Chapter 148
Renovascular Disease
Aneurysms and Arteriovenous Fistulae
Keith D. Calligaro, Matthew J. Dougherty
Renal artery aneurysms and renal arteriovenous fistulae (AVFs) are rare entities, but they are encountered frequently enough that vascular surgeons need to be well acquainted with the natural history, diagnosis, and management of these lesions. Endovascular interventions represent the newest advances in the treatment of some of these lesions. Aneurysms and AVFs are discussed separately because they rarely occur concomitantly, and their clinical course and treatment differ.
Renal Artery Aneurysms
Epidemiology
Even in referral centers, few vascular surgeons have extensive experience with the clinical management of renal artery aneurysms.1,2 Autopsy studies have revealed an incidence of 0.01% to 0.09%, which is probably an underestimation, because renal artery aneurysms may be small, intrarenal, or not specifically sought.1,3 In two angiography studies, renal artery aneurysms were documented in 0.73% (7-965) to 0.97% (83-8525) of arteriograms; in a more recent computed tomographic (CT) angiography study, the incidence was 0.7% (6-862).4–6 Conversely, these reports might overestimate the prevalence of these lesions. If renal artery aneurysms were present in almost 1% of patients undergoing abdominal aortography, vascular surgeons would be expected to have far greater experience diagnosing and treating these lesions than has been reported to date. At Pennsylvania Hospital (Philadelphia, Penn), we documented renal artery aneurysms in only 0.12% (1-845) of abdominal aortograms. Renal artery aneurysms are bilateral in about 10% of cases.1,5 If fibrodysplastic cases are omitted, there is an equal incidence in males and females.1,2,7
Because of the lack of controlled data, controversy persists regarding the indications for repair of asymptomatic renal artery aneurysms. The optimal method of repair is also controversial. Types of renal artery aneurysms, their clinical manifestations, indications for repair, and techniques of both traditional surgical and newer endovascular interventions are reviewed here.
Pathogenesis
Types of renal artery aneurysms include true (saccular and fusiform), false, dissecting, and intrarenal aneurysms.
True Aneurysms
More than 90% of true renal artery aneurysms are extraparenchymal.2,8–10 The peak incidence is in patients between the ages of 40 and 60 years. Stanley2 and Stanley et al10 have suggested that true aneurysms are probably due to either atherosclerosis or a congenital defect. Although arteriosclerotic changes have been identified in most aneurysms in patients with multiple lesions, this is not a uniform finding, suggesting that arteriosclerosis may not be the most important factor in the genesis of renal artery aneurysms. These aneurysms are more likely due to a congenital medial degenerative process with weakness of the elastic lamina.7,10 Lesions typically occur at the primary or secondary renal artery bifurcations and are rarely confined only to the main trunk of the renal artery. As discussed later, this finding makes surgical repair challenging.
Approximately 75% of true renal artery aneurysms are saccular. This type of renal artery aneurysm is usually less than 5 cm in diameter,11 although some as large as 9 cm have been reported.9,12,13 Saccular aneurysms occur almost invariably at the main renal artery bifurcation.14 Fusiform aneurysms are usually associated with atherosclerosis or are a result of a post-stenotic dilatation distal to a hemodynamically significant renal artery stenosis, the latter of which results from atherosclerosis or fibromuscular disease.7,8,14,15 Fusiform aneurysms are generally less than 2 cm in diameter and usually affect the main renal artery trunk.8
Arterial fibrodysplasia is often a direct contributor to the development of an aneurysm.2,10 Medial fibroplasia is typically associated with multiple stenoses and post-stenotic dilatation of the distal two thirds of the renal artery. Renal artery aneurysms in association with fibromuscular dysplasia are generally only a few millimeters in diameter. The typical angiographic appearance of a renal artery involved with medial fibroplasia is a “string of beads.” Larger aneurysms can also occur, however, and in one study, renal artery macroaneurysms were found in 9.2% of adults with fibromuscular dysplasia.10
A rare cause of renal artery aneurysms is Ehlers-Danlos’ syndrome. This disorder is associated with extreme arterial fragility and spontaneous rupture, and is discussed in Chapter 78.16
False Aneurysms (Pseudoaneurysms)
False aneurysms of the renal artery arise from blunt or penetrating trauma, and occasionally from iatrogenic causes such as renal artery catheterization or after nephrectomy. They represent contained ruptures of the renal artery, with only inflammatory and fibrous tissue encasing the leak.
