CHAPTER 56 Evaluation of Patients for Epilepsy Surgery
Up to 40% of epilepsy patients have medication-resistant or medically intractable seizures.1 As many as half of these patients may be candidates for curative or palliative epilepsy surgery. After successful epilepsy surgery, quality of life has been shown to improve significantly.2 The degree of success of epilepsy surgery depends largely on careful presurgical evaluation for identification and selection of the most appropriate candidates. The evaluation usually takes place in centers with a dedicated multidisciplinary team of neurologists and neurosurgeons experienced in treating patients with epilepsy, supported by electroencephalographers, neuroradiologists, neuropsychologists, psychiatrists, and paramedical staff. The aim of this chapter is to review the key elements of the presurgical evaluation and to outline some of the principles used in the decision-making process for identification of appropriate candidates for epilepsy surgery.
Initial Considerations in Choosing a Candidate for Presurgical Evaluation
We apply three fundamental criteria to identify candidates for epilepsy surgery:
In evaluating patients according to these three overarching criteria, it is important to note that intractable epilepsy is a progressive disorder that is medically, physically, and socially disabling.3 Accidental injuries as a result of seizures, psychiatric morbidity, loss of independence attributable, in part, to the inability to drive, and chronic medication toxicity all have major impacts on the quality of life of affected individuals. Recognition of this situation has gradually led to earlier consideration of epilepsy surgery, although in many series the duration of refractory epilepsy before surgery still exceeds 10 to 15 years.4
Epilepsy surgery may be proposed as a curative treatment or as a palliative treatment. Curative procedures, aimed at focal epilepsy, are almost always resective and require clear and complete identification of the epileptogenic zone (EZ). The majority of epilepsy resections target the anterior temporal lobe, including the mesial structures, and are associated with a high rate of remission. By contrast, extratemporal foci, most commonly frontal, may present additional difficulties because the EZ may be difficult to fully delineate and may involve eloquent cortex. With high-resolution neuroimaging techniques and dedicated imaging protocols, extratemporal lesions such as small heterotopias, cavernous angiomas, and focal cortical dysplasias can be visualized in up to 50% of patients, thereby dramatically improving their chance for success after epilepsy surgery. Rates of long-term freedom from seizures after resection of extratemporal lesions have improved but may still be lower than those for temporal lobe resection.5,6 In a recent meta-analysis, the median proportion of patients seizure free was 66% after temporal lobe resection, 46% after occipital or parietal resection, and 27% with frontal lobe resection.7
Palliative techniques include disconnection procedures and multiple subpial transections. Corpus callosotomy, typically performed as a two-stage procedure, may be helpful for patients with secondary generalized epilepsy syndromes and drop attacks.8 The aim of this procedure is to primarily palliate patients with atonic seizures and improve their quality of life. This procedure may offer up to 35% freedom from the most disabling seizures.7 Multiple subpial transections are thought to predominantly sever horizontal connections in the cortex while preserving columnar organization and efferent pathways.9 This technique may be used in isolation for epileptic foci lying exclusively in eloquent cortex or as an adjunct along with partial resection to approach EZs that cannot be fully removed.
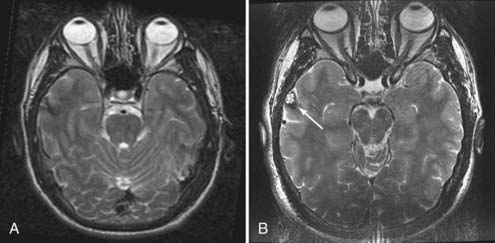
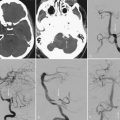
Evaluation of patients for consideration of epilepsy surgery includes a careful clinical history and physical examination, neuroimaging, and electroencephalographic (EEG) monitoring, often supplemented with detailed neuropsychological testing and psychiatric assessment when appropriate. The goal is to assess the concordance of multiple streams of data with respect to the probable EZ. Although the precise steps in the investigation vary somewhat from center to center, there is general consensus on the approach. Investigations are typically considered in phases. In general, phase I includes noninvasive recording, whereas phase II involves invasive intracranial EEG monitoring. Importantly, the investigation is an iterative process of hypothesis testing in which the results from each modality inform the interrogation of others. For example, identification of high-amplitude focal spikes on an EEG recording may prompt renewed scrutiny of magnetic resonance imaging (MRI) scans in the suspect area, thereby revealing a subtle developmental abnormality or vascular lesion (Fig. 56-1).
