Chapter 56 Evaluation and Management of Arrhythmias Associated with Congestive Heart Failure
Introduction
Both atrial and ventricular arrhythmias can not only lead to heart failure but can also occur as a consequence of heart failure. Arrhythmias commonly seen in heart failure include sinus tachycardia, atrial fibrillation (AF), premature ventricular contractions (PVCs), nonsustained ventricular tachycardia (NSVT), sustained ventricular tachycardia (VT), torsades de pointes (TdP), and ventricular fibrillation (VF) (Box 56-1).
Mechanisms
The mechanisms of arrhythmias in heart failure include re-entry, triggered activity, abnormal automaticity, and altered stretch (Box 56-2).
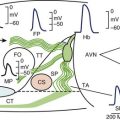
Re-entry is often seen in heart failure and may be related to conduction slowing caused by functional or anatomic barriers. The underlying mechanisms include reduced myocardial excitability and increases in resistance.1 Moreover, downregulation of the gap junction protein connexin 43 promotes cell-to-cell uncoupling, a milieu for re-entrant arrhythmia.2
Patients with heart failure often develop left ventricular hypertrophy, a compensatory mechanism for inadequate cardiac output. Left ventricular hypertrophy is associated with increased action potential duration that provides a potential for increased dispersion of repolarization leading to TdP or VF as a result of triggered activity, especially early afterdepolarization.3 Also of note, because of the downregulation of potassium (K+) currents and increased late sodium (Na+) current, the action potential duration is further prolonged, with a greater propensity for triggered activity.4 Increases in intracellular calcium (Ca2+) from enhanced sympathetic stimulation from the heart failure itself or from the use of digitalis may cause triggered arrhythmias by inducing delayed afterdepolarization.5,6
Abnormal automaticity stems from ischemia and is related to abnormal Ca2+ handling, leading to non–re-entrant arrhythmias.6
Stretch-activated channels may play an important role in arrhythmia genesis. This observation has been noted mostly in animal studies. Stretch affects both conduction and refractoriness. Any such alterations may predispose to arrhythmias in patients with heart failure. Electromechanical feedback can lead to electrical remodeling with changes in conduction and repolarization, creating heterogeneity and dispersion, with enhancement of atrial and ventricular arrhythmias.7,8
A reduction in the sinoatrial node If current (“funny” current) and its hyperpolarizing cyclic nucleotide (HCN) channel gene leads to sinus bradycardia from sinoatrial node dysfunction. In heart failure, however, increases may occur in atrial and ventricular tissue If currents that may lead to arrhythmias.9
Atrial Fibrillation
AF is quite commonly seen in patients with heart failure (30%). The tachycardia itself may predispose to heart failure but may occur as a result of heart failure. The risk of emboli is related to etiology and the CHADS 2 (Cardiac Failure, Hypertension, Age, Diabetes, Stroke [Doubled]) score. Valvular AF carries a higher risk of stroke. Compared with sinus rhythm, after adjustments for comorbidities, AF does not seem to have an independent mortality risk in heart failure.10 Mechanisms of AF in heart failure, which are complex and multi-factorial, include re-entry, triggered activity, automaticity, and activation of stretch receptors. In addition, atrial interstitial fibrosis, increased collagen synthesis, and altered connexin expression predispose to heterogeneities and electrical uncoupling and eventually AF. Of note, experimental heart failure in dogs has been shown to promote AF by causing fibrosis, interfering with local conduction, and not by altering atrial refractory period, refractoriness heterogeneity, or conduction velocity as seen with rapid atrial pacing.11
Ventricular Arrhythmias
PVCs are very common in patients with heart failure, and in those with more than 10 per hour, the incidence of NSVT is about 90%.12 In patients with prior myocardial infarction (MI), PVCs are associated with an increased risk of death, especially in those with left ventricular dysfunction. The presence of NSVT does not seem add any more risk over PVCs.13
Sustained VT and VF may account for 50% of all deaths in patients with heart failure and is often classified as sudden cardiac death (SCD). However, all SCDs may not be tachyarrhythmia related. Death from bradycardia or electromechnical dissociation is common, especially in severe heart failure.14,15 Bundle branch re-entry is a form of sustained VT that may be reproduced in the electrophysiology laboratory and is important to recognize, since this arrhythmia can be successfully treated with ablation (see below).
