24.2 The thymus258
Basic principles
The cells which circulate in the peripheral blood can be classified simply into those that are non-nucleated (erythrocytes and platelets) and those that are nucleated (leucocytes or white blood cells). White blood cells can be further subclassified into three main cell types:
• granulocytes
• monocytes
• lymphocytes.
The main function of these cells is to protect against infection. Lymphocytes and monocytes circulate around the body in blood vessels and lymphatic vessels, but they also accumulate in organised masses called lymphoid tissues. These organised masses together are known as the lymphoreticular system and the main components of this system are the lymph nodes, thymus, spleen, tonsils, adenoids, and the Peyer patches. The latter three tissues are known as mucosa-associated lymphoid tissue (MALT).
24.1. Lymph nodes
Structure and function
Lymph nodes are ovoid, encapsulated structures, which range in size from a few millimetres to a few centimetres. They are situated along the course of lymphatic vessels and tend to occur in groups where these vessels converge (e.g. the axilla, groin, neck and mediastinum). Lymph is essentially interstitial fluid containing proteins that need to return to the bloodstream, but which are prevented from doing so within the tissues because of overwhelming hydrostatic pressure. Lymph is therefore carried away from the tissues in small peripheral lymphatic vessels, which converge to form larger vessels, until eventually a single large lymphatic vessel called the thoracic duct ultimately drains the lymph into the bloodstream at the root of the neck. Before the lymph enters the bloodstream, it must pass through one or more lymph nodes. Within the lymph nodes, foreign agents and unwanted materials, which have gained access to the tissues, are entrapped and an immune response is mounted.
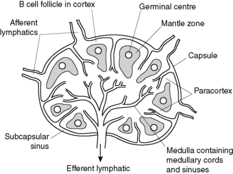
Each lymph node is divided into three main regions: the cortex, the paracortex and the medulla (Figure 66).
 |
| Figure 66 |
The cortex
The cortex is the area just beneath the capsule, and it contains spherical aggregations of B lymphocytes (B cells). These aggregations are called primary follicles. B cells are involved in humoral immunity and the primary follicle is the principal site of B cell activation in response to antigenic stimulation. Antigens that enter the lymph node are presented to the B cells in the primary follicle. Activated B cells enlarge and undergo a series of changes, resulting ultimately in the production of immunoglobulin-secreting plasma cells. Hence, after antigenic stimulation, the primary follicle enlarges and develops a pale-staining germinal centre that contains large, activated B cells. The germinal centre is surrounded by a rim of small unchallenged B cells called the mantle zone.
The paracortex
The paracortex is the area between the follicles and is rich in T lymphocytes (T cells). T cells are involved in cell-mediated immunity. Scattered histiocytic cells are also present.
The medulla
The medulla contains the medullary cords and sinuses. The sinuses are lined by macrophages, which phagocytose particulate material within the lymph. The medullary cords contain numerous plasma cells, which secrete immunoglobulins (antibodies).
Lymph enters the lymph node via multiple afferent lymphatic vessels, which perforate the fibrous capsule and empty into a slit-like space just beneath the capsule called the subcapsular sinus. From there, the lymph enters the cortex via multiple, small cortical sinuses, which penetrate into the node. In the medulla, these sinuses begin to converge into larger sinuses (the medullary sinuses) and, in turn, the medullary sinuses join to form a single efferent lymphatic vessel.
Lymphadenopathy
When there is a pathological process affecting a lymph node, it usually becomes enlarged. Lymphadenopathy is a term used to denote lymph node enlargement. Lymphadenopathy may be localised or widespread. There are two main causes:
• non-neoplastic (reactive) lymphadenopathy
• neoplastic lymphadenopathy.
Neoplasms of the lymph nodes can be divided into primary (lymphoma) or secondary (metastases).
Non-neoplastic (reactive) lymphadenopathy
Reactive lymph nodes enlarge because there is proliferation of one or more cell types within them. The various antigenic stimuli evoke the reactive proliferation of particular cell types within the lymph node, and may induce other changes. In many cases, the morphological changes induced by these aetiological agents are non-specific, such that precise diagnosis of the causal agent is not possible on histological grounds alone (blood tests may be needed). In other cases, the reactive changes are entity specific, such that the pathologist is able to make an exact diagnosis.
