39
Fetal haemolytic disease
Pathophysiology of haemolytic disease
Aetiology and predisposing factors
Prevention of haemolytic disease
Introduction
Haemolytic disease is likely to occur when maternal antibodies develop against fetal red blood cells. Red cells not infrequently cross from the fetus to the mother, either antenatally or at some intrapartum event and, if they are antigenically different from the mother’s red cells, there may be a maternal immune response with antibody production. IgG antibodies may cross in the opposite direction, back to the fetus, leading to haemolysis, anaemia, high-output cardiac failure and fetal death. There are numerous known red cell antigens but the rhesus D antigen accounts for approximately 85% of haemolytic disease.
Maternal rhesus alloimmunization exemplifies the achievements of systematic scientific and clinical research. In just 40 years, it has been possible to unravel the pathophysiology, devise useful treatments and introduce an effective means to prevent a condition which had previously caused extensive fetal morbidity and mortality. Among populations with access to an anti-D prophylaxis programme, the fully developed clinical condition of haemolytic disease of the newborn is rare.
The blood group system
Blood groups are determined by antigens on the erythrocyte cell wall. In the ABO system, the letter O is used to refer to those who lack both the A and B antigens. If the mother is group O and the fetus has paternally inherited the A or B antigen, the mother may, if exposed, develop antibodies to these fetal cells. In practice these antibodies rarely cause significant haemolytic disease and no antenatal investigations are warranted.
The next most important system is the rhesus system, which comprises at least 40 antigens, the most important of which are C, D and E. C and E have immunologically distinct isoforms which are designated ‘c’ and ‘e’, but it seems unlikely that a ‘d’ isoform exists. If there is a ‘d’ antigen, it seems to have little if any immunogenic potential and the notation ‘d’ is used to indicate the absence of ‘D’. A parent contributes one or other antigen (e.g. C or c) to the offspring from each of these three alphabetically designated pairs. An individual can therefore be homozygous or heterozygous for any of the six (e.g. Cde/cDE, cde/cdE). Those who carry the D antigen, which is inherited as an autosomal dominant, are referred to as rhesus D positive whether in the homozygous or heterozygous form.
Any of the rhesus antigens are capable of stimulating antibody formation but the D antigen is by far the most immunogenic, followed by c and E. If a rhesus negative mother has a rhesus positive baby and is at some stage sensitized to the baby’s red cells, there is a chance of anti-D antibodies developing against the fetal cells. These antibodies may cross back to the fetus and lead to fetal haemolytic anaemia.
There are other significant antigens in addition to the ABO and rhesus systems. Many of these (e.g. Ce, Fya, Jka, Cw) are poorly developed on the red cell surface and usually stimulate only low levels of antibody production, often of the IgM category (which does not cross the placenta). Some, however, will cause significant haemolytic disease.
One notable exception is the anti-Kell antibody, which has the potential to cause significant fetal anaemia and along with anti-RhD, and anti-Rhc are the most commonly reported antibodies in cases requiring intrauterine transfusion. Kell alloimmunization differs from RhD alloimmunization in that the anaemia is not solely due to haemolysis – erythroid suppression has an important role. In addition, past obstetric history cannot be relied on to predict the severity of disease in the current or subsequent pregnancies. Fortunately, the incidence of Kell alloimmunization is low (0.1–0.2%). Since only 9% of the Caucasian population is Kell-positive, the majority being heterozygous, the number of cases of haemolytic disease of the newborn is small.
Pathophysiology of haemolytic disease
Exposure to a foreign antigen leads to an antigen-specific antibody response, initially of IgM antibodies, which do not cross the placenta. On subsequent exposure, for example in a second pregnancy, the already primed Β cells produce a much larger response, this time of IgG antibodies, which do cross the placenta. In the fetal circulation, an antibody–antigen complex is formed on the red cell membrane, which provokes phagocytosis of the cell by the reticuloendothelial system and results in fetal haemolysis. This will lead to anaemia unless there is sufficient compensatory haemopoiesis from the marrow, spleen and liver.
Increasing anaemia causes progressive fetal hypoxia and acidosis leading to hepatic and cardiac dysfunction. Generalized oedema of skin develops, as well as ascites, pericardial and pleural effusions. This syndrome is known as ‘immune hydrops fetalis’ and it is potentially fatal.
Red cell haemolysis results in increased production of bilirubin, most of which passes across the placenta to the mother and is cleared by the maternal system. The fetus therefore does not become jaundiced antenatally, but after delivery its own liver is unable to metabolize bilirubin sufficiently quickly and the neonatal bilirubin level rises (Fig. 39.1). If untreated, the bilirubin can rise to levels, which endanger the nervous system and bilirubin deposition in the basal ganglia leads to a condition known as ‘kernicterus’.