Dissections
Spontaneous dissections confined to the renal artery that do not arise from the adjacent aorta are rare; however, primary dissections causing pseudoaneurysms affect the renal arteries more than any other peripheral artery.2,14,17–20 Poutasse14 and Stanley et al10 reported that 14 of 57 cases of renal artery aneurysms were due to spontaneous dissection. An intimal defect of the renal artery due to atherosclerosis is probably the underlying cause of spontaneous renal artery dissection causing aneurysms, along with dysplastic renovascular disease and trauma.8 The incidence of dissection in patients with fibrodysplastic renal arteries ranges from 0.5% to 9.0%.2,17 Dissection often extends into the branches of the renal artery and may pose particularly challenging reconstruction problems.
Traumatic renal artery dissection can occur secondary to blunt abdominal trauma or catheter-induced injury. Blunt trauma accounts for the higher prevalence of dissection in men and is more likely to result in right-sided injuries, possibly because of ptosis-related physical stresses affecting the renal pedicle.2 Blunt trauma can cause renal artery dissection by either severe stretching of the artery, with fracture of the intima, or compression of the artery against the vertebra. Renal artery dissection caused by guide wires or catheters can occur, but is rare, having been observed in only 4 of 2200 selective renal artery arteriograms.17
Intrarenal Aneurysms
Less than 10% of renal artery aneurysms are intraparenchymal.9,10 Intrarenal aneurysms are usually multiple and may be congenital, are associated with collagen vascular disease, or are posttraumatic. They may be associated with AVFs, possibly as a result of spontaneous closure of a fistula. Intrarenal aneurysms can occur with polyarteritis nodosa and are usually in the renal cortex.21,22
Clinical Manifestations and Diagnosis
Most renal artery aneurysms are asymptomatic and are found on imaging studies, such as arteriography, ultrasonography, or CT performed to investigate other intra-abdominal pathology.2,13,23 Magnetic resonance angiography (MRA) can also delineate renal artery aneurysms.99 Clinical manifestations of renal artery aneurysms include rupture, hypertension, pain, and hematuria. In one series, only 11 of 32 patients (34%) who underwent surgery for renal artery aneurysms presented with symptoms.13
The most dreaded complication of renal artery aneurysm is rupture. Patients with this complication present with manifestations similar to those of other intra-abdominal arterial ruptures, including syncope, abdominal, or flank pain, abdominal distention, and possibly, a pulsatile mass. Occasionally, an intact renal artery aneurysm presents with abdominal or flank pain, discomfort, or fullness—symptoms that are presumed to reflect acute aneurysmal expansion. Intuitively, a stable renal artery aneurysm should not cause pain or symptoms, similar to a stable abdominal aortic aneurysm, so the clinician should hesitate to attribute chronic abdominal pain to a stable, noninflammatory renal artery aneurysm.
Renal artery aneurysms may be associated with severe hypertension. Macroaneurysms were found in 2.5% of arteriograms performed for the evaluation of hypertension.10 Renal artery aneurysms may cause renovascular hypertension by distal embolization with segmental hypoperfusion, and renin-mediated vasoconstriction and fluid retention. Compression of an adjacent renal artery branch or luminal stenosis due to extensive thrombus may also lead to renin-mediated hypertension. Frequently, significant renal artery stenosis causes a post-stenotic fusiform aneurysm, and the renal artery stenosis is responsible for the hypertension. Saccular and intrarenal aneurysms are much less likely to be associated with hypertension. Nonetheless, caution should be exercised in attributing the cause of hypertension to a renal artery aneurysm, especially if intervention is being considered. Just as renal artery stenosis without an aneurysm is currently less likely being treated with intervention and more likely treated with medical management, the clinician should hesitate to recommend endovascular or surgical intervention to treat hypertension potentially due to a renal artery aneurysm. Many renal artery aneurysms may have been unjustifiably treated in the past with ill-advised over-aggressive intervention.
Patients with renal artery aneurysms caused by dissection may present with severe flank pain, hematuria, or acute hypertension, although most dissections are asymptomatic. An intravenous pyelogram may reveal nonfunction or diminished function of the involved kidney, but this is rarely the first test ordered, unless urolithiasis is considered a likely cause of the symptoms. CT angiography or MRA is essential to detect dissection.
Intrarenal aneurysms may rupture into calices.3 In addition to pain, microscopic or gross hematuria may occur. Similarly, renal artery aneurysms may rarely cause obstruction of the collecting system. Although main renal artery aneurysms may be large, they are usually not near enough to the caliceal system to cause obstruction. Intrarenal aneurysms tend to be too small to cause significant collecting duct obstruction. However, a 9-cm renal artery aneurysm has been documented to cause hydronephrosis.12
Indications for Intervention
Indications to repair a renal artery aneurysm are related to the risk of rupture, hypertension, acute dissection, and other clinical symptoms.