Phase I—Noninvasive Investigations
Routine Electroencephalographic Recording
Although the value of routine outpatient EEG studies in the decision-making process for consideration of epilepsy surgery is limited in comparison to that of other tests such as prolonged video-EEG monitoring and neuroimaging, its usefulness in the diagnosis of epilepsy and elucidation of the underlying syndrome is invaluable.10 Routine EEG findings are positive in 50% to 60% of patients with epilepsy, and the yield is increased by repeated or prolonged recordings that sample drowsiness and sleep. Even though focal slowing may signify an underlying structural abnormality, the major utility of routine EEG recordings is in the identification of interictal epileptiform discharges (IEDs) (sharp waves, spikes, or spike and wave complexes).11 Routine EEG studies aim to answer certain key questions: (1) Are there any IEDs present, and if so, how frequent are they? (2) Are the IEDs diagnostic of an idiopathic generalized syndrome that would render the patient inappropriate for resective epilepsy surgery? (3) If not, are the IEDs confined to one hemisphere or are they bilateral? (4) If the IEDs are unilateral, are they confined to one area of the hemisphere, such as the temporal region, or are they multifocal? The ideal surgical candidate would typically have a single population of well-localized IEDs, thus supporting the presence of only one EZ.
Continuous Video-Electroencephalographic Recording
Continuous computer-assisted video-EEG monitoring has become the mainstay of localization of the zone of epileptogenesis. The goals of video-EEG recording are threefold: (1) to further characterize the interictal EEG recording; (2) to detect, characterize, and quantify the patient’s habitual seizures12; and (3) to acquire physiologic data regarding seizure localization that can be correlated and compared with anatomic data obtained from neuroimaging studies. These investigations typically occur over a 5- to 7-day inpatient hospital stay (for adults) but are obviously dependent on seizure frequency and last as long as required to capture a satisfactory number of seizures.13,14 Characterizing the patient’s seizures is crucial because it allows (1) correlation of the ictal behavior with the electrographic discharge, (2) establishment of whether a patient has more than one seizure type, and (3) lateralization (which hemisphere) and localization (which area of the hemisphere) of the onset of seizures and therefore identification of the EZ. Typically, activating procedures such as sleep deprivation are used in addition to reduction or cessation of antiepileptic drugs in an attempt to increase the likelihood of seizure occurrence. In patients with nonlesional mesial temporal epilepsy, statistical analysis indicates that five concordant seizures will yield a 95% chance that the patient’s epilepsy is well lateralized (i.e., there is less than a 5% chance that five events would arise from the same side by chance alone).15 In addition to lateralization and localization, video-EEG recording of clinical seizures allows assessment of the temporal relationship between behavioral and electrical seizure onset. Prolonged delay before the first appearance of ictal EEG discharge should raise suspicion that the scalp recording may represent propagated activity from a remote site of onset.
Imaging of the Epileptogenic Zone
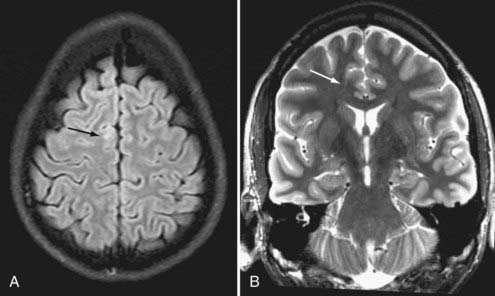
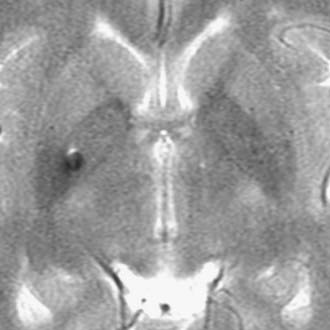
Brain MRI is the neuroimaging modality of choice for the presurgical evaluation of patients for epilepsy surgery. Hippocampal sclerosis, the most common pathology associated with temporal lobe epilepsy, is diagnosed on MRI by identification of all or some of the elements of volume loss, increased T2 signal, and loss of normal internal architecture in the affected hippocampus. Although fluid-attenuated inversion recovery (FLAIR) imaging makes signal abnormalities most obvious, true T2-weighted fast spin echo (FSE) sequences provide optimal signal-to-noise ratios for the examination of volume and architecture. In some centers, quantitative image analysis