[level-membership-for-neurosurgery-category]
22 Natural History of Giant Intracranial Aneurysms
Introduction
The first clinical series of giant intracranial aneurysms were reported in 1969 by Morley and Barr1 and Bull.2 Giant intracranial aneurysms (GIAs) are defined as those aneurysms with a greatest diameter equal to or exceeding 2.5 cm. This arbitrary designation was chosen in order to conform to the subset of the largest aneurysms in the Cooperative Study of Intracranial Aneurysms and Subarachnoid Hemorrhage.3 Likewise smaller lesions have been shown to differ significantly with regard to their rate of rupture, the incidence of presentation with mass effect, and most importantly the difficulty with surgical treatment. GIAs are relatively infrequent compared to their smaller counterparts and therefore literature support to aid in clinical decision making is not as readily available. While once thought to represent more benign lesions with a lower incidence of hemorrhage and a more optimistic natural history, it is clear now from recent, large, multicenter studies that these lesions behave aggressively and hemorrhage more frequently than their smaller counterparts.4,5
Pathologic considerations
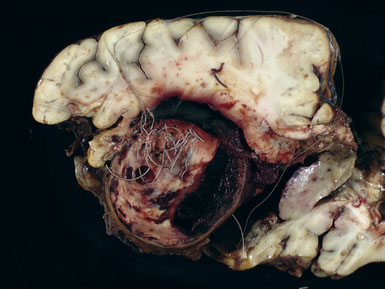
Giant aneurysms are classified as either saccular or fusiform with the vast majority being the saccular type. Giant saccular aneurysms develop as a result of hemodynamic stress at arterial bifurcations and branch points similar to smaller saccular intracranial aneurysms. Once the sac has formed, both the neck and fundus will then undergo progressive enlargement. Ferguson proposed that turbulence within the aneurysm leads to endothelial injury and subsequent platelet aggregation and fibrin deposition.6 This intraluminal cascade in giant aneurysms represents a dynamic series of events with accumulation and dissipation of platelets and fibrin-thrombus debris in an irregular fashion. Fusiform giant aneurysms most commonly result from atherosclerosis, however, may develop in patients with collagen vascular disorders such as Marfan’s syndrome, Ehlers-Danlos syndrome, and systemic lupus erythematosis. All of these diseases produce multifocal injury to the endothelial wall, resulting in weakening of an entire segment of the vessel. Fusiform lesions often present because of mass effect but not infrequently produce ischemic symptoms secondary to thromboembolic phenomena (Figure 22–1).
Epidemiology
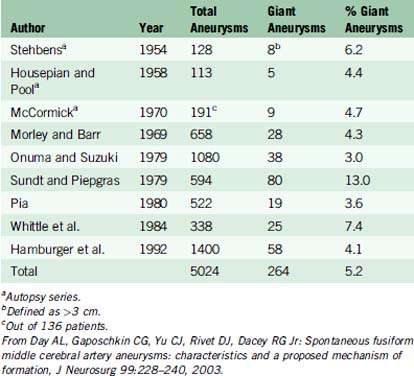
Some epidemiologic differences from smaller saccular aneurysms seem to validate the separate clinical entity of giant aneurysms. In various clinical and autopsy series, the percentage of total aneurysms that are giant ranges from 3% to 13%. In one of the largest series to date, Hamburger et al. reported 1400 intracranial aneurysms and identified 58 giant aneurysms, yielding an incidence of 4.1%.11 Based on data from three autopsy studies and six large clinical series, the aggregate incidence of giant intracranial aneurysms is 5.2%13 (Table 22–1).
Age and gender
GIAs more commonly occur in females and most commonly come to clinical attention during the fifth to sixth decade of life. In a review of the world literature, Fox identified 693 giant aneurysms of which 60% occurred in females. This review correlates with known anatomical gender distributions with the proximal internal carotid artery location in 73% of females versus 52% for the anterior communicating complex and basilar apex.7 Anson reviewed 14 series with 754 GIAs identified, finding 461 females and 295 males, yielding a 1.56:1 ratio.27 In Anson’s review, ages ranged from 6 months to 76 years with 67% of patients presenting between ages 40 and 70. This distribution parallels that of Fox’s review of the world literature, where the median age of presentation was in the sixth decade of life.