Non-specific reactive changes
Lymph nodes may react in one of five different ways to antigenic stimulation. Often, a combination of one or more of these patterns is seen.
Acute non-specific lymphadenitis This occurs when there is direct drainage of pyogenic infectious microbes into a lymph node, causing a localised acute inflammatory response. Affected lymph nodes become enlarged and tender. Microscopically, there is lymph node oedema and hyperaemia, and neutrophil polymorphs migrate from the vasculature into the nodal parenchyma. There may be progression to abscess formation.
Follicular hyperplasia This occurs when there is a B cell response, and is characterised by marked enlargement and prominence of the germinal centres.
Paracortical hyperplasia This is the result of a T cell response. The paracortex expands and encroaches on the follicles. This pattern is often encountered in reactions induced by drugs and acute viral infections.
Sinus histiocytosis This is seen if there is a marked proliferation of histiocytic cells, which normally occupy the sinuses. The sinuses become dilated and engorged with numerous histiocytes.
Granulomatous lymphadenitis This term refers to the formation of granulomas within a lymph node. A granuloma is a collection of macrophages. Causes include:
• tuberculosis
• sarcoidosis
• cat scratch disease
• Crohn’s disease
• toxoplasmosis
• reaction to foreign material.
Entity-specific reactive changes
A number of reactive conditions affecting lymph nodes induce morphological appearances which, although often complex, may be distinctive enough to enable the pathologist to make an exact diagnosis. Such conditions include certain infections (e.g. toxoplasmosis, human immunodeficiency virus (HIV) infection, Epstein–Barr virus infection), some connective tissue diseases (e.g. systemic lupus erythematosus), reactions to types of foreign material (e.g. silicone), certain drug reactions (e.g. some anticonvulsants) and conditions of uncertain aetiology (e.g. Castleman disease).
Neoplastic lymphadenopathy
Lymphoma
Lymphomas are classified into subtypes according to histological appearance and immunophenotype (i.e. the staining patterns on immunohistochemistry). Different subtypes behave differently, have different prognoses, and require different treatments. Hence, classification of lymphoma is the means by which pathologists convey meaningful information to the clinicians. There have been a number of classification systems in the past (e.g. Kiel classification, Working Formulation for Clinical Usage), all of which have had their merits. However, many entities went by different names in the different classification systems, and this was a source of confusion for both pathologists and clinicians. In addition, there have been major advances in our knowledge and understanding of lymphoid neoplasms since the 1980s, and many new entities have now been recognised. To overcome these problems, a new unifying lymphoma classification system was devised and published by the International Lymphoma Study Group in 1994, and it was called the ‘Revised European-American Classification of Lymphoid Neoplasms’ (REAL). A few years later, when the World Health Organization (WHO) was developing a new classification system for neoplastic diseases of the haematopoietic and lymphoid tissues, it adopted the REAL classification, making only a few modifications. It is the 2001 WHO Lymphoma Classification System that is currently used in clinical practice.
There are two main types of lymphoma – Hodgkin’s lymphoma and non-Hodgkin’s lymphoma. They are considered separate for two main reasons:
• Hodgkin’s lymphoma is characterised morphologically by the presence of unique neoplastic cells called Reed–Sternberg cells
• Hodgkin’s lymphoma may be associated with certain clinical symptoms.
Hodgkin’s lymphoma
Hodgkin’s lymphoma is differentiated from the other types of lymphoma by the presence of distinctive and diagnostic tumour cells called Reed–Sternberg cells (RS cells), although for the diagnosis of Hodgkin’s lymphoma to be made, the RS cells must be associated with the appropriate cellular background. Classically, the RS cell is large with a pale, bilobed nucleus and large, prominent, eosinophilic nucleoli. The nucleoli are bounded by a clear zone, giving the cell a characteristic ‘owl-eye’ appearance. The various subtypes of Hodgkin’s lymphoma are shown in Table 57.