Evaluation
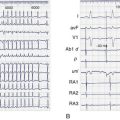
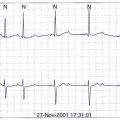
Numerous tests have been used to evaluate the excessive risks of patients with heart failure (Box 56-3). The electrocardiogram is extremely useful in determining etiologies and important prognostic factors. Sinus bradycardia in response to β-blocker therapy has been shown to be beneficial in patients with heart failure. However, sinus bradycardia may be profibrillatory in the atrium because of increased dispersion (atrial torsades) or in the ventricle, especially in the presence of class III (K+ channel blockers) antiarrhythmic drugs (see below).16 Wide QRS or bundle branch block is an independent risk factor for premature death, that is, independent of ejection fraction (EF).17,18 A fragmented QRS has been shown to be predictive of mortality and SCD in both ischemic and nonischemic cardiomyopathy. An acquired prolonged Q-T interval is also quite important and may be a harbinger of TdP, especially when associated with K+ channel blockers, bradycardia, female gender, and electrolyte imbalance.
T-wave alternans, that is, a beat-to-beat variation in T-wave morphology, although subtle, has been associated with potentially lethal arrhythmias. A more sensitive technique is microvolt T-wave alternans (MTWA). A positive test carries a poor prognosis compared with a negative test, as shown in Multicenter Automatic Defibrillator Implantation Trial II (MADIT II), in which patients with prior MI and left ventricular dysfunction were randomized to implantable cardioverter-defibrillator (ICD) or not.19 However, in the Sudden Cardiac Death in Heart Failure Trial (SCD-HeFT) substudy, a positive MTWA was not predictive of excess mortality in patients with heart failure.20 Of note, a wide QRS and the use of β-blockers may affect the results. It appears that MTWA testing should not be used to make decisions on implantation of an ICD.
Baroreflex sensitivity (BRS), a measure of autonomic nervous system activity that is independent of β-blocker usage, has been used to identify patients at high risk, especially those with previous MI. A low BRS is associated with a poor outcome. In the Autonomic Tone and Reflexes after Myocardial Infarction (ATRAMI) trial, a depressed BRS predicted cardiac mortality after MI.21
The role of the electrophysiology study (EPS) in nonischemic cardiomyopathy has not been established, and the chance of inducing a sustained ventricular arrhythmia is quite low.22 VT caused by bundle branch re-entry is sometimes seen in patients with nonischemic cardiomyopathy, and ablation of the right bundle, if electrophysiologically induced, is curative.23 Patients with ischemic cardiomyopathy and NSVT in whom sustained VT can be provoked at EPS carry a high risk of SCD compared with those in whom the arrhythmia is not inducible. However, because of a low EF, noninducibility still carries a substantial risk.24 Thus the role of EPS is limited. Left ventricular ejection fraction is the most powerful predictor of outcome and is used frequently to guide therapy, especially nonpharmacologic therapy.
Therapy
Box 56-4 provides a brief outline of the various treatment modalities for the patient with heart failure and left ventricular dysfunction.
Pharmacologic Therapy: Antiarrhythmics
β-Blockers have been consistently shown to reduce SCD and total mortality in patients with heart failure.25–27 This effect is independent of PVC suppression.28 β-Blockers are very effective in slowing the ventricular response in AF. Not all β-blockers are created equal, and those with intrinsic sympathomimetic activity may be of limited use or harmful. Noncardioselective β-blockers may be superior to cardioselective ones; in the Carvedilol or Metoprolol European Trial (COMET), carvedilol was found to be superior to metoprolol with regard to outcome in patients with heart failure. This could be explained by the low daily cardioselective dose of 89 mg of metoprolol. Of note, the incidence of AF was lower with carvedilol.