Rupture and Prevention of Rupture
Rupture of a renal artery aneurysm is an indication for emergency intervention, as it is for virtually any arterial aneurysm. Probably less than 3% of renal artery aneurysms rupture.2,10 This complication is associated with a mortality rate of approximately 10% in men and nonpregnant women.2,10,24,25 In a hemodynamically stable patient, an emergent CT scan may reveal the pathology and allow the surgeon to plan the operative repair. However, if an elderly patient presents to the emergency department with abdominal pain, a tender, distended abdomen, and severe hypotension that does not respond to fluid resuscitation, emergency exploration for a presumptive ruptured abdominal aneurysm may be indicated.
Prevention of rupture is the most common indication for intervention in cases of asymptomatic renal artery aneurysms. Traditionally, repair has been recommended for renal artery aneurysms greater than 2 cm in diameter,8,15 although this recommendation may very likely be over-aggressive, and more conservative guidelines may very well be indicated. The likelihood of rupture of a renal artery aneurysm is controversial because the natural history has not been delineated. Most reports are retrospective reviews of incidentally discovered intact renal artery aneurysms in autopsy series or collections of ruptured aneurysms that lack full details concerning their size and the presence or absence of calcification. Harrow and Sloane26 reported one of the highest rates of rupture of renal artery aneurysms, noting 14 ruptures in 100 cases. In another series of 126 renal artery aneurysms, 6 ruptured.27 Many authorities believe that there are no good data to support the belief that the larger the renal artery aneurysm, the more likely it is to rupture.1,2,8,10,28
Most other series of asymptomatic renal artery aneurysms in men and nonpregnant women report a much lower incidence of rupture. Only 1 of 62 patients with aneurysms 4 cm in diameter or smaller ruptured after follow-up from 1 to 17 years.29 None of 19 small aneurysms in another series ruptured.24 A group of 21 patients was observed for an average of 3 years without rupture.23 In another series of 18 patients with renal artery aneurysms less than 2.6 cm, who were followed for 1 to 16 years, none of the aneurysms ruptured.1 There were no ruptures in a series of 32 patients (who eventually underwent surgery) with renal artery aneurysms that ranged from 0.7 to 9 cm.13 Of 83 renal artery aneurysms found on arteriography and followed up without surgery,28 none ruptured or became symptomatic after a mean of 4.3 years.5 In a pooled analysis, there were no ruptures in more than 200 renal artery aneurysms observed for up to 17 years.7 In a recent large series of 59 renal artery aneurysms, 30 were treated conservatively and followed (all <2.0 cm), whereas 29 were treated (most with ex-vivo repair).31 Because of the very low risk of rupture and the relatively high rate of significant postoperative complications (approximately 55%), we recommend that a more conservative approach be used for these cases, and that a 2-cm guideline for intervention is too aggressive and unwarranted. There was an obvious selection bias in the follow-up of many of these aneurysms (i.e., small size), and many of the larger aneurysms were repaired.
Besides size, other factors may play a role in the consideration of elective surgery for asymptomatic renal artery aneurysms. Calcification of the aneurysm has been thought to protect against rupture. Poutasse15 suggested that a heavily calcified renal artery aneurysm may be less likely to rupture than a noncalcified or minimally calcified one. In a review of cases through 1959, 14 of 100 noncalcified aneurysms ruptured.26 In a more recent series, 15 of 18 ruptured renal artery aneurysms were noncalcified.30 However, in a series of 62 solitary aneurysms less than 4 cm in diameter, one third were not calcified, and only 1 aneurysm in the entire series ruptured in 1 to 17 years of follow-up.25 Because of these conflicting data, some authorities believe that the presence or absence of calcification is not relevant when predicting the risk of rupture.2
Most authorities agree, however, that pregnancy is associated with a significantly increased risk of rupture of a renal artery aneurysm.2,10,13,32 Pregnancy may increase the risk of rupture because of the hyperdynamic state, with increased blood volume and cardiac output; hormonal influences on the aneurysm; and increased intra-abdominal pressure due to the gravid uterus.1,10 Cohen and Shamash32 reported 18 cases of rupture during pregnancy. In another series of 18 patients who underwent surgery for renal artery aneurysms, the only two ruptures were in women at childbirth; both of these aneurysms measured only 1 cm in diameter.1 In a review of 43 ruptured renal artery aneurysms, 81% occurred in women; 21 of the 35 women in this series were younger than 40 years old, and 18 were pregnant. Of the 18 aneurysms of known size, 3 ruptured when they were less than 2 cm.38
Of note, rupture of renal artery aneurysms in pregnancy has been associated with a maternal mortality rate of 55% and a fetal death rate of 85%.32,33 Risk of renal artery rupture is small, however, even in pregnant women. In a series of 19,600 autopsies of pregnant women, no ruptured renal artery aneurysms were found.27 This report did not indicate the number of unruptured renal artery aneurysms found in this population, so the risk of rupture remains uncertain. Regardless, we agree with others that there are enough data to support an aggressive surgical or endovascular approach for pregnant women with renal artery aneurysms of any size.