| *Within the non-Hodgkin’s group, only the most common subtypes are shown. |
|||
| †The subtypes which constitute the majority of lymphoid neoplasms in adults. |
|||
| §The subtypes most common in children. |
|||
| Hodgkin’s lymphoma | • Nodular lymphocyte-predominant Hodgkin’s lymphoma | ||
| • Classical Hodgkin’s lymphoma |
• Nodular sclerosing Hodgkin’s lymphoma
• Lymphocyte-rich classical Hodgkin’s lymphoma
• Mixed-cellularity Hodgkin’s lymphoma
• Lymphocytedepleted Hodgkin’s lymphoma
|
||
| Non-Hodgkin’s lymphoma* | • B cell neoplasms |
• Precursor B cell neoplasms
• Mature B cell neoplasms
|
|
| • T/NK cell neoplasms |
• Precursor T cell neoplasms
• Mature T cell neoplasms
|
• Precursor T lymphoblastic lymphoma§
• Mycosis fungoides/Sézary syndrome
• Peripheral T cell lymphoma
• Angioimmunoblastic T cell lymphoma
• Anaplastic large-cell lymphoma
|
|
Clinical features Typical patients are young adults, the peak incidence being in the third and fourth decades of life. Patients usually present with painless lymphadenopathy, which typically affects the upper half of the body. The enlarged lymph nodes are discrete, mobile, and rubbery in consistency. Patients may also present with systemic symptoms (unexplained pyrexia, drenching night sweats, unexplained weight loss).
Staging Staging is an important determinant of treatment and prognosis. The staging system used is the Ann Arbor system:
• Stage I – involvement of a single lymph node region (I) or single extranodal organ or site (Ie).
• Stage II – involvement of two or more lymph node regions on the same side of the diaphragm (II), or one extranodal organ plus one or more lymph node regions on the same side of the diaphragm (IIe).
• Stage III – involvement of lymph regions on both sides of the diaphragm (III), which may be associated with splenic involvement (IIIs), or localised involvement of an extranodal site (IIIe), or both (IIIse).
• Stage IV – diffuse or disseminated involvement of one or more extralymphatic sites, such as the liver, lung, or bone marrow, with or without lymph node involvement.
Any of these stages may be followed by the suffix A or B, depending on whether the systemic symptoms are absent (A) or present (B). The presence of systemic symptoms is associated with a poorer prognosis.
Treatment and prognosis Factors associated with a poorer prognosis are:
• advancing age
• systemic symptoms
• advanced stage
• aggressive histological subtype (e.g. lymphocyte-depleted Hodgkin’s lymphoma)
• abnormal blood markers.
With advances in treatment during the late 1990s, the prognosis of Hodgkin’s lymphoma has generally improved, so that even with advanced disease, the 5-year disease-free survival is 60–70%.
Non-Hodgkin’s lymphoma
Within this category, there is a much wider spectrum of lymphoid neoplasms, showing a marked diversity in their histological appearance, immunophenotype, biological behaviour, response to therapy, prognosis, and clinical settings, including typical age at onset. Because of the extremely complex nature of this group of lymphoid neoplasms, only a framework for the basic understanding of non-Hodgkin’s lymphoma will be presented here as detailed descriptions of the various subtypes are not necessary in this text. The most frequently encountered subtypes are shown in Table 57. In basic terms, non-Hodgkin’s lymphoma can be subdivided according to the type of lymphoid cell of origin into B cell non-Hodgkin’s lymphoma and T cell/natural killer (NK) cell non-Hodgkin’s lymphoma. These B and T cell neoplasms can be further subdivided into precursor (lymphoblastic) neoplasms or mature (peripheral) neoplasms. Within these categories, the various distinct entities are then listed.
Diagnosis of lymphoma
In order to make a definitive diagnosis of lymphoma, a lymph node biopsy is often performed and the node examined histologically. In some cases, cytogenetic analysis is used in conjunction with histological assessment in order to make the correct diagnosis. Lymph node biopsy may be preceded by fine-needle aspiration cytology.
