Chapter 12 Aspiration Prevention and Prophylaxis
Preoperative Considerations
I Perioperative Aspiration
A Incidence
The statistical incidence of perioperative aspiration has been examined in long-term case reviews. A multicenter, prospective study of almost 200,000 operations performed in France found the overall incidence of clinically apparent aspiration to be 1.4 per 10,000 anesthesias.1 Leigh and Tytler’s 5-year survey of almost 110,000 anesthesias found 6 cases of aspiration requiring unplanned critical care.2 Warner and colleagues retrospectively reviewed more than 215,000 general anesthesias and found an incidence of aspiration of 3.1 per 10,000 cases.3 Olsson and associates examined the records of more than 175,000 anesthesias administered at one hospital over more than 13 years and reported an incidence of aspiration of 4.7 per 10,000.4 Kallar and Everett’s multicenter survey of more than 500,000 outpatient anesthesias found the incidence of aspiration to be 1.7 per 10,000.1
In their 1999 review of 133 Australian cases, Kluger and Short reported that the incidence of passive regurgitation was three times that of active vomiting and that a majority of aspiration episodes accompanied anesthesias delivered by face mask or laryngeal mask airway (LMA). Thirty-eight percent of those who aspirated developed radiographic infiltrates, more often in the right lung than in the left. The authors also noted that “a recurring theme in many incidents was one of inadequate anaesthesia leading to coughing/straining and subsequent regurgitation/vomiting.”5 In their 1999 survey of pediatric aspiration, Warner and coauthors also wrote that “nearly all cases of pulmonary aspiration … occurred in patients who gagged or coughed during airway manipulation or during induction of anesthesia.”6
Several authors have observed that only about 50% or fewer of the episodes of perioperative aspiration occur during anesthetic induction and intubation, perhaps because concern is less heightened at other times.1–9 These potentially catastrophic events also take place before induction (when the unguarded patient may be excessively sedated), during anesthesia maintenance, and during or after emergence and extubation.
B Consequences
When aspiration does occur, the subsequent clinical course can range from benign to fatal. Olsson and colleagues reported that 18% of patients who aspirated perioperatively required mechanical ventilatory support, and 5% died. All those who died had a poor preoperative physical status.4 Warner and coworkers reported that 64% of patients did not manifest coughing, wheezing, radiographic abnormalities, or a 10% decrease in arterial oxygen saturation (Sao2) from preoperative room air values during the first 2 hours after aspiration. Such patients who remained asymptomatic for 2 hours developed no respiratory sequelae. Of the patients who did manifest signs or symptoms of pulmonary aspiration within 2 hours after the event, 54% required mechanical ventilatory support for 6 hours or longer, and 25% were ventilated for more than 24 hours. Approximately 50% of those ventilated for 24 hours or longer died, generating an overall mortality rate of less than 5% of all aspirations.3
Mortality rates resulting from perioperative pulmonary aspiration have ranged from less than 5% to more than 80% in other reports.10,11 In the studies of Warner and Olsson and their coworkers, there were no deaths in healthy patients undergoing elective surgery.3,4 In their 1999 survey of 133 perioperative aspirations in Australia, Kluger and Short wrote that “the deaths following aspiration events occurred in sicker patients … although mortality can occur in healthy, younger patients.”5 Reviewing more than 85,000 Scandinavian anesthesias, Mellin-Olsen and associates noted that only 3 of 25 patients who aspirated developed serious morbidity, 2 of whom endured a prolonged course of illness but all of whom survived.8 In general, most healthy patients who aspirate only gastric fluid can expect to survive without residual respiratory impairment, albeit sometimes after a stormy postoperative course.
C Risk Factors
1 Demographic
Published surveys have associated some characteristics of patients or circumstances with an increased incidence of aspiration. Warner and colleagues noted that the relative risk of aspiration was more than four times higher for emergency surgeries compared with elective surgeries. A higher American Society of Anesthesiologists (ASA) physical status classification was also associated with a higher risk of aspiration. The incidence of aspiration increased from 1.1 per 10,000 elective anesthesias in ASA class I patients to 29.2 per 10,000 emergency anesthesias in ASA class IV and V patients. Contrary to conventional wisdom, “Age, gender, pregnancy … concurrent administration of opioids, obesity … experience … of anesthesia provider, and types of surgical procedure were not independent risk factors for pulmonary aspiration. … The most common predisposing condition in all patients was gastrointestinal obstruction.”3 Borland and coauthors also wrote that “aspiration occurred significantly more often in patients with greater severity of underlying illness.”12
Olsson and colleagues found that children and elderly persons were more likely than patients of intermediate ages to aspirate perioperatively.4 Statistically, the risk of aspiration was more than three times higher in emergency surgeries than in elective operations. The incidence of aspiration was increased more than sixfold when surgery was performed at night rather than during daylight hours. More recent studies of both adult and pediatric cases have confirmed an impressive increase in the incidence of perioperative aspiration in emergency operations,6,8 but Borland and coauthors found this increase to be only “marginally significant.”12
In Kallar and Everett’s outpatient survey,1 aspiration occurred most frequently in patients younger than 10 years of age. They also reported, “In patients with no other identifiable risk factors, 67% of aspirations occurred after difficulties in airway management or intubation.”1 In the study of Olsson and colleagues, 15 of 83 aspirations occurred in patients with no known risk factors.4 In 67% (10/15) of these cases, aspiration accompanied airway problems. In contrast to Kluger and Short’s findings,5 no patient aspirated while intubated. Although regional techniques are often favored for patients at increased risk for aspiration, elderly patients, in particular, have been reported to vomit and aspirate during subarachnoid anesthesia. Hypotension resulting from neuraxial sympathectomy can induce nausea and vomiting, and supplemental analgesics and sedatives given during lengthy operations can seriously obtund protective airway reflexes.4,13,14
Patients who are likely to have gastric contents of increased volume or acidity, elevated intragastric pressure, or decreased tone of the lower esophageal sphincter (LES) are traditionally considered to be at increased risk for perioperative pulmonary aspiration (Boxes 12-1 and 12-2).10,15 As discussed later, pregnancy combines several of these likely risk factors. Although a lengthy NPO period before elective surgery is intended to minimize the volume of gastric contents, up to 90% of fasted patients have a gastric fluid pH level lower than 2.5.11 Recent ethanol ingestion or hypoglycemic episodes stimulate gastric acid secretion, whereas tobacco inhalation temporarily lowers LES tone. LES tone has also been found to be reduced by gastric fluid acidity, caffeine, chocolate, and fatty foods.11
Box 12-1 Risk Factors for Aspiration of Gastric Contents
Box 12-2 Factors That Increase Intragastric Volume and Pressure
Surgical outpatients have traditionally been thought to carry gastric contents of expanded volume and reduced pH, possibly because of preoperative anxiety. Clinical studies, however, have not consistently confirmed this expectation.1 Furthermore, Hardy and associates contradicted several conventional notions by finding that neither gastric content volume nor pH correlated with preoperative anxiety, body mass index (BMI), ethanol or tobacco intake, or reflux history.15
2 Obesity
Obese patients were traditionally thought to pose a relatively high risk for aspiration because of their greater gastric fluid volume and acidity, intragastric pressure, and incidence of gastroesophageal reflux (GER).16 This assumption has been challenged. In 1998, Harter and colleagues studied 232 fasted, nondiabetic surgical patients who had received no relevant preoperative medication. Using conventional arbitrary criteria, they found that only 27% of obese patients, compared with 42% of the nonobese, had gastric contents of high volume and acidity. Grading obesity by BMI, they also found no association between degree of obesity and gastric fluid volume or pH.16 The presumed sluggishness of gastric emptying in obese patients has also been denied. Verdich and coworkers reported that obese and lean patients did not differ in rate of gastric emptying during the first 3 hours after a test meal.17 Lower esophageal pressure has also been shown not to differ significantly between obese and nonobese patients.16
On the other hand, the laryngoscopic challenges that arise with corpulence, along with the association between airway difficulties and aspiration episodes, appear to increase the risk of aspiration in obese patients regardless of their gastrointestinal motility. Clinical studies have both confirmed and denied a demonstrable increase in the incidence of difficult intubation (DI) in the obese.18–21 In a review of the topic, Freid noted that “obese patients … develop oxygen desaturation faster than the nonobese, and the safe apneic period is reduced from more than five minutes to less than two to three minutes in the preoxygenated state.” Importantly, he observed that “far more morbidity occurs owing to hypoxemia during difficult or failed intubation than from aspiration.”7
3 Systemic Diseases
Patients with connective tissue, neurologic, metabolic, or neuromuscular disease may be imperiled by esophageal dysfunction or laryngeal incompetence. Progressive systemic sclerosis and myotonia dystrophica have been specifically mentioned in case reports.22–24 Hardoff and coworkers found that “gastric emptying time in patients with Parkinson’s disease was delayed compared with control volunteers [and] was even slower in patients treated with levodopa.”25 Advanced age may be associated with attenuated cough or gag reflexes.
Long-standing diabetes mellitus is commonly considered to delay gastric emptying and may also compromise LES function.26 Several authors have reported a high incidence of gastroparesis and prolonged mean gastric emptying times, at least for solid foods, in diabetic patients compared with control subjects.27–30 Impairment of gastric motility was usually found to correlate with findings of autonomic neuropathy but not with peripheral neuropathy or with indices of glycemic stability.
4 Pregnancy
Pregnancy imposes a constellation of potential risk factors. The enlarging uterus increases intragastric pressure by compressing the stomach, physically delays gastric emptying by pushing the pylorus cephalad and posteriorly, and promotes GER by altering the angle of the gastroesophageal junction. Progesterone decreases the tone of the LES, and excess gastrin, produced by the placenta, promotes gastric acid secretion.26,31,32 The alterations in physique that are typical of late pregnancy can interfere with laryngoscopy and endotracheal intubation. Laryngeal and upper airway edema is also common in the parturient and can be exaggerated by preeclampsia.33
Studies of gastric emptying in pregnancy have produced somewhat inconsistent results. Wong and colleagues found that water was readily cleared from the stomachs of nonobese, nonlaboring parturients at term and wrote that “recent studies of gastric emptying in nonlaboring term women … suggest that gastric emptying is not delayed during pregnancy.”34 Chiloiro and coworkers found that gastric emptying time did not become slower with the progress of gestation but that total orocecal transit time did.35 A more common clinical concern is the parturient in labor. Scrutton and associates reported that laboring patients who consumed a light solid meal had significantly greater gastric volumes than those allowed only water.36 Although pain, in any circumstance, is thought to delay gastric emptying, Porter and coauthors stated that “pain does not appear to be the sole cause of gastric slowing in late labour since [there was] a similar delay in women in late labour who had received either epidural local anesthetic alone or no analgesia.”37
5 Pain and Analgesics
Pain and its treatment are considered to be risk factors for aspiration, notably in patients presenting with trauma. As stated by Crighton and colleagues, “circulating catecholamines have an inhibitory effect on gastric emptying, and noradrenaline release in response to painful stimuli may cause inhibition of gastric tone and emptying.”38 Patients with spinal cord or brain injuries have also been shown to manifest delayed gastric emptying of both liquid and solid contents.39–41
Administration of opioids to alleviate pain is an essential act of kindness but may further impair gastrointestinal function. Opioid receptors can be found throughout the gastrointestinal tract; human and animal studies suggest that there are central and peripheral mechanisms by which these drugs retard gastric emptying.42 Even modest intravenous doses of morphine demonstrably prolong gastric transit times in clinical studies.38,43–45
Neuraxial opioids can also prolong gastric emptying. In obstetric anesthesia, Kelly and associates “conclude[d] that the administration of fentanyl 25 mg intrathecally delays gastric emptying in labor compared with both extradural fentanyl 50 mg with bupivacaine and extradural bupivacaine alone.”46 Older reports indicated that epidural fentanyl boluses of 50 or 100 mg would retard gastric emptying. On the other hand, the addition of fentanyl (2 or 2.5 mg/mL) to dilute bupivacaine for epidural infusion during labor was not found to affect gastric motility.37,47
6 Positioning
Agnew and colleagues continuously monitored esophageal and tracheal pH in thoracotomy patients “considered to be at low risk of GER.” Twenty-eight percent of their patients not treated with a histamine2 (H2)-receptor antagonist were found to have acid reflux into the esophagus while in the lateral decubitus position, and almost 8% had acid in the trachea. Although the authors did not correlate clinical outcomes with their findings, they did advise that patients undergoing thoracotomy be considered for routine preoperative aspiration chemoprophylaxis.48
D Pathophysiology
When gastric contents enter the lungs, the resultant pulmonary pathology depends on the nature of the material aspirated (Box 12-3). Food particles small enough to enter the distal airways induce a foreign body reaction of inflammation and eventual granuloma formation. The aspiration of particulate antacids produces the same adverse response.32,49 Acid aspiration induces an inflammatory response that begins within minutes and progresses over 24 to 36 hours.49,50 In 1940, Irons and Apfelbach wrote that the “characteristic microscopic changes are intense engorgement of the alveolar capillaries, … edema, and hemorrhage into the alveolar spaces.… Another outstanding characteristic is the extensive desquamation of the lining of the bronchial tree.”51 Other authors have also described hemorrhagic pulmonary edema, intense inflammation, and derangement of the pulmonary epithelium.50,52 The membranous epithelial cells that produce surfactant are damaged or destroyed by the acid and replaced by granular epithelial cells.14 As surfactant production fails, lung units progressively collapse. Fibrin and plasma leak from the capillaries into the pulmonary interstitium and alveoli, producing the noncardiogenic pulmonary edema often referred to as adult (or acute) respiratory distress syndrome (ARDS).14,50,53,54 With effective supportive care, the acute inflammation can diminish, and epithelial regeneration can begin, within 72 hours.