Giant fusiform and dolichoectatic aneurysms probably present at a slightly younger age. Reviews from three different institutions found a mean age of presentation to be 38, 43, and 49 years with a male:female ratio of 1.5:1.8–10
Intracranial aneurysms are rare in children, with only 1% to 2% of all aneurysms occurring in the pediatric population. Of these, however, GIAs constitute a much higher proportion, are more common in males, and are less often present with hemorrhage. Ferrante reviewed 72 cases of pediatric aneurysms and found that 27% were giant and 50% were 1 to 2.5 cm in diameter.11
Location
Saccular GIAs have a similar anatomic predilection as their smaller counterparts, while the distribution is different. Similarly, the internal carotid artery (ICA) is the most common site, although GIAs occur proportionately more frequently in the vertebrobasilar system and less frequently in the anterior communicating artery (ACOM) region. Drake and Peerless reported 641 GIAs treated between 1965 and 1992 in the following locations: vertebrobasilar, 390; ICA, 178; middle cerebral artery (MCA), 64; and anterior cerebral artery (ACA), 19.12 While their data does reflect a referral bias, it supports the general trend that certain intracranial arterial segments are more vulnerable to this pathology as seen throughout the literature. Giant fusiform and dolichoectatic aneurysms have a predilection for the vertebrobasilar circulation and the MCA. These lesions, frequently referred to as “serpentine aneurysms,” are often massive, partially thrombosed lesions through which runs a serpentine channel.13
Natural history
The natural history of GIAs is dependent upon several factors, including the location of the aneurysm with respect to the subarachnoid space, the pathological form of the aneurysm (saccular vs. fusiform), the specific anatomic location, and the presence or absence of laminated thrombus and or atherosclerotic plaque within the fundus and neck of the aneurysm.14 In Morley and Barr’s original series of 28 patients, 17 had intradural aneurysms, and only four of those underwent a direct operation on their aneurysm. Of the five patients who received no definitive therapy, four died. Of the five patients with intradural aneurysms who had common carotid ligation, three died and one was disabled. They concluded that “direct surgical attack on extracavernous giant aneurysms is seldom possible or successful except in the case of MCA aneurysms.”
Peerless et al. observed 31 patients with giant intracranial aneurysms (25 saccular and six fusiform). Sixty-eight percent of patients with saccular aneurysms were dead at 2 years and 85% were dead at 5 years. If patients presenting with subarachnoid hemorrhage were excluded, the 2-year mortality rate was 62%. Only four patients who were all disabled were alive at 5 years. Of the six patients with fusiform aneurysms, four were dead at 2 years, one died at 3.5 years after diagnosis, and one remained disabled.15 Ljunggren et al. demonstrated a mortality or severe morbidity rate of 80% within 5 years in patients with untreated symptomatic GIAs.16 Hamburger et al. reported on 58 patients with GIA and showed that 7% of patients presenting without SAH died, compared with 29% of patients presenting with SAH. Only 18% of the later group were discharged home as independent compared with 50% of the former group.17 The Italian Cooperative Study on Giant Intracranial Aneurysms found that extensive or thick cisternal deposition of blood was associated with a significantly higher mortality rate than with thin or absent depositions.18 Although GIAs present more commonly with mass effect or thromboembolic events, there is no evidence that they are associated with a lower rate of rupture. It is evident that the presence of thrombus in the sac does not protect against SAH and that when extensive or complete may actually have a deleterious effect on the natural history.19–21
The most recent and comprehensive data on the natural history of GIAs comes from the International Study of Unruptured Intracranial Aneurysms Investigators (ISUIA I & II).4,5 In the retrospective arm, ISUIA I, 1449 patients with 1937 intracranial aneurysms were reviewed. The patients were divided into two groups. Group 1 patients (N = 727) had no history of SAH from a different aneurysm, while Group 2 (N = 722) had a history of a SAH from a previously repaired aneurysm. The rupture rate for aneurysms greater than 25 mm in diameter was 6% in the first year. In the prospective arm, ISUIA II, 4060 patients were assessed of which 1692 did not have aneurysmal repair. The 5-year cumulative rupture rates for patients who did not have a history of SAH was 40% for giant aneurysms located in the anterior circulation, and 50% for aneurysms located in the posterior circulation as well as the posterior communicating artery (PCOM).