techniques may be used to calculate the volume of the hippocampi to detect subtle asymmetries16; however, useful deployment of these tools is labor and resource intensive and requires a high level of technical skill and experience. In addition to hippocampal sclerosis, specific MRI epilepsy protocols involving T1-, T2-, and susceptibility-weighted sequences are used to identify congenital and acquired epileptogenic lesions, most commonly low-grade gliomas, dysembryoplastic neuroepithelial tumors, malformations of cortical development (MCDs), cavernous angiomas, and subtle posttraumatic encephalomalacia and ulegyria. Many MCDs are subtle and best appreciated by looking for blurring of the gray-white junction on high-resolution MRI (Fig. 56-2). Excellent anatomic definition of the gray-white junction is now possible with phased-array surface coils deployed in magnets with high field strength, now up to 7 T.17
Functional imaging with positron emission tomography (PET) and single-photon emission computed tomography (SPECT) offers information about ictal or interictal brain tissue metabolism. PET using [18F]fluorodeoxyglucose (FDG) typically shows regional hypometabolism around the EZ when the tracer is injected interictally. PET was therefore a very useful tool for lateralization in patients with temporal lobe epilepsy or for identification of extratemporal EZs, such as MCDs,18 in the era before the development of high-resolution MRI scanning.19 Recently, however, with the increasing anatomic definition offered by MRI, the number of patients with partial epilepsy and seemingly normal MRI findings has decreased. As a result, the value of PET in this respect has also decreased. Nevertheless, it remains a useful tool for verification of MRI and EEG findings and in patients with normal findings on MRI. Other tracers such as [11C]-flumazenil show comparable results to FDG, but with a more restricted distribution of hypometabolism, which may be advantageous, particularly with extratemporal EZs.20
SPECT images are generated by photons emitted after the intravenous injection of a radioactive isotope such as technetium 99m-hexamethylpropyleneamine oxime (99mTc-HMPAO). The isotope is taken up by brain tissue in proportion to blood flow. Therefore, when injected within the first few seconds of a partial seizure, it shows increased uptake in the area of highest blood flow. This ictal SPECT is then compared with a baseline interictal study from the same patient to detect the differences in regional perfusion. This allows identification of the area hyperperfused at the time of seizure onset. The main disadvantage of ictal SPECT is that injection of the isotope has to be performed within the first few seconds after seizure onset, which is often impractical. The advantage, however, is that after injection, the window for scanning the patient is a few hours. SPECT can also be coregistered to MRI for better anatomic correlation of the EZ, and this can be used both for guidance of intracranial electrode placement and for subsequent surgical resection in a technique called subtraction ictal SPECT coregistered to MRI (SISCOM).21
Functional MRI (fMRI) images the blood oxygen level–dependent T2 signal (BOLD or T2*), a method whereby changes in metabolism and blood flow during an active process such as a simple motor or sensory task are detected. It has two roles in the identification of candidates for epilepsy surgery. fMRI is frequently used to map eloquent cortex during the surgical planning process. In this respect, fMRI aims to replace or limit the use of more invasive techniques such as the Wada test to identify language dominance and extraoperative or perioperative direct electrical brain stimulation to perform motor, sensory, and language mapping An emerging role for fMRI is in identification of the EZ. In this application, fMRI can be combined with simultaneous EEG recordings in an attempt to detect changes in blood flow and brain metabolism during an epileptic discharge.22 With improving technology, this could be a useful tool in the future for identifying the EZ, particularly in patients with normal findings on MRI.