Box 12-3 Pathophysiology of Aspiration
The clinical features of aspiration pneumonitis have been well described for more than 60 years. Even earlier, in 1887, Becker referred to bronchopneumonia as a postoperative complication related to the inhalation of gastric contents.51 Hall, in 1940, published the first description of gastric fluid inhalation in obstetric patients. He distinguished between the aspiration of solid material, which could quickly kill by suffocation, and the aspiration syndrome produced by gastric fluid, for which he coined the term chemical pneumonitis.55 Mendelson, in 1946, described the clinical features of 66 cases of peripartum aspiration observed from 1932 to 1945. Solid food produced airway obstruction, which was quickly fatal in two instances. Otherwise, wheezing, rales, rhonchi, tachypnea, and tachycardia were prominent.56 (Subsequent reports have not found wheezing to be so universal a manifestation, occurring in about one third of aspirations.) When present, wheezing is thought to result from bronchial mucosal edema and from a reflex response to acidic airway irritation.11,50,57
Refractory hypoxemia can ensue almost immediately as bronchospasm, airway edema or obstruction, and alveolar collapse or flooding increase the effective intrapulmonary shunt fraction (Box 12-4). The awake patient may experience intense dyspnea and may cough up the pink, frothy sputum characteristic of pulmonary edema.11,32,50 More modest aspirations may not become clinically evident for several hours.32,58,59
Hemodynamic derangements can also demand therapeutic attention. As the alveolar-capillary membrane loses its integrity, plasma leaks out of the pulmonary vasculature. If the leak becomes a flood, the loss of circulating fluid volume can produce hemoconcentration, hypotension, tachycardia, and even shock.11,32 Pulmonary vasospasm may also contribute to right ventricular dysfunction.11
The radiographic evidence of pulmonary aspiration may become evident promptly, if aspiration is massive, or after a delay of several hours. There is no pattern on the chest roentgenogram that is specific for aspiration. The distribution of infiltrates depends on the volume of material inhaled and the patient’s position at the time of the event. Because of bronchial anatomy, aspiration occurring in the supine patient affects the right lower lobe most commonly, the left upper lobe least often.11,32 If pulmonary aspiration is not complicated by secondary events, improvement in symptoms can be anticipated within 24 hours, although the radiographic picture may continue to worsen for another day.32
E Determinants of Morbidity
1 pH and Volume of Aspirate
In his 1946 report, Mendelson undertook to determine the relationship between gastric fluid acidity and pulmonary morbidity. When liquid containing hydrochloric acid (HCl) was instilled into rabbits’ tracheas, the animals developed a syndrome “similar in many respects to that observed in the human following liquid aspiration,”56 with cyanosis, dyspnea, and pink, frothy sputum. On the other hand, when neutral liquid was instilled into the trachea, the rabbits endured a brief symptomatic period, “but within a few hours they [were] apparently back to normal, able to carry on rabbit activities uninhibited.”56 (Mendelson maintained a discreet silence about the nature of these uninhibited rabbit activities.)
In 1952, Teabeaut injected HCl solutions of different volumes and acidities into rabbits’ tracheas. He found that solutions with a pH higher than 2.4 caused a relatively benign tissue response similar to that induced by the intratracheal injection of water. As the pH of the injectate was reduced from 2.4 to 1.5, a progressively more severe tissue reaction was elicited. At pH 1.5, the damage was maximal and equal to that found at lower pH values.60 From this study stemmed the popular concept of the pH value of 2.5 as a threshold for chemical pneumonitis.
The determination of a critical volume of gastric contents required to produce severe aspiration pneumonitis has been even more contentious than that of a critical pH. Two teams of investigators each found that, in dogs, pulmonary injury became independent of pH as the volume of aspirate was increased from 0.5 to 4.0 mL/kg.11 A preliminary experiment by Roberts and Shirley, involving gastric fluid instillation into the right main stem bronchus of a single monkey, long ago led to the acceptance, in some quarters, of 0.4 mL/kg as the volume of gastric fluid that places a subject at risk for development of aspiration pneumonitis.61–63 Subsequent researchers challenged this number. James and colleagues demonstrated that aspirate volumes as low as 0.2 mL/kg could induce pulmonary injury if the pH of the aspirate were reduced to 1.09.64 On the other hand, Raidoo and coworkers, also studying monkeys, found that the aspiration of 0.4 to 0.6 mL/kg of fluid with a pH of 1.0 produced mild or moderate pulmonary injury, and 0.8 to 1.0 mL/kg at pH 1.0 produced severe pneumonitis, with a 50% mortality rate (3/6) at 1.0 mL/kg.61
Clearly, the volume of aspirate that is considered hazardous depends on how much morbidity or pathology must be produced to be considered significant. Arguments have also been made concerning the experimental instillation of gastric fluid into one lung versus both lungs and the reliability of gastric fluid volume measurements. In addition, even if a critical volume for aspiration pneumonitis could be reliably determined, it cannot be known how much fluid must be present in the stomach in order to deposit this critical volume into the lung or lungs.61,65 However, studies of therapeutic interventions must have criteria for defining success or failure, and threshold values for gastric fluid volume and pH are typically employed, regardless of their validity.
2 Particulate Matter
Volume and acidity are not, of course, the only determinants of sequelae when gastric contents enter the lungs. Since the report of Bond and coworkers in 1979, it has been appreciated that gastric fluid containing particulate antacids can produce severe aspiration pneumonitis, even at near-neutral pH, with wheezing, pulmonary edema, and hypoxemia requiring mechanical ventilatory support.66 Animal studies confirmed that nonparticulate gastric acid and particulate antacid solutions have similar potentials for pulmonary mischief if aspirated.58 Although blood and digestive enzymes do not appear to induce chemical pneumonitis, feculent gastric contents with a high bacterial density readily produce pneumonitis and death in animals. (Acidic gastric contents are normally sterile.) Another study demonstrated that the mucus present in the gastric fluid of dogs with intestinal obstruction produced diffuse small airway obstruction and pulmonary injury when aspirated.11
II Prevention of Aspiration
A Preoperative Fasting
The most common means of keeping gastric contents out of the lungs is to minimize the volume of such contents through preoperative fasting. However, both the utility and the necessity of adhering to traditional NPO regimens for clear liquids have been challenged. As noted by Sethi and coauthors, “the stomach can never be completely empty even after a midnight fast since it continues to secrete gastric juices.”67 The issue has been studied in both pediatric and adult surgical patients and has become particularly contentious and emotional regarding obstetric anesthesia.
1 Pediatric Patients
Conventional preoperative fasting can impose physical and emotional discomfort on children and their parents and may be difficult to enforce reliably in outpatients. Dehydration in infants and hypoglycemia in neonates may also result from prolonged NPO times.1,10 The normal stomach can empty 80% of a clear liquid load within 1 hour after ingestion. Whereas the stomach continues to secrete and reabsorb fluid throughout NPO time, ingested clear liquids are completely passed into the duodenum within 2.25 hours.68 Several researchers have therefore sought to demonstrate that children may safely be allowed to drink clear liquids until just 2 to 3 hours before elective surgery.
Van der Walt and Carter, as well as other groups, determined that healthy infants could drink limited volumes of clear liquids 3 to 4 hours before surgery with no effect on gastric content volumes.69 Splinter and colleagues found that healthy infants could drink clear liquids ad libitum until 2 hours before anesthetic induction without altering gastric fluid volume or pH. (Gastric fluid pH was quite variable, and mean pH was less than 2.5 in all groups of patients studied, regardless of NPO time.) On the other hand, milk or formula intake on the morning of surgery (4 to 6 hours before induction) was associated with the presence of curds in many of the gastric aspirates. This was considered to represent an unacceptable risk of particulate aspiration. The authors therefore concurred with previous recommendations that infants not be allowed milk or formula on the morning of surgery.70
More recently, Cook-Sather and associates studied 97 healthy infants undergoing elective surgery and found that gastric fluid volume was not increased when the fasting time for formula was reduced from 8 hours to either 6 or 4 hours.71 Schreiner and colleagues compared the gastric contents of children subjected to conventional preoperative fasting (mean NPO time, 13.5 hours) with those of children permitted clear liquids until 2 hours before anesthetic induction (mean NPO time, 2.6 hours). Gastric fluid volumes actually tended to be somewhat smaller in the children allowed to drink clear liquids up to 2 hours preoperatively, and almost all children in both groups had gastric content pH values of 2.5 or less.72 Sandhar and coauthors, studying children 1 to 14 years old, also found that clear liquid ingestion 2 to 3 hours preoperatively did not significantly increase the mean volume of gastric contents and did not increase the number of patients with gastric contents more voluminous than 0.4 mL/kg.68 Reports by Splinter, Moyao-Garcia, Maekawa, and Gombar and their colleagues all concluded that permitting children to drink nonparticulate fluids 2 to 3 hours before surgery had either no effect or a small beneficial influence on the quantity and acidity of their gastric contents.73–76
Ingestion of clear liquids alone therefore appears to pose no demonstrable hazard if taken no later than 2 hours before anesthesia by children without gastrointestinal pathology. However, solid or semisolid foods are not cleared from the stomach as rapidly as clear liquids. Meakin and associates found that a light breakfast of biscuits or orange juice with pulp, taken 2 to 4 hours before induction, did increase the volume of gastric aspirate in healthy children compared with those who had fasted for 4 hours or longer. In all fasted children, and in almost all of the fed children, the gastric content pH was 2.5 or less.77 Hyperosmolar glucose solutions are also associated with delayed gastric emptying.68
In 1999, the ASA issued the report of its Task Force on Preoperative Fasting, which included practice guidelines. These guidelines were intended to apply to healthy patients who have no known relevant risk factors or injuries and are scheduled for elective surgery. Within these limitations, the ASA Task Force “support[ed] a fasting period for clear liquids of two hours for all patients [and] a fasting period for breast milk of four hours for both neonates and infants” while considering it “appropriate to fast from intake of infant formula for six or more hours.”78
2 Adult Patients
In adult surgical patients, too, considerable evidence has demonstrated that clear liquid intake within 2 to 3 hours of anesthetic induction does not increase the risk of gastric acid aspiration. It is important to note that these studies typically involved healthy, nonpregnant, nonobese patients who were free of known gastrointestinal pathology, were not receiving opioids or other medications known to interfere with gastric emptying, and were undergoing elective surgery. The results of such studies cannot, therefore, be reliably applied to any other groups of patients.79–82
With adults, as with children, the basic arguments favoring relaxed NPO regimens for clear liquids involve their normally rapid gastric clearance. More than 90% of a 750-mL bolus of isotonic saline was found to pass from the normal stomach within 30 minutes.83 After 2 hours of fasting, the fluid in the stomach primarily represents the acid secreted by the stomach itself. Exogenous clear liquids tend to dilute endogenous gastric acid and may even accelerate gastric emptying.65,72,81 Solids, lipids, and hyperosmotic liquids are thought to delay gastric emptying, and their intake would therefore be considered ill advised before anesthetic induction.65
Several researchers have sought to correlate these theoretical considerations with clinical situations. Maltby and colleagues studied outpatients who were either kept NPO from the previous midnight or given 150 mL of water 2.5 hours before anesthetic induction. Although the mean gastric pH did not differ significantly between the two groups, the mean gastric volume was significantly less in the patients who drank than in those who fasted.83 Read and Vaughan similarly found that permitting patients to drink water ad libitum until 2 hours before surgery had no impact on gastric volume or pH but did decrease preanesthetic anxiety. Many patients had gastric pH values less than or equal to than 2.5, regardless of the time elapsed since fluid intake.84
Phillips and colleagues also determined that patients who were allowed to drink clear liquids until 2 hours before surgery had gastric volume and pH values similar to those of patients who fasted for 6 hours. Other studies have also confirmed that the ingestion of 150 mL of (pulp-free) orange juice, coffee, tea, or apple juice 2 to 3 hours before surgery has no detrimental effect on gastric pH or volume in surgical outpatients.81 (Since the publication of these reports, one can only wonder how many hours of presurgical time have been consumed in speculation regarding the pulp content of orange juice that patients have admitted to consuming.)