Essentially, all recent-onset false renal artery aneurysms should be repaired because of the high likelihood of rupture.8 In the rare case of a chronic, contained rupture of a small false aneurysm that is found months or years later, in which the pseudoaneurysm has thrombosed, careful follow-up is probably all that is warranted. Similarly, renal artery aneurysms due to fibrodysplastic disease may be associated with a higher risk of rupture because of the thin-walled nature of these aneurysms, although firm data are lacking.8 Certainly, renal artery aneurysms in men or in women beyond childbearing age that are less than 2 cm in diameter and associated with fibrodysplastic disease should be studied closely.8
In summary, our recommendation concerning the elective repair of asymptomatic renal artery aneurysms in men and in women beyond childbearing age is based on the data just presented and on the well-documented history of other abdominal arterial aneurysms. General guidelines for the repair of asymptomatic abdominal aneurysms include (1) infrarenal aortic aneurysms greater than 5 to 5.5 cm in diameter, (2) common iliac aneurysms greater than 3 cm, and (3) splenic artery aneurysms greater than 3 cm.22 Surgery is recommended for visceral artery aneurysms of any size.34 Although various hemodynamic factors may play a role in other intra-abdominal aneurysms, and despite the relative paucity of data suggesting a high risk of rupture, it seems prudent to recommend the repair of renal artery aneurysms greater than 3 cm in diameter in good-risk patients when there is reasonable certainty that nephrectomy will not be required.26,28 This guideline remains controversial, and others have taken a more conservative approach, reserving repair for aneurysms greater than 4 cm.35 As previously mentioned, any renal artery aneurysm in women of childbearing age should be repaired.
Hypertension
Although the prevalence of hypertension in patients with renal artery aneurysms is approximately 80% in several series, there is no conclusive evidence that the aneurysms themselves are the direct cause of hypertension, unless there is an associated stenosis or compression of an adjacent artery.1,10,13 In a series of 39 patients with renal artery aneurysms, 26 had diastolic hypertension, but only 9 (23%) patients had hypertension that was of renovascular origin.1 In a more recent series of 16 patients with extraparenchymal renal artery aneurysms, 75% had renovascular hypertension.37 The indication for surgical intervention for renovascular hypertension due to renal artery stenosis, secondary to atherosclerosis, continues to be the failure of medical management—namely, diastolic blood pressure greater than 90 to 100 mm Hg despite three antihypertensive medications—and the same criterion should probably be applied when a renal artery aneurysm is present. If the primary indication to repair a renal artery aneurysm is poorly controlled hypertension, we do not hesitate to obtain a nephrology consult to be sure there are firm indications for intervention. Both the stenotic artery and the aneurysm must be repaired. Our current evaluation of these patients relies primarily on the clinical scenario, the exclusion of other causes of secondary hypertension, the documentation of significant renal artery stenosis, and occasionally, the use of captopril renal scans.38
Dissection
Emergent intervention is required for dissections that cause renal artery aneurysms and threaten the viability of the kidney. Nephrectomies are frequently required, however, because of the extensive damage to the renal branch vessels and the limited time available to salvage a previously healthy kidney that cannot tolerate prolonged periods of ischemia. If hypertension is the only manifestation of a chronic dissection, and the hypertension is well controlled by blood pressure medications, or if the patient is asymptomatic and a renal artery dissection is found incidentally (without an associated aneurysm), surgery is probably not justified.10
Other Clinical Manifestations
If a patient with an intact renal artery aneurysm, as documented by CT or magnetic resonance imaging (MRI), is symptomatic—that is, experiences abdominal or flank pain or fullness—repair may be indicated. Acute onset of symptoms is more worrisome than a patient referred for months of abdominal pain or fullness, unless the renal artery aneurysm is very large. It is difficult to attribute abdominal pain to a 2-cm diameter renal artery aneurysm, and other causes need to be thoroughly investigated. Symptoms may be a harbinger of impending rupture. Embolization to the renal parenchyma may also account for these symptoms.9
Treatment: Medical, Endovascular, Surgical
Repair of a Ruptured Renal Artery Aneurysm
If emergent surgery is required for a ruptured renal artery aneurysm, a midline approach and supraceliac aortic control are generally required. A sizable juxtarenal hematoma does not allow safe aortic exposure and clamping immediately above the renal arteries. If proximal control of the renal artery itself can be obtained, the supraceliac clamp can then be removed. If the bleeding is quickly controlled, and the patient is clearly hemodynamically stable, and if the proximal and distal renal arteries lend themselves to a relatively quick and straightforward bypass, consideration can be given to reconstruction. In most cases, however, nephrectomy is required because of the instability of the patient, the prolonged ischemia of the kidney, and the technical and time-consuming nature of surgical repair with a bypass.2,10,14,29 If the aneurysm extends into the renal parenchyma or if a “bench” repair of the kidney is required, the patient is generally best treated by nephrectomy, as long as the contralateral kidney is intact with normal function. It is possible that a stable patient with a ruptured true or false renal artery aneurysm can be treated with newer endovascular techniques. Use of a stent graft may be the preferred treatment for lesions not involving the distal renal branches, as reported by Bloemsma et al.39 Routh et al40 reported thrombosis of a leaking saccular aneurysm using Gianturco coils, thrombin, and bucrylate.