Secondary tumours
The tumours most often encountered in the lymph nodes are metastatic rather than primary. Most carcinomas, melanomas, and some sarcomas have the capacity to metastasise via the lymphatic system.
24.2. The thymus
Structure and function
The thymus is an encapsulated bilobed structure, which is situated in the anterior superior mediastinum.
It is embryologically derived from the third and, occasionally, fourth pharyngeal pouches. The gland grows in size until puberty (weighing up to 50g), thereafter undergoing progressive atrophy and gradual replacement by fibrofatty tissue.
The thymus has a central role in cell-mediated immunity. During fetal development, stem cells derived from the bone marrow migrate to the thymus where they differentiate and mature into T cells. A small population of B cells is also present. The thymus has a lymphoid component and an epithelial component.
Disorders of the thymus
Agenesis and aplasia
Thymic hyperplasia (thymic follicular hyperplasia)
This condition is characterised by the presence of lymphoid follicles with germinal centres within the thymus. Thymic hyperplasia is seen in ∼70% of patients with the autoimmune condition myasthenia gravis. In these patients, the thymus appears to be the main source of the autoantibody, and thymectomy may therefore be of benefit. A degree of thymic hyperplasia may also be seen in other autoimmune disorders such as Graves’ disease, systemic lupus erythematosus, systemic sclerosis and rheumatoid arthritis.
Neoplasms of the thymus
The main types of neoplasms that can arise within the thymus are:
• thymoma
• thymic carcinoma
• lymphoma
• germ cell tumours (rare)
• neuroendocrine tumours (rare).
Thymomas
Thymomas are tumours of thymic epithelial cells. Many are asymptomatic and are only detected on chest X-ray performed for other reasons. Others become manifest clinically through local pressure symptoms, e.g. stridor, cough or dyspnoea, or through their association with myasthenia gravis. The majority of thymomas are completely benign.
Thymic carcinomas
They are usually highly aggressive.
Lymphoma
Can occur in the thymus. The thymus is not an uncommon site for Hodgkin’s lymphoma and thymic involvement by non-Hodgkin’s lymphoma, mostly T cell neoplasms or large B cell lymphomas, is frequently encountered.
Germ cell tumours
Tumours such as seminomas and teratomas may be encountered. They are derived from germ cells that have failed to migrate to the gonads during fetal life.
Neuroendocrine tumours
Thymic carcinoids may arise from neuroendocrine cells that are scattered throughout the gland.
24.3. The spleen
Structure and function
The spleen is an encapsulated organ, which is situated in the left upper quadrant of the abdomen. The spleen receives blood from the splenic artery and, in general, it is to the circulatory system what the lymph nodes are to the lymphatic system. The adult organ has two main functions:
• filtration and phagocytosis of obsolescent red blood cells and bacteria
• mounting immunological responses.
Reflecting these functions, the spleen has two main components: the white pulp, which is lymphoid tissue and is where immunological responses are mounted, and the red pulp, which is the site of blood filtration and phagocytosis.
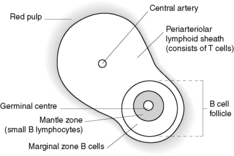
The white pulp
The splenic artery enters the spleen at the hilum and then divides to give rise to numerous central arteries, which ramify through the organ. Each central artery becomes ensheathed in lymphoid cells, which constitute the white pulp. The white pulp consists of periarteriolar lymphoid sheaths (PALS) of T cells, which are intermittently expanded by B cell follicles. The unstimulated B cell follicle consists of a nodule of small B lymphocytes surrounded by a rim of larger, marginal zone B cells. If the B cell follicle is stimulated, a germinal centre forms in the centre, surrounded by a mantle zone of small unchallenged B cells, which are surrounded by the marginal zone (Figure 67).
 |
| Figure 67 |
The red pulp
The red pulp consists of sinusoids separated by splenic cords (Figure 68). The splenic cords contain numerous macrophages. Once blood has left the central artery of the white pulp, it passes into capillaries. Blood within the capillaries can either drain directly into the sinusoids and then into the splenic veins (‘closed’ circulation), or it can first enter the splenic cords and then pass into the sinusoids through fenestrations in the sinusoid epithelium (‘open’ circulation). In the open circulation, red blood cells must undergo extreme deformation to pass from the cords into the sinusoids. Obsolescent, damaged or abnormal red blood cells have reduced deformability and, therefore, become trapped within the splenic cords where they are phagocytosed.