The safety of clear liquid ingestion before surgery does not, of course, imply that solid food may also be taken with impunity. In an early study, Miller and coworkers compared 22 adults kept NPO overnight before surgery with 23 adults permitted a light breakfast (one slice of buttered toast and tea or coffee with milk) on the morning of surgery (mean NPO time, 3.8 hours). The two groups were found not to differ significantly in mean volume or median pH of gastric contents or in the percentage of patients with a gastric pH lower than 3.0.80 Soreide and coauthors reported that the particulate elements of a light breakfast had not completely exited after 4 hours.85 Reflecting a consensus of clinical comfort, the aforementioned ASA Task Force recommended a 6-hour preoperative fast following a “light meal” and a fast of 8 hours or longer “for a meal that included fried or fatty foods or meat.”78
3 Pregnant Patients
Elkington cited a Washington state survey (1977-1981) in which none of 36 maternal deaths resulted from anesthetic complications and a North Carolina survey (1981-1985) in which only 1 of 40 maternal deaths resulted from aspiration. He did not advocate uninhibited feeding of patients in labor but rather recommended that, “For otherwise uncomplicated parturients, a nonparticulate diet should be allowed as desired.”86
Ludka and Roberts referred to a Michigan survey (1972-1984) showing that only 1 of 15 maternal deaths (0.82 per 100,000 live births) resulted from the aspiration of gastric contents, that no deaths were related to regional anesthesia, and that “failure to secure a patent airway was the primary cause of anesthesia-related maternal deaths.”87 They cited other studies indicating that women who ate during labor were less ketotic, required less analgesic medication and oxytocin, and had more active fetuses and neonates with higher Apgar scores than women who fasted during labor. They also “found that laboring women self-regulated intake. Once active labor began, women usually preferred liquids.”87
Regarding the inevitability of a full stomach in the parturient, Kallar and Everett referred to an ultrasound study in which almost two thirds of patients in labor had solid food in the stomach, regardless of how long they had fasted.1 Elkington cited a report that approximately 25% of parturients were “at risk” for aspiration pneumonitis, regardless of the duration of fasting, and that prolonged fasting was actually associated with increased gastric fluid volume at a lower pH.86 Broach and Newton contended that “administration of narcotics, not labor itself, appears to be the major factor in delaying stomach emptying.”88 In a recent review, de Souza and associates wrote the following: “It has now been proven that pregnancy by itself does not delay gastric emptying. Many investigators using different modalities have confirmed this finding. Obese term pregnant patients have also been found to have normal gastric emptying. We emphasize that these findings apply only to patients who are not in labor.”89
McKay and Mahan stated that, “among many factors that can be linked to the occurrence of aspiration, the most important appears to be faulty administration of obstetric anesthesia.” They questioned “whether parturients should be kept … on restricted liquid intake to protect them from what appears to be the basic problem: inadequate anesthesia practices.”90 The apparent implication was that the parturient should eat, drink, and be merry, for if she should aspirate, only poor anesthetic care would be to blame.
On the other hand, Chestnut and Cohen cited the Report on Confidential Enquiries into Maternal Deaths in England and Wales 1982-1984, which found that 7 of 19 anesthesia-associated maternal deaths resulted from aspiration of gastric contents into the lungs, and an ASA review of closed malpractice claims in which “maternal aspiration was the primary reason for 8% of all claims against anesthesiologists for obstetric cases.” The authors argued that “These data hardly suggest that the risk of maternal aspiration is remote.”91
In his reply to McKay and Mahan, Crawford observed that “most of the deaths from aspiration prior to the mid-1950s … were due to asphyxia, caused by respiratory obstruction with solid or semisolid material—since that time, with introduction of a firm dietary regimen for labor, only 2 of the 146 deaths noted have been in that category.” Furthermore, he contended, “There is inevitably an incidence of cesarean section and of general anesthesia in every obstetric population.… In an obstetric population the incidence of failed or difficult intubation is roughly one in 300.”92
In the work of Scrutton and colleagues, permitting parturients a “light diet” (as opposed to water only) had no effect on the course of labor or the neonatal Apgar scores but did increase the volume of gastric contents and of vomitus. They stated that “the presence of undigested food particles in the vomitus is probably of greater importance [than low pH] as a cause of mortality [and] would not support the policy of encouraging women to eat any solid food once in labour particularly when isotonic drinks appear to offer an adequate calorific alternative.”36 In their review, Ng and Smith concluded that “there is insufficient evidence to clarify changes in risk in the first 24 hours of the postpartum period, when operative procedures are common.”93
Obviously, the pregnant patient with a difficult airway cannot always be avoided, nor can general anesthesia for cesarean section, no matter how aggressively regional anesthesia is promoted. Regardless of gastric fluid volume or acidity, the presence of solid food imparts the immediate hazard of asphyxiation. Mendelson warned that “misinformed friends and relatives often urge the patient to ingest a heavy meal early in labor before going to the hospital.”56 Crawford concluded, “Grafting good anesthetic technique upon poor preparation of a patient for anesthesia is unjustifiable—there is an essential symbiosis between the two if safety is to be ensured.”92
B Preinduction Gastric Emptying
It has long been argued that the presence of a gastric tube interferes with the sphincter function of the gastroesophageal junction and promotes GER by acting as a “wick.”94 The presence of a foreign body in the pharynx could also interfere with laryngoscopy. These considerations would favor removal of the gastric tube before induction. However, in an early study by Satiani and colleagues, the incidence of “silent” GER was found to be 12% in anesthetized patients without an NG tube and 6% in patients with an NG tube in place (a statistically insignificant difference).95 Hardy wrote that “a nasogastric tube need not be withdrawn before induction of anesthesia. The tube can act as an overflow valve” and provide “a venting mechanism whereby pressure cannot build up in the stomach.”96
Dotson and associates prospectively studied the effect of NG tube size on GER in normal subjects. Attempts were made to provoke GER with a device that elevated abdominal pressure stepwise to 100 mm Hg. In this report, GER “was not detected at any level of abdominal pressure regardless of the presence or size of a nasogastric tube.”97 Salem and colleagues had previously demonstrated that “cricoid pressure is effective in sealing the esophagus around an esophageal tube against an intraesophageal pressure up to 100 cm H2O.”98 They also advocated the utility of an NG tube as a “blow-off valve” for increased intragastric pressure during induction.98 The presence of an NG tube, while allowing gastric decompression, may also hold open the LES.11,99
Vanner and Asai advised that an NG tube already inserted “should be [suctioned and] left in place [for anesthetic induction], since its presence does not reduce the efficacy of cricoid pressure.”9 On the other hand, Brock-Utne contended that “the recommendation that a nasogastric tube should be left in situ during a rapid sequence technique induction is not supported by the evidence. Clinicians who have seen aspiration of gastric contents with an nasogastric tube in situ will, no doubt, remove the nasogastric tube before anesthetic induction.”100 None of these writings addressed the usually surgical decision concerning which patients should have NG tubes placed before entering the operating room.
Some of the studies just cited would seem to indicate that an NG tube, already inserted, can be safely left in place during induction and may even have a protective benefit. Gastric decompression during surgery could also reduce the risk of regurgitation and aspiration in the postanesthetic period.96 The necessity and the utility of NG tube insertion just before induction are not so well defined. The benefits of awake gastric decompression depend, in part, on how completely the stomach can thereby be emptied. The primary drawback is patient discomfort.
Several authors have studied the thoroughness of gastric emptying attainable by gastric tube suctioning, usually in the context of comparing different methods for estimating gastric residual volume. Ong and coauthors reported, as early as 1978, that the volume of fluid obtained by orogastric suctioning correlated poorly with the gastric residual volume calculated by a dilution method, “the volume aspirated being frequently much less than the volume calculated.”101 They concluded that “aspiration through a gastric tube will not empty the stomach completely.” Mechanical decompression of the stomach before induction might therefore be of limited reliability and thus provide a false sense of security.101 Taylor and associates studied 10 obese patients in whom gastric contents were first aspirated through a 16-F multiorificed Salem Sump tube, then “completely” removed by a gastroscope. “The blind aspirated volume underestimated true total gastric volume by an average of 14.7 mL, [which] was statistically significant.… The residual content volume left in the stomach after blind aspiration varied from 4 mL to 23 mL,”102 a maximal discrepancy far less than that found by Ong and coworkers in 42 patients.101
Hardy and colleagues measured the volume of gastric fluid aspirated through an 18-F Salem Sump tube in 24 patients, then directly inspected the stomach and measured the volume of fluid remaining. The residual volume that eluded orogastric suctioning ranged from 0 to 13 mL. The authors concluded “that the volume of aspirated gastric fluid … is a very good estimate of the volume present in the stomach at the time of induction” and that gastric tube suctioning “could also be suitable to empty the stomach of its liquid contents prior to anaesthesia.”103
It can be argued that any reduction in intragastric volume and pressure before anesthetic induction is desirable and should therefore be attempted. On the other hand, as Satiani and colleagues conceded, “particulate matter [is] impossible to evacuate through the lumen of an ordinary nasogastric tube.”95 Salem and associates concluded that “placement of a nasogastric tube before anesthetic induction seems to be indicated only in patients with overdistention of the stomach.”98 Although obvious enteric obstruction is conventionally treated with gastric decompression before anesthetic induction, not every emergency or at-risk patient is subjected to NG tube insertion while awake. There is currently no consensus to dictate preinduction placement of a gastric tube in any set of patients without intestinal obstruction. In any case, gastric decompression in no way substitutes for proper perioperative management of the airway.
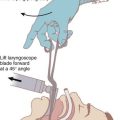
C Rapid-Sequence Induction and Cricoid Pressure
If anesthetic induction is to precede endotracheal intubation, the standard protective maneuver for 5 decades has been rapid-sequence induction (RSI) with cricoid pressure. The traditional components of this technique include preoxygenation and denitrogenation of the lungs, rapid administration of anesthetic induction and neuromuscular blocking agents with brief onset times, cricoid pressure, no manual ventilation by mask, and (one hopes) endotracheal intubation immediately after consciousness and neuromuscular transmission have been obtunded. As El-Orbany and Connolly wrote, this sequence “has achieved a status close to being a standard of care for anesthesia induction in patients with full stomachs. Despite the technique’s widespread use, there is still no agreement on how it should best be performed.”104 The practice of RSI has generated contention regarding both cricoid pressure and obligatory apnea between induction and intubation.
Abstention from positive-pressure ventilation (PPV) during RSI rests on the fear that the resulting gastric insufflation will increase the likelihood of regurgitation and aspiration of gastric contents before endotracheal intubation. The preservation of adequate oxygenation despite apnea is, of course, crucial. As noted by El-Orbany and Connolly, “Currently, some experts strongly recommend the routine use of PPV … in certain [RSI] situations. Hypoxemia can develop in obese, pregnant, pediatric, and critically ill patients … even after adequate administration of oxygen … because of their low functional residual capacity.”104 Fifty years ago, Ruben and colleagues demonstrated that “manual … ventilation did not result in gastric insufflation when airway pressures were kept <15 cm H2O. … With the application of cricoid pressure, no gastric insufflation occurred even when the inflating pressure was increased to 45 cm H2O.”105 Of course, if the patient becomes hypoxemic, manual ventilation becomes mandatory.
