Chapter 89 Isolated hepatic perfusion for extensive liver cancer
Overview
Each year, isolated hepatic metastases from a variety of primary malignancies pose a significant clinical dilemma for tens of thousands of patients. For a small percentage of patients, surgical resection or ablation is effective in controlling clinically apparent disease, but for many patients with colorectal, gastrointestinal, or neuroendocrine tumors or ocular melanoma, the number of tumors and volume of affected liver render resection and/or ablation incapable of meaningful disease control (see Chapter 81A, Chapter 81B, Chapter 81C ). In more selected circumstances, metastases arising from tumors of the breast, skin, and soft tissue can be present solely as hepatic disease, with patient quality of life and survival dictated by the ability to control liver-based disease.
Based on its unique vascular anatomy (see Chapter 1B), the liver is a favorable site for delivery of regional therapy (see Chapter 86), as complete control of circulatory inflow and outflow can be readily obtained. Additionally, established tumors in liver derive the majority of blood flow from the arterial tree, and the portal vein is maintained as the primary source of nutrient flow to the hepatic parenchyma, allowing intraarterial delivery to effectively concentrate drug within tumor-bearing areas of the liver. Animal models demonstrate that nearly 100% of blood delivered to tumors arises from the arterial circulation versus 25% for normal liver (Breedis & Young, 1954; Ridge et al, 1986). The ability to obtain complete vascular isolation also permits the delivery of clinically relevant levels of hyperthermic and/or biologic agents that would otherwise be too toxic or technically impractical to deliver. For patients with multiple hepatic metastases, the likelihood of additional subclinical disease being present within the liver increases with greater tumor volume, and thus the ability to treat the entire diseased organ through regional perfusion strategies allows the targeting of micrometastatic disease within the diseased organ.
The initial report detailing the clinical use of IHP was published in 1961 by Robert Ausman from the Roswell Park Cancer Center, describing his experience with both a porcine treatment model along with five treated patients (Ausman, 1961). In this brief report, evidence of antitumor efficacy was seen in two patients after a 60-minute perfusion with melphalan. Over the ensuing 2 decades, additional small series examined the utility of prolonged hyperthermic perfusions (Skibba & Quebbeman, 1986) without drug, as well as normothermic perfusions utilizing melphalan (Hafström et al, 1990) or mitomycin-C and 5-fluorouracil (5-FU) (Aigner et al, 1988), and were described by Skibba (Medical College of Wisconsin) and multiple European groups, respectively. The absence of long-term follow-up and the presence of significant toxicity prevented widespread adoption of this approach, until interest in the field of regional therapy was reignited by Leinhard and colleagues’ 1992 publication that detailed the successful delivery of a combination of melphalan, tumor necrosis factor (TNF), and interferon-α via hyperthermic isolated limb perfusion in a group of 29 patients with advanced extremity sarcoma or melanoma. Clinical results demonstrated a 90% complete response rate for patients with in-transit melanoma along with an 80% limb salvage rate for patients with advanced sarcoma. Of equal or greater significance was the demonstration that meticulous surgical technique could result in very effective vascular isolation with a resultant decrease in out-of-field drug exposure and associated toxicity. When this increased attention to circuit integrity and leak monitoring was applied to patients undergoing IHP, a similar decrease in systemic toxicity permitted more widespread investigation of this clinical approach.
Within the United States, a significant effort in the development and refinement of vascular isolation-perfusion techniques was initiated by Drs. Douglas Fraker and Richard Alexander within the Surgery Branch of the National Cancer Institute (NCI) (Alexander et al, 1998, 1999).
Surgical Technique
Preoperative evaluation of patients thought suitable for IHP should include an assessment of the patients’ overall cardiovascular risk factors, the extent of both intrahepatic and extrahepatic tumor malignancy, and an assessment of the liver functional status (see Chapter 2). Standard preoperative cardiac clearance should include a treadmill stress test, as the induction of venovenous stress can lead to the induction of atrial fibrillation in patients so disposed. Hepatic reserve is important to assess, as the dose-limiting toxicity observed in Phase I trials was liver based, and it was seen in patients with more than 50% of hepatic replacement with tumor or a serum bilirubin level higher than 3 mg/dL. For patients with colorectal cancer and a significant chemotherapy history that includes either oxaliplatin and/or irinotecan, a biopsy assessment of the uninvolved hepatic parenchyma must be done to rule out extensive portal inflammation, hepatic congestion, and steatohepatitis (see Chapter 65). Patients who have had extensive portal of hepatic venous dissection associated with major hepatectomy should be approached with caution.