Magnetoencephalography (MEG) is also emerging as an adjunctive, noninvasive tool for identification of the EZ. This technique measures the magnetic fields produced by electric currents in the brain, in this case epileptiform discharges. The fields generated are very weak, and hence very sensitive devices are required to detect them. Furthermore, the MEG scanner needs to be in a magnetically shielded room to eliminate outside magnetic signals. For these reasons, the device remains expensive and available only in research centers. Its main advantage is that although both MEG and EEG signals are generated by the same neurophysiologic processes, the MEG signal is not distorted by the skull and scalp. One additional advantage is that MEG is a good tool for noninvasive localization of the central sulcus with the use of somatosensory evoked magnetic fields.23 Overall, however, its value in surgical planning in relation to a seizure-free outcome has not yet been fully established.24
More recently, magnetic resonance spectroscopy (MRS) and diffusion tensor imaging (DTI) may offer additional clues to identification of the EZ in patients with MRI-negative partial epilepsy. MRS simultaneously detects and quantifies a number of brain metabolites and can therefore detect differences either between the two hippocampi, as in the case of hippocampal sclerosis, or between intralesional and perilesional tissue changes in MCD.25 In patients with hippocampal sclerosis, the typical finding in the epileptogenic hippocampus is a reduction in N– acetylaspartate and an elevation in choline-related compounds and creatine plus phosphocreatine (total creatine); the contralateral hippocampus may be normal or show a lesser degree of abnormality.26 Tractography, an important application of DTI, allows calculation of the direction of white matter tracts and may be helpful in determining the connections of the EZ to other brain areas, thus offering additional clues for identification of the EZ in patients with MRI-negative partial epilepsy.27,28
Neuropsychological Testing
Detailed neuropsychological testing is performed as part of the presurgical evaluation. Its goals are to identify epilepsy-related cognitive impairment, and it therefore complements EEG and neuroimaging in localization and lateralization of the EZ; prediction of potential postoperative deficits, particularly in memory domains, after resection of the EZ; and assessment of the patient’s mental reserve capacity.29 Comparison of the verbal and performance components of IQ testing may offer clear insight into the lateralization of dysfunctional cortex and hippocampus. Generally, a disparity of greater than 10 points between verbal IQ and performance IQ suggests significantly lateralized relative dysfunction in the dominant or nondominant hemisphere, respectively. The intracarotid sodium amobarbital procedure, also known as the Wada test, is used to determine which side of the brain is responsible for certain essential cognitive functions, namely, speech and memory. Further details related to the Wada test are provided in Chapter 57.
Surgical Decision-Making and Phase Ii (Intracranial) Investigations
Epidural Electrodes
Epidural electrodes are used infrequently and generally only for lateralization and approximate localization of the seizure onset.30 These electrodes are placed through bur holes in the skull with the electrode contact resting on the dura to provide a high-amplitude EEG signal without muscle or movement artifact. Because they do not penetrate the dura, the risk for infection is minor. These electrodes record only from the lateral convexity of the cerebral hemispheres and are limited in their spatial resolution.
Subdural Electrodes
Three different kinds of subdurals electrodes are used: grid, strip, and FO electrodes. The strips are single columns of electrodes that can be placed mainly over the lateral convexity or over the frontal or temporal lobes,31 but also in other less accessible regions such as the interhemispheric fissure. As with the other subdural electrodes, the strip is placed on the surface of the brain and records directly from the cortical surface. Although strip electrodes can be used for lateralization of seizure onset by being inserted through bur holes over, for example, the temporal lobes in each hemisphere, they are more commonly used for localization purposes in conjunction with grid electrodes, which offer additional coverage of adjacent areas of the cortical surface that abut the craniotomy site.
Grid electrodes are arrays of electrodes more than one column wide. Usually, rectangular grid arrays of 32 to 64 electrode contacts are used to maximize coverage over the craniotomy site. The site is determined from data gathered during the presurgical evaluation. Once placed, a grid is sutured to the dura to prevent motion. The grids are used to record from extratemporal regions and cannot be used to record from deeper cerebral structures such as the mesial temporal structures, which are often involved in medically refractory partial epilepsies. The main advantage of grid and strip electrodes is that they do not penetrate cerebral tissue and can record from a relatively wide area of the cortical surface. They can also be used for extraoperative cortical stimulation for mapping of specific areas of cortical function. Their disadvantages are that they have a small but significant rate of complications such as epidural and subdural bleeding, edema, or infection, often with an associated decrease in the level of consciousness and focal neurological signs.32
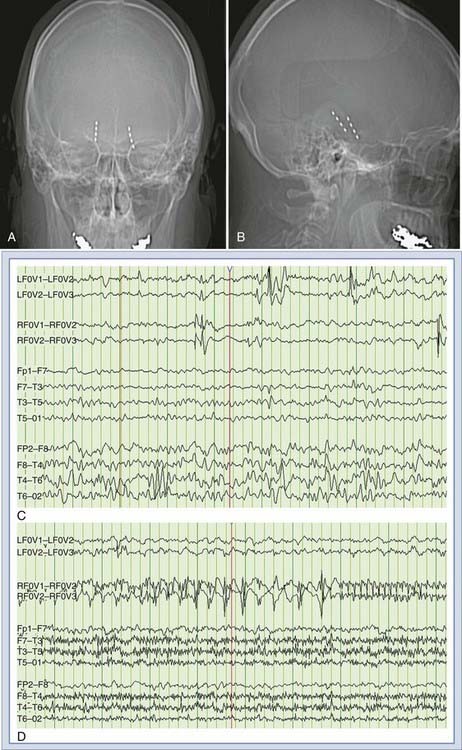
FO electrodes are intracranial electrodes introduced through the FO in the sphenoid bone, with the deepest contacts recording from the subdural space under the mesial temporal structures. FO electrodes frequently reveal striking epileptiform activity, including interictal epileptiform spikes and ictal discharges, which are not appreciated on associated scalp recordings (Fig. 56-3).