The utility of cricoid pressure has been challenged on many fronts. As described by Sellick in 1961, “The manoeuver consists in temporary occlusion of the upper end of the oesophagus by backward pressure of the cricoid cartilage against the bodies of the cervical vertebrae.… Extension of the neck and application of pressure on the cricoid cartilage obliterates the oesophageal lumen at the level of the body of the fifth cervical vertebra.… Pressure is maintained until intubation and inflation of the cuff of the endotracheal tube is completed.”106 Kopka, Herman, and colleagues noted that cricoid pressure was used as early as 1774 to avert regurgitation related to gastric distention with air during resuscitation from drowning.107,108 In Sellick’s original report of 26 high-risk cases, 23 patients neither vomited nor regurgitated at any time near induction, and in the other 3 cases the “release of cricoid pressure after intubation was followed immediately by reflux into the pharynx of gastric or oesophageal contents, suggesting that in these three cases cricoid pressure had been effective.”106
Although the LES has received considerable attention with regard to the pharmacology and pathophysiology of GER, there is also effective sphincter tone at the upper end of the esophagus. As described by Vanner and colleagues, “The upper oesophageal sphincter is formed mainly by the cricopharyngeus, a striated muscle situated behind the cricoid cartilage. The muscle tone of the cricopharyngeus creates a sphincter pressure which prevents regurgitation in the awake state.”109 These authors found that general anesthesia with neuromuscular blockade reduced the upper esophageal sphincter pressure from 38 mm Hg (while awake) to 6 mm Hg, a pressure that would typically permit passive regurgitation. Although cricoid pressure could exceed the normal awake level of upper esophageal sphincter pressure, in only half of their study patients was it applied firmly enough to do so.109
Other authors have noted the inconsistency with which Sellick’s maneuver is applied. As Stept and Safar wrote in 1970, “The attempt to close the esophagus by pressing the cricoid cartilage against the cervical vertebrae is rarely applied with proper timing, namely, starting with the onset of unconsciousness and continuing until the tracheal cuff is inflated.”110 In their study of simulated cricoid pressure, Meek and coauthors reported that target pressures could be sustained with a flexed arm for a mean of only 3.7 to 6.4 minutes and with an extended arm for 7.6 to 10.8 minutes.111
A rising chorus of skepticism concerning the efficacy of cricoid pressure has been heard. In their reviews, Kluger and Short5 and Thwaites and colleagues112 cited reports of lethal aspiration occurring despite Sellick’s maneuver. The former authors specifically questioned the “almost unerring faith in the efficacy of this manoeuvre.”5 In their prospective investigation of emergency intubations in critically ill patients, Schwartz and associates reported that “twelve patients had an unexplained infiltrate that probably resulted from aspiration.… Nine of the twelve patients had cricoid pressure applied during airway management.”113 Specifically addressing medicolegal arguments, Jackson contended that “there is no scientific validation for the commonly held belief that ‘improper application of cricoid pressure might explain any failures’ to prevent aspiration.”114
Although it is largely accepted that cricoid pressure limits the incidence of passive regurgitation, “it cannot be expected to prevent regurgitation during coughing, straining, or retching.”9 Sellick himself warned against applying cricoid pressure to a patient who is actively vomiting, lest the resulting increased pressure injure the esophagus.106 In addition, the maneuver itself can induce gagging and even vomiting in the awake patient.9 Ralph and Wareham reported a case in which “rupture of the oesophagus occurred during the application of cricoid pressure at induction of anaesthesia when the patient vomited.” Fatal mediastinitis ensued.115
It has also become more widely acknowledged that cricoid pressure can interfere with both pulmonary ventilation and endotracheal intubation. Hocking and associates reported that “cricoid pressure produced a reduction in tidal volume (VT) and an increase in peak inspiratory pressure” in 50 female patients given mechanical ventilation by mask with an oral airway. Although the changes in pressure and volume were not large, “Complete airway obstruction resulted on three occasions, all with cricoid pressure applied.”116 Saghaei and Masoodifar, in their study of 80 healthy anesthetized adults, found that bimanual cricoid pressure induced decreases in VT with significant increases in peak inspiratory pressure, blood pressure, and heart rate.117 Hartsilver and Vanner reported similar findings, concluding that “the degree of airway obstruction is related to the force applied to the cricoid cartilage. Therefore, if it is difficult to ventilate via a facemask, the amount of cricoid pressure should be reduced.”118
Cricoid pressure can also impede ventilation through an LMA. Asai and colleagues reported that standardized cricoid pressure reduced the success rate of LMA ventilation from 100% (22/22) to 14% (3/22). Under fiberoptic visualization, the 18 LMAs that had been properly positioned became displaced when cricoid pressure was applied.119 Similarly, Harry and Nolan stated that cricoid pressure reduced the success rate of endotracheal intubation through an intubating LMA from 84% to 52%.120 Using fiberoptic visualization by LMA, MacG Palmer and Ball noted that cricoid pressure commonly caused cricoid deformation or occlusion, vocal cord closure, and impairment of ventilation, especially in women.121
The application of cricoid pressure may either improve or impede conventional laryngoscopy. According to Vanner and coauthors, standard cricoid pressure usually facilitated the exposure of the glottis, and “cricoid pressure in an upward (cephalad) and backward direction was more likely to give a better view at laryngoscopy than the standard technique.”122 In a substantial minority of their patients, however, cricoid pressure had either no impact or a detrimental effect on laryngoscopy. Hillman and colleagues reported that “the change in laryngoscopic view with increasing cricoid pressure fell into one of four broad patterns: little change (11/40), gradual deterioration (10/40), improvement at low force followed by deterioration [at high force] (9/40), [and] improvement at high force (10/40).… [I]n some individuals, a force close to that currently recommended may cause a complete loss of the glottic view.”123 Other authors have contended that cricoid pressure tends to interfere with intubation with lightwand or fiberoptic techniques.124,125 On the other hand, Riad and Ansari found that “application of cricoid pressure during induction of anaesthesia for elective caesarean section neither prolongs the time nor interferes with ease of endotracheal intubation using Airtraq [a disposable optical laryngoscope].”126 In a randomized study of 700 adult surgical patients, Turgeon and colleagues concluded that “cricoid pressure applied by trained personnel does not increase the rate of failed intubation.”127
MacG Palmer and Ball concluded that “Orthodox application of cricoid pressure may … be directly implicated in the ‘can’t intubate, can’t ventilate’ scenario.”121 In their review of the current practice of RSI, Thwaites and coworkers sharpened the point of their critique by reminding us “that hypoxia can kill rapidly, while aspiration only might occur and only might kill.”112 Although Sellick’s maneuver remains a conventional element of aspiration prevention, many authors now assert that it should not be slavishly pursued to the detriment of gas exchange and airway securement.
III Medical Prophylaxis of Aspiration
A Gastroesophageal Motility
Although preparation of the patient (rational NPO strategy and perhaps gastric suctioning) and airway management are the twin pillars of aspiration prevention, pharmacologic prophylaxis has been promoted as adjunctive to patients’ safety. Because gastric contents must first pass through the esophagus before entering the pharynx and trachea, the LES has become a locus of attention. As described by Ciresi, the LES “consists of functionally but not anatomically specialized smooth muscle, about 2-4 cm in length, just proximal to the stomach. The sphincteric muscle maintains closure of the distal esophagus through a mechanism of tonic contraction … accompanied by a zone of intraluminal high pressure.”128 Normally, a cholinergic reflex loop acts to increase LES pressure when intragastric or intra-abdominal pressure rises.4 The pressure gradient between the LES and the stomach, referred to as the barrier pressure, is responsible for preventing GER (Boxes 12-5 and 12-6).13,129,130
LES function is modulated by neurohumoral influences. Cholinergic stimulation increases LES tone, whereas dopaminergic and adrenergic stimulations reduce it.13,24,129 β-Adrenergic agents and theophylline reduce LES pressure and promote GER, often with symptomatic heartburn in awake patients. β-Adrenergic blockade elevates LES pressure.131 Anticholinergics attenuate LES tone and impair the efficacy of medications given to increase LES barrier pressure.1,26,58,132,133 Although prochlorperazine raises LES pressure (presumably by an antidopaminergic effect), promethazine lowers LES pressure because of its anticholinergic properties.13 Among the wide variety of other drugs that may reduce LES tone are benzodiazepines, opioids, barbiturates, dopamine, tricyclic antidepressants, calcium channel blockers, nitroglycerin, and nitroprusside.1,26 Although succinylcholine-induced fasciculations can elevate intra-abdominal pressure, LES tone concurrently rises, and the barrier pressure is maintained or increased.1,13 Apart from pharmacologic influences, Rabey and colleagues demonstrated that “barrier pressure may be reduced after insertion of an LMA during anesthesia with spontaneous ventilation.”130
In many cases, agents that increase LES contractility also promote forward passage of gastric contents, and the factors that attenuate LES tone also retard gastric emptying. This correlation compounds pharmacologic opportunities for either protection or mischief. Opioids and anticholinergics inhibit gastrointestinal motility, increasing the volume of gastric contents available for vomiting or regurgitation.133,134 Although pain and anxiety delay gastric emptying through sympathetic stimulation, the administration of an opioid for analgesia can further retard the propulsion of gastric contents into the duodenum.26,129
1 Metoclopramide
Gastroprokinetic drugs are now available to promote gastric emptying while simultaneously enhancing LES barrier pressure. Metoclopramide is the prototypical agent in this category. The mechanisms of action proposed for metoclopramide include central antidopaminergic activity and prolactin stimulation as well as peripheral blockade of dopamine receptors and stimulation of cholinergic function in the upper gastrointestinal tract. Although metoclopramide retains its gastrokinetic effect in vagotomized subjects, atropine has been shown to interfere with this activity.128,135 Metoclopramide both raises LES contractility and barrier pressure and accelerates gastric emptying. The latter effect is achieved by intensifying gastric longitudinal muscle contraction while relaxing the gastroduodenal sphincter and increasing the coordination of gastrointestinal peristalsis. Metoclopramide has no effect on gastric acid secretion.58,128
Metoclopramide has been extensively investigated as a chemoprophylactic agent for aspiration pneumonitis in children and in adults. Several original studies of patients given metoclopramide, in a dose of 10 or 20 mg either orally (PO) or intravenously (IV), demonstrated the drug’s utility in reducing gastric residual volume.58,136,137 Gonzalez and Kallar wrote that “metoclopramide 10 mg PO or IV, in combination with Bicitra or an H2-receptor antagonist, provides the most effective control of gastric volume and pH.”58 Given PO, metoclopramide has an onset of action that reportedly varies from 30 to 60 minutes, with a duration of action of 2 to 3 hours.58 Ciresi found that metoclopramide at either 10 or 20 mg IV could reliably empty the stomach within 10 to 20 minutes.128 Manchikanti and coworkers reported that metoclopramide 10 mg IV reduced the increase in gastric volume that followed ingestion of sodium citrate and citric acid (Bicitra) but did not interfere with Bicitra’s antacid activity.136 Metoclopramide was also found to reduce the volume of gastric contents in pediatric trauma patients.138
Other researchers found metoclopramide to be less uniformly effective, especially in the context of opioid coadministration or the recent ingestion of a solid meal.132 Christensen and colleagues demonstrated no influence of metoclopramide 0.1 mg/kg on the gastric pH or volume of healthy pediatric patients.139 As a perioperative antiemetic, metoclopramide was shown to be inconsistently useful.132 Side effects attributed to metoclopramide have included somnolence, dizziness, and faintness. These problems may surface more frequently in elderly or severely ill patients.58,128 Extrapyramidal reactions are a more serious problem but reportedly occur in only 1% of subjects.128 Deehan and Dobb reported on a patient with traumatic brain injury in whom metoclopramide 10 mg IV twice induced a severe rise in intracranial pressure associated with increased cerebral blood flow.140
Metoclopramide has also been investigated in obstetric anesthesia. The drug has been shown to increase LES tone in pregnant women and therefore may be a useful prophylactic agent before cesarean section.132,135 However, studies of gastric emptying in parturient women have provided less consistent results. Metoclopramide was shown to accelerate gastric emptying in patients undergoing scheduled or urgent cesarean section.135,137 On the other hand, Cohen and associates examined 58 healthy parturients after an overnight fast and found that metoclopramide, 10 mg IV, had no significant effect on mean gastric volume or pH or on the proportion of patients with a gastric content volume exceeding 25 mL. They suggested that the drug might be more useful in the emergency setting for patients with active labor, recent food intake, pain, and anxiety.135 Maternal metoclopramide administration does produce detectable and variable neonatal blood levels of the drug but without reported effects on Apgar scores or neurobehavioral test results.135,141
2 Erythromycin
Erythromycin is a macrolide antibiotic that has been in common use for more than 50 years. Given intravenously, it has been shown to improve gastric motility in patients with diabetic gastroparesis. Enteral feedings with erythromycin also pass more quickly through the stomach than control feedings. This action is thought to arise from the stimulation of motilin receptors in gastric smooth muscle.142 Boivin and associates demonstrated that intravenous “erythromycin increased gastric emptying in a dose-response manner.… [N]ausea and stomach cramping were associated with the 3.0 mg/kg dose of erythromycin; drowsiness was associated with metoclopramide [10 mg].”143 The potential applicability of these findings to perioperative aspiration prophylaxis is interesting but unproved.
