Chapter 98A Orthotopic liver transplantation
Overview
Since the initial descriptions of orthotopic liver transplantation (OLT) in the 1960s, both the number of patients receiving transplants and the indications for the procedure have increased significantly. OLT represents the only treatment modality for many patients with a diverse spectrum of disease, with the predominant common factor being end-stage liver failure. It also has become an excellent option as curative therapy for early stage hepatocellular carcinoma (HCC). Advances in perioperative care of both donor and recipient, organ preservation methods, and surgical techniques have resulted in a 1-year overall survival of 88% for all recipients (Wolfe et al, 2010).
This chapter presents a broad overview of liver transplantation, including common criteria for recipient and donor selection (see Chapter 97A), standard operative approaches for donors and recipients (see Chapter 99), common postsurgical complications (see Chapter 100), and outcomes related to the underlying etiology of end-stage liver disease (ESLD). Specialized transplant techniques, such as split-liver and living-related donor liver transplantation, are described in Chapters 98B and 98C.
Patient Selection
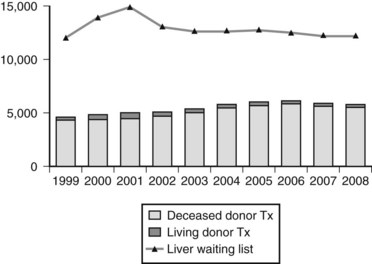
OLT represents the only curative treatment option for most patients with irreversible acute and chronic liver disease and cirrhosis, regardless of cause. Over the past 4 decades, 5-year patient survival after liver transplantation has increased from less than 50% to greater than 70% or more (Busuttil et al, 2005; Jain et al, 2000). The improvement in patient outcome has led to an expansion in the indications for transplantation and a concomitant increase in the number of patients referred to transplant centers (see Chapter 97A). The downside to this success is the persistent disparity between the number of recipients vying for a transplant and the number of donor organs available, a relatively constant supply (Fig. 98A.1).
The number of patients listed for OLT has increased sixfold since 1993, whereas the number of transplantations performed increased by only 45% during the same time. In 2008 in the United States, 15,807 patients were on the waiting list for a liver transplant, a decline from a peak of nearly 17,000 in 2002 (Wolfe et al, 2010); however, only 5817 of the patients on the waiting list received a liver allograft, and 90% of these recipients had a Model for End-Stage Liver Disease (MELD) score above 15 at transplantation (U.S. Scientific Registry of Transplant Recipients [USRTR], 2008).
Historically, the donor-to-recipient disparity leads to longer waiting times and worsening medical status with a peak waiting-list attrition rate of 187 per 1000 patient-years at risk in 1999. This rate declined by 15% and has remained steady over the last few years at 160 per 1000 patient-years at risk (Thuluvath et al, 2010). Because of the limited supply of donor organs, appropriate recipient and donor selection is paramount to improve resource use and long-term outcome.
Recipient Selection
Common indications for OLT (see Chapter 97A) include portal hypertension as manifested by variceal bleeding, ascites, encephalopathy, hyperbilirubinemia, hepatic synthetic dysfunction, and lifestyle limitations. More than 70% of liver transplantations are for noncholestatic liver disease, of which the most common etiologies are viral hepatitis (30%) and alcoholic cirrhosis (16%) (USRTR, 2008). Biliary atresia is the most common indication for liver transplantation in patients younger than 18 years of age (Busuttil et al, 2005; Goss et al, 1998). Nonalcholic steatohepatitis (NASH) is becoming an increasingly common cause of liver cirrhosis, as the prevalence of nonalcoholic fatty liver disease (NAFLD) increases; NAFLD now affects an estimated 10% of children (Schwimmer et al, 2006).
Few true, absolute contraindications to OLT exist that uniformly portend a poor patient outcome (Box 98A.1). Advanced cardiopulmonary disease, known extrahepatic malignancy, uncontrolled systemic sepsis from a source originating outside the liver, acquired immunodeficiency syndrome (AIDS), and ongoing or recent substance abuse are absolute contraindications. Many of the relative contraindications are conditions that are expected to improve after successful OLT. Examples include severe hemodynamic instability (e.g., shock) requiring multiple pharmacologic agents to maintain perfusion and severe hypoxia uncorrected by conventional intensive care measures in the context of hepatopulmonary syndrome. Other relative contraindications to OLT are extensive mesenteric venous thrombosis, morbid obesity, psychiatric disorders uncontrolled by conventional means, absence of a suitable social support network, and extremes of age (Jain et al, 2000; Loinaz et al, 2002; Rustgi et al, 2004).
Increasingly, advances in surgical technique and medical supportive care are overcoming obstacles and comorbid conditions formerly considered absolute contraindications to transplantation, including portal vein thrombosis (PVT), human immunodeficiency virus (HIV) infection, and advanced age. Patients who receive liver transplants now have higher rates of diabetes mellitus (DM), renal insufficiency, and morbid obesity (Thuluvath et al, 2010). Although pretransplantation insulin-dependent DM is not a contraindication to transplantation, evidence suggests that better risk stratification of these patients is warranted (Thuluvath, 2005). Likewise, patients with extreme body mass indexes (<18.5 or >40) might be at higher risk for posttransplantation complications (Dick et al, 2009). Morbid obesity is also associated with an increased risk of infectious complications and posttransplantation malignancy.
Although HIV infection has historically been considered a contraindication to OLT, the advent of highly active antiretroviral therapy (HAART) has turned HIV into a chronic condition, and patients are now living long enough to suffer the morbidity and mortality of other diseases, including ESLD. Therefore some patients with HIV may be considered for liver transplantation, if their CD4 T-cell count is greater than 200 and their HIV RNA viral load is less than 50 copies/µL within 12 months prior to transplantation (Di Benedetto et al, 2008). It should be noted that hepatitis C virus (HCV) coinfection with HIV predicts worse outcomes after OLT, with faster and higher rates of HCV recurrence (Di Benedetto et al, 2008).
The etiology of liver failure may be predictive of outcome after OLT, although the correlation between preoperative risk and graft survival sometimes varies. Jain and colleagues (2000) examined outcomes after OLT in 4000 consecutive patients treated at the University of Pittsburgh. Patients with metabolic or autoimmune liver disease experienced 10-year survival rates greater than 60%, whereas the survival rates for viral hepatitis or alcohol-induced cirrhosis were 40% to 50%, and patients transplanted for advanced hepatic malignancy had only a 22% survival at 10 years. Results for all patients were better in the most recent era. Earlier contradictory reports notwithstanding, recipient age probably does not influence transplantation results significantly (Gayowski et al, 1998; Ploeg et al, 1993; Totsuka et al, 2004). Thorough screening for medical comorbidities commonly found in older populations—such as lifestyle-limiting cardiopulmonary disease, systemic vascular disease, and chronic renal insufficiency—is crucial to successful OLT in patients older than 60 years.
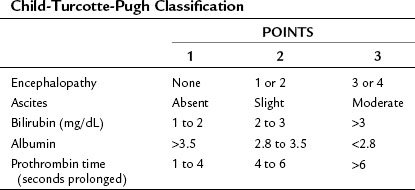
The recipient selection process has undergone extensive revisions to ensure equitable allocation of a scarce resource (cadaveric donor livers) while attempting to avoid futile transplantation. Before February 2002, recipients awaiting OLT were prioritized based on the Child-Turcotte-Pugh (CTP) scoring system (Table 98A.1), time on the waiting list, and patient location (e.g., intensive care unit). Waiting lists grew under this system, and it became increasingly clear that these parameters were not good measures of disease severity.
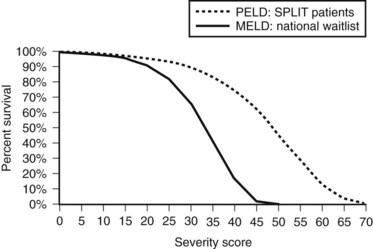
In 2000, the Department of Health and Human Services issued a guideline stating that the allocation of livers for transplantation should be based primarily on medical urgency (Freeman et al, 2002). The United Network of Organ Sharing (UNOS), which administers the Organ Procurement and Transplantation Network, commissioned several subcommittees to examine new methods for allocation. The result was the universal adoption of the MELD criteria (Table 98A.2) as a measure of the potential recipient’s necessity for OLT. Concurrent revision of the pediatric liver allocation process produced the Pediatric End-Stage Liver Disease (PELD) model, which is discussed further in Chapter 98C. Per a policy change in 2005, PELD is applied to potential transplant recipients younger than 12 years. Adolescents aged 12 to 17 years are stratified using the MELD system.
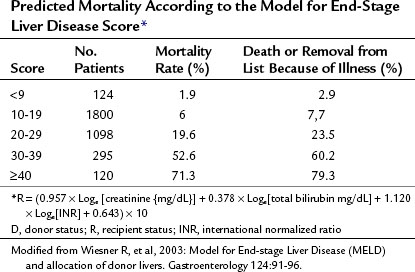
In contrast to its predecessor, the CTP score, MELD has been validated extensively as a predictor for 3-month mortality from chronic liver disease (Malinchoc et al, 2000; Wiesner et al, 2003a). It incorporates serum creatinine, bilirubin, and international normalized ratio (INR) as markers for risk of death; relies on objective and readily available blood tests; and practically eliminates time on the waiting list from consideration. Using MELD criteria, rates of death while awaiting OLT and removal from the waiting list for being too sick have decreased (Thuluvath et al, 2010). Median time to transplant in 2007 was 361 days.
As shown in Figure 98A.2, the use of MELD criteria for predicting mortality from ESLD results in an appropriate correlation between severity score and actuarial survival at 3 months. Survival benefit analysis of liver transplant recipients stratified by MELD score demonstrates that the risks involved in transplantation are equivalent or less than the risks associated with remaining a transplant candidate on the waiting list with a MELD score over 15 (Merion et al, 2005). The MELD-based allocation process is dynamic and amenable to subtle changes to reflect an evolving understanding of the diverse pathology and physiology seen in patients treated with OLT.
Special considerations for certain subsets of patients with liver disease are ongoing. Early in the initial evaluation of the MELD criteria, it was recognized that patients with HCC and early cirrhosis should be prioritized lower on the waiting list, when they could potentially benefit most from transplantation (e.g., HCC at an early tumor stage; see Chapter 97D). Conceivably, patients with HCC but low MELD scores could develop progressive disease, which would eliminate OLT as a treatment option. Since the most recent revisions to MELD, patients with stage II HCC currently are allocated 22 MELD priority points. Criteria for selecting HCC patients for transplantation are discussed further in this chapter, including those with more advanced stage disease. Guidelines also exist for awarding priority points for other conditions, such as hepatopulmonary syndrome, portopulmonary hypertension, hepatic artery thrombosis after liver transplant, pediatric hepatoblastoma, inborn errors of metabolism, familial amyloidosis, and primary oxaluria; however, the process is highly variable by region (USRTR, 2008).
Since 2002, the number of patients on the waiting list with exception points for any condition has more than doubled, with close to 900 on the list in 2008 (Thuluvath et al, 2010). Increasingly, stage I and II hilar cholangiocarcinoma is becoming an acceptable indication for OLT, and uniform criteria for obtaining MELD exception points have been proposed (Gores et al, 2006; see Chapter 97E). Eligible patients must be treated according to a specific protocol that requires neoadjuvant chemoradiation, followed by a staging laparotomy to confirm absence of N1 disease (Rea et al, 2005).
Donor Selection
Increased demand for donor organs, as depicted in Figure 98A.1, has resulted in the concomitant increase in the use of less than ideal donors. The theoretic ideal donor is an otherwise healthy, hemodynamically stable young person who suffers an irreversible cerebral insult that results in brain death; this situation is rarely realized. Although marginal donors are being used more frequently, absolute contraindications remain, including known extracranial malignancy (except for basal or squamous cell cancer of the skin), overwhelming sepsis, hepatic cirrhosis, hepatic macrosteatosis greater than 60%, and HIV infection. Generally, hepatitis B virus (HBV) surface antigen and core antibody positivity in the donor also constitute absolute contraindications (Wachs et al, 1995), although many centers consider transplantation of core antibody-positive livers for HBV-positive recipients, if the graft seems otherwise suitable, and even into non-HBV recipients in selected circumstances. Advanced donor age may adversely affect graft survival, and in practice, donors older than 80 years usually are excluded from consideration.
Data that demonstrate differential effects on specific recipient groups, such as HCV-positive recipients, remain inconclusive (Doyle et al, 2008; Lake et al, 2005; Russo et al, 2004). Although liver allografts with 30% to 60% steatosis are utilized in select groups of recipients, it should be recognized that greater than 60% steatosis significantly increases the risk of primary nonfunction (Urena et al, 1999). A donor risk index (DRI) has been developed to help quantify and stratify allografts by predicted failure risk (Feng et al, 2006).
As a result of the shortage of cadaveric donors, the use of living donors has increased in the United States, from 1996 with a peak in 2001; it has since declined steadily, and LDLTs now represent only 4% of liver transplantations performed (see Chapter 98B). A shift in demographics has also been seen in recipients of LDLT: patients age 50 to 64 years represent the largest group (USRTR, 2008). LDLT is appealing, because it allows elective transplantation after optimizing the health of the recipient, reduces cold ischemic times, and potentially shortens waiting times. The disadvantages include morbidity and rare mortality in the donor population (Brown et al, 2003). The most common practice in adults is transplantation of the right hepatic lobe. Despite initial data indicating worse outcomes in LDLT, a trend has been seen toward improved graft survival and overall survival in the United States in the last 10 years (Thuluvath et al, 2010). Although LDLT from an adult to a child has become well accepted and standardized, adult-adult LDLT continues to evolve, and the place of this approach as part of the overall picture of liver transplant options is not fully defined.