The natural history of patients presenting with symptoms related to mass effect or ischemia from giant aneurysms is less clear. In giant paraclinoid aneurysms presenting with visual loss, the deterioration usually progresses without treatment.22,23 Patients with symptoms of brainstem compression due to giant dolichoectatic verterbobasilar artery aneurysms also appear to have a poor prognosis without treatment. In a report by Michael, all seven patients with giant posterior circulation aneurysms treated with observation died between 2 months and 2 years after diagnosis.24 The Italian Cooperative Study analyzed 25 patients with untreated giant aneurysms and reported mortality in seven cases (28%), which was double the mortality rate in the 86 treated cases. Morbidity was seen in 12 of the observed cases due to progressive expansion of the aneurysm mass.
The natural history of giant aneurysms presenting with mass effect is usually one of progressive enlargement. Sonntag and Stein demonstrated progressive enlargement on successive angiograms in two of 13 cases being non-surgically managed.25 Growth can be extremely rapid with one case in the literature documenting formation in less than 3 months.26 There is no evidence that the presence of extensive thrombus improves the natural history of giant aneurysms. Despite the lack of luminal filling, it has been demonstrated that even extensive intra-aneursmal thrombosis does not protect against SAH. Furthermore, the presence of extensive or even complete thrombosis within a giant aneurysm may increase the risk of compressive symptoms.27
The risk of distal ischemic symptoms may actually increase in partially or extensively thrombosed giant aneurysms, contributing to a more ominous natural history. Sutherland and Peerless noted distal thromboembolism in 59% of cases in their series of giant aneurysms containing thrombus.28 In addition, it is also possible for thrombus to propagate into the parent artery and cause occlusion. The risk of thromboembolic stroke from thrombosed giant aneurysms seems to be greatest in cases of fusiform or dolichoectatic vertebrobasilar artery aneurysms.29
Intracavernous aneurysms
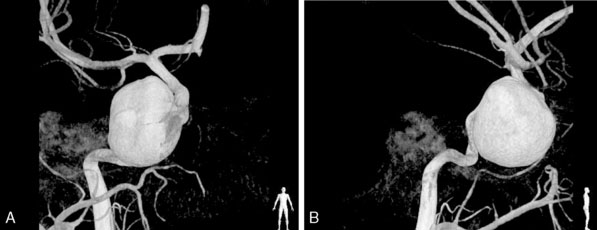
Intracavernous aneurysms generally contain sacs located extradurally, and therefore, for the purposes of discussing natural history and clinical manifestations, are considered as a separate and unique clinical entity. They most commonly occur in middle aged women with a history of hypertension. Approximately 21% of intracavernous aneurysms are bilateral and 15% are giant, with intracavernous aneurysms accounting for 3% to 39% of all giant aneurysms.30 Unlike their intradural counterparts these lesions are associated with a more benign natural history.31 Intracavernous aneurysms may be discovered as incidental findings or as a result of compressive symptoms of the cranial nerves traversing the cavernous sinus (Figure 22–2).
When an intracavernous aneurysm ruptures, it most commonly produces a carotid cavernous fistula, which is the cause of most high-flow, spontaneous carotid cavernous fistulas.32 This presentation, however, is more common with smaller intracavernous aneurysms than in those with giant sacs. The wall of the intracavernous internal carotid artery bulges into the sphenoid sinus in 71% of specimens and bone is absent between the ICA and sphenoid mucosa in 4%.33 Because of this anatomic relationship, an intracavernous aneurysm may rupture into the sphenoid sinus and produce epistaxis, which may be life-threatening.