Elective Repair of Renal Artery Aneurysm
Open surgical and endovascular repair are options to repair renal artery aneurysms. Even in the case of elective surgery, repair of a renal artery aneurysm is usually more challenging than revascularization for renal artery stenosis. Most renal artery aneurysms extend past the bifurcation of the main renal arteries and frequently extend into the renal parenchyma. Associated renal artery stenosis may need to be repaired in conjunction with the aneurysm. For in-situ repairs of a renal artery aneurysm, the left kidney can be exposed through a retroperitoneal approach with a transverse left supraumbilical incision. The right kidney can be exposed through a transperitoneal approach with a Kocher maneuver to reflect the right colon and duodenum medially or with a subcostal incision.
Several methods have been used to repair renal artery aneurysms. The most straightforward technique for saccular aneurysms involves aneurysmorrhaphy with primary repair or patching. In three combined series of patients who underwent surgical repair of renal artery aneurysms, approximately one third (6 of 18, 3 of 10, and 6 of 23) of the aneurysms were able to be repaired in this manner.1,9,13 If this technique is not possible, we and others prefer autologous tissue bypasses, such as a saphenous vein bypass, if the graft can be anastomosed to the distal part of the main trunk of the renal artery or to the most proximal branches.8,13 The most common renal arterial reconstruction is an end-to-side anastomosis of a small renal artery branch to the main renal artery, or a side-to-side anastomosis of two small renal arteries to create a common inflow channel with a single, larger diameter lumen, which can then be anastomosed to the renal artery or vein. Because the small branches of the main renal artery are often involved with the aneurysm, a branched autologous graft is preferred to reconstruct these lesions. The internal iliac artery is an excellent choice in these reconstructions because of its multiple small side branches.28,41 Alternatively, the saphenous vein also functions well. The proximal anastomosis of the graft is usually the infrarenal aorta. Useful alternative reconstructions include a splenorenal bypass for a left-sided renal artery aneurysm and hepatorenal bypass for a right-sided aneurysm.
If multiple branch vessels are involved, and especially if the cause of the renal artery aneurysm is dissection resulting in a friable vessel, extracorporeal or bench surgery may be required.42,43 This technique is recommended when renal ischemia is projected to exceed 45 minutes or when exposure of small renal branches is required. Ex-vivo surgery requires nephrectomy, followed by hypothermic perfusion of the kidney with a heparinized renal preservation solution. The kidney can then be autotransplanted to its original bed, as Dean et al44 and Crutchley et al45 prefer, or to the iliac fossa. For renal autotransplantation into the iliac fossa, a flank incision with a retroperitoneal approach is used for exposure of the kidney, ureter, and iliac artery. Gonadal and adrenal veins are divided to obtain an adequate length of renal vein. If the reconstruction can be safely performed by placing the kidney on the anterior abdominal wall, the ureter does not need to be divided. The procedure is occasionally best performed at a separate table after dividing the ureter and removing the kidney from the operative field. Perfusion is carried out through the main renal artery to preserve the kidney while selected branches are individually repaired and other branches are perfused. The kidney may be perfused with a heparinized crystalloid solution, such as Collins solution or lactated Ringer’s solution with heparin 1000 U/L with 12.5 g of mannitol, while the kidney is wrapped with gauze and placed in a chilled solution at 4° C.8,40,46 The use of continuous pulsatile perfusion is controversial.40 Ex-vivo repair is also discussed in Chapter 145.