Class III drugs such as sotalol and dofetilide can suppress both supraventricular and ventricular arrhythmias and may be considered safe, although they have no beneficial effects on mortality. However, the risk of a proarrhythmic effect is high in patients with heart failure, so extreme caution must be exercised. Amiodarone is unique and is a very complex antiarrhythmic agent with the capability of blocking Na+, Ca2+, and K+ channels. Amiodarone is also an α- and β-blocker and is very effective in suppressing atrial and ventricular arrhythmias. Proarrhythmia is not often seen with this compound. However, it has many noncardiac adverse effects, so its use may be limited. It shows no benefit on survival in patients with prior MI, those with prior MI and left ventricular dysfunction, those with heart failure, and those with AF and heart failure.12,29–31 Dronedarone has a molecular structure similar to that of amiodarone but without the iodine.
In Antiarrhythmic Trial with Dronedarone in Heart Failure (ANDROMEDA), dronedarone was associated with increased mortality caused by worsening heart failure.32 Of note, however, in the recently published Placebo-Controlled, Double-Blind, Parallel Arm Trial to Assess the Efficacy of Dronedarone 400 mg bid for the Prevention of Cardiovascular Hospitalization or Death from Any Cause in Patients with Atrial Fibrillation/Atrial Flutter (ATHENA) trial, dronedarone was effective in reducing cardiovascular hospitalization or death.33
The Ca2+ channel blockers are of no benefit and may be harmful in patients with heart failure.34,35
Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers, in addition to β-blockers, are mandated in the treatment algorithm of patients with heart failure. These agents suppress both ventricular and supraventricular arrhythmias.36 They are thought to accomplish this effect by structural, functional, and electrical remodeling. Incidentally, two large prospective trials have shown that angiotensin receptor blockers have no benefit in patients with diastolic heart failure.37,38
The aldosterone receptor blockers aldactone and eplerenone have been shown to improve survival in patients with heart failure.39,40 Both drugs reduce the incidence of SCD and may be related to a decrease in the re-entrant mechanism because of a reduction of fibrosis.
Ranolazine is a very complex antianginal compound with complex antiarrhythmic activities. It can block not only the K+ and Ca2+ currents but also the late Na+ current, thereby decreasing proarrhythmia; it also has potential antiarrhythmic properties. At this time, no trials have been performed using this agent in patients with heart failure. However, ranolazine suppresses malignant ventricular arrhythmias, as shown in the Metabolic Efficiency with Ranolazine for Less Ischemia in Non–ST Segment Elevation Acute Coronary Syndrome–Thrombolysis in Myocardial Infarction (MERLIN-TIMI) trial.41
Numerous trials have shown that “statins” suppress the incidence of AF. This is especially true in the postoperative period, as shown in the Atorvastatin for Reduction of Myocardial Dysrhythmia After Cardiac Surgery (ARMYDA-3) trial, in which atorvastatin was shown to be superior to placebo.42 In retrospective analyses, statins also lower the recurrence of VT or VF in patients with ICDs.43,44
In the Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico Prevenzione (GISSI) trial, n-3 polyunsaturated fatty acids (PUFAs) have been shown to reduce SCD in patients with recent MI.45 In the recently published GISSI-HF trial, PUFA reduced all-cause mortality and cardiovascular admissions.46 Of note, PUFAs attenuate heart failure–associated atrial structural remodeling and AF promotion without changing atrial refractory periods and electrical remodeling.47
In patients at high risk for AF, warfarin is the drug of choice since the risk of embolization in these patients is unacceptably high. For patients not suitable for warfarin therapy, the combination of clopidogrel and aspirin has been shown to be superior to aspirin alone, in the recent publication of the Atrial Fibrillation Clopidogrel Trial With Irbesartan for Prevention of Vascular Events (ACTIVE) trial.48
Nonpharmacologic Therapy
Revascularization with coronary artery bypass grafting (CABG) has been shown to reduce SCD.49 It is unclear whether this is related to suppression of arrhythmia. Data on revascularization in patients with heart failure and arrhythmia suppression are not available. The ongoing Surgical Treatment of Ischemic Heart Failure (STICH) trial may provide some answers. In the CABG PATCH trial, patients with prior MI and left ventricular dysfunction were randomized to CABG or to CABG with ICD.50 The survival rates were the same in both groups, which suggests that revascularization possibly provides protection from arrhythmic death. Data on the effects of percutaneous coronary intervention on arrhythmias in patients with heart failure are not available.