Implantation of FO electrodes is a minimally invasive procedure that in selected patients may provide significantly more information than scalp recordings and can preclude the need for more invasive depth electrodes.33 They have the additional advantage that they can be implanted “on the fly” in the course of a routine scalp investigation when required, without a lot of advanced planning. They are most useful in cases of suspected mesial temporal lobe epilepsy in which the phase I investigation failed to show unilateral seizure onsets. The advantage is that because the FO is a natural opening, implantation of FO electrodes does not involve disruption of the cranium or scalp. The disadvantage is that they record only from mesial temporal structures, thus offering biased information in cases in which the seizure onset is remote and therefore not captured by the FO electrodes but captured as it propagates through the mesial temporal structures. The complications related to FO implantation are relatively minor and transitory and include temporary facial pain and hypoesthesia in the trigeminal territory, temporomandibular joint dysfunction, and retromandibular hematoma.34
Intracerebral Depth Electrodes
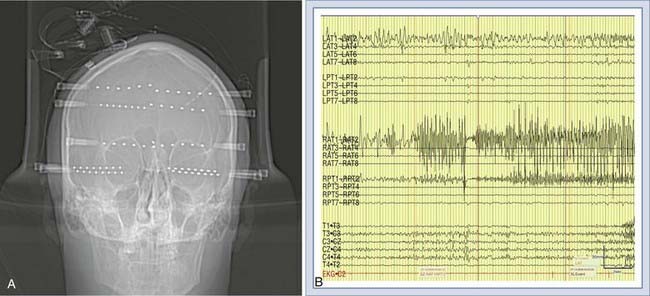
Intracerebral or depth electrodes are flexible electrodes with multiple contact points that are placed stereotactically via bur holes into deep brain structures, usually the hippocampus, amygdala, and orbitofrontal and cingulate regions (Fig. 56-4).
Common methods for placement include the orthogonal, occipital, and parasagittal approaches.35 Bilateral depth electrodes, like FO electrodes, are particularly useful for lateralization, especially in cases in which the scalp EEG recording is occluded by movement artifact at seizure onset. Unlike FO electrodes, however, multiple depth electrodes may be inserted and stereotactically targeted as clinically required. They are therefore better suited than FO electrodes for studying patients in whom the EZ may not be mesial temporal. They can also be used for localization in conjunction with ipsilateral subdural grids and strips, for example, in patients with suspected dual pathology in the same hemisphere involving a candidate neocortical region and the mesial temporal structures.36 The indications for depth electrode placement are expanding as neuroimaging becomes more advanced and more subcortical lesions, such as MCDs, are identified. The major complications of depth electrodes include hemorrhage and infection, although the rates are relatively lower when only bur holes rather than a craniotomy are used.
After implantation of the intracranial electrodes, antiepileptic drug doses are decreased and eventually stopped to increase the likelihood of recording seizures. The use of MRI-compatible intracranial electrodes allows accurate visualization of the anatomic location of the electrodes. Specialized software systems that can combine the patient’s segmented brain, postimplantation MRI scans, intracranial EEG findings, and results from cortical stimulation into one integrated environment of three-dimensional visualization have been developed to facilitate the planning of epilepsy surgery.37
Alarcon G, Kissani N, Dad M, et al. Lateralizing and localizing values of ictal onset recorded on the scalp: evidence from simultaneous recordings with intracranial foramen ovale electrodes. Epilepsia. 2001;42:1426-1437.
Barnett GH, Burgess RC, Awad IA, et al. Epidural peg electrodes for the presurgical evaluation of intractable epilepsy. Neurosurgery. 1990;27:113-115.
Blatt DR, Roper SN, Friedman WA. Invasive monitoring of limbic epilepsy using stereotactic depth and subdural strip electrodes: surgical technique. Surg Neurol. 1997;48:74-79.