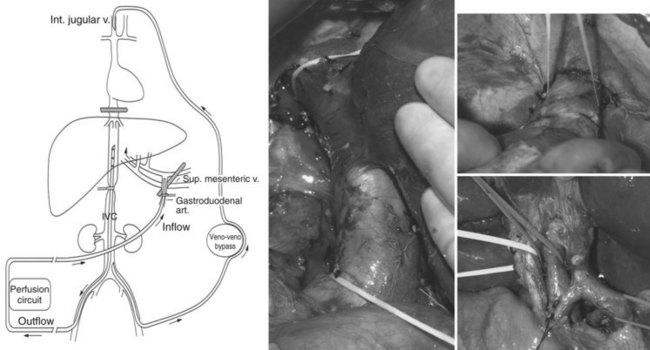
Preparation of the liver for perfusion includes completion of a cholecystectomy and full mobilization the liver (Alexander, 2005; Libutti et al, 2000). All lateral attachments are taken down so that the vena cava is fully visualized, and all retroperitoneal venous tributaries are taken down to ensure there will be no leak of chemotherapy from the isolated segment of the retrohepatic inferior vena cava (IVC). The duodenum is mobilized, and the IVC is mobilized from the renal veins to the hepatic veins. The right adrenal vein is suture ligated and divided, but both phrenic veins are preserved. The hepatic artery is identified, and the gastroduodenal artery (GDA) is mobilized from its origin for a length of 2 cm to serve as the arterial inflow catheterization site. Nodal tissue in the porta hepatis is dissected to allow clamping of the portal structures, with the hepatic artery clamp proximal to the takeoff of the GDA. Minor accessory right and left hepatic vessels may be ligated, but replaced or accessory arteries of a significant size are prepared for cannulation along with the GDA.
After completion of dissection, the patient is heparinized to an activated clotting time longer than 400 seconds. An external venovenous bypass circuit is established by placing a cannula into the left femoral vein and advancing it into the infrarenal IVC and then advancing a second cannula through the internal jugular vein into the superior vena cava (SVC). This allows maintenance of the systemic circulation by actively shunting IVC blood during treatment. Once the venous bypass has been established, the intrahepatic (IHP) circuit is constructed (see Fig. 89.1), and the GDA is ligated distally. The inflow perfusion cannula is positioned in the proximal GDA; once it has been secured, a cross clamp is placed across the entire porta hepatis, including the common hepatic artery, bile duct, and portal vein. The perfusion outflow cannula is inserted into the retrohepatic IVC via a percutaneous cannulation of the right femoral vein, and a tourniquet is placed around the cannula at the level of the suprarenal infrahepatic IVC. The suprahepatic IVC is then cross clamped, completing the vascular isolation of the liver, and perfusion is initiated; the perfusate consists of approximately 500 mL Ringer’s lactate to which 2 U packed red blood cells are added.
Once perfusion is initiated, flow through the isolated circuit is maintained between 400 and 600 mL/min. The routine use of leak monitors has been abandoned, as detectable leak is rare in the presence of a stable circuit reservoir (Barker et al, 1995). Temperature probes are placed into the anatomic right and left lobes of the liver, and the perfusate is heated to maintain hepatic hyperthermia of 40° C. Once hyperthermia is obtained, and perfusion parameters are stable, melphalan (1.5 mg/kg ideal body weight) is administered into the arterial limb of the isolated perfusion circuit over 5 minutes. The liver is perfused for 1 hour, after which time the circuit is flushed with 2000 mL saline and 500 mL colloid to flush all chemotherapy from the hepatic vasculature. The portal and suprahepatic IVC clamps are removed to allow native blood flow to be restored. The GDA is either suture ligated or a hepatic arterial infusion pump is placed. The venovenous bypass circuit is halted, and both the femoral vein and right internal jugular vein catheters are removed; anticoagulation is reversed with protamine and 2 U fresh frozen plasma.
Postoperative care focuses on the maintenance of normal coagulation profiles and standard fluid resuscitation (see Chapter 22). Heparin should be avoided, as high levels of heparin-induced antibodies are common (Masucci et al, 1999); this phenomenon is rare but can lead to devastating consequences, if heparin is administered in the early postoperative setting.