B Reduction of Gastric Acid Content
Chemoprophylaxis of aspiration pneumonitis can also include the inhibition of gastric acid secretion or the neutralization of HCl already in the stomach. The former should eventually increase the pH and reduce the volume of gastric contents but has no effect on acidic fluid already in place. The latter should elevate gastric fluid pH but may also increase gastric fluid volume. The aspiration of particulate antacids can pose hazards equivalent to those of gastric acid inhalation, as previously described. In 1982, Eyler and colleagues demonstrated severe pulmonary pathology in rabbits resulting from aspiration of a commercial particulate antacid.144 Oral antacid prophylaxis should therefore include only soluble, nonparticulate agents.
1 Neutralization of Gastric Acid
The clear antacid solutions most commonly studied are sodium citrate (0.3 molar solution) and Bicitra. The pH of sodium citrate solutions typically exceeds 7.0, whereas that of Bicitra is 4.3.136 Manchikanti and associates compared surgical outpatients given Bicitra 15 or 30 mL PO with a matched control group. All patients studied were nonobese and NPO for at least 8 hours. Of the control patients, 88% had a gastric content pH of 2.5 or less, in contrast to 32% of those given Bicitra 15 mL and only 16% of those given Bicitra 30 mL.136
Sodium citrate has been evaluated as a sole prophylactic agent in a variety of surgical settings, with inconsistent results. Kuster and colleagues found that sodium citrate 30 mL, taken shortly before elective surgery, resulted in gastric fluid pH values greater than 3.5 in 95% of patients.145 Colman and coworkers administered 15 or 30 mL of sodium citrate to 30 parturient women before emergency cesarean section. All 15 patients given 30 mL sodium citrate and 14 of 15 given 15 mL had gastric pH values of 2.5 or higher.146 In other reports, however, sodium citrate failed to alter gastric fluid pH in surgical patients. In a 0.3 molar solution, 30 mL may be more consistently effective than 15 mL, but it may still not have prolonged effects in patients with rapid gastric emptying.13,147,148 Antacid prophylaxis may therefore be adequate at the induction of anesthesia but inadequate at the time of awakening. Larger volumes of sodium citrate can induce nausea and vomiting or diarrhea.136
2 Inhibition of Gastric Acid Secretion
a H2-Receptor Blockade
Gastric acid production is strongly modulated by the action of H2 receptors. H2-receptor blockade inhibits basal acid secretion as well as that stimulated by the presence of gastrin or food. Both H2 antagonists and anticholinergic agents block the neural stimulation of gastric acid secretion.128 However, this beneficial anticholinergic effect is overridden by the inhibition of gastrointestinal motility, so that gastric volume is not reduced and gastric pH is elevated only inconsistently.58 Although H2 antagonists do not delay gastric emptying, their inhibition of acid secretion tends to correlate inconsistently with both the timing and the magnitude of maximal drug concentration in the plasma.93 Various H2 antagonists have been evaluated in both surgical and obstetric settings, with different doses and routes of administration, and with and without other prophylactic medications, to produce an expansive volume of findings.
Cimetidine
Given before elective surgery, a variety of cimetidine regimens can ensure that most patients have gastric fluid volume or pH values, or both, in the safe range, as defined by the investigators. These usually effective regimens include cimetidine 300 mg PO at bedtime followed by 300 mg PO or intramuscularly (IM) on the morning of surgery; cimetidine 300 to 600 mg PO given 1.5 to 2 hours preoperatively; and cimetidine 200 mg IV given 1 hour before surgery. In one study, cimetidine most reliably produced gastric content safety when combined with preoperative metoclopramide.1 The reliability of oral cimetidine is generally improved if the drug is administered both the night before and on the morning of anesthesia.11
A gastric fluid pH of 2.5 or lower has been found in 5% to 35% of patients treated with single 300-mg doses of cimetidine given PO, IM, or IV in different studies. Significant elevation of gastric pH requires 30 to 60 minutes to become evident after the IV administration of cimetidine and 60 to 90 minutes after IM or PO dosing. Effective inhibition of gastric acid secretion persists for 4 to 6 hours.58,132 Papadimitriou and colleagues administered cimetidine 400 mg IV to 20 patients facing emergency surgery. Compared with 10 such patients given placebo treatment, those receiving cimetidine were found to have significantly lower mean gastric acidity, but the range of pH values was 1.6 to 7.2.149
Cimetidine chemoprophylaxis has also been evaluated in obstetric anesthesia. In a study of 100 patients undergoing emergency cesarean section, cimetidine 200 mg IM was administered when surgical delivery was decided upon, followed by oral intake of a 0.3-molar solution of sodium citrate 30 mL just before induction. None of these patients had a gastric fluid pH lower than 2.7, and only 1 of 100 had a gastric fluid pH lower than 3.0.90 Cimetidine administered in this fashion would most likely reduce gastric acidity by the time of extubation, whereas sodium citrate would be required to neutralize the acid already present.
Although cimetidine has a well-established record of safety when administered for perioperative aspiration prophylaxis, there are potential and observed side effects. The rapid IV infusion of large doses (e.g., 400 to 600 mg) has reportedly induced both hypotension and dangerous ventricular dysrhythmias.129,132 Smith and colleagues advised that IV cimetidine be infused over at least a 10-minute period.150 Other side effects sporadically associated with cimetidine include confusion, dizziness, headaches, and diarrhea, although these have not been reported to occur with single-dose preoperative administration.128,129,132
Cimetidine competitively inhibits the hepatic mixed-function oxidase system (cytochrome P450 enzyme) and also reduces hepatic perfusion.129,131,132 As a result, cimetidine may elevate the blood concentrations of other drugs that are cleared by the liver, including warfarin, propranolol, diazepam, theophylline, phenytoin, meperidine, bupivacaine, and lidocaine. Clinically, this seems to be a greater concern with long-term use than with one- or two-dose administration.128,132,146
Ranitidine
After cimetidine, ranitidine emerged as the next option for H2 blockade. Ranitidine is considered to exert little or no inhibition of hepatic enzymes, has a longer duration of action (6 to 8 hours) than cimetidine, and has an efficacy greater than or equal to that of the original H2 blocker. Effective onset times for the two drugs appear to be similar.1,58,132,146,151 Smith and coauthors reported that ranitidine 50 mg, given IV over 2 minutes to 20 critically ill patients, led to variable, transient reductions in mean arterial pressure and systemic vascular resistance. These hemodynamic effects occurred less frequently and were of lesser degree and duration than those resulting from cimetidine 200 mg, similarly administered. Previous sporadic case reports associated significant bradycardia with the IV administration of either cimetidine or ranitidine.150
In the study of adult surgical outpatients by Maltby and colleagues, ranitidine 150 mg PO, given 2.5 hours before anesthetic induction, significantly decreased gastric residual volume and significantly increased gastric fluid pH compared with placebo. In no patient was there the conventional (but arbitrary) at-risk combination of gastric pH lower than 2.5 and gastric volume greater than 25 mL.83 McAllister and associates treated adult patients with a single oral dose of ranitidine 300 mg, given 2 hours before surgery, and found both a significant increase in mean gastric fluid pH and a significant decrease in mean gastric fluid volume compared with placebo treatment. Noting the occasional patient with gastric fluid pH lower than 2.5, the authors cautioned that “it is unsafe to assume that H2 antagonists will always eliminate the risk of acid aspiration pneumonitis.”152 Single-dose IV administration of ranitidine, 40 to 100 mg, has also been found to reliably generate gastric fluid pH values greater than 2.5 in adults, manifesting a greater efficacy than that of cimetidine 300 mg IV.1
Sandhar and coworkers evaluated the efficacy of a single oral dose of ranitidine, 2 mg/kg, given 2 to 3 hours before surgery to patients aged 1 to 14 years. Although ranitidine significantly reduced both the volume and the acidity of gastric contents compared with placebo, 6 of 44 children receiving ranitidine did have gastric fluid pH values of 2.5 or lower.68 These findings confirmed those of a similar study of Goudsouzian and Young,153 although other authors have not demonstrated such a consistent reduction in gastric fluid volume.1,83
Papadimitriou and associates compared ranitidine 150 mg IV with cimetidine 400 mg IV and with placebo given 1 hour before anesthetic induction to emergency surgical patients. Ranitidine and cimetidine caused similar reductions in gastric volume and acidity; only the reductions in acidity were statistically significant. Although the mean pH values in the cimetidine and ranitidine groups were similar, only ranitidine consistently produced safe gastric pH values (all of which were 5.0 or higher).149 Vila and colleagues, evaluating H2 antagonists in morbidly obese surgical patients, concluded that ranitidine was superior to cimetidine in elevating gastric fluid pH.154 A literature review cited by Gonzalez and Kallar also found that ranitidine more reliably ensured that gastric fluid pH would exceed 2.5 than did cimetidine, although neither agent consistently reduced gastric fluid volume into the range considered safe by study authors.58
The effect of PO ranitidine (150 mg, given 2 to 3 hours before the scheduled time of surgery) with or without PO metoclopramide (10 mg, given 1 hour before surgery) and/or sodium citrate (30 mL on call to the operating room) on gastric fluid volume and pH was measured in 196 elective surgical inpatients. Although no combination guaranteed a safe combination of fluid volume and pH, a single oral dose of ranitidine was statistically as effective as triple prophylaxis.155 In pediatric patients, both Gombar and Maekawa and their colleagues showed that PO ranitidine (either 2 mg/kg or 75 mg) effectively elevated gastric fluid pH with no appreciable effect on gastric volume.76,156 In his study of the IV administration of these drugs 15 minutes before anesthetic induction, Hong concluded that “prophylactic ranitidine (50 mg IV) and metoclopramide (10 mg IV) may be an easy and useful method to decrease the volume while increasing the pH of gastric contents.”157
Ranitidine has also been evaluated for prophylactic use in obstetric anesthesia. Rout and colleagues evaluated the efficacy of ranitidine 50 mg IV given to laboring patients when cesarean section was decided upon. A control group received no H2-antagonist therapy, but all patients were given 30 mL of 0.3-molar sodium citrate shortly before induction. At the time of induction, 4% of those given only sodium citrate had a gastric fluid volume greater than 25 mL along with a pH lower than 3.5, compared with 2.3% of those given both citrate and ranitidine (P = 0.05).151 At the time of extubation, 5.6% of those given only sodium citrate were considered to be at risk by the preceding criteria, compared with only 0.3% of those given both citrate and ranitidine (P < 0.05). In a recent Cochrane Database Systems Review, Paranjothyand coworkers concluded that, although “the quality of the evidence was poor, … the findings suggest that the combination of antacids plus H2 antagonists is superior to antacids alone in increasing gastric pH [in patients undergoing caesarean section]. When a single agent is used, antacids alone are superior to H2 antagonists.”158
Others