Blum D. Prevalence of bilateral partial seizure foci and implications for electroencephalographic telemetry monitoring and epilepsy surgery. Electroencephalogr Clin Neurophysiol. 1994;91:329-336.
Cascino GD. Surgical treatment for extratemporal epilepsy. Curr Treat Options Neurol. 2004;6:257-262.
Cascino GD. Surgical treatment for epilepsy. Epilepsy Res. 2004;60:179-186.
Cascino GD. Video-EEG monitoring in adults. Epilepsia. 2002;43(suppl 3):80-93.
Chen Q, Lui S, Li CX, et al. MRI-negative refractory partial epilepsy: role for diffusion tensor imaging in high field MRI. Epilepsy Res. 2008.
Chugani HT, Shields WD, Shewmon DA, et al. Infantile spasms: I. PET identifies focal cortical dysgenesis in cryptogenic cases for surgical treatment. Ann Neurol. 1990;27:406-413.
Cross JH, Connelly A, Jackson GD, et al. Proton magnetic resonance spectroscopy in children with temporal lobe epilepsy. Ann Neurol. 1996;39:107-113.
Engel JJr, Kuhl DE, Phelps ME, et al. Comparative localization of epileptic foci in partial epilepsy by PCT and EEG. Ann Neurol. 1982;12:529-537.
Engel JJr, Wiebe S, French J, et al. Practice parameter: temporal lobe and localized neocortical resections for epilepsy: report of the Quality Standards Subcommittee of the American Academy of Neurology, in association with the American Epilepsy Society and the American Association of Neurological Surgeons. Neurology. 2003;60:538-547.
Fitzsimons M, Browne G, Kirker J, et al. An international survey of long-term video/EEG services. J Clin Neurophysiol. 2000;17:59-67.
Fountas KN, Smith JR. Subdural electrode–associated complications: a 20-year experience. Stereotact Funct Neurosurg. 2007;85:264-272.
Gotman J, Benar CG, Dubeau F. Combining EEG and FMRI in epilepsy: methodological challenges and clinical results. J Clin Neurophysiol. 2004;21:229-240.
Helmstaedter C. Neuropsychological aspects of epilepsy surgery. Epilepsy Behav. 2004;5(suppl 1):S45-S55.
Jeha LE, Najm I, Bingaman W, et al. Surgical outcome and prognostic factors of frontal lobe epilepsy surgery. Brain. 2007;130:574-584.
Knake S, Triantafyllou C, Wald LL, et al. 3T phased array MRI improves the presurgical evaluation in focal epilepsies: a prospective study. Neurology. 2005;65:1026-1031.
Kwan P, Brodie MJ. Early identification of refractory epilepsy. N Engl J Med. 2000;342:314-319.
Lau M, Yam D, Burneo JG. A systematic review on MEG and its use in the presurgical evaluation of localization-related epilepsy. Epilepsy Res. 2008;79:97-104.
Morrell F, Whisler WW, Bleck TP. Multiple subpial transection: a new approach to the surgical treatment of focal epilepsy. J Neurosurg. 1989;70:231-239.
Rahimi SY, Park YD, Witcher MR, et al. Corpus callosotomy for treatment of pediatric epilepsy in the modern era. Pediatr Neurosurg. 2007;43:202-208.
Ryvlin P, Bouvard S, Le Bars D, et al. Clinical utility of flumazenil-PET versus [18F]fluorodeoxyglucose-PET and MRI in refractory partial epilepsy. A prospective study in 100 patients. Brain. 1998;121:2067-2081.
Schuele SU, Luders HO. Intractable epilepsy: management and therapeutic alternatives. Lancet Neurol. 2008;7:514-524.
So EL. Integration of EEG, MRI, and SPECT in localizing the seizure focus for epilepsy surgery. Epilepsia. 2000;41(suppl 3):S48-S54.
Spencer SS, Spencer DD, Williamson PD, et al. Combined depth and subdural electrode investigation in uncontrolled epilepsy. Neurology. 1990;40:74-79.
Sutherling WW, Crandall PH, Darcey TM, et al. The magnetic and electric fields agree with intracranial localizations of somatosensory cortex. Neurology. 1988;38:1705-1714.
Tellez-Zenteno JF, Dhar R, Wiebe S. Long-term seizure outcomes following epilepsy surgery: a systematic review and meta-analysis. Brain. 2005;128:1188-1198.