When performed for proper indications by well-trained surgeons, repair of renal artery aneurysms should be associated with low morbidity and mortality.2,9,46,47 English et al48 reported a 1.7% perioperative mortality rate for surgical repair in 62 patients with 72 renal artery aneurysms, with a 4-year patency of 96% and cured or improved hypertension in three quarters of patients. Pfeiffer et al,49 from Germany, reported similar excellent long-term surgical results in a series of 94 patients. In another series of 12 patients operated on for renal artery aneurysms, there was no mortality, and only 1 patient required reoperation for ureteral stenosis.47 A more recent series of 26 renal artery aneurysms that required reconstruction of first- or second-order branches in 25 cases were repaired primarily with in-situ techniques. Autogenous vein bypasses resulted in a mortality of 0% with long-term patency of 95% at an average follow-up of 99 months (range, 1-300 months).50 Ex-vivo repairs have been shown to be safe and effective by Dean et al,44 Crutchley et al,45 and others.42 Murray et al41 reported a series of 11 patients with renal artery aneurysms successfully treated using ex-vivo repair. In another series of eight aneurysms, all were successfully repaired with the ex-vivo technique, without deaths or complications.49 In a review of ex-vivo repairs, postoperative mortality rates ranged from 0% to 9.6%.51 Use of bifurcated internal iliac artery autografts was also highly successful in a series of 11 patients, most with fibrodysplastic aneurysms, who were treated by in-situ or bench repair.41 Finally, in a series of 35 repairs of renal artery aneurysms treated by in-situ repair, ex-vivo repair, or nephrectomy, there was no mortality and only one postoperative graft occlusion.13 Recent technologic and surgical developments have also allowed less invasive surgical repair of complex renal artery aneurysms, and both laparoscopic and robot-assisted techniques have been described.52,53 A series of five renal artery aneurysms, albeit small-diameter ones measuring less than 3 cm, were treated with robot-assisted laparoscopic in-situ aneurysmectomy followed by revascularization without complications.54
An alternative approach to the treatment of renal artery aneurysms includes the use of endovascular techniques.28,55–57 Degenerative renal artery aneurysms have been treated with transcatheter embolization with detachable platinum coils that occlude the aneurysms, but maintain renal flow.56–58 One patient in whom a renal artery aneurysm occurred after percutaneous renal biopsy was also successfully treated by embolization.59 In a small series of endovascular repairs, Klein et al56 treated 12 renal artery aneurysms using selective endovascular embolization with nondetachable microcoils or Guglielmo’s detachable coils. Eight aneurysms were located in the bifurcation of the main renal artery, two were in the main renal artery, and two were intrarenal. All 12 aneurysms were successfully occluded, with only 2 minor complications. These authors concluded that endovascular treatment of renal artery aneurysms with microcoils is as safe as surgical treatment and less invasiave.56 In another series, 13 patients with renal artery aneurysms were successfully treated using a variety of endovascular techniques with combinations of coil embolization, liquid embolization, stenting, and stent grafts with no complications after an average follow-up of 43 months (range, 13-103 months).60 A larger series of 18 renal artery aneurysms were also treated with endovascular intervention.61 Sixteen of the 18 aneurysms were embolized utilizing adjunctive techniques to protect embolizing into the main renal artery. There was a 100% technical success rate, with only one aneurysm showing residual minor flow into the sac and with no long-term complications. Alternatively, several authors have recently reported using ethylene vinyl alcohol copolymer to ablate renal artery aneurysms.62,63 With this technique, the main renal artery is protected with an angioplasty balloon while the polymer is infused into the aneurysmal sac, achieving controlled exclusion. Flow-diverting stents have been used to treat six renal artery aneurysms by reducing flow in the aneurysm sac and promoting thrombosis while maintaining flow in the main artery and branches.64
Over the past 5 years there have also been more numerous reports of endovascular stent graft exclusion as definitive treatment for renal artery aneurysms. Many aneurysms extend to branch vessels, making this approach risky or ill-advised, but it may be ideal for aneurysms of the main renal artery not involving branches. Advances in stent graft technology, with smaller diameter devices and lower profile delivery systems, have made definitive treatment of renal artery aneurysms feasible. A review of the English literature analyzing use of stent grafts to treat renal artery aneurysms up to April 2010 yielded 22 cases with a technical success rate of 91% (20 cases) without any deaths or significant morbidity reported.65 Although most published series have been small, results have been excellent, and this approach will undoubtedly become more common as technology improves.66–70
Fibromuscular Dysplasia
Post-stenotic dilatation resulting from fibromuscular disease can be treated by balloon angioplasty of the stenotic lesion; in these cases, the primary indication for treatment is the stenotic lesion. When the lesion extends into the branches of the main renal artery, surgery can yield excellent results. Dean et al44 reported 24 patients with fibromuscular disease, many of whom had branch aneurysms; all but one did well.