DCD livers are increasingly being used and now represent 5% of all liver-only transplants (USRTR, 2008). Compared with historic results with brain-dead donors, outcomes using DCD allografts are less optimal, with 1- and 3-year graft survivals of 70% and 60%, 10-year graft survival of 44% for all patients, and a primary nonfunction rate of almost 12% (Abt et al, 2004; Bernat et al, 2006; D’Alessandro et al, 2000; de Vera et al, 2009; Mateo et al, 2006; Thuluvath et al, 2010). The graft survival rate improves when donor warm ischemia time is less than 30 minutes and cold ischemia time is less than 10 hours (Mateo et al, 2006); however, DCD grafts are associated with an increased risk of nonanastomotic intrahepatic biliary strictures (ischemic cholangiopathy) and posttransplantation hepatic artery stenosis (Chan et al, 2008; Pine et al, 2009). Given the risks inherent in DCD allografts, they should be used cautiously in select groups of recipients.
Operative Techniques
The first published description of human liver transplantation was by Starzl and colleagues in 1963 at the University of Colorado. In this seminal paper, the dismal outcomes of three OLT recipients were described, including one intraoperative death from uncorrectable coagulopathy and two survivors of 7 and 22 days. In addition to the pioneering conceptual framework and implementation of liver transplantation, several advanced techniques are presented in this reference, including the use of grafts from non–heart-beating donors, venovenous bypass in the recipients, choledochocholedochostomy, and coagulation monitoring using thromboelastography (TEG). Many of these concepts remain or have reentered the realm of liver transplantation more than 40 years after their initial description. Based largely on the initial body of work by Starzl and colleagues, this section describes the surgical procedures commonly used at Washington University in St. Louis during OLT (see Chapter 99).
Donor Hepatectomy
Management of a cadaveric organ donor begins preoperatively, immediately after identification of a candidate and after evaluation by trained transplant coordinators. After brain death, severe physiologic derangements can occur, and physiologic instability increases in proportion to the length of time between declaration of death and organ procurement (Nygaard et al, 1990). The progression from brain death to somatic death results in the loss of 10% to 20% of potential donors (Wood et al, 2004).
Complications that commonly occur in a brain-dead donor include hypotension, the requirement for multiple transfusions, disseminated intravascular coagulopathy (DIC), diabetes insipidus (DI), pulmonary edema and hypoxia, acidosis, and arrhythmias and cardiac arrest. Intravascular volume repletion to normovolemia is the cornerstone of management; however, vasopressors or inotropic agents often are necessary to achieve an adequate perfusion pressure. The use of low-dose arginine vasopressin allows a reduction in the dosing of α-adrenergic agents, which may impair end-organ perfusion (Pennefather et al, 1995). Directed therapy using a pulmonary artery catheter can improve outcome in patients with brain death–induced or traumatic cardiac dysfunction (Wheeldon et al, 1995). A thorough review of the medical management of potential organ donors can be found elsewhere (Wood et al, 2004).
Surgical techniques for procuring abdominal organs from brain-dead, heart-beating donors have been described previously (Farmer et al, 2000; Merkel et al, 1972; Starzl et al, 1984, 1987). A midline incision from the suprasternal notch to the pubis is performed, followed by sternotomy and entry into the peritoneum. The abdomen is inspected for any evidence of malignancy or gross gastrointestinal ischemia, which would preclude transplantation. Procurement proceeds in several phases: warm dissection and cannulation, exsanguination with cold perfusion and organ removal, and back-table dissection and organ preparation.
Procurement in a DCD donor requires a slight modification (Bernat et al, 2006; D’Alessandro et al, 2000). In the controlled setting, the donor is brought to the operating room, and support is withdrawn. Heparin is administered to reduce risk of thrombus formation in the graft (Bernat et al, 2006). Apnea and cessation of circulation ensue after a variable amount of time, at which point of death is declared. It is important to note that 10% of potential donors do not die within 2 hours of withdrawal of support; these patients are not candidates for subsequent organ donation and are transferred back to the intensive care unit (ICU) and are allowed to die (Cooper et al, 2004). Circulation is unlikely to resume after 2 minutes of complete cessation; a minimum waiting period of 2 minutes is required, and a 5-minute interval between asystole; pronunciation of death prior to further intervention is strongly encouraged (Bernat et al, 2006).
The goal now becomes rapid reperfusion of the organs for procurement with cold preservation solution; this usually is accomplished via a quick midline laparotomy and cannulation of the aorta. Alternatively, some centers use cannulae placed before death in the femoral artery and vein. Less than 30 minutes of donor warm ischemia time is generally considered acceptable (Bernat et al, 2006). Hepatectomy is performed as in a standard brain-dead donor, commonly followed by a back-table flush. Cadaveric split-liver and living-related liver transplantation techniques are addressed in Chapters 98B and 98C.
Recipient Hepatectomy
Next, the portal vein is skeletonized proximally to just above the confluence of the splenic and superior mesenteric vein. At this point, further dissection is influenced by the use of temporary portocaval shunting or venovenous bypass; both techniques allow decompression of the splanchnic circulation, which reduces bowel edema during the anhepatic phase. When venovenous bypass is used, the portal vein is cannulated, and bypass is instituted as previously described (Shaw et al, 1984). A potential disadvantage of venovenous bypass is the added complexity and potential complications associated with the bypass process (e.g., thrombosis of the bypass circuit). We prefer portocaval shunting with an end-to-side anastomosis between the divided portal vein and infrahepatic IVC. The shunt is kept in place until the suprahepatic caval anastomosis is completed.
Temporary portocaval shunting in conjunction with the piggyback technique may improve intraoperative hemodynamic stability and renal function and may reduce the transfusion requirement (Arzu et al, 2008; Davila et al, 2008; Figueras et al, 2001). If neither venovenous bypass nor temporary portocaval shunting is used, the portal vein simply can be clamped proximally, ligated in the hilum, and divided; however, this method may be associated with a severe reduction in venous return, up to a 50% decrease in arterial blood pressure, mesenteric venous hypertension and associated organ failure, and potential increased hemodynamic instability resulting in intraoperative mortality (Hoffmann et al, 2009).
With the portal vein bypassed or clamped, exposure for the infrahepatic dissection and vascular control of the IVC is easily obtained. The piggyback technique leaves the recipient retrohepatic IVC intact and requires ligation and division of all retrohepatic caval branches (Tzakis et al, 1989). The advantage of this approach is that native caval flow is maintained, and venovenous bypass is not required; the disadvantage is that division of retrohepatic caval branches can be tedious and time consuming. The donor suprahepatic IVC is anastomosed to the confluence of the right, middle, and left hepatic veins, which are joined in a common cuff. Vascular control is achieved by clamping the hepatic veins at their point of entry into the vena cava.
Alternatively, if a bicaval technique is used, the recipient’s native retrohepatic IVC is removed with the native liver. The retrohepatic IVC is mobilized out of the retroperitoneum from the left side. The right triangular ligament is taken down, and the retrohepatic IVC is dissected from the right side. The adrenal vein is ligated in the traditional bicaval approach; this dissection frees up the retrohepatic IVC above the hepatic veins to allow application of infrahepatic and suprahepatic IVC clamps. The recipient liver is sharply excised with care taken to leave cuffs of IVC above and below the liver. This technique allows for true orthotopic placement of the donor graft. Retrospective analysis of the bicaval and piggyback techniques suggest that safety and outcomes are comparable (Nishida et al, 2006).
Recipient Implantation
OLT requires three or four vascular anastomoses in the following order: 1) suprahepatic IVC; 2) infrahepatic IVC, if a bicaval technique is used; 3) portal vein; and 4) hepatic artery. Alternatively, the use of the piggyback technique allows just a single caval (end-to-side) anastomosis, with simple ligation of the donor infrahepatic IVC; this results in a reduction in the duration of the anhepatic phase (Hosein Shokouh-Amiri et al, 2000). When the liver is fully reperfused, reconstruction of the biliary tract begins.
Adequate cuffs of suprahepatic and infrahepatic IVC are essential for reconstruction (Starzl et al, 1979). Anastomoses of these cuffs require reconstruction of the posterior walls from within the lumen using a running 3-0 polypropylene suture. The anterior layer is sutured externally using either an interrupted or a continuous technique. Another advantage of the piggyback technique is the use of a side-biting vascular clamp on the IVC at the level of ostia of the hepatic veins. Although this clamp may impair venous return to the heart to some degree during a clamp time of 15 to 30 minutes, it generally produces greater hemodynamic stability during the anhepatic phase than does complete caval occlusion, which requires a bicaval procedure (Moreno-Gonzalez et al, 2003). After the hepatic donor vena cava has been anastomosed to the recipient IVC, the side-biting clamp can be moved to the graft side of the anastomosis, restoring complete venous return, while the remaining vascular connections are performed.
The final vascular anastomosis is arterial. The key principle of hepatic arterialization is to ensure pulsatile inflow through a large caliber vessel over a short length (Farmer et al, 2000). The technique involves the use of a fine (7-0) monofilament suture in a running or interrupted fashion. The vessel ends are frequently spatulated and are sewn from the outside and rotated to achieve the most precise anastomosis. The presence of aberrant hepatic arterial anatomy is encountered in 10% to 30% of grafts, and preservation of these vessels is essential for successful engraftment. Reconstruction of the aberrant vessels to obtain a single inflow vessel is imperative and takes place during the back table preparation of the graft as described earlier.
Recipient inflow is obtained from a branch off the celiac trunk, usually the proper or common hepatic artery. When adequate arterial inflow cannot be obtained by this artery, the use of an arterial conduit is recommended. Inflow originating from the infrarenal and supraceliac aorta has been described; both methods provide excellent inflow, and the choice is based on technical considerations and surgeon preference. The best choice of conduit is usually the donor iliac vessels. When donor vessels are unavailable or inadequate, prosthetic conduits can be used, typically polytetrafluoroethylene (PTFE). In the perioperative period, aortic conduits are associated with increased operating time, greater transfusion requirements, and respiratory and renal failure (Nikitin et al, 2008). In addition, several cases of internal hernias with small bowel volvulus around the intraperitoneal conduit have been reported (Nishida et al, 2002).
Prior to anastomosis, both the donor and recipient bile ducts should be trimmed sharply to remove devitalized tissues, and brisk bleeding should be evident from the cut ends. The recipient bile duct should be cleared of sludge and stones. Accommodation for size mismatch with ductoplasty of the larger duct or spatulation of both ducts has been described (Buczkowski et al, 2007; Nissen & Klein, 2009). The choledochocholedochostomy and choledochojejunostomy anastomoses are accomplished with the use of fine absorbable monofilament suture in a single-layer closure.
Whether to stent the biliary anastomosis during OLT is subject to debate (Barkun et al, 2003; Bawa et al, 1998; Johnson et al, 2000). Proponents believe that decompression of the biliary tree reduces the rate of clinically significant leaks, whereas others point to data that suggest a higher rate of biliary stricture from internal stents. At our institution, biliary stenting is performed selectively based on individual case circumstances and surgeon preferences.
Complications
Primary Nonfunction
Primary nonfunction is defined as early graft failure after OLT in the absence of identified technical complications. Clinical presentation varies, but patients typically are seen with alterations in mental status, diminished bile production, coagulopathy, markedly elevated transaminases, and metabolic acidosis. Multiorgan failure ensues, with oliguria and hypoxia occurring frequently. The reported rate of primary nonfunction varies between 1% to 7% of all OLTs (Jain et al, 2000; Johnson et al, 2007; Kamath et al, 1991; Kemmer et al, 2007; Taner et al, 2008; Totsuka et al, 2004). Retransplantation is the treatment of choice and typically is required within 72 hours. Some authors also report the distinctly separate category of initial poor function, in which allografts might have a chance for functional recovery; however, no consensus for a clear definition exists.
The etiology of primary nonfunction is unclear but is likely multifactorial. Factors commonly reported to be associated with nonfunction include prolonged cold and warm ischemic times, donation after cardiac death, intraoperative systemic hypotension (mean arterial pressure <40 mm Hg), severity of donor steatosis, advanced donor age, and recipient factors such as portal vein thrombosis (PVT), renal failure, dependence on life support, and status 1 listing (de Vera et al, 2009; Fernandez-Merino et al, 2003; Johnson et al, 2007; Marsman et al, 1996; Nair et al, 2002; Ploeg et al, 1993; Reich et al, 2003; Sharma et al, 2010; Strasberg et al, 1994). Although it has been proven that severe macrosteatosis (>60%) increases risk of graft failure, the influence of moderate steatosis (30% to 60%) remains unclear (Yoo et al, 2003a), and such allografts can be used with careful selection (Doyle et al, 2010).
Donor liver biopsy to quantify graft steatosis should be performed before implantation of any graft that appears marginal (D’Alessandro et al, 1991). As would be expected, the likelihood of primary nonfunction is increased in the presence of multiple risk factors (e.g., steatotic graft with extended total ischemic time) and in allografts with several marginal donor criteria (Pokorny et al, 2005; Salizzoni et al, 2003).