A voluminous body of evidence thus documents the general safety and efficacy of preoperative cimetidine and ranitidine in ameliorating the acidity and volume of gastric contents. Newer agents, such as famotidine (10 mg PO) and nizatidine, have also been evaluated, with generally favorable results.154,159 Wajima and colleagues reported that nizatidine 300 mg PO was uniformly effective in maintaining gastric content pH above 2.5 and volume below 25 mL when given 2 hours before surgery.160
On the basis of the presumably high ratio of benefit to risk, H2-blocking agents have been recommended for surgical patients who have an increased likelihood of inhaling gastric contents.1,65 However, given the infrequency of perioperative aspiration pneumonitis, documentation of the actual clinical benefit of such practice has yet to be provided. In the review by Warner and coauthors of more than 215,000 general anesthesias in adults, 35 patients with acknowledged risk factors did aspirate perioperatively. Of these 35, 17 had been given prophylactic medication. In this small sample, aspiration prophylaxis produced no discernible difference in the incidence of pulmonary complications.3 In general, the routine preoperative use of H2 antagonists is not considered either essential or cost-effective. As stated by Kallar and Everett, “It has yet to be proven that prophylaxis against acid aspiration changes morbidity or mortality in healthy patients having elective surgery.”1
b Proton Pump Inhibition
Proton pump inhibitors (PPIs) constitute a newer class of agents for the suppression of gastric acid production. Acetylcholine, histamine, and gastrin all stimulate HCl secretion by the gastric parietal cell. Although these agonists stimulate different populations of receptors, their mechanisms of action all eventually result in the formation of cyclic adenosine monophosphate (cAMP). cAMP activates the proton pump, H+,K+-adenosine triphosphatase (H+,K+-ATPase), which exchanges intraluminal potassium ions for intracellular hydrogen ions. Hydrogen ions are thereby secreted from the parietal cell into gastric fluids.68,152 Omeprazole, the prototype PPI, is actually a pro-drug that is absorbed in the small intestine and is activated in the highly acidic milieu of the gastric parietal cell. Activated omeprazole then remains in the parietal cell for up to 48 hours, inhibiting the proton pump in a prolonged manner.141,161–163 Inhibition of gastric acid secretion can be nearly complete, with no discernible side effects. A single dose of omeprazole, 20 to 40 mg, reduces gastric acidity for up to 48 hours. On the other hand, PPIs are characterized by variable first-pass metabolism with resulting inconsistencies in the plasma concentration after any given oral dose. As is the case with H2 antagonists, there is also an unpredictable relationship between peak plasma concentration and peak inhibition of gastric acid production.93
Omeprazole has been evaluated as a preoperative agent for the chemoprophylaxis of aspiration pneumonitis. Bouly and colleagues gave omeprazole 40 mg PO to healthy patients either the evening before or 2 hours before elective surgery. Although mean gastric fluid pH was significantly higher with omeprazole treatment than with placebo, 6 of 30 patients receiving omeprazole had gastric fluid pH values lower than 2.5 at the time of induction. Omeprazole significantly reduced gastric fluid volume compared with placebo.161
Omeprazole has also been evaluated in obstetric anesthesia. In a study by Orr and associates, all 15 patients who received omeprazole and also metoclopramide 20 mg IM at least 20 minutes before elective cesarean section had gastric fluid pH values greater than 2.5, both on induction and at extubation. When omeprazole 80 mg PO was given only on the morning of elective cesarean section, 2 of 33 patients had a gastric fluid pH lower than 2.5 at induction, 1 of whom also had a gastric fluid volume greater than 40 mL. All gastric fluid pH values were greater than 2.5 at extubation. Of 16 patients who also received metoclopramide 20 mg IM at least 20 minutes before elective cesarean section, 2 still had gastric fluid pH values lower than 2.5 (with gastric fluid volumes less than 25 mL) on induction, but all gastric fluid pH values exceeded 2.5 at extubation.141
In general, the PPIs have been found most effective when given in two doses, one on the night before and one on the morning of surgery.93 Given the dwindling proportion of patients hospitalized before elective surgery, however, two-dose regimens for chemoprophylaxis would seem somewhat impractical. Furthermore, Nishina and coauthors reported that a single preoperative oral dose of ranitidine was more effective in reducing gastric acid content than two-dose regimens of rabeprazole or lansoprazole.164 On the other hand, Pisegna and colleagues found that “pantoprazole (40 mg IV) decreased gastric acid output and volume, and increased pH within 1 hour of dosing. Effects were sustained for up to 12 hours following single-dose administration.”165
IV Conclusions
An impressive array of pharmacologic agents can now be employed to promote antegrade gastric emptying, inhibit GER, and reduce the acid content of gastric fluids. These drugs have an established record of safety and offer the reasonable expectation of rendering gastric fluid less threatening to the lungs. However, because of the low incidence of clinically significant perioperative aspiration, it may not be possible to demonstrate statistically that the use of these agents actually improves patients’ outcomes. In reference to gastric prokinetic drugs, antacids, and inhibitors of acid secretion, the ASA Task Force concluded that “the routine preoperative use of [such medications] in patients who have no apparent increased risk for pulmonary aspiration is not recommended.”78 Chemoprophylaxis is only an adjunct to and not a substitute for otherwise sound clinical practice. It is, of course, less desirable to have aspirated and survived than never to have aspirated at all.
V Clinical Pearls
• Regurgitation and aspiration can result from “light” anesthesia, coughing, and gagging in the patient not intubated.
• About half of all cases of perioperative aspiration occur at times other than anesthetic induction.
• Patients manifesting no evidence of respiratory impairment for 2 hours after a known or suspected aspiration episode are highly unlikely to become significantly symptomatic later.
• Delayed gastric emptying in diabetic patients correlates with the presence of autonomic, but not peripheral, neuropathy.
• Both pain and opiates significantly retard gastric emptying.
• Aspiration of particulate antacids can induce a severe granulomatous pneumonitis.
• Ingestion of clear liquids 2 to 3 hours before anesthetic induction does not appear to increase the risk of gastric content aspiration in patients with no gastrointestinal pathology.
• Cricoid pressure can compromise ventilation by either mask or LMA and can either facilitate or interfere with direct laryngoscopy.
• Nonparticulate antacids rapidly raise gastric fluid pH in most patients, with an inconsistent duration of action.
• H2 antagonists more reliably reduce gastric fluid acidity than gastric fluid volume.
• Metoclopramide reduces gastric fluid volume in most patients without consistently affecting gastric fluid pH.
All references can be found online at expertconsult.com.
3 Warner MA, Warner ME, Weber JG. Clinical significance of pulmonary aspiration during the perioperative period. Anesthesiology. 1993;78:56–62.
7 Fried EB. The rapid sequence induction revisited: Obesity and sleep apnea syndrome. Anesthesiol Clin North Am. 2005;23:551–554.
56 Mendelson CL. The aspiration of stomach contents into the lungs during obstetric anesthesia. Am J Obstet Gynecol. 1946;52:191.
78 American Society of Anesthesiologists Task Force on Preoperative Fasting. Practice guidelines for preoperative fasting and the use of pharmacologic agents to reduce the risk of pulmonary aspiration: Application to healthy patients undergoing elective procedures. Anesthesiology. 1999;90:896–905.
89 de Souza DG, Doar LH, Mehta SH, et al. Aspiration prophylaxis and rapid sequence induction for elective cesarean delivery: Time to reassess old dogma? Anesth Analg. 2010;110:1503–1505.
92 Crawford JS. How can aspiration of vomitus in obstetrics best be prevented? Commentary: Setting the record straight. Birth. 1988;15:230–235.
93 Ng A, Smith G. Gastroesophageal reflux and aspiration of gastric contents in anesthetic practice. Anesth Analg. 2001;93:494–513.
104 El-Orbany M, Connolly LA. Rapid sequence induction and intubation: Current controversy. Anesth Analg. 2010;111:1318–1325.
106 Sellick BA. Cricoid pressure to control regurgitation of stomach contents during induction of anaesthesia. Lancet. 1961;2:404.
158 Paranjothy S, Griffiths JD, Broughton KH, et al. Interventions at caesarean section for reducing the risk of aspiration pneumonitis (review). Cochrane Database Syst Rev. (1):2010. CD004943
1 Kallar SK, Everett LL. Potential risks and preventive measures for pulmonary aspiration: New concepts in preoperative fasting guidelines. Anesth Analg. 1993;77:171–182.
2 Leigh JM, Tytler JA. Admissions to the intensive care unit after complications of anaesthetic techniques over 10 years. Anaesthesia. 1990;45:814–820.
3 Warner MA, Warner ME, Weber JG. Clinical significance of pulmonary aspiration during the perioperative period. Anesthesiology. 1993;78:56–62.
4 Olsson GL, Hallen B, Hambraeus-Jonzon K. Aspiration during anaesthesia: A computer-aided study of 185,358 anaesthetics. Acta Anaesthesiol Scand. 1986;30:84–92.
5 Kluger MT, Short TG. Aspiration during anaesthesia: A review of 133 cases from the Australian Anaesthetic Incident Monitoring Study (AIMS). Anaesthesia. 1999;54:19–26.
6 Warner MA, Warner ME, Warner DO, et al. Perioperative pulmonary aspiration in infants and children. Anesthesiology. 1999;90:66–71.
7 Fried EB. The rapid sequence induction revisited: Obesity and sleep apnea syndrome. Anesthesiology Clin North Am. 2005;23:551–564.
8 Mellin-Olsen J, Fasting S, Gisvold SE. Routine preoperative gastric emptying is seldom indicated: A study of 85,594 anaesthetics with special focus on aspiration pneumonia. Acta Anaesthesiol Scand. 1996;40:1184–1188.
9 Vanner RG, Asai T. Safe use of cricoid pressure. Anaesthesia. 1999;54:1–3.
10 Cotes CJ. NPO after midnight for children: A reappraisal. Anesthesiology. 1990;72:589–592.
11 DePaso WJ. Aspiration pneumonia. Clin Chest Med. 1991;12:269–284.
12 Borland LM, Sereika SM, Woelfel SK, et al. Pulmonary aspiration in pediatric patients during general anesthesia: Incidence and outcome. J Clin Anesth. 1998;10:95–102.
13 Aitkenhead AR. Anaesthesia and the gastrointestinal system. Eur J Anaesthesiol. 1988;5:73–112.
14 LoCicero J. Bronchopulmonary aspiration. Surg Clin North Am. 1989;69:71–76.
15 Hardy JF, Lepage Y, Bonneville-Chouinard N. Occurrence of gastroesophageal reflux on induction of anaesthesia does not correlate with the volume of gastric contents. Can J Anaesth. 1990;37:502–508.
16 Harter RL, Kelly WB, Kramer MG, et al. A comparison of the volume and pH of gastric contents of obese and lean surgical patients. Anesth Analg. 1998;86:147–152.
17 Verdich C, Madsen JL, Toubro S, et al. Effect of obesity and major weight reduction on gastric emptying. Int J Obes Relat Metab Disord. 2000;24:899–905.
18 Juvin P, Lavaut E, Dupont H, et al. Difficult tracheal intubation is more common in obese than in lean patients. Anesth Analg. 2003;97:595–600.
19 Rose DK, Cohen MM. The airway: Problems and predictions in 18,500 patients. Can J Anaesth. 1994;41:372–383.
20 Bond A. Obesity and difficult intubation. Anaesth Intensive Care. 1993;21:828–830.
21 Brodsky JB, Lemmens HJ, Brock-Utne JG, et al. Morbid obesity and tracheal intubation. Anesth Analg. 2002;94:732–736.
22 Hannon VM, Cunningham AJ, Hutchinson M, et al. Aspiration pneumonia and coma: An unusual presentation of dystrophica myotonia. Can Anaesth Soc J. 1986;33:803–806.
23 Roberts JG, Sabar R, Gianoli JA, et al. Progressive systemic sclerosis: Clinical manifestations and anesthetic considerations. J Clin Anesth. 2002;14:474–477.
24 Russin SJ, Adler AG. Pulmonary aspiration. Postgrad Med. 1989;85:155–161.
25 Hardoff R, Sula M, Tamir A, et al. Gastric emptying time and gastric motility in patients with Parkinson’s disease. Mov Disord. 2001;16:1041–1047.