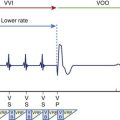
ICD therapy has consistently shown survival benefits in patients with heart failure and left ventricular dysfunction. This is related to a reduction in SCD, as a result of tachyarrhythmias.51,52 Of interest, SCD-HeFT did not show clear benefit in New York Heart Association class III heart failure but only in class II. In MADIT II, benefit was seen in patients with prior infarction and left ventricular dysfunction. A disconnect may exist between the degree of heart failure and benefit from ICD therapy. The SCD-HeFT substudy of AF did not show any survival benefit with ICD therapy. Moreover, in those with sinus rhythm at baseline, the ICD increased AF incidence.10
Cardiac resynchronization therapy (CRT) has been shown to reduce hospitalization and mortality in patients with heart failure.53,54 Responders to CRT have shown a significant reduction in ventricular arrhythmias and ICD shocks compared with nonresponders.55 The reduction in arrhythmia density is possibly related to reverse remodeling. A recent study on patients with predominantly class 2 congestive heart failure showed mortality reduction with CRT and ICD compared with ICD alone.56
Frequent PVCs may contribute to left ventricular dysfunction. Ablation of these isolated foci can abolish these PVCs with resolution of the cardiomypathy.57 Sustained monomorphic VT that recurs despite antiarrhythmic therapy can be successfully ablated. However, the 1-year mortality rate is still around 18%.58,59
Pulmonary vein isolation has been shown to have superior beneficial effects over AV node ablation in selected patients with AF and heart failure. Improvements were seen in the quality of life, 6-minute walk, and EF.60
Key References
Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure, for the Sudden Cardiac Death in Heart Failure Trial (SCD-HEFT) Investigators. N Engl J Med. 2005;352:225-237.
Bristow MR, Saxon LA, Boehmer J, et al. Cardiac-Resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure, for the Comparison of Medical Therapy, Pacing, and Defibrillation in Heart Failure (COMPANION) Investigators. N Engl J Med. 2004;350:2140-2150.
DiBiase L, Gasparini M, Lunati M, et al. Antiarrhythmic effect of reverse ventricular remodeling induced by cardiac resynchronization therapy. J Am Coll Cardiol. 2008;52:1442-1449.
Gold MR, Ip JH, Costantini O, et al. Role of microvolt T-Wave alternans in assessment of arrhythmia vulnerability among patients with heart failure and systolic dysfunction: Primary results from the T-Wave alternans sudden cardiac death in Heart Failure Trial Substudy. Circulation. 2008;118:2022-2028.
Hohnloser SH, Crijns HJ, Eickels M, et al. Effect of dronedarone on cardiovascular events in atrial fibrillation, for the ATHENA Investigators. N Eng J Med. 2009;360:668-678.
Kober L, Torp-Pedersen C, McMurray JJ, et al. Increased mortality after dronedarone therapy for severe heart failure, for the Dronedarone Study Group. N Eng J Med. 2008;358:2678-2687.