Hepatic Artery Thrombosis
Hepatic artery thrombosis (HAT) can be broadly divided into early (acute) and late (delayed) clinical presentation, with different etiologies, patient manifestations, and treatments. Although there is no formal consensus, early HAT is typically defined as occurring within the first 1 to 2 months after OLT and has a mean incidence of 2.9% in adult OLT patients, with a significantly higher incidence of 8% to 10% in the pediatric population (Bekker et al, 2009; Farmer et al, 2007). Early HAT carries a high risk of graft loss and death—53% and 33%, respectively (Bekker et al, 2009).
Technical, donor, and recipient factors contribute to an increased risk of HAT (Bekker et al, 2009; Del Gaudio et al, 2005; Duffy et al, 2009; Jurim et al, 1995; Soin et al, 1996; Vivarelli et al, 2004). Donor factors may include small-caliber vessels, aberrant anatomy that requires complex arterial reconstruction, use of aortic conduits, or cytomegalovirus (CMV) seropositivity donor-recipient mismatch. Variable recipient anatomy (e.g., replaced hepatic artery from superior mesenteric artery [SMA]) also influences risk of HAT.
Some reports suggest that endovascular procedures—intraarterial thrombolysis, percutaneous transluminal angioplasty, and endoluminal stenting—can be successful in hepatic arterial revascularization (Singhal et al, 2010); however, a majority of patients eventually require retransplantation (Bekker et al, 2009; Duffy et al, 2009). Because of the severity of this complication, some authors advocate the routine use of postoperative duplex US.
Late HAT is typically more indolent, frequently because a previously undiagnosed stricture allows collateral circulation to develop. Clinical presentation may include fever secondary to perihepatic abscess or biliary leak, biliary strictures, or cholangitis, although in some cases patients may be asymptomatic with no clinical consequences (Gunsar et al, 2003). Contributing factors are active tobacco abuse; coagulation abnormalities, such as factor V Leiden; cerebrovascular accident as donor cause of death; donor death at an age greater than 50 years; recipient CMV positivity; and use of donor iliac interposition graft (Del Gaudio et al, 2005; Gunsar et al, 2003; Pascual et al, 1997; Pungpapong et al, 2002; Stewart et al, 2009; Vivarelli et al, 2004).
Treatment may be attempted with endoscopic or percutaneous biliary decompression, stenting, or even systemic anticoagulation. Retransplantation is required less for late HAT than for early HAT. Because postoperative antiplatelet therapy may reduce the rate of late HAT in high-risk patients (Vivarelli et al, 2007), it is our practice to prescribe daily aspirin (81 mg) for all patients.
Hepatic Artery Stenosis
Hepatic artery stenosis (HAS) without thrombosis is also a recognized complication of liver transplantation, with an incidence of 4% to 11% (da Silva et al, 2008). DCD allografts have been implicated as a risk factor (Pine et al, 2009). Initial presentation typically includes a mild increase in aminotransferase levels with or without associated graft dysfunction or biliary complications. Doppler US demonstrates increased resistance in hepatic arterial flow and is often diagnostic, but confirmation by arteriography is the gold standard. Therapeutic options include angioplasty with or without stenting. Restenosis can occur in up to one third of patients within 1 year after stenting (Ueno et al, 2006).
Portal Vein Thrombosis
PVT occurs less frequently than HAT after OLT, with an incidence less than 2% in adult recipients and 10% in pediatric recipients (Duffy et al, 2009; Lerut et al, 1987; Millis et al, 1996), although PVT adversely affects overall survival after OLT (Duffy et al, 2009). Low portal flow, small-diameter veins (<5 mm), preexisting PVT in the recipient, donor-recipient vessel size mismatch, and the use of vascular grafts for reconstruction are known risk factors for developing PVT (Cheng et al, 2004).
PVT is typically symptomatic, and patients can present with acute hepatic failure, as with HAT, or with the sequelae of portal hypertension, such as increasing ascites, splenomegaly, and variceal hemorrhage (Duffy et al, 2009). Diagnosis is made using duplex US or contrast-enhanced CT portal venography.
Depending on the timeliness of diagnosis and the acuity of the patient, several treatment options exist. In patients with fulminant hepatic failure from PVT, reexploration and attempted portal revascularization are performed. These patients sometimes require retransplantation, particularly in the rare setting of combined PVT and HAT (total absence of hepatic inflow). The use of a portocaval shunt to augment flow through the reconstructed portal vein has been described (Bakthavatsalam et al, 2001), as has the use of transjugular intrahepatic portocaval shunt (TIPS) in conjunction with thrombolytics (Ciccarelli et al, 2001). Systemic anticoagulation may be sufficient in patients with preserved graft function (Duffy et al, 2009). Symptoms of portal hypertension but preserved graft function often can be managed medically with standard therapies for ascites in combination with variceal banding or sclerotherapy; however, the graft salvage rate with these alternative strategies is typically much less than 50% (Duffy et al, 2009).
Portal Vein Stenosis
Portal vein stenosis, in contrast to PVT, is frequently diagnosed on routine screening US in asymptomatic patients. The most common area of stenosis is the extrahepatic portal venous anastomotic site. Narrowing of the main portal vein diameter by more than 50%, presence of a poststenotic jet, or lack of visualized flow on Doppler imaging are diagnostic. Many stenoses can now be managed with percutaneous transhepatic balloon angioplasty and stenting (Woo et al, 2007).
Biliary Complications
Biliary complications are the most common type of posttransplantation complication, and they arise in 7% to 29% of liver recipients. Patients with HAT, living-donor allografts, and DCD livers are at higher risk for biliary complications (Maluf et al, 2005; Pine et al, 2009; Zajko et al, 1988). Historically, Roux-en-Y choledochojejunostomy was associated with higher complication rates than choledochocholedochostomy (O’Connor et al, 1995); however, this remains a controversial topic in transplantation techniques. All patients found to have a biliary complication should undergo US to evaluate HAT as a contributing factor.
Biliary strictures occur twice as frequently as anastomotic biliary leaks and can be classified as anastomotic or nonanastomotic (ischemic cholangiopathy) (Balsells et al, 1995; Qian et al, 2004). Most anastomotic strictures are managed endoscopically with balloon dilation and stenting. Ischemic cholangiopathy more commonly results from bile duct ischemia or an immune response, appears later and affects multiple sites, tends to be more difficult to manage, and is rarely amenable to endoscopic treatment. Nonanastomotic strictures are associated with a higher rate of graft failure requiring retransplantation (Chan et al, 2008; de Vera et al, 2009; Pine et al, 2009). A multidisciplinary approach to diagnosis and management of biliary complications is necessary and can result in higher patient and graft survival rates (Verran et al, 1997).
In an asymptomatic patient with stable liver function, biliary complications can often be managed nonoperatively. Bile collections should be drained percutaneously under US or CT guidance, and most leaks are controlled with endoscopic stent placement across the anastomosis (Shah et al, 2004). Major leaks or total disruption usually requires operative conversion to a choledochojejunostomy or hepaticojejunostomy, an option that can result in long-term biliary patency (Langer et al, 2009).
Numerous studies have evaluated the use of T-tubes and internal stents for prophylactic biliary decompression. Several have identified T-tube use as an independent risk factor for increased postoperative biliary complications (e.g., biliary-cutaneous fistulae and cholangitis), and their use has not been shown to reduce the need for posttransplantation interventional procedures (Qian et al, 2004; Scatton et al, 2001; Sotiropoulos et al, 2009). Consequently, T-tubes are much less commonly used in standard adult OLT in the United States; however, a recent prospective European trial did not find evidence of increased biliary complications with T-tubes (Weiss et al, 2009). The effectiveness of internal stenting in reducing biliary complications also remains inconclusive (Welling et al, 2008).
Infection
Infection is the most common cause of death after OLT at all time points (Jain et al, 2000), directly causing 28% of all deaths in liver transplant patients. An autopsy series of OLT patients showed an even higher infection-related mortality rate of 64% (Torbenson et al, 1998). Two thirds of OLT recipients experience at least one serious infectious episode (Winston et al, 1995), despite ever-improving prophylactic regimens. Intraabdominal infections occur in 40% of recipients with a higher incidence in patients with other noninfectious surgical complications (Reid et al, 2009).
Liver transplant patients are immunocompromised as a result of antirejection medications, they are also malnourished, chronically ill, have received multiple blood-product transfusions, and have undergone lengthy and complex surgical procedures—all of which are risk factors for infectious complications. Additionally, the risk of posttransplantation infection is further increased by the prevalence of morbid obesity in the recipient population (Dick et al, 2009).
Diagnosis of infection can be complicated by a relative absence of symptoms, as infection frequently manifests only as a leukocytosis (Reid et al, 2009). As part of the pretransplantation evaluation, recipients should undergo an infectious disease workup and should receive appropriate vaccines. Appropriate antimicrobial prophylaxis can reduce the postoperative infection rate by more than half, depending on the pathogen. The current prophylactic regimen used at Washington University in St. Louis is shown in Box 98A.2.
Box 98A.2 Antimicrobial Prophylaxis for Liver Transplantation Patients at Washington University in St. Louis
Bacterial pathogens are the most common infectious agent in the early postoperative period, and the proportion of posttransplantation infection as a result of bacteremia has increased significantly (Singh et al, 2004). DM and serum albumin less than 3 mg/dL are independent risk factors for bacteremia. Although gram-negative bacilli remain frequent causes of postoperative infection after OLT, gram-positive cocci, such as methicillin-resistant Staphylcoccus aureus (MRSA) and vancomycin-resistant enterococci (VRE), have become increasingly prevalent at many transplant centers (Papanicolaou et al, 1996; Reid et al, 2009; Singh et al, 2004). Colonization with MRSA and VRE before transplantation increases the risk of developing infection after OLT with the same organism and, in the case of VRE colonization, is associated with an increased mortality risk (Russell et al, 2008).
Typically presenting in the first 2 to 3 weeks after transplantation, 25% of OLT patients are reported to develop MRSA bacteremia, with colonization of indwelling vascular catheters accounting for about half of the cases (Singh et al, 2000a). Vascular catheters account for one quarter of bacteremia for all pathogens combined. Other sources of post-OLT bacteremia include—in decreasing order of frequency—pneumonia, biliary infections, abdominal sources, and surgical wound infections (Singh et al, 2000b, 2004). The emergence of drug-resistant organisms resulting from overuse of common antibiotics has made the treatment of infected liver transplant recipients increasingly difficult.
CMV is the most common viral pathogen encountered by OLT recipients, although incidence of CMV infection has declined as a result of improved prophylaxis (Singh et al, 2004). Intraoperative hypothermia is a known risk factor for developing CMV infection regardless of prophylaxis (Paterson et al, 1999). Without appropriate prophylaxis, the overall incidence in this population is commonly 50% to 60%, with clinically apparent infections usually occurring 3 to 12 weeks after transplant (Farmer et al, 2000).
The routine use of ganciclovir (Cytovene) has been shown to dramatically reduce CMV infection after OLT in several randomized, controlled trials (Gane et al, 1997; Winston, 1995; Winston & Busuttil, 2003, 2004). The standard regimen includes an induction with intravenous ganciclovir followed by prophylaxis with oral ganciclovir for 3 to 6 months. Some studies suggest that valganciclovir, which can be administered with once-daily dosing because of improved bioavailability, might be as efficacious and safe as ganciclovir in the prevention of CMV infection in low-risk, but not high-risk, liver recipients (Jain et al, 2005; Park et al, 2006; Shiley et al, 2009); prolonged treatment (≥100 days) can be costly and might be unnecessary in low-risk patients (e.g., seronegative donor and recipient). Ganciclovir-resistant CMV has been observed in 20% of solid-organ transplant recipients treated with ganciclovir, with presentation typically observed late in the first year (Limaye et al, 2000). Our current strategy is to reserve prolonged treatment with ganciclovir for seronegative patients receiving a seropositive graft or in patients with evidence of active CMV infection.
Invasive fungal infections usually are caused by Candida or Aspergillus species, Cryptococcus, and non-Aspergillus mycelial fungi, with an incidence of 5% to 9% in OLT patients (Pappas et al, 2010; Singh, 2000). Invasive candidiasis and aspergillosis tend to occur relatively early in the postoperative period, whereas Cryptococcus infection becomes apparent several months to years after OLT (Pappas et al, 2010). Traditionally, Candida albicans has been the most common fungal pathogen.
Risk factors for developing fungemia include use of broad-spectrum antibiotics or immunosuppressant medications, retransplantation or other reoperative intervention, CMV infection, renal failure, and overall severity of medical illness (Cruciani et al, 2006). Routine prophylaxis with antifungal agents—such as fluconazole, itraconazole, and amphotericin B—significantly reduces fungal infection and infection-related mortality after liver transplantation; however, it does not improve overall mortality (Cruciani et al, 2006). Historically, fluconazole was almost universally included in antimicrobial prophylaxis, but this subsequently led to an increase in reported Aspergillus infections, because Aspergillus is not susceptible to fluconazole.
Currently, a majority of transplant centers provide antifungal prophylaxis only to high-risk patients, including those requiring retransplantation or reexploration, dialysis, mechanical ventilation, or a prolonged ICU stay and those subject to and graft failure or colonization with Candida (Singh et al, 2008). Although prophylaxis against Aspergillus is not supported by the literature (Braun et al, 1998; Singh 2000), voriconazole, amphotericin B, and caspofungin may be used alone or in combination for treatment of invasive aspergillosis (Singh et al, 2008), which is associated with a 40% 1 year mortality rate after infection (Pappas et al, 2010).