Nonsaccular vertebrobasilar aneurysms
This class of aneurysms warrants a separate discussion because of the unique natural history associated with these lesions. Frequently referred to as fusiform or dolichoectatic aneurysms, they are relatively uncommon compared to their saccular counterparts. In addition, they are different with respect to their clinical presentation and treatment options. These aneurysms usually cause symptoms by compressing adjacent brainstem or cranial nerves or by launching emboli from mural thrombus within the aneurysm leading to ischemia in vascular territories distal to the aneurysm. Subarachnoid hemorrhage occurs with less frequency compared to saccular aneurysms. The hemorrhage rate for dolichoectatic and fusiform aneurysms is not well understood and no clear consensus exists in the world literature regarding the predicted behavior of these aneurysms. Drake and others have reported that the number of patients with fusiform and dolichoectatic aneurysms that present with subarachnoid hemorrhage is lower than for patients harboring saccular anerurysms.12,34–36 Other series have demonstrated a rupture rate quite similar to that of saccular aneurysms.37 However, because fusiform aneurysms are more likely to be diagnosed from symptoms related to compression or ischemia, there is a lower incidence of SAH at presentation.
This discrepancy in the natural history is accounted for predominantly by small case series and retrospective reviews. Flemming et al. were the first group to prospectively review the natural history of fusiform and dolichoectatic vertebrobasilar aneurysms in their analysis of 159 patients from the Mayo Clinic.38 Patients most commonly presented initially because of symptoms associated with mass effect (22%), related ischemic stroke (28%), or hemorrhage (3%). After a mean follow-up interval of 4.4 years, they calculated the annual prospective risk of rupture to be 0.9% overall and up to 2.3% per year for certain morphological subtypes based upon their own classification system. Sixty-five patients died during follow-up. These deaths were found to be aneurysm related in 45% of patients, with the most common mechanism being ischemic cerebral infarction or thrombosis.
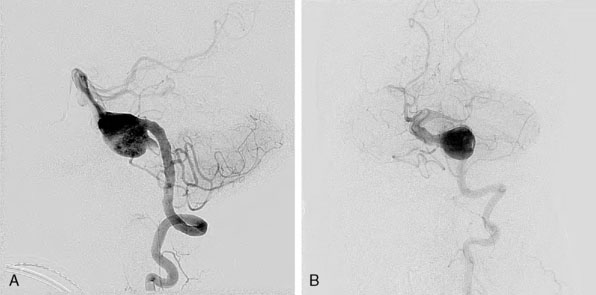
O’shaughnessy and colleagues reported the progressive growth of a giant dolichoectatic vertebrobasilar artery aneurysm after complete Hunterian ligation of the posterior circulation. This report is the first such case in the English literature in which progressive lesion expansion occurred after complete Hunterian proximal arterial occlusion of the posterior circulation.39 They demonstrated lesional expansion despite eliminating the principal orthograde inflow and proposed an alternative theory to conventional thinking of aneurysm growth and subsequent expansion as a consequence of hemodynamic stress and proposed that in vertebrobasilar dolichoectasia a more likely explanation arises from a progressive, expansile arteriopathy throughout the arterial wall that occurs independent of hemodynamic stress (Figure 22–3).
1 Morley T.P., Barr H.W.K. Giant intracranial aneurysms: diagnosis, course and management. Clin Neurosurg. 1969;16:73-94.
2 Bull J. Massive aneurysms at the base of the brain. Brain. 1969;92:535-570.
3 Locksley H.B. Report on the Cooperative Study of Intracranial Aneurysms and Subarachnoid Hemorrhage. Section V, Part II. Natural history of subarachnoid hemorrhage, intracranial aneurysms and arteriovenous malformations. J Neurosurg. 1966;25:321-368.
4 Wiebers D.O., Whisnant J.P., Huston J.3rd, et al. International Study of Unruptured Intracranial Aneurysms Investigators. Lancet. 2003;362:103-110.
5 International Study of Unruptured Intracranial Aneurysms Investigators. Unruptured intracranial aneurysms—risk of rupture and risks of surgical intervention. N Engl J Med. 1998;339:1725-1733.
6 Ferguson G.G. Physical factors in the initiation, growth and rupture of human intracranial saccular aneurysms. J Neurosurg. 1972;37:666-667.
7 Fox J.L. Intracranial Aneurysms. New York: Springer-Verlag, 1983.
8 Little J.R., Louis P., Weistein M., et al. Giant fusiform aneurysms of the cerebral arteries. Stroke. 1981;12:183-188.
9 Suzuki S., Takahashi T., Ohkuma H., et al. Management of giant serpentine aneurysms of the middle cerebral artery. Review of the literature and report of a case successfully treated by STA-MCA anastomosis only. Acta Neurochir (Wien). 1992;117:23-39.