Pharmacokinetic analyses performed during early phase IHP trials have demonstrated that complete vascular isolation is routinely achieved with no detectable levels of melphalan in the systemic circulation (Lans et al, 2001). A small amount of systemic drug observed in the immediate postperfusion period has been addressed by unclamping the portal clamp for 10 seconds during the washout phase, facilitating clearing of the portal circulation. A transient significant elevation of aspartate and alanine transaminases was universally observed but was self-limited and resolved within 7 days. Biliary congestion and cholestatic jaundice is rare when proper patient selection is maintained with regard to underlying liver disease and tumor volume. Overall operative mortality rate is 4% across single-institution experiences (Alexander, 2005), and operative times and blood loss have decreased with the implementation of less invasive catheterization techniques, with operative times of less than 5 hours and mean operative blood loss less than 500 mL. An additional 500 mL of blood loss results from the removal of melphalan-contaminated hepatic circuit blood. Recent dose-escalation studies performed by Zeh and Bartlett at the University of Pittsburgh have demonstrated effective isolation and safety utilizing both 5-FU and oxaliplatin (Colville et al, 2010).
Results
Colorectal Cancer (See Chapters 81A and 86)
The greatest volume of data regarding clinical effectiveness of IHP can be found for patients with metastatic colorectal cancer treated at two institutions: the Surgery Branch of the NCI in Bethesda, MD, and the Leiden Medical Center in the Netherlands. In a series of 154 patients reported by Van Iersel and colleagues (2008), 105 patients were deemed eligible for IHP at laparotomy. A 50% response rate with a median survival of 11.4 months was reported in treated patients, with improved survival associated with the delivery of adjuvant systemic chemotherapy. The primary site of progressive disease was intrahepatic, either in isolation (68%) or associated with other systemic progression (18%).
Alexander and colleagues (2005, 2009) reported a series of 120 patients treated with melphalan-based IHP over an 11-year period ending in 2005 at the NCI in Bethesda. The majority of patients received melphalan alone (n = 69) or in combination with TNF (n = 41), with an additional 10 patients receiving TNF alone. The latter small cohort of patients did not appear to show any clinically meaningful benefit from IHP. Overall, this was a heavily pretreated patient group: 74 patients had received systemic or liver-directed therapy for their metastatic disease. A median overall survival (OS) of 17.4 months was achieved, with a 2-year survival rate of 34% and an overall response rate of 61%. Five treatment-related mortalities were reported, but three of these events were associated with the Phase I dose-escalation portion of the program. A multivariate analysis of factors associated with hepatic progression-free and OS demonstrated that a preoperative carcinoembryonic antigen (CEA) level less than 30 and the ability to successfully administer postoperative hepatic arterial embolization (HAI) therapy with fluorodeoxyuridine (FUDR) (see Chapter 86) were associated with significant gains in both outcomes. Of interest, neither response rate nor duration of survival was impacted negatively with a history of previous chemotherapy, contrary to observations made in other second-line approaches.
Recently, Zeh and colleagues (2009) published a Phase I dose-escalation trial that examined the use of oxaliplatin administered via the IHP circuit, with dosing based upon ideal body weight. Dose-limiting hepatic venoocclusive disease was noted at 60 mg/m2, establishing the maximally tolerated dose of 40 mg/m2, at which minimal hepatic toxicity was observed (Zeh et al, 2009). Although only 10 patients were included in this report, the assessment of overall response at 6 months was 66% based on Response Evaluation Criteria in Solid Tumors (RECIST). An additional Phase I trial examining dose escalation of 5-FU paired with fixed-dose oxaliplatin is ongoing (Table 89.1).
