa to the egg membrane, a rapid electrical depolarization produces the first block to polyspermy in the egg. This is followed by a wave of Ca++ that causes the cortical granules to release their contents into the perivitelline space and ultimately to inactivate the sperm receptors in the zona pellucida.
After fusion of the spermatozoa to the egg membrane, a rapid electrical depolarization produces the first block to polyspermy in the egg. This is followed by a wave of Ca++ that causes the cortical granules to release their contents into the perivitelline space and ultimately to inactivate the sperm receptors in the zona pellucida.
 Sperm penetration stimulates a rapid intensification of respiration and metabolism of the egg.
Sperm penetration stimulates a rapid intensification of respiration and metabolism of the egg.
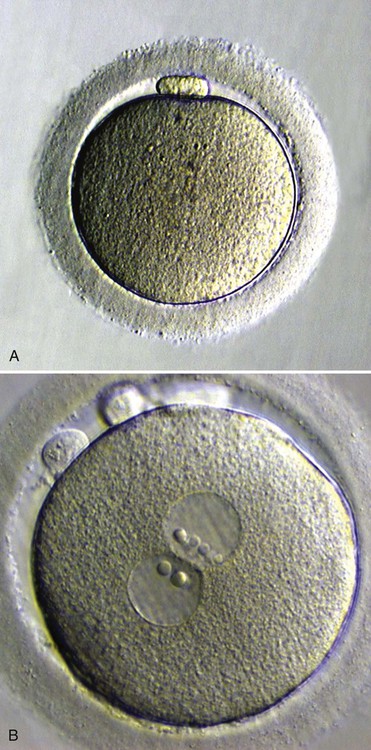
 Within the egg, the nuclear material of the spermatozoon decondenses and forms the male pronucleus. At the same time, the egg completes the second meiotic division, and the remaining nuclear material becomes surrounded by a membrane, to form the female pronucleus.
Within the egg, the nuclear material of the spermatozoon decondenses and forms the male pronucleus. At the same time, the egg completes the second meiotic division, and the remaining nuclear material becomes surrounded by a membrane, to form the female pronucleus.
 After DNA replication, the male and female pronuclei join, and their chromosomes become organized for a mitotic division. Fertilization is complete, and the fertilized egg is properly called a zygote.
After DNA replication, the male and female pronuclei join, and their chromosomes become organized for a mitotic division. Fertilization is complete, and the fertilized egg is properly called a zygote.
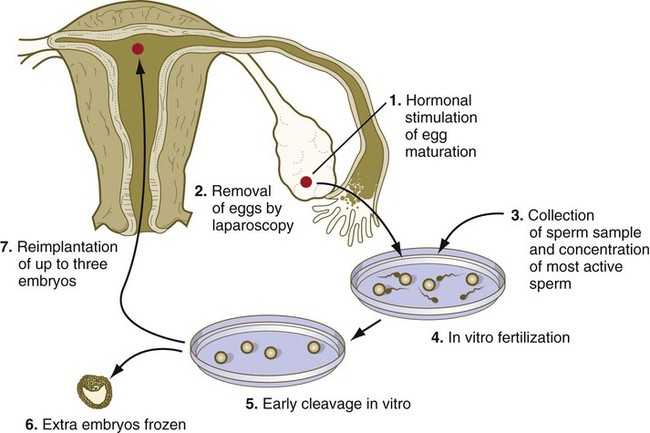
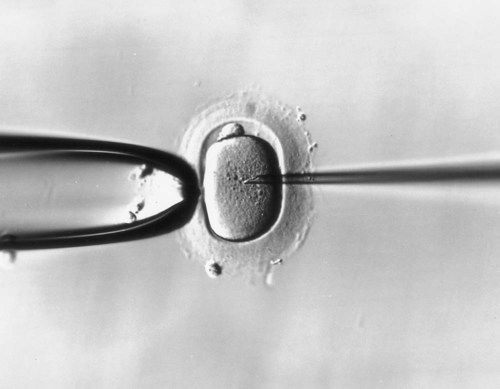
 Treatment of infertility by in vitro fertilization and embryo transfer is a multistage process involving stimulating gamete production by drugs such as clomiphene citrate, obtaining eggs by laparoscopic techniques in the woman, storing gametes by freezing, performing in vitro fertilization and culture of embryos, preserving the embryo, and transferring the embryo to the mother (Clinical Correlation 2.1).
Treatment of infertility by in vitro fertilization and embryo transfer is a multistage process involving stimulating gamete production by drugs such as clomiphene citrate, obtaining eggs by laparoscopic techniques in the woman, storing gametes by freezing, performing in vitro fertilization and culture of embryos, preserving the embryo, and transferring the embryo to the mother (Clinical Correlation 2.1).
 Other techniques used for the treatment of infertility are gamete intrafallopian transfer (GIFT), which is the transfer of gametes directly into the uterine tube, and zygote intrafallopian transfer (ZIFT), with is the transfer of zygotes into the uterine tube. These techniques can be used with biological and surrogate mothers.
Other techniques used for the treatment of infertility are gamete intrafallopian transfer (GIFT), which is the transfer of gametes directly into the uterine tube, and zygote intrafallopian transfer (ZIFT), with is the transfer of zygotes into the uterine tube. These techniques can be used with biological and surrogate mothers.