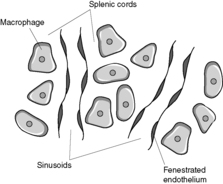 |
| Figure 68 |
Macroscopically, the cut surface of the spleen is red (red pulp) dotted with grey specks (white pulp).
Disorders of the spleen
Congenital abnormalities
An accessory (supernumerary) spleen is not uncommon, the most usual location being the hilum of the spleen. Congenital absence of the spleen (asplenia) and polysplenia are rare and are often associated with other congenital defects.
Splenomegaly
This term refers to enlargement of the spleen. With very few exceptions, pathological states in the spleen are manifest as splenomegaly, and there are numerous causes. However, apart from being an indicator of an underlying disorder, splenomegaly may itself cause problems by inducing portal hypertension (because of increased blood flow though the organ) and hypersplenism. Hypersplenism is characterised by splenomegaly, a deficiency in one or more of the cellular elements in the blood, and correction of the cytopenia(s) by splenectomy.
Infection
The morphological changes which occur in the spleen during infection often vary depending on the particular infecting organism. In acute non-specific splenitis due to sepsis, the spleen becomes very soft and, when sliced, the substance of the spleen typically flows out from the cut surface. In infectious mononucleosis the splenic capsule is particularly vulnerable to rupture. In malaria, the spleen can reach a massive size and is markedly congested.
Congestive splenomegaly
Persistent elevation of the splenic or portal venous blood pressure can cause splenomegaly. The pressure may be raised due to prehepatic, hepatic or post-hepatic causes. Prehepatic causes include thrombosis of the splenic or portal veins. The most important hepatic cause is cirrhosis. Post-hepatic causes include thrombosis of the hepatic veins (Budd–Chiari syndrome), and raised inferior vena cava pressure associated with right heart failure.
The spleen may be considerably enlarged and the capsule is often thickened and fibrotic. The cut surface is dark red with inconspicuous white pulp. Microscopically, the increased venous pressure is reflected by distension of the sinusoids by red blood cells. Collagen may be laid down in the basement membranes of distended sinusoids, resulting in impairment of blood flow from the cords to the sinusoids. The red blood cells are therefore exposed to the cord macrophages for much longer, and hypersplenism may result. The elevated venous pressure can lead to sinusoid rupture and intraparenchymal haemorrhage. Organisation of these areas gives rise to small brown nodules called Gamna–Gandy bodies, which are visible with the naked eye.
Lymphohaematogenous neoplasms
The spleen is not infrequently involved in a number of lymphoid and haematological neoplasms. Each type of neoplasm is associated with a different pattern of splenic involvement. For example, in chronic myeloid leukaemia, the red pulp is filled with mature myeloid precursors, and in Hodgkin’s lymphoma, the white pulp is preferentially involved with the formation of expansile nodules.
Non-lymphohaematogenous tumours of the spleen are rare, but include hamartomas, lymphangiomas and haemangiomas.
Splenic rupture
Rupture of the spleen is most commonly the result of blunt trauma, such as can occur during a car accident when the steering wheel may inflict a severe blow to the upper abdomen. Spontaneous splenic rupture may occur if the organ is enlarged and abnormally soft, as in infectious mononucleosis. Rupture is followed by extensive intraperitoneal haemorrhage. Prompt splenectomy is necessary to prevent death from hypovolaemic shock.