Table 89.1 Patient and Treatment Characteristics of 120 Patients with Metastatic Colorectal Cancer Undergoing Intrahepatic Perfusion
| Number of patients | 120 |
| Age in years (range) | 52 (22-74) |
| Female/male | 41 : 79 |
| Tumor Characteristics | |
| Number of metastatic lesions, median (range) | 8 (1 to 50) |
| Percentage of liver replaced by tumor, median (range) | 20% (5% to 75%) |
| Preoperative CEA Level | |
| Normal | 14 |
| High, stable | 15 |
| High, increasing | 81 |
| Unknown | 10 |
| Type of Liver Metastases | |
| Synchronous | 79 |
| Metachronous | 41 |
| Prior Chemotherapy | |
| Chemotherapy for established liver tumors (5-FU/LV and CPT-11 or HAI) | 74 |
| Chemotherapy as adjuvant (5-FU/LV, at least 4 cycles) | 20 |
| None | 26 |
| Perfusion and Operative Data, Median (Range) | |
| Total melphalan (mg) dosed at 1.5 mg/kg | 105 (69-160) |
| Tumor necrosis factor dose (mg) | 1 (0.3-2.0) |
| Perfusion flow rate (mL/min) | 800 (550-1350) |
| Perfusion pressure (mm Hg) | 145 (70-255) |
| Bypass flow rate (mL/min) | 1888 (1300-2520) |
| Central liver temperature (°C) | 40 (39-40.8) |
| Estimated blood loss (L) | 2.0 (0.5-5.0) |
CEA, carcinoembryonic antigen; FU/LV, fluorouracil/leucovorin; CPT-11, irinotecan; HAI, hepatic arterial infusion
From Alexander HR Jr, et al, 2009: Analysis of factors associated with outcome in patients undergoing isolated hepatic perfusion for unresectable liver metastases from colorectal center. Ann Surg Oncol 16:1852-1859.
Ocular Melanoma (See Chapter 81C)
Ocular melanoma has the unique ability to metastasize to the liver in the absence of clinically apparent extrahepatic disease, with liver failure being a significant source of mortality in this patient population. Of the approximately 4000 patients who have ocular melanoma develop on an annual basis in the United States, nearly 50% will develop metastatic disease; in 80%, the liver will be the sole or life-limiting site of spread. The group from the surgery branch at the NCI reported an overall response rate of 62%, including a 10% complete response in 29 patients treated with melphalan alone via the IHP circuit (Alexander et al, 2003). OS for all treated patients was 12 months, with a median progression-free survival (PFS) of 8 months. In those patients who achieved a response by RECIST criteria, the median duration of response was 10 months, with the majority of patients (66%) experiencing hepatic progression as the initial site of progressive disease. Transient, self-limited grade 3 or 4 toxicity was observed in noted in 19 patients, with no treatment mortalities; however, patient-selection criteria limited treatment to individuals with less than 50% hepatic replacement with tumor, and elevations in preoperative lactate dehydrogenase were observed as a strong predictor of poor outcome.
Several European groups reported similar experiences in cohorts of patients with metastatic ocular melanoma, including Noter and colleagues (2004) and Rizell and colleagues (2008); both supported the results of Alexander and others. Noter and colleagues at the Leiden University Medical Center treated 8 patients with a fixed dose of 200 mg melphalan for 60 minutes; they reported a 50% response rate and median progression-free and OS of 6.7 and 9.9 months, respectively, and 2-year survival was 37.5%. A larger series of 27 patients, with a response rate of 71% and a median OS of 12.6 months, was reported by Rizell. Multiple series consistently report recurrent liver disease to be the greatest threat to patient survival, even after significant treatment responses.
Hepatocellular Carcinoma (See Chapters 80, 86, and 88)
Ku and colleagues (1998) from Kobe, Japan, reported long-term IHP with 60 to 150 mg/m2 doxorubicin in 28 consecutive patients with multiple advanced HCC. In this group of stage IV patients, the main tumor diameter averaged 7.4 cm, and 17 of 28 patients had macroscopic vascular invasion to the portal vein. The overall response rate was 63% with 5 complete remissions and 12 partial responses. The OS rate was 67% and 39% at 1 and 5 years, respectively. A similar tumor response rate of 64% has been reproduced by the current updating series with 79 patients (Ku et al, 2003), which represents the largest single-institution experience with IHP. In this expanded series, with a mortality rate of 4%, the 1- and 5-year survival rates of stage IVa patients were 81% and 20%, respectively.
The Kobe group also reported results of repetitive IHP combined with reductive hepatectomy for 25 patients with multiple advanced HCCs (Ku et al, 2004) previously deemed to be unresectable. This dual-treatment strategy of surgical resection plus IHP is based on several oncologic advantages: First, a large HCC frequently accompanies feeding arteries from adjacent organs, and these collateral arteries are eliminated at the time of surgical resection. Second, reductive hepatectomy results in decreasing the vascular bed of the liver, which is theoretically beneficial to IHP, because the relative dose rate to the residual tumors is increased. Third, in HCC patients with tumor thrombi in the portal trunk, a life-threatening sequence of events, such as massive ascites, variceal hemorrhage, and eventual hepatic failure, frequently occurs. Such urgent situations could be prevented by the first-stage surgery. In a series of 35 patients with multiple advanced HCCs, the complete response rate was 46% with a partial response rate of 36%; in patients with the disease confined to the liver, the 5-year survival rate reached 51% (Ku et al, 2004).