Infection with the protozoan Pneumocystis carinii occurs in 3% to 11% of liver transplant patients in the absence of prophylaxis (Singh, 2000). Because T-cell immunity is the primary defense against this organism, prolonged use of corticosteroids or muromonab-CD3 monoclonal antibodies and active CMV infection increase the risk of infection. Trimethoprim-sulfamethoxazole (single strength, once daily) offers highly effective prophylaxis at a low cost and with minimal side effects. Because the risk of P. carinii infection is eightfold higher in the first year after OLT than in subsequent years, prophylaxis generally is continued only during these initial 12 months (Gordon et al, 1999).
Rejection
Rejection can sometimes be a major hurdle to long-term survival after OLT. Previously reported to occur in 40% to 70% of OLT patients (Klintmalm, 1991), the development of novel immunosuppressive regimens has reduced the lifetime rejection rate to less than 20% (McAlister et al, 2001). It is becoming increasingly clear that certain subpopulations of patients receiving OLT can be weaned completely from all immunosuppressive agents without experiencing rejection (Devlin et al, 1998; Mazariegos et al, 1997). The University of Pittsburgh experience shows that immunosuppression can be withdrawn successfully in almost one third of patients, although it is unknown exactly what traits allow such weaning.
Hyperacute rejection is rarely seen today. It typically occurs in the setting of ABO incompatibility, with a reported graft failure rate of 46% observed in 51 patients transplanted across ABO type (Demetris et al, 1988). Hyperacute rejection is mediated by preformed antibodies in the recipient and is directed against the graft endothelium. These antibodies produce activation of the innate immune system via the complement cascade, ultimately leading to rapid graft destruction. Retransplantation is the only treatment option.
Although acute liver rejection usually occurs within the first 4 weeks after transplantation, it can occur later. Patients with acute liver rejection are seen with signs of fever, malaise, right upper quadrant abdominal pain, and elevated liver transaminases. Elevation in liver enzymes is often detected before physical symptoms or signs occur. Percutaneous liver biopsy reveals portal inflammation with predominantly mononuclear cells, bile duct inflammation and injury, centrilobular necrosis, and lobular inflammation. Differentiation from other pathologic processes can be difficult. In contrast to other solid-organ transplants, such as kidney and heart, the occurrence of an acute rejection episode does not seem to reduce the overall graft survival, if treatment is initiated promptly (Wiesner et al, 1998).
Today, only about 2% of patients experience chronic rejection after liver transplantation, although the incidence is higher in patients with autoimmune disease (Wiesner et al, 2003). The etiology of chronic rejection is multifactorial, characterized by progressive loss of bile ducts, obliteration of medium-sized and large hepatic arterioles, and cellular portal infiltration (Farmer et al, 2000). Standardized histopathologic evaluation of acute and chronic rejection using the Banff schema (Tables 98A.3 and 98A.4) allows objective decision making and facilitates comparisons of natural histories among patients (Banff Consensus, 1997; Demetris et al, 2000; Racusen et al, 2003).
Table 98A.3 Rejection Activity Index from the Banff Schema for Acute Hepatic Rejection
| Category | Criteria | Score |
|---|---|---|
| Portal inflammation | Mostly lymphatic inflammation involving a minority of the triads | 1 |
| Expansion of most triads by a mixed infiltrate containing lymphocytes, neutrophils, and eosinophils | 2 | |
| Marked expansion of most or all triads by a mixed infiltrate containing numerous blasts, with spillover into periportal parenchyma | 3 | |
| Bile duct damage | Minority of ducts infiltrated by inflammatory cells, with only mild reactive changes in epithelial cells | 1 |
| Most or all ducts infiltrated by inflammatory cells, with occasional degenerative duct changes, such as nuclear pleomorphism, disorder polarity, and vacuolization | 2 | |
| As above, with most or all ducts showing degenerative changes | 3 | |
| Venous endothelial inflammation | Subendothelial lymphocytic infiltration of some portal or hepatic venules | 1 |
| Subendothelial infiltration involving most or all portal or hepatic venules | 2 | |
| As above, with perivenular inflammation extending into surrounding parenchyma and associated hepatocyte necrosis | 3 |
This index has a range from 0 to 9, classified as follows: 0 to 3, minimal acute rejection; 4 to 6, mild acute rejection; 7 to 9, moderate to severe acute rejection.
Modified from the Banff Consensus, 1997: schema for grading liver allograft rejection: an international consensus document. Hepatology 25:658-663.
Table 98A.4 Banff Schema for Chronic Hepatic Rejection
| Structure | Early Chronic Rejection | Late Chronic Rejection |
|---|---|---|
| Small bile ducts (<60 µm) | Degenerative changes involving most ducts: increased nuclear-to-cytoplasmic ratio, nuclear hyperchromasia, uneven nuclear spacing, ducts partially lined with epithelium | Degenerative changes in remaining bile ducts |
| Bile duct loss in <50% of portal tracts | Bile duct loss in >50% of portal tracts | |
| Terminal hepatic venules | Intimal/luminal inflammation | Focal obliteration |
| Lytic zone 3 necrosis and inflammation | Variable inflammation | |
| Mild perivenular fibrosis | Severe (bridging) fibrosis | |
| Portal tract hepatic arterioles | Occasional loss involving <25% of portal tracts | Loss involving >25% of portal tracts |
| Large perihilar hepatic artery branches | Intimal inflammation, focal foam-cell deposition | Luminal narrowing by subintimal foam cells and fibrointimal proliferation |
| Large perihilar bile ducts | Inflammation damage, focal foam-cell deposition | Mural fibrosis |
| Other | “Transition” hepatitis with spotty necrosis of hepatocytes | Sinusoidal foam cell accumulation, marked cholestasis |
Modified from Demetris A, et al, 2000: Update of the International Banff Schema for Liver Allograft Rejection: working recommendations for the histopathologic staging and reporting of chronic rejection: an international panel. Hepatology 31:792-799.
Metabolic and Systemic Complications
With the increase in overall survival after OLT, a growing recognition has emerged of associated long-term medical sequelae and their impact on patient health. Prolonged use of corticosteroids can produce hyperlipidemia, obesity, DM, arterial hypertension, and mineral-deficient bone disease; commonly used antirejection drugs induce similar derangements. Cardiovascular disease then becomes a leading cause of death in patients who survive more than 3 years after liver transplantation, accounting for more than half of the reported mortality in some series (Asfar et al, 1996; Pruthi et al, 2001). Predictors of perioperative cardiac events include pre-OLT history of stroke, and perioperative mortality was increased with a history of coronary artery disease (CAD) and postoperative sepsis (Safadi et al, 2009).
Renal Dysfunction
Liver transplant recipients have a high risk of developing posttransplant chronic renal failure, with an incidence of 18% at 5 years (Ojo et al, 2003). The average functional decline in renal function is a 38% decrease in glomerular filtration rate, and it is correlated with time elapsed since transplantation (decline by 36 mL/min/1.73m2) (Bucuvalas et al, 2006; Karie-Guigues et al, 2009). The use of calcineurin inhibitors (CNIs) is associated with increased risk of renal failure in patients after OLT, although this decline may be partially attenuated with concomitant administration of mycophenolate mofetil and calcineurin dose reduction (Karie-Guigues et al, 2009). Other risk factors for the onset of post-OLT renal dysfunction include age and gender of the recipient, history of HCV infection, DM, and pretransplantation renal insufficiency, CAD, and primary nonfunction (Ojo et al, 2003; Pawarode et al, 2003). Mortality following OLT is four times greater in recipients who develop posttransplantation renal failure (Ojo et al, 2003).
Posttransplantation Metabolic Syndrome
Posttransplantation metabolic syndrome (PTMS) is becoming recognized as an important entity in liver transplant recipients. Prevalence in the post-OLT population is twice that of the estimated 24% prevalence in the general population (Bianchi et al, 2008; Hanouneh et al, 2008; Laryea et al, 2007; Pagadala et al, 2009). Posttransplantation obesity develops in 60% of patients, with most excess weight gain occurring in the first year (Muñoz et al, 1991). The cause of weight gain is multifactorial: corticosteroids and cyclosporine, and to a lesser extent tacrolimus, are known to correlate significantly with the incidence of obesity (Canzanello et al, 1997). Development of PTMS does not seem to be influenced by the etiology of the recipient’s ESLD (Bianchi et al, 2008); however, liver transplant recipients who develop PTMS appear to be at higher risk for vascular events (Laryea et al, 2007) and are at higher risk for development of allograft steatosis (Dumortier et al, 2010).
Diabetes Mellitus
De novo DM occurs in up to one third of previously nondiabetic liver transplant recipients (Navasa et al, 1996; Sheiner et al, 2000). Corticosteroids are known to induce insulin resistance, and the occurrence of graft rejection necessitating increased steroid dosing is a risk factor for developing diabetes. Tacrolimus and other immunosuppressants also increase insulin resistance. Diabetes in most patients is transient and generally resolves within the first year, as immunosuppressive medications are tapered.
Infection with HCV is another known risk factor for the development of posttransplantation diabetes, and this risk is correlated with increased HCV viral load (Delgado-Borrego et al, 2008). Although the mechanisms underlying the relationship between HCV and diabetes are not fully understood, altered insulin sensitivity is likely to play a role. Pretransplantation glucose intolerance, as determined by formal glucose challenge testing, is present in 53% of candidates awaiting liver transplantation (Blanco et al, 2001). Although some degree of glucose intolerance may improve with OLT, insulin-dependent diabetes almost never resolves after transplantation (Shields et al, 1999; Stegall et al, 1995). Additionally, insulin-dependent DM is a known risk factor for lower survival following transplantation, and CAD is an independent predictor of worse outcomes (Yoo & Thuluvath, 2002).
Neurologic Complications
Neurologic complications occur with greater frequency in recipients of liver allografts than in other solid-organ recipients (Senzolo et al, 2009). The aggregate prevalence of neurologic sequelae is 25%, although such have occurred in more than 60% of patients in some series (Amodio et al, 2007; Bronster et al, 2000; Emiroglu et al, 2006; Ghaus et al, 2001; Lewis & Howdle, 2003; Saner et al, 2006).
Posttransplantation encephalopathy is the most common neurologic complication, followed by seizures (Bronster et al, 2000; Lewis & Howdle, 2003; Saner et al, 2006; Senzolo et al, 2009). Encephalopathy can be caused by anoxia, sepsis, medications (especially CNIs), primary graft nonfunction, renal failure, rejection, and central pontine myelinolysis (CPM) (Erol et al, 2007). Infection, stroke, and CPM are also common causes of seizures (Senzolo et al, 2009). Other neurologic complications include posterior leukoencephalopathy, cerebellar syndrome, focal deficits, headache, tremor, sleep disorders, and peripheral neuropathy. Risk factors for neurologic complications include an operative time longer than 10 hours, high CTP score, and a history of hepatic encephalopathy (Dhar et al, 2008). Older age and higher MELD scores pretransplantation are associated with increased risk of tacrolimus-related neurotoxicity (DiMartini et al, 2008). In cases of CNI-related neurotoxicity, patients can frequently be switched to a different drug within the same class with successful resolution of symptoms (Emiroglu et al, 2006; Erol et al, 2007). Most central nervous system (CNS) complications (80%) occur within 1 month after OLT but may be seen up to several years after transplantation (Bronster et al, 2000). The incidence of neurologic complications in LDLT recipients (17%) approximates that of cadaveric allograft recipients, and outcomes from these complications appear to be similar (Saner et al, 2010).
Immunosuppressant Toxicity
CNIs can cause significant renal and neurotoxicity. Use of sirolimus as an alternative immunosuppressant in patients with these adverse effects may allow patients to recover from CNI toxicity without significant risk of rejection (Di Benedetto et al, 2008; Morard et al, 2007).
Outcomes
Survival after OLT has improved markedly as a result of continuing refinements in organ procurement and preservation, recipient selection, surgical and anesthetic techniques, perioperative care, and long-term immunosuppression. Overall patient survival at 10 years is 60% in the United States, with an unadjusted 10-year graft survival of 54% (Thuluvath et al, 2010). In select patient populations, some institutions have achieved near zero short-term mortality (Broering et al, 2004; Sugawara et al, 2002).
Recipient factors associated with reduced survival include recipient age over 65 years, preexisting CAD, insulin-dependent DM, renal insufficiency, and extreme body mass index (BMI) (Busuttil et al, 2005; Dick et al, 2009; Nair et al, 2002; Thuluvath et al, 2010; Yoo & Thuluvath, 2002). Other factors that adversely affect survival include conditions that require urgent transplantation, such as hepatic malignancy as the indication for transplantation, donor hospitalization longer than 6 days, cerebrovascular accident as donor cause of death, prolonged warm (>45 minutes) and cold (>10 hours) ischemic time, and retransplantation (Busuttil et al, 2005).
Deceased-donor split allografts have not been shown to adversely affect outcomes in large series (Busuttil et al, 2005), and LDLT over the last decade has had better long-term outcomes than deceased-donor transplantation (Thuluvath et al, 2010). The overall rate of retransplantation for recipients of DDLT was 7.8% in 2007, representing the lowest level in the last decade (USRTR, 2008). Outcomes after liver transplantation for certain disease processes are discussed below.