10 Anson J.A., Lawton M.T., Spetzler R.F. Characteristics and surgical treatment of dolichoectatic and fusiform aneurysms. J Neurosurg. 1996;84:185-193.
11 Ferrante L., Fortuna A., Celli P., et al. Intracranial arterial aneurysms in early childhood. Surg Neurol. 1988;29:39-56.
12 Drake C.G., Peerless S.J. Giant fusiform intracranial aneurysms: review of 120 patients treated surgically from 1965 to 1992. J Neurosurg. 1997;87:141-162.
13 Day A.L., Gaposchkin C.G., Yu C.J., Rivet D.J., Dacey R.G.Jr. Spontaneous fusiform middle cerebral artery aneurysms: characteristics and a proposed mechanism of formation. J Neurosurg. 2003;99:228-240.
14 Barrow D.L., Alleyne C. Natural History of Giant Intracranial Aneurysms and Indications for Intervention. Clin Neurosurg. 1995;42:214-244.
15 Peerless S.J., Wallace M.D., Drake C.G. Giant intracranial aneurysms. In: Youmans J.R., editor. Neurological Surgery. ed 3. Philidelphia: WB Saunders; 1990:1742-1763.
16 Ljunggren B., Brandt L., Sundbarg G., et al. Early management of aneurysmal subarachnoid hemorrhage. Neurosurgery. 1982;11:412-418.
17 Hamburger C., Schoenberger J., Lange M. Management and prognosis of intracranial giant aneurysms. A report of 58 cases. Neurosurg Rev. 1992;15:97-103.
18 Rosta L., Battaglia R., Pasqualin A., et al. Italian Cooperative Study on Giant Intracranial Aneurysms. 2: Radiological Data. Acta Neurochir Suppl (Wien). 1988;42:53-59.
19 Drake C.G. Giant intracranial aneurysms: experience with surgical treatment in 174 patients. Clin Neurosurg. 1979;26:12-95.
20 Khurana V.G., Wijdicks E.F.M., Parisi J.E., et al. Acute deterioration from thrombosis and rerupture of a giant intracranial aneurysm. Neurology. 1999;52:1697-1699.
21 Khurana V.G., Piepgras D.G., Whisnant J.P. Ruptured giant intracranial aneurysms. Part I. A study of rebleeding. J Neurosurg. 1998;88:425-429.
22 Ferguson C.G., Drake Cg. Carotid-ophthalmic aneurysms: visual abnormalities in 32 patients and the results of treatment. Surg Neurol. 1981;16:1-8.
23 Heros R.C., Nelson P.B., Ojemann R.G., et al. Large and giant paraclinoid aneurysms: surgical techniques, complications and results. Neurosurgery. 1983;12:153-163.
24 Michael W.F. Posterior fossa aneurysms simulating tumors. J Neurol Neurosurg Psychiatry. 1974;37:218-223.
25 Sonntag V.K.H., Yuan R.H., Stein B.M. Giant intracranial aneurysms: a review of 13 cases. Surg Neurol. 1977;8:81-84.
26 Fried L.C., Yballe A. Rapid formation of a giant aneurysm: case report. J Neurol Neurosurg Psychiatry. 1972;35:527-530.
27 Anson J.A. Epidemiology and natural history. In: Awad I.A., Barrow D.L., editors. Giant Intracranial Aneurysms. Park Ridge, IL: American Association of Neurological Surgeons, 1995.
28 Sutherland G.R., King M.E., Peerless S.J., et al. Platelet interaction with giant intracranial aneurysms. J Neurosurg. 1982;56:53-61.
29 Nishizaki T., Tamaki N., Takeda N., et al. dolichoectatic basilar artery: a review of 23 cases. Stroke. 1986;17:1277-1281.
30 Berenstein A., Ransohof J., Kupersmith M., et al. Transvascular treatment of giant aneurysms of the cavernous carotid and vertebral arteries. Surg Neurol. 1984;21:3-12.