Renal Arteriovenous Malformations and Fistulae
Arteriovenous malformations (AVMs) and AVFs are uncommon lesions that can be associated with hematuria, hypertension, renal dysfunction, high-output congestive heart failure, and even rupture. More than 200 cases have been reported since the first description in 1928.72 Fistulae may be congenital or acquired. Multiple diagnostic modalities are now available, although conventional selective arteriography remains critical in therapeutic interventions. Many asymptomatic lesions do not require treatment. In the past, symptomatic lesions were treated surgically, but endovascular treatment has now supplanted surgery in most cases.
Epidemiology and Pathogenesis
Congenital Arteriovenous Malformations
True congenital AVMs of the kidney are rare, with an incidence of only 0.04%.73 In a large series, only 1 congenital AVM was noted in 30,000 autopsies.82 These lesions represent approximately one fourth of all renal AVFs.74,75 The right kidney is involved more often than the left, and although multiple lesions can occur, a single focus is more common.42 The angiographic appearance of the lesions is similar to that of AVMs elsewhere, with large coils of dilated vessels. Piquet et al76 described a single artery feeding all but advanced cases; others described multiple connections of arterial branches and venous tributaries.72
An early “blush” is noted and correlates with the degree of arteriovenous shunting, which is variable. These lesions have been described as cirsoid or varix-like, and are generally focal and located in the renal medulla. AVMs are not neoplastic, but enlargement presumably can occur because of vessel dilatation and hypertrophy associated with high-flow volume from arteriovenous shunting. Symptomatic AVMs have been reported in pregnancy,72,77 and it is thought that the hyperdynamic state of the gravida leads to increased AVM flow and symptoms. Histologically, involved vessels have irregular fibrosis or intimal hyperplasia, as well as medial hypertrophy. Focal intraparenchymal hemorrhage may be noted in the lamina propria beneath the transitional epithelium of the collecting system.78
Acquired Arteriovenous Fistulae
Acquired AVFs may occur spontaneously. Spontaneous AVFs have been documented in association with fibromuscular dysplasia79 and are thought to develop when a dysplastic or aneurysmal renal artery erodes into a neighboring vein.80 This may also occur with renal malignancy, and significant arteriovenous shunting is a hallmark of renal cell carcinoma.81 With arteriography, it can be difficult to differentiate a renal malignancy from a congenital or acquired AVF, although CT and MRI generally reveal a mass distinct from the renal parenchyma in malignancy. As with AVMs, symptoms depend on the degree of shunting.
Traumatic AVFs are the most common lesions, accounting for more than 70% of all renal AVFs.72 These lesions may occur after nephrectomy, and are related to erosion of the arterial stump into the vein with mass ligature,82–84 after renal artery angioplasty,72 after blunt85 or penetrating83 trauma, after nephrostomy,85 and most commonly, after percutaneous renal biopsy. With the routine use of needle biopsy for the diagnosis of rejection in renal allografts, the incidence of acquired AVFs has grown, although only 1% to 2% of patients who undergo needle biopsy develop symptomatic AVFs.87,88 However, the true incidence of AVF is 15% to 18% when arteriography is routinely used.89,90 Similarly, Ozbek et al91 found AVFs in 8 of 64 patients (12.5%) monitored by color duplex ultrasonography, whereas only 5% developed AVFs in the study of Rollino et al.92 In the prospective study of Merkus et al,89 who used routine color duplex surveillance, 10% of patients who underwent biopsy developed AVFs. In their series, the development of AVFs correlated with bleeding dysfunction (elevated bleeding time or diminished platelet count), supporting the idea that inadequate intraparenchymal hemostasis led to the development of a channel between an artery and vein that subsequently enlarges. Others have reported fewer fistulae and bleeding complications with the use of automated small-gauge needles rather than the standard 14-gauge core biopsy technique.93,94
Clinical Presentation
The majority of both congenital and acquired AVFs do not produce clinical symptoms, and many lesions are noted incidentally in studies done for other reasons. Some patients are discovered to have AVFs when undergoing radiographic evaluation for vague abdominal or flank symptoms. The most common symptom of congenital AVM is hematuria, which occurs in 72% of cases.82 Hematuria occurs when subepithelial varices erode transitional epithelium into the collecting system. A dramatic presentation with massive hematuria can occur,36,77,95 although minor or microscopic hematuria is more common. Hypertension occurs in congenital AVMs and is also the primary abnormality in most acquired AVFs that are described as symptomatic. The hypertension is renin-mediated, based on diminished glomerular filtration pressure distal to the fistula because of arterial “steal.”75,86,96 Renal dysfunction is usually not noted except in transplant patients, in whom diminished parenchymal flow in the solitary kidney is not masked by a functional contralateral kidney.97
Although AVFs are generally painless, intermittent perilumbar discomfort has been reported in some patients.81,84 This discomfort is generally associated with hematuria and may represent as renal colic. Additionally, dyspnea and other symptoms of congestive heart failure may be the primary complaint in some patients; this is more common with acquired lesions, and only with those having a large communication between the artery and vein. This “high-output” type of heart failure manifests as tachycardia, left ventricular hypertrophy, and cardiomegaly, and a palpable thrill in the flank. A continuous abdominal bruit is a hallmark of acquired AVFs and is frequently noted with congenital AVMs as well. Retroperitoneal or intra-abdominal hemorrhage rarely occurs with AVMs and AVFs.36,98 However, patients with rupture present with severe abdominal and flank pain and shock, a clinical picture indistinguishable from ruptured abdominal aortic aneurysms.