Biliary Atresia
The long-term primary hepatic salvage rate after portoenterostomy in infants with biliary atresia ranges from 20% to 45% (Davenport et al, 2004; Otte et al, 1994; Schreiber et al, 2007; see Chapter 40). The remaining infants develop progressive biliary cirrhosis and ultimately require liver transplantation. Biliary atresia is the most common indication for OLT in children younger than 18 years, and the best results are obtained in patients referred for transplantation early, after only a single attempted portoenterostomy. Approximately 80% of children transplanted for biliary atresia have had a prior biliary drainage procedure (Utterson et al, 2005; Visser et al, 2004). Despite this high rate of transplantation, portoenterostomy remains a viable bridge to definitive therapy, allowing infants to grow.
Transplantation is a durable solution for biliary atresia, and this recipient population obtains the best survival rates, with 10-year patient survival greater than 80% and graft survival greater than 67% (Barshes et al, 2005; D’Alessandro et al, 2007; Diem et al, 2003; Farmer et al, 2007; Schreiber et al, 2007; Visser et al, 2004). The use of split-liver cadaveric grafts and living related transplantation has expanded the potential donor pool, extending OLT to an increased number of children with biliary atresia, without a significant increase in post-OLT mortality (Barshes et al, 2005; Chen et al, 2006; Yersiz et al, 2003).
Additionally, use of living-donor liver allografts significantly shortens time on the waiting list (Visser et al, 2004); however, technical variant grafts may increase the risk of graft failure (Utterson et al, 2005). Graft failure and vascular complications account for the majority of early post-OLT deaths, whereas rejection remains the leading cause of late death in these patients (Barshes et al, 2005).
Primary Sclerosing Cholangitis and Primary Biliary Cirrhosis
Primary sclerosing cholangitis (PSC) is a chronic cholestatic liver disease of unknown origin that is frequently associated (70% to 80%) with inflammatory bowel disease, usually ulcerative colitis (UC; see Chapter 41). Patients with PSC also are at risk for the development of cholangiocarcinoma. Although medical palliation exists, the only cure for PSC is liver transplantation. Outcomes after OLT for PSC are excellent, with 5-year patient survival rates ranging from 80% to 85% (Goss et al, 1997; Ricci et al, 1997; Solano et al, 2003) and 10-year survival at 70% (Busuttil et al, 2005). Whether the incidental discovery of cholangiocarcinoma in the explanted liver affects patient survival is controversial (Goss et al, 1997; Solano et al, 2003). Approximately 9% to 11% of patients develop recurrent PSC after transplantation.
Primary biliary cirrhosis (PBC) is an autoimmune disease typically characterized by the development of circulating antimitochondrial antibodies (AMAs). Patients with PBC generally have better long-term survival (>7 years) than patients transplanted for PSC (Maheshwari et al, 2004). They are more prone to chronic rejection, however, and are less likely to be weaned from immunosuppression (MacQuillan et al, 2003). PBC likely recurs in a few patients after OLT, although long-term follow-up data are lacking, and this rate appears to be much less common than for PSC recurrence. Posttransplantation AMA titers are not predictive of disease recurrence; a liver biopsy specimen showing granulomatous destructive cholangitis remains the gold standard (Faust, 2001).
Alcoholic Cirrhosis
Although often producing ESLD in conjunction with viral hepatitis, alcoholic cirrhosis is one of the leading indications for OLT in the United States (Amersi et al, 1998; Thuluvath et al, 2010). Early reports showing equivalent outcomes in patients with alcoholic cirrhosis compared with patients with nonalcoholic cirrhosis resulted in widespread application of OLT for this indication (Bird et al, 1990; Starzl et al, 1988). Medical outcomes in this group continue to be good (Busuttil et al, 2005; DiMartini et al, 1998; Lim & Keeffe, 2004).
Recidivism occurs in roughly 20% to 30% of patients transplanted for alcoholic cirrhosis (Biselli et al, 2010; Pageaux et al, 2003; Rowley et al, 2010). Although there is little convincing evidence that resumption of drinking adversely affects graft or patient survival after OLT, there are ethical concerns over possibly misallocating donor organs to active substance abusers (Lim & Keeffe, 2004). Relapse might also serve as a proxy marker for patients who are likely to have poor nutrition and overall health as well as poor compliance with their immunosuppressive regimen and, therefore, increased risk of rejection (Pageaux et al, 2003). Most transplant centers require potential transplant candidates to participate in a support program, undergo psychologic evaluation, and remain abstinent from substance abuse, with documented random drug and alcohol testing for at least 6 months before placement on the waiting list. Multivariate analysis of alcoholic patients with cirrhosis who have undergone OLT showed that abstinence for 6 months or more before OLT is the strongest predictor for nonrelapse after transplantation (Miguet et al, 2004).
Hepatitis B Virus
Since the standardization of HBV treatment with hepatitis B immunoglobulin (HBIG) and antiviral agents, the rate of recurrent HBV infection in liver transplant recipients has declined from 80% in untreated patients to less than 10% of treated patients at 2 years (Roche & Samuel, 2004; Saab et al, 2009; Todo et al, 1991; see Chapter 64). Current treatment strategies include reducing or eliminating active viral replication before transplantation through the administration of lamivudine or adefovir, in combination with other agents, then combining one of these with HBIG after OLT indefinitely. This approach has resulted in similar outcomes for patients without recurrence compared with patients undergoing OLT for other reasons (Kim et al, 2004; Roche et al, 2003; Steinmuller et al, 2002); however, HBIG administration can be costly and cumbersome, and resistance to lamivudine is reported in 20% of patients after treatment for 1 year.
Although combination prophylaxis with lamivudine and HBIG is highly effective, other methods to combat HBV recurrence are being investigated. Alternative regimens include combination antiviral agents (e.g., lamivudine and adefovir) without routine use of HBIG (Nath et al, 2006). HBV recurrence portends a poor outcome, with a 5-year survival rate of 47% (Saab et al, 2009). For patients with HCC at transplantation, recurrent HBV infection is also significantly associated with a recurrence of HCC after transplantation (Saab et al, 2009).
Hepatitis C Virus
Since its definitive identification in 1989, HCV has been determined to be a major cause of chronic liver disease and cirrhosis, accounting for 37% to 41% of OLT performed in the United States (Thuluvath et al, 2010). Approximately 2% of the general population carries the virus, and 50% to 60% of these individuals develop chronic liver disease (Farmer et al, 2000; see Chapter 64).
Short-term and intermediate-term patient survival after OLT for HCV is not significantly different from HCV-negative patients who receive transplants for other nonmalignant reasons, although graft survival is significantly reduced compared with allografts for alcoholic liver disease (Biselli et al, 2010; Boker et al, 1997; Ghobrial et al, 2001). Unadjusted 10-year survival rates are lower for HCV-positive recipients in the United States (Thuluvath et al, 2010). Recurrent infection is nearly universal, and chronic hepatitis develops in most patients after OLT (Gordon et al, 2009). The clinical course is similar to nontransplanted HCV-positive patients, with only 13% developing hepatic fibrosis (Saab et al, 2009). In these patients with recurrent hepatitis C cirrhosis, 5-year survival decreases to 30% (Saab et al, 2005). Recipients coinfected with HIV tend to experience faster and more aggressive recurrence of HCV (Di Benedetto et al, 2008).
In contrast to HBV, no approved therapies are currently available for prevention of recurrent HCV infection after liver transplantation. Outcomes for transplantation of HCV recipients with extended-criteria livers are significantly worse, and donor age has been reported to affect graft survival adversely (Lake et al, 2005; Machicao et al, 2004; Russo et al, 2004; Thuluvath et al, 2010); however, older donors can be safely used, with similar results in HCV-positive recipients with short cold and warm ischemia times and careful donor selection (Doyle et al, 2008).
Hepatocellular Carcinoma (See Chapters 80 and 97D)
HCC was one of the first documented indications for OLT (Starzl et al, 1968). The decision to utilize transplantation for this disease is based on the oncologic premise of performing a complete resection (total hepatectomy) with wide surgical margins to effect a cure and to remove potential future sites of tumor formation in the diseased remnant liver. Improved selection of appropriate candidates with HCC for OLT has led to improved outcomes and consequently has more than doubled the number of patients with HCC on the waiting list (Thuluvath et al, 2010).
Liver transplantation in the setting of extrahepatic malignancy is contraindicated. Conversely, the finding of incidental HCC in the explanted specimen after transplantation for a nonmalignant indication does not alter patient or graft outcome significantly (Cillo et al, 2004). The difficulty has been the selection of patients for OLT with malignant disease confined to the liver but at an advanced stage.
In their series examining liver transplantation for HCC, Mazzaferro and colleagues (1996) obtained a 75% 4-year actuarial survival using specific guidelines in highly selected patients. Subsequently termed the Milan criteria for OLT, patients under this protocol were offered transplantation if they had a solitary tumor less than 5 cm in diameter or no more than three nodules, each less than 3 cm in diameter (stage II). Largely as a result of this report, UNOS adopted these criteria for placing potential recipients on the waiting list, limiting transplantation to patients with stage I (single tumor up to 2 cm in diameter) or stage II disease.
Currently, MELD exception points (22 points) are awarded only on a routine basis to patients with stage II HCC, who account for 8% to 9% of all deceased-donor liver recipients today (USRTR, 2008). Patients with more advanced HCC can only receive additional MELD priority points through special appeal to regional review boards of transplant surgeons and physicians. Although not equaling the excellent results of Mazzaferro and colleagues (1996), reported 5-year survival after OLT for HCC generally ranges from 60% to 75% in several large, more recent series (Goodman et al, 2005; Yao et al, 2001; Yoo et al, 2003).
Several centers have attempted to expand liver transplantation to patients with tumors exceeding the Milan criteria and have met with varying degrees of success. The University of California–San Francisco (UCSF) criteria permit transplantation for patients with solitary tumors smaller than 6.5 cm diameter, fewer than three tumors, and a maximum diameter less than 4.5 cm or total cumulative diameter less than 8 cm (Yao et al, 2001).
Prospective trials currently are examining multimodal treatment for HCC, whereby patients undergo chemoembolization or other ablative techniques to downstage the tumor clinically, followed by transplantation. We require patients with stage III or IV HCC to undergo tumor downstaging, usually with TACE, to meet Milan criteria and with a minimum period of 3 months of observation without evidence of extrahepatic tumor on restaging. Our results using this strategy in highly selected patients are equivalent to patients initially meeting Milan criteria without the need for downstaging (Chapman et al, 2008). Currently, 58% of patients receiving MELD exception points for HCC have undergone TACE or radiofrequency ablation while on the waiting list, with the majority receiving TACE (Thuluvath et al, 2010). Although no prospective trials demonstrate a long-term oncologic benefit to pretransplantation TACE, retrospective data strongly suggest that this may be beneficial, and we consider this a routine practice in our HCC transplant candidates (Bharat et al, 2006).
Cholangiocarcinoma (See Chapters 50C and 97E)
No effective medical treatment has been found for cholangiocarcinoma. Surgical extirpation is feasible in less than 30% of all patients and produces 5-year survival rates less than 30%, even with complete resection (Jarnagin et al, 2001; Rea et al, 2004). Additionally, patients with PSC, a known risk factor for cholangiocarcinoma, are often poor candidates for resection because of concurrent cirrhosis. Historically, the use of OLT for treatment of cholangiocarcinoma has been associated with 5-year survival rates of 30% and even lower rates in patients with incidentally discovered cholangiocarcinoma at transplantation (Becker et al, 2008). Surgeons at the Mayo Clinic reported successful results using strict treatment protocols for performing OLT in patients with localized, node-negative hilar cholangiocarcinoma, which included the use of neoadjuvant chemoradiation, staging laparotomy, resection to negative margins, and OLT (see Chapter 97E). Using these or similar protocols, 5-year survival rates of 72% have been achieved in a highly selected group of patients, many of whom had PSC (Heimbach et al, 2004; Rea et al, 2009; Sudan et al, 2002). Retrospective analysis revealed improved outcomes for patients enrolled in the Mayo protocol compared with those of patients undergoing major hepatic resection (Rea et al, 2005); however, these outcomes have been difficult to achieve at other institutions, and neoadjuvant therapy is associated with higher rates of arterial and venous complications after transplantation (Mantel et al, 2007).
Fulminant Hepatic Failure (See Chapters 72 and 97C)
The cohort that undergoes OLT for fulminant hepatic failure comprises a diverse group of patients and disease etiologies. By definition, these patients have no previous demonstrable liver disease, yet they present acutely with encephalopathy; synthetic dysfunction, such as coagulopathy; and jaundice. Most commonly, the etiology is never determined, but fulminant hepatic failure (FHF) can be caused by drugs, viruses, toxins, or other liver injuries (Hoofnagle et al, 1995). Although artificial liver support systems are under development, the only widely available cure for acute liver failure is OLT. Limited donor organ availability leads to the relatively high waiting-list mortality rate of 377 deaths per 1000 patient-years at risk. MELD scores tend not to predict outcome accurately in patients with FHF (Kremers et al, 2004); in this setting, outcome after OLT is below average, with approximately 67% of patients surviving to 5 years (Farmer et al, 2003).