26 Ruffalo RL. Aspiration pneumonitis: Risk factors and management of the critically ill patient. DICP. 1990;24:S12–S16.
27 Caballero-Plasencia AM, Muros-Navarro MC, Martin-Ruiz JL, et al. Gastroparesis of digestible and indigestible solids in patients with insulin-dependent diabetes mellitus or functional dyspepsia. Dig Dis Sci. 1994;39:1409–1415.
28 Ishihara H, Singh H, Giesecke AH. Relationship between diabetic autonomic neuropathy and gastric contents. Anesth Analg. 1994;78:943–947.
29 Merio R, Festa A, Bergmann H, et al. Slow gastric emptying in type I diabetes: Relation to autonomic and peripheral neuropathy, blood glucose, and glycemic control. Diabetes Care. 1997;20:419–423.
30 Vaisman N, Weintrob N, Blumenthal A, et al. Gastric emptying in patients with type I diabetes mellitus. Ann N Y Acad Sci. 1999;873:506–511.
31 Ezri T, Szmuk P, Stein A, et al. Peripartum general anaesthesia without tracheal intubation: Incidence of aspiration pneumonia. Anaesthesia. 2000;55:421–426.
32 Hollingsworth HM, Irwin RS. Acute respiratory failure in pregnancy. Clin Chest Med. 1992;13:723–740.
33 Morgan M. Anaesthetic contribution to maternal mortality. Br J Anaesth. 1987;59:842–855.
34 Wong CA, Loffredi M, Ganchiff JN. Gastric emptying of water in term pregnancy. Anesthesiology. 2002;96:1395–1400.
35 Chiloiro M, Darconza G, Piccioli E, et al. Gastric emptying and orocecal transit time in pregnancy. J Gastroenterol. 2001;36:538–543.
36 Scrutton MJ, Metcalfe GA, Lowy C, et al. Eating in labour: A randomised controlled trial assessing the risks and benefits. Anaesthesia. 1999;54:329–434.
37 Porter JS, Bonello E, Reynolds F. The influence of epidural administration of fentanyl infusion on gastric emptying in labour. Anaesthesia. 1997;52:1151–1156.
38 Crighton IM, Martin PH, Hobbs GJ, et al. A comparison of the effects of intravenous tramadol, codeine, and morphine on gastric emptying in human volunteers. Anesth Analg. 1998;87:445–449.
39 Kao CH, ChangLai SP, Chieng PU, et al. Gastric emptying in head-injured patients. Am J Gastroenterol. 1998;93:1108–1112.
40 Kao CH, ChangLai SP, Chieng PU, et al. Gastric emptying in male neurologic trauma. J Nucl Med. 1998;39:1798–1801.
41 Lockey DJ, Coats T, Parr MJ. Aspiration in severe trauma: A prospective study. Anaesthesia. 1999;54:1097–1098.
42 Murphy DB, Sutton JA, Prescott LF, et al. Opioid-induced delay in gastric emptying: A peripheral mechanism in humans. Anesthesiology. 1997;87:765–770.
43 Hammas B, Thorn SE, Wattwil M. Propofol and gastric effects of morphine. Acta Anaesthesiol Scand. 2001;45:1023–1027.
44 Murphy DB, Sutton A, Prescott LF, et al. A comparison of the effects of tramadol and morphine on gastric emptying in man. Anaesthesia. 1997;52:1224–1229.
45 Yuan CS, Foss JF, O’Connor M, et al. Effects of low-dose morphine on gastric emptying in healthy volunteers. J Clin Pharmacol. 1998;38:1017–1020.
46 Kelly MC, Carabine UA, Hill DA, et al. A comparison of the effect of intrathecal and extradural fentanyl on gastric emptying in laboring women. Anesth Analg. 1997;85:834–838.
47 Zimmermann DL, Breen TW, Fick G. Adding fentanyl 0.0002% to epidural bupivacaine 0.125% does not delay gastric emptying in laboring parturients. Anesth Analg. 1996;82:612–616.
48 Agnew NM, Kendall JB, Akrofi M, et al. Gastroesophageal reflux and tracheal aspiration in the thoracotomy position: Should ranitidine premedication be routine? Anesth Analg. 2002;95:1645–1649.
49 Kirsch CM, Sanders A. Aspiration pneumonia: Medical management. Otolaryngol Clin North Am. 1988;21:677–689.
50 Dal Santo G. Acid aspiration: Pathophysiological aspects, prevention, and therapy. Int Anesthesiol Clin. 1986;24:31–52.
51 Irons EE, Apfelbach CW. Aspiration bronchopneumonia. JAMA. 1940;115:584–587.
52 Veddeng OJ, Myhre ESP, Risoe C, et al. Haemodynamic effects of selective positive end-expiratory pressure after unilateral pulmonary hydrochloric acid-aspiration in dogs. Intensive Care Med. 1992;18:356–361.
53 Pennza PT. Aspiration pneumonia, necrotizing pneumonia, and lung abscess. Emerg Med Clin North Am. 1989;7:279–307.
54 Sohma A, Brampton WJ, Dunnill MS, et al. Effect of ventilation with positive end-expiratory pressure on the development of lung damage in experimental acid aspiration pneumonia in the rabbit. Intensive Care Med. 1992;18:112–117.
55 Hall CC. Aspiration pneumonitis: An obstetric hazard. JAMA. 1940;114:728–733.
56 Mendelson CL. The aspiration of stomach contents into the lungs during obstetric anesthesia. Am J Obstet Gynecol. 1946;52:191–205.
57 Hollingsworth HM. Wheezing and stridor. Clin Chest Med. 1987;8:231–240.
58 Gonzalez ER, Kallar SK. Reducing the risk of aspiration pneumonitis. DICP. 1989;23:203–208.
59 Smith BE. Anesthetic emergencies. Clin Obstet Gynecol. 1985;28:391–404.
60 Teabeaut JR. Aspiration of gastric contents. Am J Pathol. 1952;28:51–67.
61 Raidoo DM, Rocke DA, Brock-Utne JG, et al. Critical volume for pulmonary acid aspiration: Reappraisal in a primate model. Br J Anaesth. 1990;65:248–250.
62 Roberts RB, Shirley MA. Antacid therapy in obstetrics. Anesthesiology. 1980;53:83.
63 Roberts RB, Shirley MA. Reducing the risk of acid aspiration during cesarean section. Anesth Analg. 1974;53:859–868.
64 James CF, Modell JH, Gibbs CP, et al. Pulmonary aspiration: Effects of volume and pH in the rat. Anesth Analg. 1984;63:655–658.
65 Goresky GV, Maltby JR. Fasting guidelines for elective surgical patients. Can J Anaesth. 1990;37:493–495.
66 Bond VK, Stoelting RK, Gupta CD. Pulmonary aspiration syndrome after inhalation of gastric fluid containing antacids. Anesthesiology. 1979;51:452–453.
67 Sethi AK, Chatterji C, Bhargava SK, et al. Safe pre-operative fasting times after milk or clear fluid in children: A preliminary study using real-time ultrasound. Anaesthesia. 1999;54:51–59.
68 Sandhar BK, Goresky GV, Maltby JR, et al. Effects of oral liquids and ranitidine on gastric fluid volume and pH in children undergoing outpatient surgery. Anesthesiology. 1989;71:327–330.
69 Van der Walt JH, Carter JA. The effect of different pre-operative feeding regimens on plasma glucose and gastric volume and pH in infancy. Anaesth Intensive Care. 1986;14:352–359.
70 Splinter WM, Schaefer JD, Bonn GE. Unlimited clear fluid ingestion by infants up to 2 hours before surgery is safe. Can J Anaesth. 1990;37:S95.
71 Cook-Sather SD, Harris KA, Chiavacci R, et al. A liberalized fasting guideline for formula-fed infants does not increase average gastric fluid volume before elective surgery. Anesth Analg. 2003;96:965–969.
72 Schreiner MS, Triebwasser A, Keon TP. Ingestion of liquids compared with preoperative fasting in pediatric outpatients. Anesthesiology. 1990;72:593–597.
73 Splinter WM, Schaefer JD. Unlimited clear fluid ingestion two hours before surgery in children does not affect volume or pH of stomach contents. Anaesth Intensive Care. 1990;18:522–526.
74 Moyao-Garcia D, Corrales-Fernandez MA, Blanco-Rodriguez G, et al. Benefits of oral administration of an electrolyte solution interrupting a prolonged preoperatory fasting period in pediatric patients. J Pediatr Surg. 2001;36:457–459.
75 Maekawa N, Mikawa K, Yaku H, et al. Effects of 2-, 4-, and 12-hour fasting intervals on preoperative gastric fluid pH and volume, and plasma glucose and lipid homeostasis in children. Acta Anaesthesiol Scand. 1993;37:783–787.
76 Gombar S, Dureja J, Kiran S, et al. The effect of pre-operative intake of oral water and ranitidine on gastric fluid volume and pH in children undergoing elective surgery. J Indian Med Assoc. 1997;95:166–168.
77 Meakin G, Dingwall AE, Addison GM. Effects of fasting and oral premedication on the pH and volume of gastric aspirate in children. Br J Anaesth. 1987;59:678–682.
78 American Society of Anesthesiologists Task Force on Preoperative Fasting. Practice guidelines for preoperative fasting and the use of pharmacologic agents to reduce the risk of pulmonary aspiration: Application to healthy patients undergoing elective procedures. Anesthesiology. 1999;90:896–905.
79 Lindahl SG. Not only towards enhanced preoperative comfort. Anesth Analg. 2001;93:1091–1092.
80 Miller M, Wishart HY, Nimmo WS. Gastric contents at induction of anaesthesia: Is a 4-hour fast necessary? Br J Anaesth. 1983;55:1185–1188.
81 Phillips S, Hutchinson S, Davidson T. Preoperative drinking does not affect gastric contents. Br J Anaesth. 1993;70:6–9.
82 Strunin L. How long should patients fast before surgery? Time for new guidelines. Br J Anaesth. 1993;70:1–3.
83 Maltby JR, Sutherland AD, Sale JP, et al. Preoperative oral fluids: Is a five-hour fast justified prior to elective surgery? Anesth Analg. 1983;65:1112–1116.
84 Read MS, Vaughan RS. Allowing pre-operative patients to drink: Effects on patients’ safety and comfort of unlimited oral water until 2 hours before anaesthesia. Acta Anaesthesiol Scand. 1991;35:591–595.
85 Soreide E, Hausken T, Soreide JA, et al. Gastric emptying of a light hospital breakfast: A study using real time ultrasonography. Acta Anaesthesiol Scand. 1996;40:549–553.
86 Elkington KW. At the water’s edge: Where obstetrics and anesthesia meet. Obstet Gynecol. 1991;77:304–308.
87 Ludka LM, Roberts CC. Eating and drinking in labor. J Nurse Midwifery. 1993;38:199–207.
88 Broach J, Newton N. Food and beverages in labor: Part II. The effects of cessation of oral intake during labor. Birth. 1988;15:88–92.
89 de Souza DG, Doar LH, Mehta SH, et al. Aspiration prophylaxis and rapid sequence induction for elective cesarean delivery: Time to reassess old dogma? Anesth Analg. 2010;110:1503–1505.
90 McKay S, Mahan C. How can aspiration of vomitus in obstetrics best be prevented? Birth. 1998;15:222–235.
91 Chestnut DH, Cohen SE. At the water’s edge: Where obstetrics and anesthesia meet. Obstet Gynecol. 1991;77:965–967.
92 Crawford JS. How can aspiration of vomitus in obstetrics best be prevented? Commentary: Setting the record straight. Birth. 1988;15:230–235.
93 Ng A, Smith G. Gastroesophageal reflux and aspiration of gastric contents in anesthetic practice. Anesth Analg. 2001;93:494–513.
94 Stone SB. Efficacy of cimetidine in decreasing the acidity and volume of gastric contents: Inadequacy of nasogastric suction and endotracheal intubation [letter]. J Trauma. 1981;21:996–997.
95 Satiani B, Bonner JT, Stone HH. Factors influencing intraoperative gastric regurgitation. Arch Surg. 1978;113:721–723.