Other Histologies
Phase II trials examining the utility of IHP for patients with advanced gastrointestinal neuroendocrine tumors (see Chapter 81B) and primary hepatobiliary tumors (see Chapter 50A, Chapter 50B, Chapter 50C, Chapter 50D ) were reported by the NCI group. Grover and colleagues (2004) described a group of 13 patients who were treated with melphalan (n = 10), melphalan plus TNF (n = 2), or TNF alone (n = 1). A single treatment-associated mortality was observed, along with a response of 50% and a median OS of 48 months. Nine patients with either HCC or cholangiocarcinoma were treated under the same conditions. Baseline tumor volume was significant, with a median percent hepatic replacement of 41%. Overall response rate (all positive response) was 66%, with median PFS and OS of 7.7 and 16.3 months, respectively (Feldman et al, 2004).
Percutaneous Hepatic Perfusion
The greatest weakness of IHP technology is the inability to repeat the procedure secondary to extensive surgical scarring around the IVC, made more acute by the observation that intrahepatic recurrence is common even after significant clinical responses. First reports of a less invasive technique of liver perfusion were published by Curley and colleagues (1993, 1994); utilizing percutaneously catheters and hepatic venous filtration, 80% of doxorubicin administered via the hepatic artery was effectively filtered from the hepatic venous circulation. Ravikumar and colleagues (1994) reported a similar series of 28 patients who underwent 58 treatments with either 5-FU or doxorubicin, but neither series established a clear utility for the technology, and further studies were not completed.
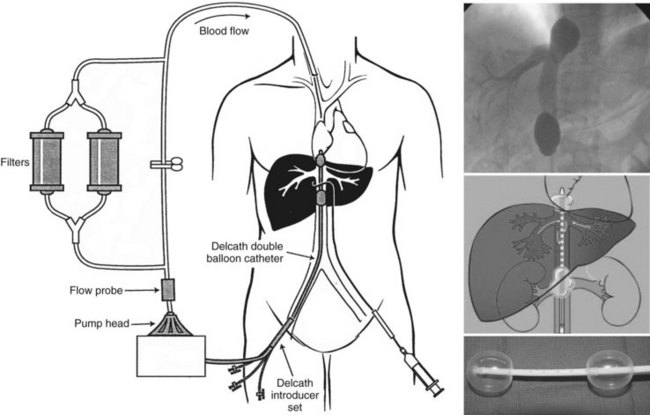
A systematic examination of a refined percutaneous hepatic perfusion (PHP) system was initiated at the Surgery Branch of the NCI in 2001 that utilized melphalan in a formal dose-escalation protocol with results published in 2005. The PHP system uses a double-balloon IVC catheter system (Delcath Systems, New York, NY) to isolate hepatic venous outflow and allow high-dose infusion of chemotherapy to the liver (Fig. 89.2). The main component of the system is a 16-Fr polyethylene double-balloon catheter with one large lumen and three accessory lumina. The two low-pressure occlusion balloons are inflated independently through separate lumina. The cephalic balloon blocks the IVC superior to the hepatic veins, and the caudal balloon obstructs the IVC inferior to the hepatic veins, allowing complete isolation of hepatic venous outflow. The span between the two occlusion balloons consists of a fenestrated segment that feeds into the large, central lumen, which exits the catheter from the proximal end. The additional lumen enters the catheter at a point inferior to the caudal balloon and exits at the distal tip; it serves as a channel for a guidewire and also allows some blood flow from the infrarenal IVC to the right atrium. During the procedure, melphalan is infused through a catheter percutaneously inserted into the hepatic artery. The melphalan perfuses the liver and exits the organ through the hepatic veins. Hepatic venous effluent is collected using the occlusion balloon catheter, and melphalan-dosed blood from the central lumen is pumped through an extracorporeal circuit that consists of a centrifugal pump and hemoperfusion drug-filtration cartridges. Melphalan flowing through the circuit is filtered with two activated-carbon filter cartridges arranged in parallel, and filtered blood is returned to systemic circulation via a venous return sheath inserted into the internal jugular vein. Treatments are administered with patients under general anesthesia (Figs. 89.3 and 89.4)
The completed Phase I study reported 28 patients who underwent 74 treatments (Pingpank et al, 2005). Twelve patients were treated in an initial feasibility cohort at 2.0 mg/kg prior to a 16-patient dose escalation to 3.5 mg/kg, after which dose-limiting bone marrow toxicity was observed in 2 of 6 treated patients. Maximum tolerated dose (MTD) was established at 3.0 mg/kg for a total of four planned treatments per patient. Examination of the venous filtration system revealed a consistent 78.5% melphalan extraction. At the MTD, grades 3 and 4 hepatic and hematologic complications, neutropenia and thrombocytopenia, were observed in 19% and 66% of treated patients, respectively. An overall response rate of 30% was reported across multiple tumor types and at all dose levels, with a 50% response rate noted in the 10 patients with metastatic ocular melanoma.