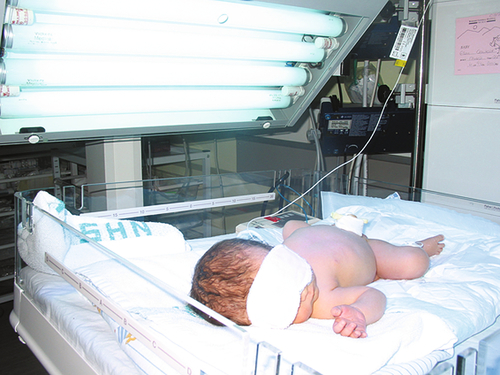
Fig. 39.1Baby delivered at 37 weeks because of high anti-D levels.
The bilirubin level rose steeply in the first 24 h but, with phototherapy, exchange transfusion was avoided.
Although most fetal bilirubin is readily cleared, some also passes into the fetal urine and then into the amniotic fluid. The level of amniotic bilirubin is therefore an indicator of the severity of the haemolysis.
Incidence
The incidence varies widely. Before the availability of anti-D prophylaxis, rhesus haemolytic disease was common in populations where there was a high prevalence of Rh(D)-negative individuals and where high parity caused an accumulation of alloimmunized women. In the UK, 17% of the population is Rh(D)-negative; 55% of Caucasian males are heterozygous for the RhD antigen. Assuming random mating without intervention, around two-thirds of Rh(D)-negative mothers would be expected to carry a rhesus-positive fetus. Approximately 10% of pregnant women are therefore at risk of developing anti-D antibodies.
Since the effective use of prophylactic anti-D, the perinatal mortality from haemolytic disease has fallen from around 46/100 000 to 1.9/100 000. Newly sensitized cases are detected at a rate of approximately 1/1000 maternities.
Aetiology and predisposing factors
Transfer of fetal erythrocytes to the maternal circulation during pregnancy (fetomaternal haemorrhage) may occur without any obvious predisposing event, and about 75% of women may be found to have fetal red cells circulating at some stage during the pregnancy or delivery. Fetomaternal haemorrhage is more likely, however, with disruption of the placental bed and this may occur with:
![]() miscarriage and ectopic pregnancy
miscarriage and ectopic pregnancy
![]() invasive intrauterine procedures, e.g. amniocentesis, CVS, fetal shunts
invasive intrauterine procedures, e.g. amniocentesis, CVS, fetal shunts
![]() external cephalic version
external cephalic version
![]() abdominal trauma
abdominal trauma
![]() antepartum haemorrhage
antepartum haemorrhage
![]() labour and delivery, particularly with delivery of the placenta.
labour and delivery, particularly with delivery of the placenta.
An immune response may, but does not inevitably, follow fetomaternal haemorrhage. The response depends in part on the volume of blood, its antigenic potential, and on the maternal responsiveness. ABO incompatibility between mother and fetus may paradoxically offer some protection, as the transfused cells are likely to be haemolysed by circulating maternal antibodies, reducing the risk of immunization. This observation illustrates the mechanism for the use of prophylactic anti-D immunoglobulin in that exogenous anti-D is used to bind and lyse any rhesus-positive fetal red cells that reach the maternal circulation.
Prevention of haemolytic disease
Anti-D prophylaxis
The most effective preventive measure is the use of intramuscular anti-D to provide passive immunization of non-sensitized women around the time of exposure. A rhesus negative mother who has a potentially sensitizing event (see Box 39.1) before 20 weeks’ gestation should be given 250 IU of anti-D as soon as possible after the event, and certainly within 72 h if possible. At more than 20 weeks, the dose is 500 IU. At delivery a sample of fetal cord blood should be rhesus grouped and, if positive, a film made of the mother’s blood for Kleihauer testing. The Kleihauer test estimates the volume of fetomaternal transfusion and allows an appropriate dose of anti-D to be calculated.
Immunization can occur as a result of silent fetomaternal haemorrhage, therefore prophylaxis is likely to be more effective if anti-D is given to all non-sensitized rhesus-negative mothers routinely in the third trimester (either 500 IU at 28 and 34 weeks, or a single larger dose early in the third trimester). This is standard practice in most areas.
Surprisingly, there is some evidence of benefit from anti-D prophylaxis even for women already mildly sensitized, in that subsequent children appear less severely affected by haemolytic disease than would otherwise have been expected.
Prediction of at-risk pregnancies
Routine maternal screening
All pregnant women at their first visit have serum sent for ABO and Rh(D) grouping with screening for irregular antibodies. The maternal serum level of any antibody discovered (usually anti-D) is used as an initial screening test for further action. There are regional variations, but an example of when to check for antibody levels is shown in Table 39.1.