96 Hardy J-F. Large volume gastroesophageal reflux: A rationale for risk reduction in the perioperative period. Can J Anaesth. 1988;35:162–173.
97 Dotson RG, Robinson RG, Pingleton SK. Gastroesophageal reflux with nasogastric tubes: Effect of nasogastric tube size. Am J Respir Crit Care Med. 1994;149:1659–1662.
98 Salem MR, Joseph NJ, Heyman HJ, et al. Cricoid compression is effective in obliterating the esophageal lumen in the presence of a nasogastric tube. Anesthesiology. 1985;63:443–446.
99 Khawaja IT, Buffa SD, Brandstetter RD. Aspiration pneumonia. Postgrad Med. 1992;92:165–168.
100 Brock-Utne J. Gastroesophageal reflux and aspiration of gastric contents. Anesth Analg. 2003;94:762–763.
101 Ong BY, Palahniuk RJ, Cumming M. Gastric volume and pH in out-patients. Can Anaesth Soc J. 1978;25:36–39.
102 Taylor WJ, Champion MC, Barry AW, et al. Measuring gastric contents during general anaesthesia: Evaluation of blind gastric aspiration. Can J Anaesth. 1989;36:51–54.
103 Hardy J-F, Plourde G, Lebrun M, et al. Determining gastric contents during general anaesthesia: Evaluation of two methods. Can J Anaesth. 1987;34:474–477.
104 El-Orbany M, Connolly LA. Rapid sequence induction and intubation: Current controversy. Anesth Analg. 2010;111:1318–1325.
105 Ruben H, Knudsen EJ, Carugati G. Gastric inflation in relation to airway pressure. Acta Anesthesiol Scand. 1961;5:107–114.
106 Sellick BA. Cricoid pressure to control regurgitation of stomach contents during induction of anaesthesia. Lancet. 1961;2:404–406.
107 Kopka A. The introduction of cricoid pressure. Anaesthesia. 2002;57:827.
108 Herman NL, Carter B, Van Decar TK. Cricoid pressure: Teaching the recommended level. Anesth Analg. 1996;83:859–863.
109 Vanner RG, O’Dwyer JP, Pryle BJ, et al. Upper oesophageal sphincter pressure and the effect of cricoid pressure. Anaesthesia. 1992;47:95–100.
110 Stept WJ, Safar P. Rapid induction-intubation for prevention of gastric-content aspiration. Anesth Analg. 1970;49:633–636.
111 Meek T, Vincent A, Duggen JE. Cricoid pressure: Can protective force be sustained? Br J Anaesth. 1998;80:672–674.
112 Thwaites AJ, Rice CP, Smith I. Rapid sequence induction: A questionnaire survey of its routine conduct and continued management during a failed intubation. Anaesthesia. 1999;54:376–381.
113 Schwartz DE, Matthay MA, Cohen NH. Death and other complications of emergency airway management in critically ill adults: A prospective investigation of 297 tracheal intubations. Anesthesiology. 1995;82:367–376.
114 Jackson SH. Efficacy and safety of cricoid pressure needs scientific validation. Anesthesiology. 1996;84:751–752.
115 Ralph SJ, Wareham CA. Rupture of the oesophagus during cricoid pressure. Anaesthesia. 1991;46:40–41.
116 Hocking G, Roberts FL, Thew ME. Airway obstruction with cricoid pressure and lateral tilt. Anaesthesia. 2001;56:825–828.
117 Saghaei M, Masoodifar M. The pressor response and airway effects of cricoid pressure during induction of general anesthesia. Anesth Analg. 2001;93:787–790.
118 Hartsilver EL, Vanner RG. Airway obstruction with cricoid pressure. Anaesthesia. 2000;55:208–211.
119 Asai T, Barclay K, Power I, et al. Cricoid pressure impedes placement of the laryngeal mask airway. Br J Anaesth. 1995;74:521–525.
120 Harry RM, Nolan JP. The use of cricoid pressure with the intubating laryngeal mask. Anaesthesia. 1999;54:656–659.
121 MacG Palmer JH, Ball DR. The effect of cricoid pressure on the cricoid cartilage and vocal cords: An endoscopic study in anaesthetised patients. Anaesthesia. 2000;55:263–268.
122 Vanner RG, Clarke P, Moore WJ, et al. The effect of cricoid pressure and neck support on the view at laryngoscopy. Anaesthesia. 1997;52:896–900.
123 Hillman DR, Platt PR, Eastwood PR. The upper airway during anaesthesia. Br J Anaesth. 2003;91:31–39.
124 Asai T, Murao K, Shingu K. Cricoid pressure applied after placement of laryngeal mask impedes subsequent fibreoptic tracheal intubation through mask. Br J Anaesth. 2000;85:256–261.
125 Smith CE, Boyer D. Cricoid pressure decreases ease of tracheal intubation using fibreoptic laryngoscopy (WuScope System). Can J Anaesth. 2002;49:614–619.
126 Riad W, Ansari T. Effect of cricoid pressure on the laryngoscopic view by Airtraq in elective caesarean section: A pilot study. Eur J Anaesth. 2009;26:981–982.
127 Turgeon AG, Nicole PC, Trepanier CA, et al. Cricoid pressure does not increase the rate of failed intubation by direct laryngoscopy in adults. Anesthesiology. 2005;102:315–319.
128 Ciresi SA. Gastrointestinal pharmacology review and anesthetic application to the combat casualty. Mil Med. 1989;154:555–559.
129 Nimmo WS. Aspiration of gastric contents. Br J Hosp Med. 1985;34:176–179.
130 Rabey PG, Murphy PJ, Langton JA, et al. Effect of the laryngeal mask airway on lower oesophageal sphincter pressure in patients during general anaesthesia. Br J Anaesth. 1992;69:346–348.
131 Barish CF, Wu WC, Castell DO. Respiratory complications of gastroesophageal reflux. Arch Intern Med. 1985;145:1882–1888.
132 McCammon RL. Prophylaxis for aspiration pneumonitis. Can Anaesth Soc J. 1986;33:S47–S53.
133 Randell T, Saarvinaara L, Oikkonen M, et al. Oral atropine enhances the risk for acid aspiration in children. Acta Anaesthesiol Scand. 1991;35:651–653.
134 Todd JG, Nimmo WS. Effect of premedication on drug absorption and gastric emptying. Br J Anaesth. 1983;55:1189–1193.
135 Cohen SE, Jasson J, Talafre M-L, et al. Does metoclopramide decrease the volume of gastric contents in patients undergoing cesarean section? Anesthesiology. 1984;61:604–607.
136 Manchikanti L, Grow JB, Colliver JA, et al. Bicitra (sodium citrate) and metoclopramide in outpatient anesthesia for prophylaxis against aspiration pneumonitis. Anesthesiology. 1985;63:378–384.
137 Shaughnessy AF. Potential uses for metoclopramide. DICP. 1985;19:723–728.
138 Splinter WM, Schreiner MS. Preoperative fasting in children. Anesth Analg. 1999;89:80–89.
139 Christensen S, Farrow-Gillespie A, Lerman J. Effects of ranitidine and metoclopramide on gastric fluid pH and volume in children. Br J Anaesth. 1990;65:456–460.
140 Deehan S, Dobb GJ. Metoclopramide-induced raised intracranial pressure after head injury. J Neurosurg Anesthesiol. 2002;14:157–160.
141 Orr DA, Bill KM, Gillon KRW, et al. Effects of omeprazole, with and without metoclopramide, in elective obstetric anaesthesia. Anaesthesia. 1993;48:114–119.
142 Berne JD, Norwood SH, McAuley CE, et al. Erythromycin reduces delayed gastric emptying in critically ill trauma patients: A randomized, controlled trial. J Trauma. 2002;53:422–425.
143 Boivin MA, Carey MC, Levy H. Erythromycin accelerates gastric emptying in a dose-response manner in healthy subjects. Pharmacotherapy. 2003;23:5–8.
144 Eyler SW, Cullen BF, Murphy ME, et al. Antacid aspiration in rabbits: A comparison of Mylanta and Bicitra. Anesth Analg. 1982;61:288–292.
145 Kuster M, Naji P, Gabi K, et al. Die intraoperative, direkte und kontinuirliche pH-Messung im Magen nach Vorbehandlung mit Ranitidin oder Natriumcitrat. Anaesthesist. 1989;38:59–64.
146 Colman RD, Frank M, Loughnan BA, et al. Use of i.m. ranitidine for the prophylaxis of aspiration pneumonitis in obstetrics. Br J Anaesth. 1988;61:720–729.
147 Joyce TH. Prophylaxis for pulmonary acid aspiration. Am J Med. 1987;83:S46–S52.
148 Ormezzano X, Francois TP, Viaud JY, et al. Aspiration pneumonitis prophylaxis in obstetric anaesthesia: Comparison of effervescent cimetidine-sodium citrate mixture and sodium citrate. Br J Anaesth. 1990;64:503–506.
149 Papadimitriou L, Kandiloros A, Lakiotis K, et al. Protecting against the acid aspiration syndrome in adult patients undergoing emergency surgery. Hepatogastroenterology. 1992;39:560–561.
150 Smith CL, Bardgett DM, Hunter JM. Haemodynamic effects of the i.v. administration of ranitidine in the critically ill patient. Br J Anaesth. 1987;59:1397–1402.
151 Rout CC, Rocke DA, Gouws E. Intravenous ranitidine reduces the risk of acid aspiration of gastric contents at emergency cesarean section. Anesth Analg. 1993;76:156–161.
152 McAllister JD, Moote CA, Sharpe MD, et al. Random double-blind comparison of nizatidine, famotidine, ranitidine, and placebo. Can J Anaesth. 1990;37:S22.
153 Goudsouzian NG, Young ET. The efficacy of ranitidine in children. Acta Anaesthesiol Scand. 1987;31:387–390.
154 Vila P, Valles J, Canet J, et al. Acid aspiration prophylaxis in morbidly obese patients: Famotidine vs. ranitidine. Anaesthesia. 1991;46:967–969.
155 Maltby JR, Elliott RH, Warnell I, et al. Gastric fluid volume and pH in elective surgical patients: Triple prophylaxis is not superior to ranitidine alone. Can J Anaesth. 1990;37:650–655.
156 Maekawa N, Nishina K, Mikawa K, et al. Comparison of pirenzepine, ranitidine, and pirenzepine-ranitidine combination for reducing preoperative gastric fluid acidity and volume in children. Br J Anaesth. 1998;80:53–57.
157 Hong JY. Effects of metoclopramide and ranitidine on preoperative gastric contents in day-case surgery. Yonsei Med J. 2006;47:315–318.
158 Paranjothy S, Griffiths JD, Broughton KH, et al. Interventions at caesarean section for reducing the risk of aspiration pneumonitis [review]. Cochrane Database Syst Rev. (1):2010. CD004943
159 Trekova NA, Iavorovskii AG, Shmyrin MM, et al. Use of H2-receptor blocker famotidine in anesthesiology for cardiopulmonary bypass surgery. Anesteziol Reanimatol. 2002;1:16–19.
160 Wajima Z, Shitara T, Inoue T, et al. The effect of previous administration of nizatidine on the neuromuscular effects of vecuronium and the effect of nizatidine on gastric secretion. Anaesth Intensive Care. 2000;28:46–48.
161 Bouly A, Nathan N, Feiss P. Comparison of omeprazole with cimetidine for prophylaxis of acid aspiration in elective surgery. Eur J Anaesthesiol. 1993;10:209–213.
162 Ewart MC, Yau G, Gin T, et al. A comparison of the effects of omeprazole and ranitidine on gastric secretion in women undergoing elective caesarean section. Anaesthesia. 1990;45:527–530.
163 Moore J, Flynn RJ, Sampaio M, et al. Effect of single-dose omeprazole on intragastric acidity and volume during obstetric anaesthesia. Anaesthesia. 1989;44:559–562.
164 Nishina K, Mikawa K, Takao Y, et al. A comparison of rabeprazole, lansoprazole, and ranitidine for improving preoperative gastric fluid property in adults undergoing elective surgery. Anesth Analg. 2000;90:717–721.
165 Pisegna JR, Karlstadt RG, Norton JA, et al. Effect of preoperative intraveonous pantoprazole in elective-surgery patients: A pilot study. Dig Dis Sci. 2009;54:1041–1049.