Uijl SG, Leijten FS, Arends JB, et al. Decision-making in temporal lobe epilepsy surgery: the contribution of basic non-invasive tests. Seizure. 2008;17:364-373.
Van Paesschen W, Sisodiya S, Connelly A, et al. Quantitative hippocampal MRI and intractable temporal lobe epilepsy. Neurology. 1995;45:2233-2240.
Velasco TR, Sakamoto AC, Alexandre VJr, et al. Foramen ovale electrodes can identify a focal seizure onset when surface EEG fails in mesial temporal lobe epilepsy. Epilepsia. 2006;47:1300-1307.
Velis D, Plouin P, Gotman J, et al. for the ILAE DMC Subcommittee on Neurophysiology. Recommendations regarding the requirements and applications for long-term recordings in epilepsy. Epilepsia. 2007;48:379-384.
Wang Y, Agarwal R, Nguyen D, et al. Intracranial electrode visualization in invasive pre-surgical evaluation for epilepsy. Conf Proc IEEE Eng Med Biol Soc. 2005;1:952-955.
Widdess-Walsh P, Diehl B, Najm I. Neuroimaging of focal cortical dysplasia. J Neuroimaging. 2006;16:185-196.
Wiebe S, Blume WT, Girvin JP, et al. Effectiveness and Efficiency of Surgery for Temporal Lobe Epilepsy Study Group. A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med. 2001;345:311-318.
Woermann FG, McLean MA, Bartlett PA, et al. Quantitative short echo time proton magnetic resonance spectroscopic imaging study of malformations of cortical development causing epilepsy. Brain. 2001;124:427-436.
Wyler AR, Ojemann GA, Lettich E, et al. Subdural strip electrodes for localizing epileptogenic foci. J Neurosurg. 1984;60:1195-1200.
1 Kwan P, Brodie MJ. Early identification of refractory epilepsy. N Engl J Med. 2000;342:314-319.
2 Wiebe S, Blume WT, Girvin JP, et al. Effectiveness and Efficiency of Surgery for Temporal Lobe Epilepsy Study Group. A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med. 2001;345:311-318.
3 Cascino GD. Surgical treatment for epilepsy. Epilepsy Res. 2004;60:179-186.
4 Engel JJr, Wiebe S, French J, et al. Practice parameter: temporal lobe and localized neocortical resections for epilepsy: report of the Quality Standards Subcommittee of the American Academy of Neurology, in association with the American Epilepsy Society and the American Association of Neurological Surgeons. Neurology. 2003;60:538-547.
5 Tellez-Zenteno JF, Dhar R, Wiebe S. Long-term seizure outcomes following epilepsy surgery: a systematic review and meta-analysis. Brain. 2005;128:1188-1198.
6 Cascino GD. Surgical treatment for extratemporal epilepsy. Curr Treat Options Neurol. 2004;6:257-262.
7 Jeha LE, Najm I, Bingaman W, et al. Surgical outcome and prognostic factors of frontal lobe epilepsy surgery. Brain. 2007;130:574-584.
8 Rahimi SY, Park YD, Witcher MR, et al. Corpus callosotomy for treatment of pediatric epilepsy in the modern era. Pediatr Neurosurg. 2007;43:202-208.
9 Morrell F, Whisler WW, Bleck TP. Multiple subpial transection: a new approach to the surgical treatment of focal epilepsy. J Neurosurg. 1989;70:231-239.
10 Uijl SG, Leijten FS, Arends JB, et al. Decision-making in temporal lobe epilepsy surgery: the contribution of basic non-invasive tests. Seizure. 2008;17:364-373.
11 Schuele SU, Luders HO. Intractable epilepsy: management and therapeutic alternatives. Lancet Neurol. 2008;7:514-524.
12 Velis D, Plouin P, Gotman J, et al. for the ILAE DMC Subcommittee on Neurophysiology. Recommendations regarding the requirements and applications for long-term recordings in epilepsy. Epilepsia. 2007;48:379-384.
13 Cascino GD. Video-EEG monitoring in adults. Epilepsia. 2002;43(suppl 3):80-93.
14 Fitzsimons M, Browne G, Kirker J, et al. An international survey of long-term video/EEG services. J Clin Neurophysiol. 2000;17:59-67.
15 Blum D. Prevalence of bilateral partial seizure foci and implications for electroencephalographic telemetry monitoring and epilepsy surgery. Electroencephalogr Clin Neurophysiol. 1994;91:329-336.