31 Linskkey M.E., Sekhar L.N., Hirsch W.L., et al. Aneurysms of the intracavernous carotid artery: natural history and indications for treatment. Neurosurgery. 1990;6:933-938.
32 Barrow D.L., Spector R.H., Braun I.F., et al. Classification and treatment of spontaneous carotid cavernous fistulas. J Neurosurg. 1985;56:248-256.
33 Harris F.S., Rhoton A.L. Anatomy of the cavernous sinus. A microsurgical study. J Neurosurg. 1976;45:169-180.
34 Anson J.A., Lawton M.T., Spetzler R.F. Chararacteristics and surgical treatment of dolichoectatic and fusiform aneurysms. J Neurosurg. 1996;84:185-193.
35 Nishizaki T., Tamaki N., Takeda N., et al. Dolichoectatic basilar artery: a review of 23 cases. Stroke. 1986;17:1277-1281.
36 Anson J.A. Treatment strategies for intracranial fusiform aneurysms. Neurosurg Clin North Am. 1998;9:743-753.
37 Vates G.E., Auguste K.I., Lawton M.T. Fusiform, dolichoectatic and dissecting aneurysms: diagnosis and management. In: LeRoux P., Winn H.R., Newell D., editors. Management of Cerebral Aneurysms. Philadelphia: Saunders; 2003:689-709.
38 Flemming K.D., Wiebers D.O., Brown R.D., et al. Prospective risk of hemorrhage in patients with vertebrobasilar nonsaccular intracranial aneurysm. J Neurosurg. 2004;101:82-87.
39 O’shaughnessy B.A., Getch C.C., Bendok B.R., et al. Progressive growth of a giant dolichoectatic vertebrobasilar artery aneurysm after complete Hunterian occlusion of the posterior circulation: case report. Neurosurgery. 2004;55:1223.
[/level-membership-for-neurosurgery-category][not-level-membership-for-neurosurgery-category]
22 Natural History of Giant Intracranial Aneurysms
Introduction
The first clinical series of giant intracranial aneurysms were reported in 1969 by Morley and Barr1 and Bull.2 Giant intracranial aneurysms (GIAs) are defined as those aneurysms with a greatest diameter equal to or exceeding 2.5 cm. This arbitrary designation was chosen in order to conform to the subset of the largest aneurysms in the Cooperative Study of Intracranial Aneurysms and Subarachnoid Hemorrhage.3 Likewise smaller lesions have been shown to differ significantly with regard to their rate of rupture, the incidence of presentation with mass effect, and most importantly the difficulty with surgical treatment. GIAs are relatively infrequent compared to their smaller counterparts and therefore literature support to aid in clinical decision making is not as readily available. While once thought to represent more benign lesions with a lower incidence of hemorrhage and a more optimistic natural history, it is clear now from recent, large, multicenter studies that these lesions behave aggressively and hemorrhage more frequently than their smaller counterparts.4,5
Pathologic considerations
Giant aneurysms are classified as either saccular or fusiform with the vast majority being the saccular type. Giant saccular aneurysms develop as a result of hemodynamic stress at arterial bifurcations and branch points similar to smaller saccular intracranial aneurysms. Once the sac has formed, both the neck and fundus will then undergo progressive enlargement. Ferguson proposed that turbulence within the aneurysm leads to endothelial injury and subsequent platelet aggregation and fibrin deposition.6 This intraluminal cascade in giant aneurysms represents a dynamic series of events with accumulation and dissipation of platelets and fibrin-thrombus debris in an irregular fashion. Fusiform giant aneurysms most commonly result from atherosclerosis, however, may develop in patients with collagen vascular disorders such as Marfan’s syndrome, Ehlers-Danlos syndrome, and systemic lupus erythematosis. All of these diseases produce multifocal injury to the endothelial wall, resulting in weakening of an entire segment of the vessel. Fusiform lesions often present because of mass effect but not infrequently produce ischemic symptoms secondary to thromboembolic phenomena (Figure 22–1).
Epidemiology
Some epidemiologic differences from smaller saccular aneurysms seem to validate the separate clinical entity of giant aneurysms. In various clinical and autopsy series, the percentage of total aneurysms that are giant ranges from 3% to 13%. In one of the largest series to date, Hamburger et al. reported 1400 intracranial aneurysms and identified 58 giant aneurysms, yielding an incidence of 4.1%.11 Based on data from three autopsy studies and six large clinical series, the aggregate incidence of giant intracranial aneurysms is 5.2%13 (Table 22–1).