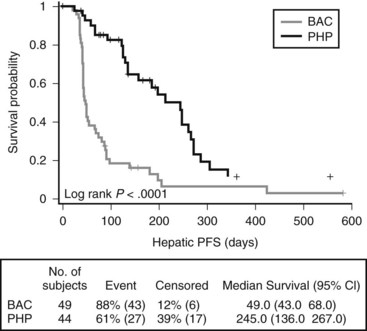
Two subsequent trials have been reported, including a Phase II cohort of 23 patients with metastatic gastrointestinal neuroendocrine tumors. The overall response rate was 79% in the 19 evaluable patients, with median hepatic PFS and OS of 39 and 40 months, respectively (Pingpank et al, 2008). A recently completed Phase III random-assignment trial that compared PHP with melphalan to standard of care for patients with hepatic metastatic melanoma has been reported. The primary end point of this trial was hepatic PFS, and 93 patients were randomized at 10 institutions, with 27 of 49 patients crossing over from the standard-of-care arm at hepatic progression. Median hepatic (245 days vs. 49 days; P < .001; hazard ratio [HR] .301) and overall (186 days vs. 46 days; P < .001; HR, 0.404) PFS were significantly improved on the experimental (PHP) arm of the study. OS was not significantly improved, in part because of the high crossover rate. Overall response rate was 34.1% (15 of 44) for PHP vs. 2.0 % (1 of 49) for best alternative care (BAC) (P < .001) (Pingpank et al, 2010).
Aigner KR, Walther H, Link KH. Isolated liver perfusion with MMC/5-FU: surgical technique, pharmacokinetics, clinical results. Contr Oncol. 1988;29:229-246.
Alexander HR. Isolation perfusion. In: DeVita, VT, Hellman, S, Rosenberg, SA. Cancer: Principles and Practice of Oncology. Philadelphia: Lippincott Williams & Wilkins; 2005:2316-2322.
Alexander HR, Bartlett DL, Libutti SK. National Cancer Institute experience with regional therapy for unresectable primary and metastatic cancer of the liver or peritoneal cavity. In: Markman, M, editor. Regional Chemotherapy: Clinical Research and Practice (Current Clinical Oncology). Totowa, NJ: Humana Press; 1999:127-150.
Alexander HR, et al. Isolated hepatic perfusion with tumor necrosis factor and melphalan for unresectable cancers confined to the liver. J Clin Oncol. 1998;16:1479-1489.
Alexander HRJr, et al. Hyperthermic isolated hepatic perfusion using melphalan for patients with ocular melanoma metastatic to liver. Clin Cancer Res. 2003;9:6343-6349.
Alexander HRJr, et al. Isolated hepatic perfusion for the treatment of patients with colorectal cancer liver metastases after irinotecan-based therapy. Ann Surg Oncol. 2005;12:138-144.
Alexander HRJr, et al. Analysis of factors associated with outcome in patients undergoing isolated hepatic perfusion for unresectable liver metastases from colorectal center. Ann Surg Oncol. 2009;16:1852-1859.
Ausman RK. Development of a technic for isolated perfusion of the liver. NY State J Med. 1961;61:3393-3397.
Barker WC, et al. Continuous intraoperative external monitoring of perfusate leak using I-131 human serum albumin during isolated perfusion of the liver and limbs. Eur J Nucl Med. 1995;22:1242-1248.
Breedis C, Young G. Blood supply of neoplasms of the liver. Am J Pathol. 1954;30:969-985.
Colville H, et al. In vitro circuit stability of 5-fluorouracil and oxaliplatin in support of hyperthermic isolated hepatic perfusion. J Extra Corpor Technol. 2010;42:75-79.