Luu M, Stevenson WG, Stevenson LW, et al. Diverse mechanism of unexpected cardiac arrest in advanced heart failure. Circulation. 1989;80:1675-1680.
Moss AJ, Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction, for the Multicenter Automatic Defibrillator Implantation Trial II Investigators. N Eng J Med. 2002;346:877-883.
Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure, The Randomized Aldactone Evaluation Study Investigators. N Eng J Med. 1999;341(10):709-717.
Poole-Wilson PA, Swedberg K, Cleland JGF, et al. Comparison of carvedilol and metoprolol on clinical outcomes in patients with chronic heart failure. Results of the Carvedilol or Metoprolol European Trial, for the COMET Investigators. Lancet. 2003;362:7-13.
Roy D, Talajic M, Nattel S, et al. Rhythm control versus rate control for atrial fibrillation and heart failure. N Engl J Med. 2008;358:2667-2677.
Singh SN, Fisher SG, Carson PE, Fletcher RD. Prevalence and significance of nonsustained ventricular tachycardia in patients with premature ventricular contractions and heart failure treated with vasodilator therapy. J Am Coll Cardiol. 1998;32:942-947.
Singh SN, Fletcher RD, Fisher SG, et al. Amiodarone in patients with congestive heart failure and asymptomatic ventricular arrhythmia. Survival Trial of Antiarrhythmic Therapy in Congestive Heart Failure. N Eng J Med. 1995;333(2):77-82.
Zabel M, Koller BS, Sachs F, Franz MR. Stretch-induced voltage changes in the isolated beating heart: Importance of the timing of stretch and implications for stretch-activated ion channels. Cardiovasc Res. 1996;32:120-130.
1 Howgwei J, Lyon AR, Akar FG. Arrhythmia mechanisms in the failing heart. Pacing Cardiol Electrophysiol. 2008;31:1048-1056.
2 Wiegerinck RF, Veen TA, Belterman CH, et al. Transmural dispersion of refractoriness and conduction velocity is associated with heterogeneously reduced connexin43 in a rabbit model of heart failure. Heart Rhythm. 2008;5:1178-1185.
3 Tomaselli GF, Beuckemann DF, Calkins HG, et al. Sudden cardiac death in heart failure: The role of abnormal repolarization. Circulation. 1994;90:2534-2539.
4 Tomaselli GF, Eduardo M. Electrophysiological remodeling in hypertrophy and heart failure. Cardiovasc Res. 1999;42:270-283.
5 Akar FG, Tomaselli GF. Ion channels as novel therapeutic targets in heart failure. Annu Med. 2005;37:44-54.
6 Lo R, Hsia HH. Ventricular arrhythmias in heart failure patients. Cardiol Clin. 2008;26:381-403.
7 Zabel M, Koller BS, Sachs F, Franz MR. Stretch-induced voltage changes in the isolated beating heart: Importance of the timing of stretch and implications for stretch-activated ion channels. Cardiovasc Res. 1996;32:120-130.
8 Ninio DM, Saint DA. The role of stretch-activated channels in atrial fibrillation and the impact of intracellular acidosis. Prog Biophys Mol Biol. 2008;97(2-3):401-416.
9 Nattel S, Maguy A, LeBouter S, Yeh YH. Arrhythmogenic ion-channel remodeling in the heart: Heart failure, myocardial infarction and atrial fibrillation. Physiol Rev. 2007;87:425-456.
10 Singh SN, Poole J, Anderson J, et al. Role of amiodarone or implantable cardioverter/defibrillator in patients with atrial fibrillation and heart failure, for the SCD-HeFT Investigators. Am Heart J. 2006;152:974.e7-974.e11.
11 Li D, Fareh S, Leung TK, Nattel S. Promotion of atrial fibrillation by heart failure in dogs: Atrial remodeling of a different sort. Circulation. 1999;100:87-95.
