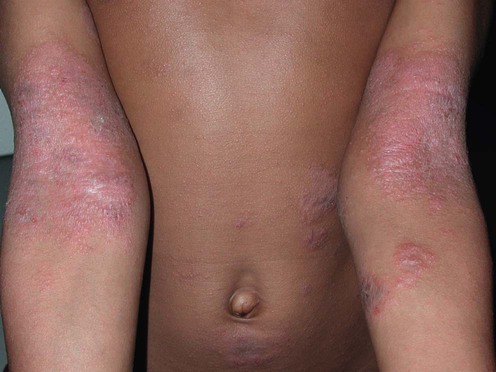
Atopic dermatitis
Management strategy
Interventional education
Comprehensive multispecialty ‘eczema clinics’ are beginning to test the utility of intensive education as a distinct component of long-term AD management. This model, with its longer appointments, focused educational curricula, patient support networks, and the ability to elicit patient/family feedback, parallels strategies shown to be effective for managing asthma, diabetes, and other chronic diseases. Supporters hope to empower patients and caregivers and improve both clinical and quality of life outcomes. Long-term comparative evaluation is required to examine the cost-effectiveness and suitability of these educational programs. Other learning modalities beyond in-clinic disease education may be useful to assist with disease management, including hand-outs, internet-based written material and instructional videos (e.g., www.eczemacenter.org; www.nationaleczema.org; www.eczema.org).
Referral, quality of life issues, and educational resources
Emotional stress can exacerbate AD in some patients who may respond to frustration, anxiety, embarrassment, or other psychologically stressful events with a perceived increase in pruritus and subsequent scratching. This may be particularly important in the adolescent population, where even very mild skin disease may be considered ‘disfiguring.’ In some cases, patients may use scratching for secondary gain and in others scratching has simply become habitual. Relaxation, biofeedback, and behavioral modification techniques may be helpful in such patients. Psychosocial evaluation and counseling should be considered in families where emotional triggers appear to function as obstacles to disease management, or where quality of life is clearly affected. Therapeutic education has been shown beneficial in managing AD, and may be accomplished through extended in-office discussion, written handouts, eczema ‘school programs’ and/or web resources, which include video training modules (e.g., www.eczemacenter.org). Families may also benefit from support groups such as the US National Eczema Association (www.nationaleczema.org) and the UK National Eczema Society (www.eczema.org).
First-line therapies
Second-line therapies
Topical calcineurin inhibitors and malignancies in pediatric patients: a literature review.
Manthripragada A, 2011. Presented at: U.S. Food and Drug Administration Pediatric Advisory Committee Meeting, May 16. Available at: http://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/PediatricAdvisoryCommittee/UCM255411.pdf. Accessed September 20, 2012.
Third-line therapies






 Education
Education Emollients
Emollients Topical corticosteroids
Topical corticosteroids Wet wraps
Wet wraps Topical immunomodulators
Topical immunomodulators Antimicrobials
Antimicrobials Antihistamines/anxiolytics
Antihistamines/anxiolytics Barrier repair products
Barrier repair products Avoidance of triggers
Avoidance of triggers Dust mite reduction
Dust mite reduction Timing of solid food introduction
Timing of solid food introduction Phototherapy
Phototherapy Azathioprine
Azathioprine Cyclosporine
Cyclosporine Methotrexate
Methotrexate Mycophenolate mofetil
Mycophenolate mofetil Interferon-γ
Interferon-γ Efaluzimab
Efaluzimab Systemic corticosteroids
Systemic corticosteroids