Nonalcoholic Steatohepatitis (See Chapter 65)
Cirrhosis secondary to nonalcoholic steatohepatitis (NASH) is a rapidly increasing indication for liver transplantation, accounting for approximately 5% of transplantations performed in 2007 (USRTR, 2008). NASH, a stepwise progression in the natural history of nonalcoholic fatty liver disease (NAFLD), is associated with obesity. Data gathered to study the effects of BMI on outcomes of OLT are inconclusive. Although it has been shown that morbidly obese recipients are at higher risk for PTMS and infectious complications, it has not been definitively shown to affect long-term outcomes (Leonard et al, 2008; Malik et al, 2009). Independent risk factors for development of graft steatosis after OTL include pretransplantation graft steatosis, post-OLT obesity, hypertension, DM, hyperlipidemia, use of tacrolimus, and alcoholic cirrhosis as the primary indication for liver transplantation (Dumortier et al, 2010).
Retransplantation
The rate of retransplantation has been gradually declining, reaching a low of 7.8% in 2007 (USRTR, 2008). In particular, the rate of retransplantation for HCV dropped to 5% in 2008 (Thuluvath et al, 2010). The most common indications for retransplantation include primary nonfunction (46% of retransplants), HAT (29%), acute and chronic rejection (7%), and recurrent disease (Busuttil et al, 2005; Jain et al, 2000; Lang et al, 2008; Marti et al, 2008; Thuluvath et al, 2010). Use of DCD allografts is associated with a 13% retransplantation rate, which has remained stable over the last decade (Thuluvath et al, 2010).
Outcome after retransplantation is generally worse than after initial OLT but is slowly improving, with a 10-year graft survival up to 69% in some centers (Marti et al, 2008); however, this varies significantly by primary indication (Adam & Hoti, 2009; Thuluvath et al, 2010). Patients requiring retransplantation are at increased risk for infection, HAT, and acute renal failure (Uemura et al, 2007). Retransplantation for primary nonfunction yields 54% to 60% patient survival at 5 years (Kemmer et al, 2007; Uemura et al, 2007). Recurrent HCV is associated with worse outcomes following retransplantation overall, and most centers avoid retransplantation for this indication; however, a recent multicenter analysis demonstrated similar 3-year survival rates in a highly select group of patients with recurrent HCV compared with non-HCV retransplant recipients (McCashland et al, 2007). Factors portending a poor prognosis include preoperative mechanical ventilation, HCV infection, elevated creatinine and bilirubin, and prolonged donor cold ischemia (Markmann et al, 1999; Yoo et al, 2003).
Conclusion
1 Organized regional and national networks designed for the early identification of potential organ donors and rapid procurement in select candidates
2 Improved surgical and anesthetic techniques, allowing previous obstacles to OLT to be overcome (e.g., PVT, severe coagulopathy)
3 Novel antirejection and antimicrobial agents, with increasing emphasis on achieving minimal immunosuppression in the shortest time possible
4 Increased vigilance for the occurrence of acute complications after OLT, with the development of rapid screening tests for HAT, PVT, and primary nonfunction
Abt PL, et al. Survival following liver transplantation from non–heart-beating donors. Ann Surg. 2004;239:87-92.
Adam R, Hoti E. Liver transplantation: the current situation. Semin Liver Dis. 2009;29:3-18.
Amersi F, Farmer DG, Busuttil RW. Fifteen-year experience with adult and pediatric liver transplantation at the University of California, Los Angeles. Clin Transpl. 1998:255-261.
Amodio P, et al. Neurological complications after orthotopic liver transplantation. Dig Liver Dis. 2007;39:740-747.
Arzu GD, et al. Temporary porto-caval shunt utility during orthotopic liver transplantation. Transplant Proc. 2008;40:1937-1940.
Asfar S, et al. An analysis of late deaths after liver transplantation. Transplantation. 1996;61:1377-1381.
Bakthavatsalam R, et al. Rescue of acute portal vein thrombosis after liver transplantation using a cavoportal shunt at re-transplantation. Am J Transplant. 2001;1:284-287.
Balsells J, et al. Evolution of biliary complications in liver transplantation without biliary drainage. Transplant Proc. 1995;27:2339-2340.
Banff Consensus. Banff schema for grading liver allograft rejection: an international consensus document. Hepatology. 1997;25:658-663.
Barkun JS, et al. Do biliary endoprostheses decrease biliary complications after liver transplantation? Transplant Proc. 2003;35:2435-2437.
Barshes NR, et al. Orthotopic liver transplantation for biliary atresia: the U.S. experience. Liver Transpl. 2005;11:1193-1200.
Bawa SM, et al. Biliary reconstruction with or without an internal biliary stent in orthotopic liver transplantation: a prospective randomised trial. Transpl Int. 1998;11(Suppl 1):S245-247.
Becker NS, et al. Outcomes analysis for 280 patients with cholangiocarcinoma treated with liver transplantation over an 18-year period. J Gastrointest Surg. 2008;12:117-122.
Bekker J, Ploem S, de Jong KP. Early hepatic artery thrombosis after liver transplantation: a systematic review of the incidence, outcome and risk factors. Am J Transplant. 2009;9:746-757.
Bernat JL, et al. Report of a National Conference on Donation after cardiac death. Am J Transplant. 2006;6:281-291.
Bharat A, et al. Pre-liver transplantation locoregional adjuvant therapy for hepatocellular carcinoma as a strategy to improve long-term survival. J Am Coll Surg. 2006;203:411-420.
Bianchi G, et al. Metabolic syndrome in liver transplantation: relation to etiology and immunosuppression. Liver Transpl. 2008;14:1648-1654.
Bird GL, et al. Liver transplantation in patients with alcoholic cirrhosis: selection criteria and rates of survival and relapse. Br Med J. 1990;301:15-17.
Biselli M, et al. Long term follow-up and outcome of liver transplantation for alcoholic liver disease: a single center case-control study. J Clin Gastroenterol. 2010;44:52-57.
Blanco JJ, et al. Liver transplantation in cirrhotic patients with diabetes mellitus: midterm results, survival, and adverse events. Liver Transpl. 2001;7:226-233.
Boker KH, et al. Long-term outcome of hepatitis C virus infection after liver transplantation. Hepatology. 1997;25:203-210.
Braun F, et al. Is liposomal amphotericin B (ambisome) an effective prophylaxis of mycotic infections after liver transplantation? Transplant Proc. 1998;30:1481-1483.
Broering DC, et al. One hundred thirty-two consecutive pediatric liver transplants without hospital mortality: lessons learned and outlook for the future. Ann Surg. 2004;240:1002-1012. discussion 1012
Bronster DJ, et al. Central nervous system complications in liver transplant recipients: incidence, timing, and long-term follow-up. Clin Transplant. 2000;14:1-7.
Brown RSJr, et al. A survey of liver transplantation from living adult donors in the United States. N Engl J Med. 2003;348:818-825.
Bucuvalas JC, et al. Outcomes after liver transplantation: keep the end in mind. J Pediatr Gastroenterol Nutr. 2006;43(Suppl 1):S41-48.
Buczkowski AK, et al. Spatulated end-to-end bile duct reconstruction in orthotopic liver transplantation. Clin Transplant. 2007;21:7-12.
Busuttil RW, et al. Analysis of long-term outcomes of 3200 liver transplantations over two decades: a single-center experience. Ann Surg. 2005;241:905-916. discussion 916-908
Canzanello VJ, et al. Evolution of cardiovascular risk after liver transplantation: a comparison of cyclosporine A and tacrolimus (FK506). Liver Transpl Surg. 1997;3:1-9.
Chan EY, et al. Ischemic cholangiopathy following liver transplantation from donation after cardiac death donors. Liver Transpl. 2008;14:604-610.
Chapman WC, et al. Outcomes of neoadjuvant transarterial chemoembolization to downstage hepatocellular carcinoma before liver transplantation. Ann Surg. 2008;248:617-625.
Chen CL, et al. Living donor liver transplantation for biliary atresia: a single-center experience with first 100 cases. Am J Transplant. 2006;6:2672-2679.
Cheng YF, et al. Risk factors for intraoperative portal vein thrombosis in pediatric living donor liver transplantation. Clin Transplant. 2004;18:390-394.
Ciccarelli O, et al. Transjugular intrahepatic portosystemic shunt approach and local thrombolysis for treatment of early posttransplant portal vein thrombosis. Transplantation. 2001;72:159-161.
Cillo U, et al. Liver transplantation for the treatment of moderately or well-differentiated hepatocellular carcinoma. Ann Surg. 2004;239:150-159.
Cooper JT, et al. Donation after cardiac death: the University of Wisconsin experience with renal transplantation. Am J Transplant. 2004;4:1490-1494.
Cruciani M, et al. Antifungal prophylaxis in liver transplant patients: a systematic review and meta-analysis. Liver Transpl. 2006;12:850-858.
D’Alessandro AM, et al. The predictive value of donor liver biopsies for the development of primary nonfunction after orthotopic liver transplantation. Transplantation. 1991;51:157-163.
D’Alessandro AM, et al. Liver transplantation from controlled non–heart-beating donors. Surgery. 2000;128:579-588.
D’Alessandro AM, et al. Liver transplantation in pediatric patients: twenty years of experience at the University of Wisconsin. Pediatr Transplant. 2007;11:661-670.
da Silva RF, et al. Prevalence, treatment, and outcomes of the hepatic artery stenosis after liver transplantation. Transplant Proc. 2008;40:805-807.
Davenport M, et al. The outcome of the older (> or = 100 days) infant with biliary atresia. J Pediatr Surg. 2004;39:575-581.
Davila D, Bartlett A, Heaton N. Temporary portocaval shunt in orthotopic liver transplantation: need for a standardized approach? Liver Transpl. 2008;14:1414-1419.
de Vera ME, et al. Liver transplantation using donation after cardiac death donors: long-term follow-up from a single center. Am J Transplant. 2009;9:773-781.
Delgado-Borrego A, et al. Prospective study of liver transplant recipients with HCV infection: evidence for a causal relationship between HCV and insulin resistance. Liver Transpl. 2008;14:193-201.
Del Gaudio M, et al. Outcome of hepatic artery reconstruction in liver transplantation with an iliac arterial interposition graft. Clin Transplant. 2005;19:399-405.
Demetris A, et al. Update of the International Banff Schema for Liver Allograft Rejection: working recommendations for the histopathologic staging and reporting of chronic rejection—an international panel. Hepatology. 2000;31:792-799.
Demetris AJ, et al. Antibody-mediated rejection of human orthotopic liver allografts: a study of liver transplantation across ABO blood group barriers. Am J Pathol. 1988;132:489-502.
Devlin J, et al. Defining the outcome of immunosuppression withdrawal after liver transplantation. Hepatology. 1998;27:926-933.
Dhar R, Young GB, Marotta P. Perioperative neurological complications after liver transplantation are best predicted by pre-transplant hepatic encephalopathy. Neurocrit Care. 2008;8:253-258.
Di Benedetto F, et al. Human immunodeficiency virus and liver transplantation: our point of view. Transplant Proc. 2008;40:1965-1971.
Dick AA, et al. Liver transplantation at the extremes of the body mass index. Liver Transpl. 2009;15:968-977.
Diem HV, et al. Pediatric liver transplantation for biliary atresia: results of primary grafts in 328 recipients. Transplantation. 2003;75:1692-1697.
DiMartini A, et al. Outcome of liver transplantation in critically ill patients with alcoholic cirrhosis: survival according to medical variables and sobriety. Transplantation. 1998;66:298-302.
DiMartini A, et al. Age, Model for End-stage Liver Disease score, and organ functioning predict posttransplant tacrolimus neurotoxicity. Liver Transpl. 2008;14:815-822.
Doyle MB, et al. Liver transplant for hepatitis C virus: effect of using older donor grafts on short- and medium-term survival. Arch Surg. 2008;143:679-685. discussion 685
Doyle MB, et al. Short- and long-term outcomes after steatotic liver transplantation. Arch Surg. 2010;145(7):653-660.
Duffy JP, et al. Vascular complications of orthotopic liver transplantation: experience in more than 4,200 patients. J Am Coll Surg. 2009;208:896-903. discussion 903-895
Dumortier J, et al. Non-alcoholic fatty liver disease in liver transplant recipients: another story of “seed and soil.”. Am J Gastroenterol. 2010;105:613-620.
Emiroglu R, et al. Tacrolimus-related neurologic and renal complications in liver transplantation: a single-center experience. Transplant Proc. 2006;38:619-621.
Erol I, et al. Neurological complications of liver transplantation in pediatric patients: a single center experience. Pediatr Transplant. 2007;11:152-159.
Farmer DG, et al. Orthotopic liver transplantation. In: Blumgart, L, Fong, Y. Surgery of the Liver and Biliary Tract. Philadelphia: Saunders; 2001:2071-2085.
Farmer DG, et al. Liver transplantation for fulminant hepatic failure: experience with more than 200 patients over a 17-year period. Ann Surg. 2003;237:666-675. discussion 675-666
Farmer DG, et al. Predictors of outcomes after pediatric liver transplantation: an analysis of more than 800 cases performed at a single institution. J Am Coll Surg. 2007;204:904-914. discussion 914-906
Faust TW. Recurrent primary biliary cirrhosis, primary sclerosing cholangitis, and autoimmune hepatitis after transplantation. Liver Transpl. 2001;7:S99-S108.
Feng S, et al. Characteristics associated with liver graft failure: the concept of a donor risk index. Am J Transplant. 2006;6:783-790.
Fernandez-Merino FJ, et al. Impact of donor, recipient, and graft features on the development of primary dysfunction in liver transplants. Transplant Proc. 2003;35:1793-1794.