12 Singh SN, Fletcher RD, Fisher SG, et al. Amiodarone in patients with congestive heart failure and asymptomatic ventricular arrhythmia. Survival Trial of Antiarrhythmic Therapy in Congestive Heart Failure. N Engl J Med. 1995;333(2):77-82.
13 Singh SN, Fisher SG, Carson PE, Fletcher RD. Prevalence and significance of nonsustained ventricular tachycardia in patients with premature ventricular contractions and heart failure treated with vasodilator therapy. J Am Coll Cardiol. 1998;32:942-947.
14 Luu M, Stevenson WG, Stevenson LW, et al. Diverse mechanism of unexpected cardiac arrest in advanced heart failure. Circulation. 1989;80:1675-1680.
15 Mitchell LB, Pineda EA, Titus JL, et al. Sudden death in patients with implantable cardioverter defibrillators. J Am Coll Cardiol. 2002;39:1323-1328.
16 Zipes ST. Cesium-induced atrial tachycardia degenerating into atrial fibrillation in dogs: Atrial torsades de pointes? J Cardiovasc Electrophysiol. 1998;9:970-975.
17 Iuliano S, Fisher SG, Karasik PE, et al. QRS duration and mortality in patients with congestive heart failure. Am Heart J. 2002;143:1085-1091.
18 Das MK, Zipes DP. Fragmented QRS: A predictor of mortality and sudden cardiac death. Heart Rhythm. 2009;6:S8-S14.
19 Bloomfield DM, Steinman RC, Namerow PB, et al. Microvolt T-wave alternans distinguishes between patients likely and patients not likely to benefit from implanted cardiac defibrillator therapy: A solution to the Multicenter Automatic Defibrillator Implantation Trial (MADIT) II conundrum. Circulation. 2004;110:1885-1889.
20 Gold MR, Ip JH, Costantini O, et al. Role of microvolt T-wave alternans in assessment of arrhythmia vulnerability among patients with heart failure and systolic dysfunction: Primary results from the T-wave alternans sudden cardiac death in Heart Failure Trial Substudy. Circulation. 2008;118:2022-2028.
21 LaRovere MT, Bigger JT, Marcus FI, et al. Baroreflex sensitivity and heart-rate variability I prediction of total cardiac mortality after myocardial infarction. ATRAMI (Autonomic Tone and Reflexes After Myocardial Infarction) Investigators. Lancet. 1998;35(9101):478-484.
22 Milner PG, Dimarco JP, Lerman BB. Electrophysiologic evaluation of sustained tachyarrhythmias in idiopathic dilated cardiomyopathy. Pacing Clin Electrophysiol. 1988;11:562.
23 Tchou P, Jazayeri M, Denker S, et al. Transcatheter electrical ablation of right bundle branch: A method of treating macroreentrant ventricular tachycardia due to bundle branch reentry. Circulation. 1988;8:246.
24 Buxton AE, Lee KL, Fisher JD, et al. A randomized study of the prevention of sudden death in patients with coronary artery disease. Multicenter Unsustained Tachycardia Trial Investigators. N Engl J Med. 1999;341(25):1882-1890.
25 CIBIS-II Investigators and Committees. The cardiac insufficiency bisoprolol study-II (CIBIS=II): A randomized trial. Lancet. 1999;353:9-13.
26 MERIT-HF Study Group. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL randomized intervention trial in congestive heart failure. Lancet. 1999;353:2001-2007.
27 Poole-Wilson PA, Swedberg K, Cleland JGF, et al. Comparison of carvedilol and metoprolol on clinical outcomes in patients with chronic heart failure. Results of the Carvedilol or Metoprolol European Trial, for the COMET Investigators. Lancet. 2003;362:7-13.
28 Ryden L, Ariniego R, Ainman K, et al. A double-blind trial of metoprolol in acute myocardial infarction: Effects on ventricular tachyarrhythmia. N Engl J Med. 1983;308:614-618.