Age and gender
GIAs more commonly occur in females and most commonly come to clinical attention during the fifth to sixth decade of life. In a review of the world literature, Fox identified 693 giant aneurysms of which 60% occurred in females. This review correlates with known anatomical gender distributions with the proximal internal carotid artery location in 73% of females versus 52% for the anterior communicating complex and basilar apex.7 Anson reviewed 14 series with 754 GIAs identified, finding 461 females and 295 males, yielding a 1.56:1 ratio.27 In Anson’s review, ages ranged from 6 months to 76 years with 67% of patients presenting between ages 40 and 70. This distribution parallels that of Fox’s review of the world literature, where the median age of presentation was in the sixth decade of life.
Giant fusiform and dolichoectatic aneurysms probably present at a slightly younger age. Reviews from three different institutions found a mean age of presentation to be 38, 43, and 49 years with a male:female ratio of 1.5:1.8–10
Intracranial aneurysms are rare in children, with only 1% to 2% of all aneurysms occurring in the pediatric population. Of these, however, GIAs constitute a much higher proportion, are more common in males, and are less often present with hemorrhage. Ferrante reviewed 72 cases of pediatric aneurysms and found that 27% were giant and 50% were 1 to 2.5 cm in diameter.11
Location
Saccular GIAs have a similar anatomic predilection as their smaller counterparts, while the distribution is different. Similarly, the internal carotid artery (ICA) is the most common site, although GIAs occur proportionately more frequently in the vertebrobasilar system and less frequently in the anterior communicating artery (ACOM) region. Drake and Peerless reported 641 GIAs treated between 1965 and 1992 in the following locations: vertebrobasilar, 390; ICA, 178; middle cerebral artery (MCA), 64; and anterior cerebral artery (ACA), 19.12 While their data does reflect a referral bias, it supports the general trend that certain intracranial arterial segments are more vulnerable to this pathology as seen throughout the literature. Giant fusiform and dolichoectatic aneurysms have a predilection for the vertebrobasilar circulation and the MCA. These lesions, frequently referred to as “serpentine aneurysms,” are often massive, partially thrombosed lesions through which runs a serpentine channel.13
Natural history
The natural history of GIAs is dependent upon several factors, including the location of the aneurysm with respect to the subarachnoid space, the pathological form of the aneurysm (saccular vs. fusiform), the specific anatomic location, and the presence or absence of laminated thrombus and or atherosclerotic plaque within the fundus and neck of the aneurysm.14 In Morley and Barr’s original series of 28 patients, 17 had intradural aneurysms, and only four of those underwent a direct operation on their aneurysm. Of the five patients who received no definitive therapy, four died. Of the five patients with intradural aneurysms who had common carotid ligation, three died and one was disabled. They concluded that “direct surgical attack on extracavernous giant aneurysms is seldom possible or successful except in the case of MCA aneurysms.”
Peerless et al. observed 31 patients with giant intracranial aneurysms (25 saccular and six fusiform). Sixty-eight percent of patients with saccular aneurysms were dead at 2 years and 85% were dead at 5 years. If patients presenting with subarachnoid hemorrhage were excluded, the 2-year mortality rate was 62%. Only four patients who were all disabled were alive at 5 years. Of the six patients with fusiform aneurysms, four were dead at 2 years, one died at 3.5 years after diagnosis, and one remained disabled.15 Ljunggren et al. demonstrated a mortality or severe morbidity rate of 80% within 5 years in patients with untreated symptomatic GIAs.16 Hamburger et al. reported on 58 patients with GIA and showed that 7% of patients presenting without SAH died, compared with 29% of patients presenting with SAH. Only 18% of the later group were discharged home as independent compared with 50% of the former group.17 The Italian Cooperative Study on Giant Intracranial Aneurysms found that extensive or thick cisternal deposition of blood was associated with a significantly higher mortality rate than with thin or absent depositions.18
[/not-level-membership-for-neurosurgery-category]