Figueras J, et al. Temporary portocaval shunt during liver transplantation with vena cava preservation: results of a prospective randomized study. Liver Transpl. 2001;7:904-911.
Freeman RBJr, et al. The new liver allocation system: moving toward evidence-based transplantation policy. Liver Transpl. 2002;8:851-858.
Gane E, et al. Randomised trial of efficacy and safety of oral ganciclovir in the prevention of cytomegalovirus disease in liver-transplant recipients. The Oral Ganciclovir International Transplantation Study Group [corrected]. Lancet. 1997;350:1729-1733.
Gayowski T, et al. Orthotopic liver transplantation in high-risk patients: risk factors associated with mortality and infectious morbidity. Transplantation. 1998;65:499-504.
Ghaus N, Bohlega S, Rezeig M. Neurological complications in liver transplantation. J Neurol. 2001;248:1042-1048.
Ghobrial RM, et al. A 10-year experience of liver transplantation for hepatitis C: analysis of factors determining outcome in over 500 patients. Ann Surg. 2001;234:384-393. discussion 393-384
Goodman J, et al. Liver transplantation for hepatocellular carcinoma: expanding special priority to include stage III disease. Arch Surg. 2005;140:459-464. discussion 464
Gordon FD, Kwo P, Vargas HE. Treatment of hepatitis C in liver transplant recipients. Liver Transpl. 2009;15:126-135.
Gordon SM, et al. Should prophylaxis for Pneumocystis carinii pneumonia in solid organ transplant recipients ever be discontinued? Clin Infect Dis. 1999;28:240-246.
Gores GJ, et al. Model for End-stage Liver Disease (MELD) exception for cholangiocarcinoma or biliary dysplasia. Liver Transpl. 2006;12:S95-S97.
Goss JA, et al. Orthotopic liver transplantation for primary sclerosing cholangitis: a 12-year single center experience. Ann Surg. 1997;225:472-481. discussion 481-473
Goss JA, et al. Long-term results of pediatric liver transplantation: an analysis of 569 transplants. Ann Surg. 1998;228:411-420.
Gunsar F, et al. Late hepatic artery thrombosis after orthotopic liver transplantation. Liver Transpl. 2003;9:605-611.
Hanouneh IA, et al. The significance of metabolic syndrome in the setting of recurrent hepatitis C after liver transplantation. Liver Transpl. 2008;14:1287-1293.
Heimbach JK, et al. Liver transplantation for unresectable perihilar cholangiocarcinoma. Semin Liver Dis. 2004;24:201-207.
Hoffmann K, et al. Is veno-venous bypass still needed during liver transplantation? A review of the literature. Clin Transplant. 2009;23:1-8.
Hoofnagle JH, et al. Fulminant hepatic failure: summary of a workshop. Hepatology. 1995;21:240-252.
Hosein Shokouh-Amiri M, et al. Choice of surgical technique influences perioperative outcomes in liver transplantation. Ann Surg. 2000;231:814-823.
Jain A, et al. Long-term survival after liver transplantation in 4,000 consecutive patients at a single center. Ann Surg. 2000;232:490-500.
Jain A, et al. Does valganciclovir hydrochloride (Valcyte) provide effective prophylaxis against cytomegalovirus infection in liver transplant recipients? Transplant Proc. 2005;37:3182-3186.
Jarnagin WR, et al. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann Surg. 2001;234:507-517. discussion 517-509
Johnson MW, et al. Internal biliary stenting in orthotopic liver transplantation. Liver Transpl. 2000;6:356-361.
Johnson SR, et al. Primary nonfunction (PNF) in the MELD Era: an SRTR database analysis. Am J Transplant. 2007;7:1003-1009.
Jurim O, et al. Reduced-size grafts—the solution for hepatic artery thrombosis after pediatric liver transplantation? J Pediatr Surg. 1995;30:53-55.
Kamath GS, et al. Primary nonfunction of the liver graft: when should we retransplant? Transplant Proc. 1991;23:1954.
Karie-Guigues S, et al. Long-term renal function in liver transplant recipients and impact of immunosuppressive regimens (calcineurin inhibitors alone or in combination with mycophenolate mofetil): the TRY study. Liver Transpl. 2009;15:1083-1091.
Kemmer N, et al. Long-term analysis of primary nonfunction in liver transplant recipients. Transplant Proc. 2007;39:1477-1480.
Kim WR, et al. Outcome of liver transplantation for hepatitis B in the United States. Liver Transpl. 2004;10:968-974.
Klintmalm GB. Rejection therapies. Dig Dis Sci. 1991;36:1431-1433.
Kremers WK, et al. MELD score as a predictor of pretransplant and posttransplant survival in OPTN/UNOS status 1 patients. Hepatology. 2004;39:764-769.
Lake JR, et al. Differential effects of donor age in liver transplant recipients infected with hepatitis B, hepatitis C and without viral hepatitis. Am J Transplant. 2005;5:549-557.
Lang H, et al. Incidence of liver retransplantation and its effect on patient survival. Transplant Proc. 2008;40:3201-3203.
Langer FB, et al. Outcome of hepaticojejunostomy for biliary tract obstruction following liver transplantation. Clin Transplant. 2009;23:361-367.
Laryea M, et al. Metabolic syndrome in liver transplant recipients: prevalence and association with major vascular events. Liver Transpl. 2007;13:1109-1114.
Leonard J, et al. The impact of obesity on long-term outcomes in liver transplant recipients-results of the NIDDK liver transplant database. Am J Transplant. 2008;8:667-672.
Lerut J, et al. Complications of venous reconstruction in human orthotopic liver transplantation. Ann Surg. 1987;205:404-414.
Lewis MB, Howdle PD. Neurologic complications of liver transplantation in adults. Neurology.. 2003;61:1174-1178.
Lim JK, Keeffe EB. Liver transplantation for alcoholic liver disease: current concepts and length of sobriety. Liver Transpl. 2004;10:S31-S38.
Limaye AP, et al. Emergence of ganciclovir-resistant cytomegalovirus disease among recipients of solid-organ transplants. Lancet. 2000;356:645-649.
Loinaz C, et al. Liver transplantation in patients with portal thrombosis: results in 76 patients. Transplant Proc. 2002;34:248-249.
Machicao VI, et al. Donor age affects fibrosis progression and graft survival after liver transplantation for hepatitis C. Transplantation. 2004;77:84-92.
MacQuillan GC, Neuberger J. Liver transplantation for primary biliary cirrhosis. Clin Liver Dis. 2003;7:941-956. ix
Maheshwari A, Yoo HY, Thuluvath PJ. Long-term outcome of liver transplantation in patients with PSC: a comparative analysis with PBC. Am J Gastroenterol. 2004;99:538-542.
Malik SM, et al. Outcome after liver transplantation for NASH cirrhosis. Am J Transplant. 2009;9:782-793.
Malinchoc M, et al. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology. 2000;31:864-871.
Maluf DG, et al. Adult living donor versus deceased donor liver transplantation: a 6-year single center experience. Am J Transplant. 2005;5:149-156.
Mantel HT, et al. Vascular complications after orthotopic liver transplantation after neoadjuvant therapy for hilar cholangiocarcinoma. Liver Transpl. 2007;13:1372-1381.
Markmann JF, et al. A simple model to estimate survival after retransplantation of the liver. Transplantation. 1999;67:422-430.
Marsman WA, et al. Use of fatty donor liver is associated with diminished early patient and graft survival. Transplantation. 1996;62:1246-1251.
Marti J, et al. Optimization of liver grafts in liver retransplantation: a European single-center experience. Surgery. 2008;144:762-769.
Mateo R, et al. Risk factors for graft survival after liver transplantation from donation after cardiac death donors: an analysis of OPTN/UNOS data. Am J Transplant. 2006;6:791-796.
Mazariegos GV, et al. Weaning of immunosuppression in liver transplant recipients. Transplantation. 1997;63:243-249.
Mazzaferro V, et al. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med. 1996;334:693-699.
McAlister VC, et al. Orthotopic liver transplantation using low-dose tacrolimus and sirolimus. Liver Transpl. 2001;7:701-708.
McCashland T, et al. Retransplantation for hepatitis C: results of a U.S. multicenter retransplant study. Liver Transpl. 2007;13:1246-1253.
Merion RM, et al. The survival benefit of liver transplantation. Am J Transplant. 2005;5:307-313.
Merkel FK, Jonasson O, Bergan JJ. Procurement of cadaver donor organs: evisceration technique. Transplant Proc. 1972;4:585-589.
Miguet M, et al. Predictive factors of alcohol relapse after orthotopic liver transplantation for alcoholic liver disease. Gastroenterol Clin Biol. 2004;28:845-851.
Millis JM, et al. Portal vein thrombosis and stenosis in pediatric liver transplantation. Transplantation. 1996;62:748-754.
Morard I, et al. Conversion to sirolimus-based immunosuppression in maintenance liver transplantation patients. Liver Transpl. 2007;13:658-664.
Moreno-Gonzalez E, et al. Advantages of the piggy back technique on intraoperative transfusion, fluid consumption, and vasoactive drug requirements in liver transplantation: a comparative study. Transplant Proc. 2003;35:1918-1919.
Muñoz SJ, et al. Hyperlipidemia and obesity after orthotopic liver transplantation. Transplant Proc. 1991;23:1480-1483.
Nair S, Verma S, Thuluvath PJ. Pretransplant renal function predicts survival in patients undergoing orthotopic liver transplantation. Hepatology. 2002;35:1179-1185.
Nath DS, et al. Hepatitis B prophylaxis post-liver transplant without maintenance hepatitis B immunoglobulin therapy. Clin Transplant. 2006;20:206-210.
Navasa M, et al. Diabetes mellitus after liver transplantation: prevalence and predictive factors. J Hepatol. 1996;25:64-71.
Nikitin D, et al. Twenty years of follow-up of aortohepatic conduits in liver transplantation. Liver Transpl. 2008;14:1486-1490.
Nishida S, et al. Internal hernia of the small bowel around infrarenal arterial conduits after liver transplantation. Clin Transplant. 2002;16:334-338.
Nishida S, et al. Piggyback technique in adult orthotopic liver transplantation: an analysis of 1067 liver transplants at a single center. HPB (Oxford). 2006;8:182-188.
Nissen NN, Klein AS. Choledocho-choledochostomy in deceased donor liver transplantation. J Gastrointest Surg. 2009;13:810-813.
Nygaard CE, Townsend RN, Diamond DL. Organ donor management and organ outcome: a 6-year review from a Level I trauma center. J Trauma. 1990;30:728-732.
O’Connor TP, Lewis WD, Jenkins RL. Biliary tract complications after liver transplantation. Arch Surg. 1995;130:312-317.
Ojo AO, et al. Chronic renal failure after transplantation of a nonrenal organ. N Engl J Med. 2003;349:931-940.
Otte JB, et al. Sequential treatment of biliary atresia with Kasai portoenterostomy and liver transplantation: a review. Hepatology. 1994;20:41S-48S.
Pagadala M, et al. Posttransplant metabolic syndrome: an epidemic waiting to happen. Liver Transpl. 2009;15:1662-1670.
Pageaux GP, et al. Alcohol relapse after liver transplantation for alcoholic liver disease: does it matter? J Hepatol. 2003;38:629-634.
Papanicolaou GA, et al. Nosocomial infections with vancomycin-resistant Enterococcus faecium in liver transplant recipients: risk factors for acquisition and mortality. Clin Infect Dis. 1996;23:760-766.
Pappas PG, et al. Invasive fungal infections among organ transplant recipients: results of the Transplant-Associated Infection Surveillance Network (TRANSNET). Clin Infect Dis. 2010;50:1101-1111.
Park JM, et al. Efficacy and safety of low-dose valganciclovir in the prevention of cytomegalovirus disease in adult liver transplant recipients. Liver Transpl. 2006;12:112-116.
Pascual M, et al. Anticardiolipin antibodies and hepatic artery thrombosis after liver transplantation. Transplantation. 1997;64:1361-1364.
Paterson DL, et al. Intraoperative hypothermia is an independent risk factor for early cytomegalovirus infection in liver transplant recipients. Transplantation. 1999;67:1151-1155.
Pawarode A, Fine DM, Thuluvath PJ. Independent risk factors and natural history of renal dysfunction in liver transplant recipients. Liver Transpl. 2003;9:741-747.
Pennefather SH, et al. Use of low-dose arginine vasopressin to support brain-dead organ donors. Transplantation. 1995;59:58-62.
Pine JK, et al. Liver transplantation following donation after cardiac death: an analysis using matched pairs. Liver Transpl. 2009;15:1072-1082.
Ploeg RJ, et al. Risk factors for primary dysfunction after liver transplantation—a multivariate analysis. Transplantation. 1993;55:807-813.
Pokorny H, et al. Influence of cumulative number of marginal donor criteria on primary organ dysfunction in liver recipients. Clin Transplant. 2005;19:532-536.
Pruthi J, et al. Analysis of causes of death in liver transplant recipients who survived more than 3 years. Liver Transpl. 2001;7:811-815.
Pungpapong S, et al. Cigarette smoking is associated with an increased incidence of vascular complications after liver transplantation. Liver Transpl. 2002;8:582-587.
Qian YB, et al. Risk factors for biliary complications after liver transplantation. Arch Surg. 2004;139:1101-1105.
Racusen LC, et al. Antibody-mediated rejection criteria: an addition to the Banff 97 classification of renal allograft rejection. Am J Transplant. 2003;3:708-714.