29 Cairns J, Connolly S, Roberts R, Gent M, for the Canadian Amiodarone Myocardial Infarction Arrhythmia Trial Investigators. Randomized trial of outcome after myocardial infarction in patients with frequent or repetitive ventricular premature depolarizations. CAMIAT. Lancet. 1997;349:675-682.
30 Julian DG, Camm AJ, Frangin G, et al. Randomised trial of effect of amiodarone on mortality in patients with left ventricular dysfunction after recent myocardial infarction. EMIAT. Lancet. 1997;349:667-674.
31 Roy D, Talajic M, Nattel S, et al. Rhythm control versus rate control for atrial fibrillation and heart failure. N Engl J Med. 2008;358:2667-2677.
32 Kober L, Torp-Pedersen C, McMurray JJ, et al. Increased mortality after dronedarone therapy for severe heart failure, for the Dronedarone Study Group. N Eng J Med. 2008;358:2678-2687.
33 Hohnloser SH, Crijns HJ, Eickels M, et al. Effect of dronedarone on cardiovascular events in atrial fibrillation, for the ATHENA Investigators. N Eng J Med. 2009;360:668-678.
34 Hansen JF. Treatment with verapamil after an acute myocardial infarction. Review of the Danish studies on verapamil in myocardial infarction (DAVIT I and II). Drugs. 1991;42(Suppl 2):43-53.
35 Goldstein RE, Boccuzzi SJ, Cruess D, Nattel S. Diltiazem increases late-onset congestive heart failure in postinfarction patients with early reduction in ejection fraction. The Adverse Experience Committee; and the Multicenter Diltiazem Postinfarction Research Group. Circulation. 1991;83(1):52-60.
36 Makkar KM, Sanoski CA, Spinler SA. Role of angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers and aldosterone antagonists in the prevention of atrial and ventricular arrhythmias. Pharacotherapy. 2009;29:31-48.
37 Yusuf S, Pfeffer MA, Swedberg K, et al. Effects of candesartan in patients with chronic heart failure and preserved left ventricular ejection fraction: The CHARM Preserved Trial. Lancet. 2003;362:777-781.
38 Massie BM, Carson PE, McMurray JJ, et al. Irbesartan in patients with heart failure and preserved ejection fraction, for the I-PRESERVE Investigators. N Engl J Med. 2008;359:2456-2467.
39 Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure, The Randomized Aldactone Evaluation Study Investigators. N Engl J Med. 1999;341(10):709-717.
40 Pitt B, Remme W, Zannad F, et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction, for the Eplerenone Post-Acute Myocardial Infarction Heart Failure Efficacy and Survival Study Investigators. N Engl J Med. 2003;348(14):1309-1321.
41 Scirica BM, Morrow DA, Hod H, et al. Effect of ranolazine, an antianginal agent with novel electrophysiological properties, on the incidence of arrhythmias in patients with non-ST segment elevation acute coronary syndrome. Circulation. 2007;116(15):1647-1652.
42 Patti G, Chello M, Candura D, et al. Randomized trial of atorvastatin for reduction of post-operative atrial fibrillation in patients undergoing cardiac surgery: Results of the ARMYDA-3 (Atorvastatin for Reduction of Myocardial Dysrhythmia After cardiac surgery) study. Circulation. 2006;114:1455-1461.
43 Sutter J, Tavernier R, DeBuyzere M, et al. Lipid lowering drugs and recurrences of life-threatening ventricular arrhythmias in high-risk patients. J Am Coll Cardiol. 2000;36(3):766-775.
44 Mitchell LB, Powell JL, Gillis AM, Kehl V, Hallstrom AP, the AVID Investigators. Are lipid-lowering drugs also antiarrhythmic drugs? An analysis of the antiarrhythmics versus implantable defibrillators (AVID) trial. J Am Coll Cardiol. 2003;42:81-87.
