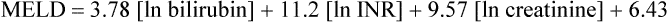Hepatic physiology and preoperative evaluation
Summary
A liver biopsy remains the gold standard for the diagnosis, grading, and staging of liver disease. Liver function tests may be useful for the differential diagnosis of liver disease, as depicted in Table 49-1.
Table 49-1
Liver Function Tests and Differential Diagnosis
| Hepatic Dysfunction | Bilirubin | Transaminase Enzymes | Alkaline Phosphatase | Causes |
| Prehepatic | Increased unconjugated fraction | Normal | Normal | Hemolysis Hematoma resorption Bilirubin overload from transfusion of red blood cells |
| Intrahepatic (hepatocellular) | Increased conjugated fraction | Markedly increased | Normal to slightly increased | Viral Drugs Sepsis Hypoxemia Cirrhosis |
| Posthepatic (cholestatic) | Increased conjugated fraction | Normal to slightly increased | Markedly increased | Stones Sepsis |
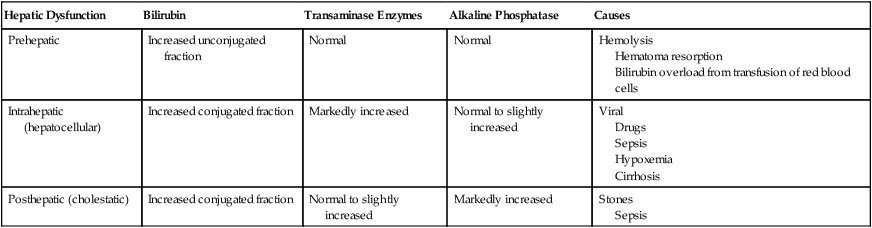
Reprinted with permission from Marschall KE. Diseases of the liver and biliary tract. In: Hines RA, Marschall KE, eds. Stoelting’s Anesthesia and Co-Existing Disease, 5th ed. Philadelphia: Churchill Livingstone; 2008:259-278.