Rea DJ, et al. Major hepatic resection for hilar cholangiocarcinoma: analysis of 46 patients. Arch Surg. 2004;139:514-523. discussion 523-515
Rea DJ, et al. Liver transplantation with neoadjuvant chemoradiation is more effective than resection for hilar cholangiocarcinoma. Ann Surg. 2005;242:451-458. discussion 458-461
Rea DJ, et al. Transplantation for cholangiocarcinoma: when and for whom? Surg Oncol Clin N Am. 2009;18:325-337. ix
Reich DL, et al. Association of intraoperative hypotension and pulmonary hypertension with adverse outcomes after orthotopic liver transplantation. J Cardiothorac Vasc Anesth. 2003;17:699-702.
Reid GE, et al. Early intra-abdominal infections associated with orthotopic liver transplantation. Transplantation. 2009;87:1706-1711.
Ricci P, et al. A prognostic model for the outcome of liver transplantation in patients with cholestatic liver disease. Hepatology. 1997;25:672-677.
Roche B, Samuel D. Evolving strategies to prevent HBV recurrence. Liver Transpl. 2004;10:S74-S85.
Roche B, et al. HBV DNA persistence 10 years after liver transplantation despite successful anti-HBS passive immunoprophylaxis. Hepatology. 2003;38:86-95.
Rowley AA, et al. The psychiatric diagnosis of alcohol abuse and the medical diagnosis of alcoholic related liver disease: effects on liver transplant survival. J Clin Psychol Med Settings. 2010;17:195-202.
Russell DL, et al. Outcomes of colonization with MRSA and VRE among liver transplant candidates and recipients. Am J Transplant. 2008;8:1737-1743.
Russo MW, et al. Impact of donor age and year of transplant on graft survival in liver transplant recipients with chronic hepatitis C. Am J Transplant. 2004;4:1133-1138.
Rustgi VK, et al. Impact of body mass index on graft failure and overall survival following liver transplant. Clin Transplant. 2004;18:634-637.
Saab S, et al. Mortality predictors in liver transplant recipients with recurrent hepatitis C cirrhosis. Liver Int. 2005;25:940-945.
Saab S, et al. Recurrence of hepatocellular carcinoma and hepatitis B reinfection in hepatitis B surface antigen–positive patients after liver transplantation. Liver Transpl. 2009;15:1525-1534.
Safadi A, et al. Perioperative risk predictors of cardiac outcomes in patients undergoing liver transplantation surgery. Circulation. 2009;120:1189-1194.
Salizzoni M, et al. Marginal grafts: finding the correct treatment for fatty livers. Transpl Int. 2003;16:486-493.
Saner F, et al. Neurological complications after cadaveric and living donor liver transplantation. J Neurol. 2006;253:612-617.
Saner FH, et al. Neurologic complications in adult living donor liver transplant patients: an underestimated factor? J Neurol. 2010;257:253-258.
Scatton O, et al. Randomized trial of choledochocholedochostomy with or without a T tube in orthotopic liver transplantation. Ann Surg. 2001;233:432-437.
Schreiber RA, et al. Biliary atresia: the Canadian experience. J Pediatr. 2007;151:659-665. 665 e651
Schwimmer JB, et al. Prevalence of fatty liver in children and adolescents. Pediatrics.. 2006;118:1388-1393.
Senzolo M, Ferronato C, Burra P. Neurologic complications after solid organ transplantation. Transpl Int. 2009;22:269-278.
Shah JN, et al. Endoscopic management of biliary complications after adult living donor liver transplantation. Am J Gastroenterol. 2004;99:1291-1295.
Sharma R, et al. Surgical complications following liver transplantation in patients with portal vein thrombosis—a single-center perspective. J Gastrointest Surg. 2010;14:520-527.
Shaw BWJr, et al. Venous bypass in clinical liver transplantation. Ann Surg. 1984;200:524-534.
Sheiner PA, et al. Long-term medical complications in patients surviving > or = 5 years after liver transplant. Transplantation. 2000;69:781-789.
Shields PL, et al. Poor outcome in patients with diabetes mellitus undergoing liver transplantation. Transplantation. 1999;68:530-535.
Shiley KT, et al. Increased incidence of cytomegalovirus infection in high-risk liver transplant recipients receiving valganciclovir prophylaxis versus ganciclovir prophylaxis. Liver Transpl. 2009;15:963-967.
Singh N. The current management of infectious diseases in the liver transplant recipient. Clin Liver Dis. 2000;4:657-673. ix
Singh N, et al. Methicillin-resistant Staphylococcus aureus: the other emerging resistant gram-positive coccus among liver transplant recipients. Clin Infect Dis. 2000;30:322-327.
Singh N, et al. Predicting bacteremia and bacteremic mortality in liver transplant recipients. Liver Transpl. 2000;6:54-61.
Singh N, et al. Bacteremias in liver transplant recipients: shift toward gram-negative bacteria as predominant pathogens. Liver Transpl. 2004;10:844-849.
Singh N, et al. Antifungal management practices in liver transplant recipients. Am J Transplant. 2008;8:426-431.
Singhal A, et al. Endovascular treatment of hepatic artery thrombosis following liver transplantation. Transpl Int. 2010;23:245-256.
Soin AS, et al. Donor arterial variations in liver transplantation: management and outcome of 527 consecutive grafts. Br J Surg. 1996;83:637-641.
Solano E, et al. Liver transplantation for primary sclerosing cholangitis. Transplant Proc. 2003;35:2431-2434.
Sotiropoulos GC, et al. Orthotopic liver transplantation: T-tube or not T-tube? Systematic review and meta-analysis of results. Transplantation. 2009;87:1672-1680.
Starzl TE, et al. Homotransplantation of the liver in humans. Surg Gynecol Obstet. 1963;117:659-676.
Starzl TE, et al. Orthotopic homotransplantation of the human liver. Ann Surg. 1968;168:392-415.
Starzl TE, et al. Development of a suprahepatic recipient vena cava cuff for liver transplantation. Surg Gynecol Obstet. 1979;149:77.
Starzl TE, et al. A flexible procedure for multiple cadaveric organ procurement. Surg Gynecol Obstet. 1984;158:223-230.
Starzl TE, et al. An improved technique for multiple organ harvesting. Surg Gynecol Obstet. 1987;165:343-348.
Starzl TE, et al. Orthotopic liver transplantation for alcoholic cirrhosis. JAMA. 1988;260:2542-2544.
Stegall MD, et al. Metabolic complications after liver transplantation. Diabetes, hypercholesterolemia, hypertension, and obesity. Transplantation. 1995;60:1057-1060.
Steinmuller T, et al. Increasing applicability of liver transplantation for patients with hepatitis B–related liver disease. Hepatology. 2002;35:1528-1535.
Stewart ZA, et al. Increased risk of graft loss from hepatic artery thrombosis after liver transplantation with older donors. Liver Transpl. 2009;15:1688-1695.
Strasberg SM, et al. Selecting the donor liver: risk factors for poor function after orthotopic liver transplantation. Hepatology. 1994;20:829-838.
Sudan D, et al. Radiochemotherapy and transplantation allow long-term survival for nonresectable hilar cholangiocarcinoma. Am J Transplant. 2002;2:774-779.
Sugawara Y, et al. Living donor liver transplantation in adults: recent advances and results. Surgery. 2002;132:348-352.
Taner CB, Bathala V, Nguyen JH. Primary nonfunction in liver transplantation: a single-center experience. Transplant Proc. 2008;40:3566-3568.
Thuluvath PJ. When is diabetes mellitus a relative or absolute contraindication to liver transplantation? Liver Transpl. 2005:S25-S29.
Thuluvath PJ, et al. Liver transplantation in the United States, 1999-2008. Am J Transplant. 2010;10:1003-1019.
Todo S, et al. Orthotopic liver transplantation for patients with hepatitis B virus–related liver disease. Hepatology. 1991;13:619-626.
Torbenson M, et al. Causes of death in autopsied liver transplantation patients. Mod Pathol. 1998;11:37-46.
Totsuka E, et al. Analysis of clinical variables of donors and recipients with respect to short-term graft outcome in human liver transplantation. Transplant Proc. 2004;36:2215-2218.
Tzakis A, Todo S, Starzl TE. Orthotopic liver transplantation with preservation of the inferior vena cava. Ann Surg. 1989;210:649-652.
Uemura T, et al. Liver retransplantation for primary nonfunction: analysis of a 20-year single-center experience. Liver Transpl. 2007;13:227-233.
Ueno T, et al. Clinical outcomes from hepatic artery stenting in liver transplantation. Liver Transpl. 2006;12:422-427.
Urena MA, et al. An approach to the rational use of steatotic donor livers in liver transplantation. Hepatogastroenterology. 1999;46:1164-1173.
U.S. Scientific Registry of Transplant Recipients, 2008: Annual Report of the U.S. Organ Procurement and Transplantation Network and the Scientific Registry of Transplant Recipients: Transplant Data 1998-2007. Rockville, MD, U.S. Department of Health and Human Services, Health Resources and Services Administration, Healthcare Systems Bureau, Division of Transplantation.
Utterson EC, et al. Biliary atresia: clinical profiles, risk factors, and outcomes of 755 patients listed for liver transplantation. J Pediatr. 2005;147:180-185.
Verran DJ, et al. Biliary reconstruction without T tubes or stents in liver transplantation: report of 502 consecutive cases. Liver Transpl Surg. 1997;3:365-373.
Visser BC, et al. The influence of portoenterostomy on transplantation for biliary atresia. Liver Transpl. 2004;10:1279-1286.
Vivarelli M, et al. Ischemic arterial complications after liver transplantation in the adult: multivariate analysis of risk factors. Arch Surg. 2004;139:1069-1074.
Vivarelli M, et al. Can antiplatelet prophylaxis reduce the incidence of hepatic artery thrombosis after liver transplantation? Liver Transpl. 2007;13:651-654.
Wachs ME, et al. The risk of transmission of hepatitis B from HBsAg(−), HBcAb(+), HBIgM(−) organ donors. Transplantation. 1995;59:230-234.
Weiss S, et al. Biliary reconstruction using a side-to-side choledochocholedochostomy with or without T-tube in deceased donor liver transplantation: a prospective randomized trial. Ann Surg. 2009;250:766-771.
Welling TH, et al. Biliary complications following liver transplantation in the Model for End-stage Liver Disease era: effect of donor, recipient, and technical factors. Liver Transpl. 2008;14:73-80.
Wheeldon DR, et al. Transforming the “unacceptable” donor: outcomes from the adoption of a standardized donor management technique. J Heart Lung Transplant. 1995;14:734-742.
Wiesner R, et al. Model for End-stage Liver Disease (MELD) and allocation of donor livers. Gastroenterology. 2003;124:91-96.
Wiesner RH, et al. Acute hepatic allograft rejection: incidence, risk factors, and impact on outcome. Hepatology. 1998;28:638-645.
Wiesner RH, et al. Recent advances in liver transplantation. Mayo Clin Proc. 2003;78:197-210.
Winston DJ. Prevention of cytomegalovirus disease in transplant recipients. Lancet. 1995;346:1380-1381.
Winston DJ, Busuttil RW. Randomized controlled trial of oral ganciclovir versus oral acyclovir after induction with intravenous ganciclovir for long-term prophylaxis of cytomegalovirus disease in cytomegalovirus-seropositive liver transplant recipients. Transplantation. 2003;75:229-233.
Winston DJ, Busuttil RW. Randomized controlled trial of sequential intravenous and oral ganciclovir versus prolonged intravenous ganciclovir for long-term prophylaxis of cytomegalovirus disease in high-risk cytomegalovirus-seronegative liver transplant recipients with cytomegalovirus-seropositive donors. Transplantation. 2004;77:305-308.
Winston DJ, Emmanouilides C, Busuttil RW. Infections in liver transplant recipients. Clin Infect Dis.. 1995;21:1077-1089. quiz 1090-1071
Wolfe RA, Roys EC, Merion RM. Trends in organ donation and transplantation in the United States, 1999-2008. Am J Transplant. 2010;10:961-972.
Woo DH, et al. Management of portal venous complications after liver transplantation. Tech Vasc Interv Radiol. 2007;10:233-239.
Wood KE, et al. Care of the potential organ donor. N Engl J Med. 2004;351:2730-2739.
Yao FY, et al. Liver transplantation for hepatocellular carcinoma: expansion of the tumor size limits does not adversely impact survival. Hepatology. 2001;33:1394-1403.
Yersiz H, et al. One hundred in situ split-liver transplantations: a single-center experience. Ann Surg. 2003;238:496-505. discussion 506-497
Yoo HY, Molmenti E, Thuluvath PJ. The effect of donor body mass index on primary graft nonfunction, retransplantation rate, and early graft and patient survival after liver transplantation. Liver Transpl. 2003;9:72-78.
Yoo HY, Thuluvath PJ. The effect of insulin-dependent diabetes mellitus on outcome of liver transplantation. Transplantation. 2002;74:1007-1012.
Yoo HY, et al. The outcome of liver transplantation in patients with hepatocellular carcinoma in the United States between 1988 and 2001: 5-year survival has improved significantly with time. J Clin Oncol. 2003;21:4329-4335.
Zajko AB, et al. Percutaneous transhepatic cholangiography rather than ultrasound as a screening test for postoperative biliary complications in liver transplant patients. Transplant Proc. 1988;20:678-681.