45 Marchioli R, Barzi F, Bomba E, et al. Early protection against sudden death by n-3 polyunsaturated fatty acids after myocardial infarction: Time-course analysis of the results of the Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico (GISSI)-Prevenzione, on behalf of the GISSI-Prevenzione Investigators. Circulation. 2002;105:1897-1903.
46 GISSI-HF Investigators. Effect of n-3 polyunsaturated fatty acids in patients with chronic heart failure (The GISSI-HF trial): A randomised double-blind, placebo-controlled trial. Lancet. 2008;372:1223-1230.
47 Sakabe M, Shiroshita-Takeshita A, Maguy A, et al. Omega-3 polyunsaturated fatty acids prevent atrial fibrillation associated with heart failure but not atrial tachycardia remodeling. Circulation. 2007;116:2101-2109.
48 The ACTIVE Investigators. Effect of clopidogrel added to aspirin in patients with atrial fibrillation. N Engl J Med. 2009;360(20):2066-2078.
49 Holmes DRJr, Davis KB, Mock MB, et al. Risk factor profiles of patients with sudden cardiac death and death from other cardiac causes: A report from the Coronary Artery Surgery Study. J Am Coll Cardiol. 1989;13:524-530.
50 Bigger JTJr. Prophylactic use of implanted cardiac defibrillators in patients at high risk for ventricular arrhythmias after coronary-artery bypass graft surgery. Coronary Artery Bypass Graft (CABG) Patch Trial Investigators. N Engl J Med. 1997;337(22):1569-1575.
51 Moss AJ, Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction, for the Multicenter Automatic Defibrillator Implantation Trial II Investigators. N Engl J Med. 2002;346:877-883.
52 Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure, for the Sudden Cardiac Death in Heart Failure Trial (SCD-HEFT) Investigators. N Engl J Med. 2005;352:225-237.
53 Bristow MR, Saxon LA, Boehmer J, et al. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure, for the Comparison of Medical Therapy, Pacing, and Defibrillation in Heart Failure (COMPANION) Investigators. N Engl J Med. 2004;350:2140-2150.
54 Cleland JG, Daubert JC, Erdmann E, et al. Longer-term effects of cardiac resynchronization therapy on mortality in heart failure [the Cardiac REsynchroniztion-Heart Failure (CARE-HF) trial extension phase], on behalf of the CARE-HF Investigators. Eur Heart J. 2006;27:1928-1932.
55 DiBiase L, Gasparini M, Lunati M, et al. Antiarrhythmic effect of reverse ventricular remodeling induced by cardiac resynchronization therapy. J Am Coll Cardiol. 2008;52:1442-1449.
56 Tang AS, Wells GA, Talajic M, et al. Cardiac-resynchronization therapy for mild-to-moderate heart failure, Resynchronization-Defibrillation for Ambulatory Heart Failure Trial Investigators. N Engl J Med. 2010;363(25):2385-2395.
57 Chugh SS, Shen WK, Luria DM, Smith HC. First evidence of premature ventricular complex-induced cardiomyopathy: A potentially reversible cause of heart failure. J Cardiovasc Electrophysiol. 2000;3:328-329.
58 Kottkamp H, Hindricks G, Chen X, et al. Radiofrequency catheter ablation of sustained ventricular tachycardia in idiopathic dilated cardiomyopathy. Circulation. 1995;92(5):1159-1168.
59 Stevenson WG, Wilber DF, Natale A, et al. Irrigated radiofrequency catheter ablation guided by electroanatomic mapping for recurrent ventricular tachycardia after myocardial infarction: The Multicenter Thermocool Ventricular Tachycardia Ablation Trial, Multicenter Thermocool VT Ablation Trial Investigators. Circulation. 2008;118(25):2773-2782.
60 Khan MN, Jais P, Cummings J, et al. Pulmonary-vein isolation for atrial fibrillation in patients with heart failure, for the PABA-CHF Investigators. N Engl J Med. 2008;359:1778-1785.