Self-assessment: questions
One best answer question
2. A 60-year old man died at home. He had a history of increasing shortness of breath. A coroner’s post mortem was carried out to determine the cause of death. The main findings were a fish-mouth deformity of the mitral valve, left atrial dilatation and right ventricular hypertrophy, markedly congested and oedematous lungs, a ‘nutmeg liver’ and a mildly enlarged spleen containing small, firm, brown nodules. The most likely diagnosis is:
a. carcinomatosis
b. infective endocarditis
c. sarcoidosis
d. pulmonary thromboembolism
e. congestive cardiac failure
True-false questions
1. The following statements are correct:
a. lymph nodes are only found in the neck
b. after passing through lymph nodes, lymph enters the bloodstream via the thoracic duct
c. lymph node enlargement always indicates a malignant process
d. acute viral infections cause paracortical expansion
e. the primary follicles within a lymph node are composed of T cells
2. The following statements are correct:
a. lymphomas may affect the gastrointestinal tract
b. Hodgkin’s lymphoma is one of the most common forms of malignancy in young adults
c. lymph nodes involved by Hodgkin’s lymphoma may become painful on consumption of alcohol
d. non-Hodgkin’s lymphomas are characterised by the presence of Reed–Sternberg cells
e. the definitive diagnosis of lymphoma requires histological assessment of a lymph node biopsy specimen
3. The following statements regarding the thymus are correct:
a. the thymus is situated in the anterior superior mediastinum
b. thymomas are tumours of thymic T cells
c. thymic hyperplasia may be associated with myasthenia gravis
d. DiGeorge syndrome is associated with thymic enlargement
e. the thymus is most active in late adult life
4. The following statements regarding the spleen are correct:
a. the spleen is composed of white pulp and red pulp
b. malaria is associated with development of massive splenomegaly
c. splenomegaly may be associated with leucopenia
d. spontaneous rupture of the spleen refers to splenic rupture when there is no underlying pathology
e. splenectomy should be supplemented by vaccination against Pneumococcus
5. The following statements are true:
a. reactive lymphadenopathy is usually self-limiting
b. lymph node enlargement associated with pyrexia and night sweats is always due to infection
c. lymphoma hardly ever occurs in young people
d. the diagnosis of lymphoma is made on clinical grounds alone
e. high-grade lymphomas always respond poorly to treatment
Case history questions
Case history 1
A 32-year-old woman presented to her general practitioner with unexplained weight loss and drenching night sweats for the last 6months. On examination she has right-sided cervical lymphadenopathy.
2. What further information would you seek from the history and examination to help determine the diagnosis?
3. What tests might you perform to establish the diagnosis?
Viva questions
1. How are lymphomas classified?
2. How might thymic tumours present clinically?
3. What are the main causes of congestive splenomegaly?
Self-assessment: answers
One best answer
2. e. Congestive cardiac failure. The fish-mouth deformity to the mitral valve denotes mitral stenosis. This leads to increased pressure in the left atrium (causing left atrial dilatation) and pulmonary veins (causing raised pulmonary venous pressure, pulmonary oedema and right ventricular hypertrophy). The ‘nutmeg liver’ represents chronic passive venous congestion of the liver and the small, firm, brown nodules in the spleen are most likely to be Gamna–Gandy bodies seen in longstanding severe congestive splenomegaly. All of the changes in the cardiac chambers, lungs, liver and spleen can be explained by the haemodynamic disturbances caused by severe mitral stenosis. The history of shortness of breath is consistent with cardiac failure. Carcinomatosis would appear as multiple tumour nodules at many sites. The characteristic vegetations observed on cardiac valves complicated by infective endocarditis were not seen in this case. Sarcoidosis can cause hepatosplenomegaly, but the changes described in the heart do not fit with this diagnosis. Thromboembolism can be seen in patients with mitral stenosis through the increased risk of atrial fibrillation (leading to thrombus formation), but since the thrombus would form in the left atrium it would enter the systemic circulation, not the pulmonary circulation. Systemic thromboembolism could lead to infarction of the spleen, but splenic infarcts usually appear pale and wedge shaped.
True-false answers
1.
a. False.
b. True.
c. False.
d. True.
e. False. Primary follicles are composed predominantly of B cells.
2.
a. True. The gastrointestinal tract contains mucosa-associated lymphoid tissue within its wall, which may be involved by lymphoma.
b. True.
c. True.
d. False. Reed–Sternberg cells are seen in Hodgkin’s lymphoma.
e. True.