Table 39.1
Possible screening programme for antibodies in haemolytic disease of the newborn
| ABO + RhD group and antibody screen | At 10–16 weeks | |
| All pregnant women (whether D +ve or D −ve) | RhD group and antibody screen | At 28–36 weeks |
| Patients identified with alloantibodies Anti-D, c or Kell related | Antibody screen ± titre and quantification (D,c) | At least monthly to 28 weeks, then 2-weekly to term |
| Other antibodies | Antibody screen ± titre | At 28–36 weeks, thereafter, depending on the titre |
Having identified an irregular antibody in the maternal circulation, the next stage is to assess the likely rhesus status of the baby by establishing the genotype of the putative father and remembering that the D antigen is inherited as an autosomal dominant. If the father is found to be d/d it is likely that anti-D antibodies in a rhesus-negative woman have developed from exposure to some other source of incompatible red cells, for example a previous blood transfusion or the fetus of a previous partner. Assuming confident paternity, the fetus will be unaffected and further specific action is unnecessary. If the father is homozygous for the D antigen, it follows that the fetus will also be rhesus D positive and will be at risk of haemolytic disease. Where the father is believed to be D/d, half of his offspring will be rhesus positive. In this situation it is important to establish the fetal blood group to determine whether or not the pregnancy is at risk. This can be done non-invasively by examining cell-free fetal DNA present in a maternal blood sample. Accuracies of 100% are reported for RhD genotyping. DNA amplification of amniotic fluid should only be performed for fetal genotyping if the patient is already undergoing amniocentesis for other indications.
Clinical significance of the antibody
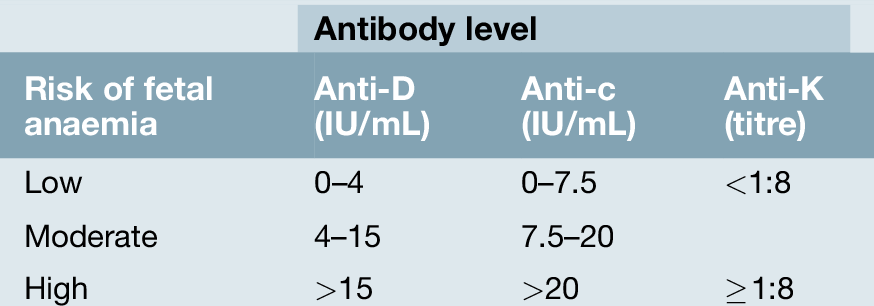
Red cell antigens vary in their likelihood of stimulating an immune response, with the D antigen being the most immunogenic. Apart from c and Kell most of the other antigens are less likely to lead to significant clinical problems and are not discussed further in this section. The D antibody titre in maternal serum and an actual quantification can be measured. The level is recognized to correlate well with disease severity. Severe disease is rare if the maternal antibody level is < 4 IU/mL, and additional specific intervention is probably not required. The risk of significant problems is only moderate between 4 and 15 IU/mL but above 15 IU/mL there is a risk of severe anaemia in 50% of fetuses. These higher levels call for further investigation, described below, and the management of these relatively uncommon cases should be centralized within specialized units. A sudden rise in levels, rather than a particular absolute level, is also likely to be significant.
Fetal assessment and therapy
Clinical symptoms and signs of fetal haemolytic anaemia occur late, are easily missed, and are of little help in the management. In advanced disease fetal movements are reduced or even absent, and there may be fetal growth restriction. In severe cases, there may be fetal hydrops with signs of ascites, pericardial and pleural effusions, and oedema of the skin. Polyhydramnios, which is associated with fetal hydrops, may also be detected. Cardiotocography may reveal an unreactive pattern or even decelerations, but again only in advanced disease. A sinusoidal fetal heart pattern is thought to be fairly specific for severe anaemia. All of the above represent the ‘end-stage’ of the process of progressive fetal anaemia. It is critical in the management of known alloimmunized pregnancies to detect the fetus that will benefit from intrauterine therapy before the fetal condition is seriously compromised. A rise in antibody titre or quantification should prompt non-invasive monitoring as described below.