16 Van Paesschen W, Sisodiya S, Connelly A, et al. Quantitative hippocampal MRI and intractable temporal lobe epilepsy. Neurology. 1995;45:2233-2240.
17 Knake S, Triantafyllou C, Wald LL, et al. 3T phased array MRI improves the presurgical evaluation in focal epilepsies: a prospective study. Neurology. 2005;65:1026-1031.
18 Chugani HT, Shields WD, Shewmon DA, et al. Infantile spasms: I. PET identifies focal cortical dysgenesis in cryptogenic cases for surgical treatment. Ann Neurol. 1990;27:406-413.
19 Engel JJr, Kuhl DE, Phelps ME, et al. Comparative localization of epileptic foci in partial epilepsy by PCT and EEG. Ann Neurol. 1982;12:529-537.
20 Ryvlin P, Bouvard S, Le Bars D, et al. Clinical utility of flumazenil-PET versus [18F]fluorodeoxyglucose-PET and MRI in refractory partial epilepsy. A prospective study in 100 patients. Brain. 1998;121:2067-2081.
21 So EL. Integration of EEG, MRI, and SPECT in localizing the seizure focus for epilepsy surgery. Epilepsia. 2000;41(suppl 3):S48-S54.
22 Gotman J, Benar CG, Dubeau F. Combining EEG and FMRI in epilepsy: methodological challenges and clinical results. J Clin Neurophysiol. 2004;21:229-240.
23 Sutherling WW, Crandall PH, Darcey TM, et al. The magnetic and electric fields agree with intracranial localizations of somatosensory cortex. Neurology. 1988;38:1705-1714.
24 Lau M, Yam D, Burneo JG. A systematic review on MEG and its use in the presurgical evaluation of localization-related epilepsy. Epilepsy Res. 2008;79:97-104.
25 Woermann FG, McLean MA, Bartlett PA, et al. Quantitative short echo time proton magnetic resonance spectroscopic imaging study of malformations of cortical development causing epilepsy. Brain. 2001;124:427-436.
26 Cross JH, Connelly A, Jackson GD, et al. Proton magnetic resonance spectroscopy in children with temporal lobe epilepsy. Ann Neurol. 1996;39:107-113.
27 Widdess-Walsh P, Diehl B, Najm I. Neuroimaging of focal cortical dysplasia. J Neuroimaging. 2006;16:185-196.
28 Chen Q, Lui S, Li CX, et al. MRI-negative refractory partial epilepsy: role for diffusion tensor imaging in high field MRI. Epilepsy Res. 2008;80:83-89.
29 Helmstaedter C. Neuropsychological aspects of epilepsy surgery. Epilepsy Behav. 2004;5(suppl 1):S45-S55.
30 Barnett GH, Burgess RC, Awad IA, et al. Epidural peg electrodes for the presurgical evaluation of intractable epilepsy. Neurosurgery. 1990;27:113-115.
31 Wyler AR, Ojemann GA, Lettich E, et al. Subdural strip electrodes for localizing epileptogenic foci. J Neurosurg. 1984;60:1195-1200.
32 Fountas KN, Smith JR. Subdural electrode–associated complications: a 20-year experience. Stereotact Funct Neurosurg. 2007;85:264-272.
33 Alarcon G, Kissani N, Dad M, et al. Lateralizing and localizing values of ictal onset recorded on the scalp: evidence from simultaneous recordings with intracranial foramen ovale electrodes. Epilepsia. 2001;42:1426-1437.
34 Velasco TR, Sakamoto AC, Alexandre VJr, et al. Foramen ovale electrodes can identify a focal seizure onset when surface EEG fails in mesial temporal lobe epilepsy. Epilepsia. 2006;47:1300-1307.
35 Blatt DR, Roper SN, Friedman WA. Invasive monitoring of limbic epilepsy using stereotactic depth and subdural strip electrodes: surgical technique. Surg Neurol. 1997;48:74-79.
36 Spencer SS, Spencer DD, Williamson PD, et al. Combined depth and subdural electrode investigation in uncontrolled epilepsy. Neurology. 1990;40:74-79.
37 Wang Y, Agarwal R, Nguyen D, et al. Intracranial electrode visualization in invasive pre-surgical evaluation for epilepsy. Conf Proc IEEE Eng Med Biol Soc. 2005;1:952-955.