3.
a. True.
b. False. Thymomas are tumours of thymic epithelial cells.
c. True.
d. False.
e. False. The thymus undergoes progressive atrophy after puberty.
4.
a. True.
b. True.
c. True. Splenomegaly may induce hypersplenism.
d. False. Spontaneous splenic rupture refers to non-traumatic rupture. If rupture is spontaneous, an underlying pathology predisposing to rupture must be considered.
e. True. Patients who have had a splenectomy have a life-long increased risk of infection with Pneumococcus and other encapsulated bacteria. Hence they should be considered for vaccination.
5.
a. True.
b. False. Hodgkin’s disease can present in a similar way.
c. False.
e. False.
Case history answers
Case history 1
1. There are two main causes of lymph node enlargement – a reactive process and a neoplastic process. In this case the lymphadenopathy is associated with a 6-month history of unexplained weight loss and drenching night sweats. This should raise the possibility of Hodgkin’s lymphoma, and the age of the patient and apparent involvement of only one group of nodes in the upper part of the body is supportive of this diagnosis. However, this combination of symptoms and findings is also seen during the course of some infections. Although an acute infection is unlikely with this long history, a chronic infection should be considered. For example, tuberculosis could cause pyrexia with night sweats, weight loss and cervical lymphadenopathy. Secondary involvement of the lymph nodes by a malignant tumour is unlikely in this age group.
2. Comment: Ask questions to determine if an infective process could be causing the symptoms, e.g. chronic cough, pharyngitis, laryngitis, etc. You should also ask if there has been any recent travel abroad where an unusual infection, such as tuberculosis, could have been contracted. Remember that lymph nodes affected by Hodgkin’s lymphoma may be painful on consumption of alcohol, so you should enquire about this. All systems should be examined in the physical examination to exclude an infective aetiology. Remember that a low-grade pyrexia may be present in Hodgkin’s lymphoma. Lymph nodes involved by an infective process may be tender on palpation. Lymph nodes affected by Hodgkin’s lymphoma are said to be discrete, mobile, and rubbery in consistency. You would also need to examine for lymphadenopathy elsewhere on the body, and see if there is any associated splenomegaly or hepatomegaly. Remember that the spleen may be enlarged in some infective processes, most notably infectious mononucleosis.
3. If the possibility of lymphoma is not excluded on history or examination, the next step is either to remove part, or all, of an affected lymph node for formal histological assessment or to perform a fine-needle aspiration (FNA) and examine the extracted cells for any signs of a neoplastic process. If signs of neoplasia are present on FNA, the lymph node must then be removed for formal histological assessment and diagnosis. Other tests may aid the diagnosis of infections. To diagnose tuberculosis, a chest X-ray should be the performed but ultimately the bacillus must be isolated in a tissue specimen, usually either sputum or a biopsy. Serology may be helpful in the diagnosis of some infections, e.g. infectious mononucleosis.
Viva answers
1. The classification of lymphoma has been a hot topic lately. The classification system now used by most clinicians is the WHO classification, which is based on the REAL classification. Lymphomas are divided into two main types – Hodgkin’s lymphoma and non-Hodgkin’s lymphoma. Within these two groups there are a number of separate entities that differ in their histological appearance, immunophenotype, clinical behaviour and prognosis (see Table 57).
2. Many thymic tumours are discovered incidentally during the course of thoracic surgery or imaging studies. Thymic abnormalities are actively sought in patients with myasthenia gravis. When they reach a certain size they produce local pressure symptoms relating to their location in the anterior mediastinum, e.g. stridor, cough or dyspnoea.
3. Congestive splenomegaly arises when there is persistent or chronic venous congestion. The venous congestion may be systemic (such as in right-sided cardiac failure) or localised, but traditionally the causes of congestive splenomegaly are divided into three categories:
• prehepatic causes
• hepatic causes
• post-hepatic causes.
Prehepatic causes include obstruction of the extrahepatic portal vein or splenic vein by thrombus or an inflammatory process. By far the most common hepatic cause is cirrhosis. Post-hepatic causes include right-sided cardiac failure and thrombosis of the hepatic veins (Budd–Chiari syndrome).