Curley SA, et al. Reduction of systemic drug exposure after hepatic arterial infusion of doxorubicin with complete hepatic venous isolation and extracorporeal chemofiltration. Surgery. 1993;114:579-585.
Curley SA, et al. Complete hepatic venous isolation and extracorporeal chemofiltration as treatment for human hepatocellular carcinoma: a phase I study. Ann Surg Oncol. 1994;1:389-399.
Feldman ED, et al. Treatment of unresectable primary hepatic malignancies using hyperthermic isolated hepatic perfusion. J Gastrointest Surg. 2004;8:200-207.
Grover AC, et al. Isolated hepatic perfusion for the treatment of patients with advanced liver metastases from pancreatic and gastrointestinal neuroendocrine neoplasms. Surgery. 2004;136:1176-1182.
Hafström L, et al. The pharmacokinetics of melphalan in regional hyperthermic liver perfusion. Reg Cancer Treat. 1990;3:23-25.
Ku Y, et al. Induction of long-term remission in advanced hepatocellular carcinoma with percutaneous isolated liver chemoperfusion. Ann Surg. 1998;227:519-526.
Ku Y, et al. Percutaneous isolated hepatic perfusion for advanced hepatocellular carcinoma established with the aid of interventional radiology technique. Geka. 2003;65:1158-1167.
Ku Y, et al. Reductive surgery plus percutaneous isolated hepatic perfusion for multiple advanced hepatocellular carcinoma. Ann Surg. 2004;239:53-60.
Lans TE, et al. Role of tumor necrosis factor (TNF) on toxicity and cytokine production following isolated hepatic perfusion (IHP). Clin Cancer Res. 2001;7:784-790.
Libutti SK, et al. Technique and results of hyperthermic isolated hepatic perfusion with tumor necrosis factor and melphalan for the treatment of unresectable hepatic malignancies. J Am Coll Surg. 2000;191:519-530.
Lienard D, et al. High-dose recombinant tumor necrosis factor alpha in combination with interferon gamma and melphalan in isolation perfusion of the limbs for melanoma and sarcoma. J Clin Oncol. 1992;10:52-60.
Masucci IP, et al. Thrombocytopenia after isolated limb or hepatic perfusions with melphalan: the risk of heparin-induced thrombocytopenia. Ann Surg Oncol. 1999;6:476-480.
Noter SL, et al. Isolated hepatic perfusion with high-dose melphalan for the treatment of uveal melanoma metastases confined to the liver. Melanoma Res. 2004;14:67-72.
Pingpank JF, et al. Phase I study of hepatic arterial melphalan infusion and hepatic venous hemofiltration using percutaneously placed catheters in patients with unresectable hepatic malignancies. J Clin Oncol. 2005;23:3465-3474.
Pingpank JF, et al. High dose intra-arterial melphalan delivered via percutaneous hepatic perfusion (PHP) for patients with unresectable hepatic metastases from primary neuroendocrine tumors. Proc AHPBA. 2008. March
Pingpank JF, et al. A phase III random assignment trial comparing percutaneous hepatic perfusion with melphalan (PHP-mel) to standard of care for patients with hepatic metastases from metastatic ocular or cutaneous melanoma. Proc ASCO. 2010. (LBA #8512)
Ravikumar TS, et al. Percutaneous hepatic vein isolation and high-dose hepatic arterial infusion chemotherapy for unresectable liver tumors. J Clin Oncol. 1994;12:2723-2736.
Ridge JA, Sigurdson ER, Daly JM. Fluorodeoxyuridine uptake by human colorectal hepatic metastases after hepatic artery infusion. Surgery. 1986;100:285-291.
Rizell M, et al. Isolated hepatic perfusion for liver metastases of malignant melanoma. Melanoma Res. 2008;18:120-126.
Skibba JL, Quebbeman EJ. Tumoricidal effects and patient survival after hyperthermic liver perfusion. Arch Surg. 1986;121:1266-1271.
Van Iersel LB, et al. Isolated hepatic melphalan perfusion of colorectal liver metastases: outcome and prognostic factors in 154 patients. Ann Oncol. 2008;19:1127-1134.
Zeh HJ3rd, et al. A phase I study of hyperthermic isolated hepatic perfusion with oxaliplatin in the treatment of unresectable liver metastases from colorectal cancer. Ann Surg Oncol. 2009;16:385-394.