Non-invasive testing
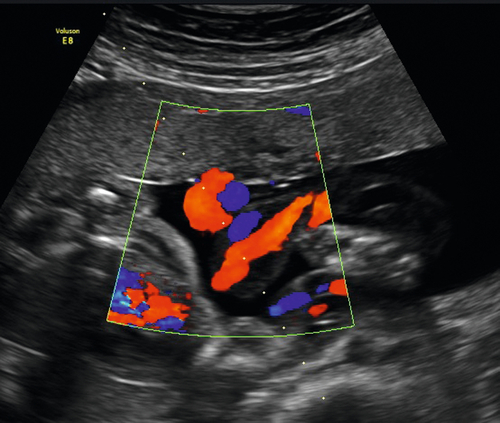
Historically, serial amniocentesis and measurement of amniotic fluid bilirubin levels was the method of assessing at-risk pregnancies. However, this has been superseded by non-invasive testing utilizing fetal middle cerebral artery (MCA) Doppler studies (Fig. 39.2). A hyperkinetic circulation correlates very well with the degree of fetal anaemia, and can be used to predict the requirement for further therapy. Table 39.2 indicates the level for different antibodies at which there is a moderate or severe risk of anaemia, and once these levels are reached MCA Doppler monitoring should be commenced. The peak systolic velocity (PSV) of the MCA is measured, and, if > 1.5 multiples of the median for gestational age, is predictive of moderate to severe fetal anaemia in 100% of cases for a false positive rate of 12%. The test is reliable up to 34 weeks’ gestation. Once moderate to severe anaemia is predicted, intrauterine therapy should commence. Non-invasive monitoring with MCA Doppler avoids the procedure-related loss rate associated with amniocentesis, and exacerbation of the degree of sensitization that results from invasive procedures.
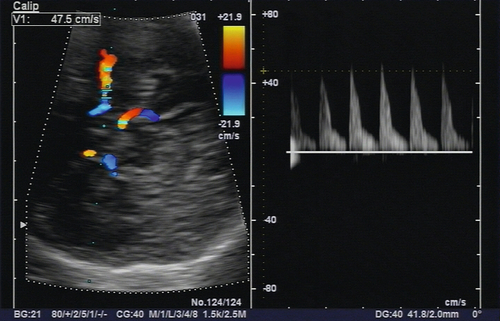
This non-invasive assessment can be used to determine the need for intrauterine transfusion.
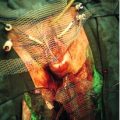
Fetal blood sampling and intrauterine transfusion
Access to the fetal circulation is achieved by inserting a needle into the baby’s cord, ideally at its point of placental insertion (Fig. 39.3) or alternatively, the intrahepatic portion of the umbilical vein. This enables an immediate haemoglobin (or haematocrit) estimation to be made, and facilitates simultaneous intrauterine transfusion (IUT) of blood. Group O rhesus-negative blood is cross-matched to the mother’s own serum prior to the procedure and, if the haemoglobin/haematocrit is low, a calculated volume can be transfused during the same sampling procedure. IUT carries the risk of cord haematoma, fetal bradycardia, intrauterine death, and further sensitization of the mother to fetal red cell antigens. A procedure-related loss rate in the region of 1% is quoted. It is likely that multiple IUTs will be required owing to the relentlessly progressive anaemia that occurs in alloimmunized pregnancies.
If an intravascular transfusion is technically too difficult, for example at very early gestations (< 18 weeks), it is possible to inject the red cells directly into the peritoneal cavity, from which they are subsequently absorbed by the fetal lymphatic system. This results in a slower increase in fetal haematocrit than direct intravascular transfusion.
Additional measures
In severe early cases, when hydropic change occurs before fetal transfusion is technically possible, repeated maternal plasma exchange may reduce the levels of maternal antibody. The technique requires special equipment to separate red cells from plasma and is both time-consuming and expensive. Maternal immunosuppression has also been tried in very severe cases.
Delivery
All babies with haemolytic disease should be delivered in a specialist unit with full neonatal intensive care facilities. If ultrasound assessment is normal and the antibody level relatively low, a conservative approach with delivery at term is appropriate. If premature delivery is anticipated, maternal corticosteroid therapy is warranted. Babies in whom mild anaemia is suspected, or who have been successfully treated with IUT, can be delivered vaginally, unless there are other obstetric indications for caesarean section. For cases managed with IUT, labour is induced at around 35 weeks’ gestation. If a hydropic fetus requires delivery, this should be by caesarean section. Experienced paediatric attendance at delivery is essential and cord blood must be collected for assessment of haemoglobin, platelets, blood grouping, bilirubin and direct Coombs’ testing. The neonate may require intensive support with measures to control anaemia, hyperbilirubinaemia and any associated cardiorespiratory problems.
Prognosis
For mildly affected fetuses in whom intrauterine therapy is unnecessary, the outlook, in experienced units, is excellent. Survival rates for non-hydropic fetuses undergoing IUT are ≥ 90%, compared with approximately 75% if hydrops is present.
Long-term sequelae
Early reports suggested serious neurological impairments, including cerebral palsy, abnormal development and hearing problems, especially in those children who were transfused in utero. Recent experience is very much more reassuring and suggests there are few, if any, additional risks beyond the well-recognized hazards of prematurity.