Diagnosis
Excretory urography is performed in many patients presenting with hematuria or flank pain. A filling defect may be noted in the kidney, and dilated vessels can compress the collecting system, although these findings are not specific. Although intravenous pyelography is helpful to exclude more common causes of hematuria, such as nephrolithiasis, it is of limited use in the diagnosis of AVFs.
CT can usually define AVFs and AVMs within the kidney, but it is not always possible to differentiate these lesions from other hypervascular abnormalities (e.g., renal cell carcinoma). Similarly, radionuclide imaging can demonstrate early augmented perfusion, but differentiation from malignancy is not possible.99 In contrast, ultrafast CT with angiographic reconstruction has significantly improved the noninvasive imaging of AVFs and AVMs.100 Likewise, contrast-enhanced MRA allows three-dimensional reconstruction that can provide visualization not possible with conventional angiography.101
Color duplex imaging is also of growing importance in the diagnosis of AVMs and AVFs. Because it is inexpensive and noninvasive, it is the ideal study for screening purposes. Color duplex imaging has been used liberally to assess for AVFs after percutaneous renal biopsy.89,102,103 Marked turbulence is noted on color examination, and Doppler spectral analysis reveals an elevation of peak systolic flow velocity and a larger increase in end-diastolic flow velocity compared with the normal renal artery, with a resultant low resistive index.91,102
Arteriography has been the definitive diagnostic modality for renal AVMs and AVFs. Rapid opacification of the inferior vena cava is noted. Depending on the size of the fistula, the nephrogram may be diminished distal to the AVF. With congenital AVMs, multiple segmental and interlobar arteries communicate with varix-like veins, whereas a single arterial communication is generally present with acquired AVFs.82 Although it is a relatively expensive and invasive diagnostic study, arteriography alone offers the opportunity for definitive therapy.
Treatment: Medical, Surgical, Endovascular
The majority of both congenital and acquired AVFs do not cause symptoms and do not require treatment. However, patients may become symptomatic many years after the occurrence or diagnosis of AVFs and should be closely observed for the development of hypertension, hematuria, or high-output cardiac failure. Additionally, most AVFs occurring after percutaneous renal biopsy close spontaneously.86,89,91,103,104 This is particularly true of AVFs discovered early after biopsy by color duplex ultrasonography. Thus, periodic duplex surveillance along with clinical follow-up for the development of hypertension or renal insufficiency is indicated. If a postbiopsy AVF persists at 1 year, it is not likely to close spontaneously,87 although intervention should still be delayed until the development of symptoms.89,96 Spontaneous regression of AVMs unrelated to trauma has also been reported.105,106 Although hypertension related to an AVF may be readily controlled with angiotensin-converting enzyme inhibitors,79 the long-term effect on renal function is not known. In most published reports, patients with hypertension have undergone surgical or endovascular therapy; thus, the natural history of medically treated patients with hypertension secondary to AVFs remains undefined.
For patients with symptomatic AVFs, surgery has been the standard treatment for many years, and may still be the best option in certain circumstances.107,108 Except for very peripheral lesions, a transperitoneal approach is preferred to establish proximal arterial and venous control at the renal pedicle. Because of the frequent presence of thin-walled dilated veins and channels, surgery can be challenging. Ex-vivo repair of a large, complex renal artery aneurysm causing an AVF in a patient with fibromuscular dysplasia has been reported.109 With surgery, ligation of the feeding vessel or vessels alone is often not possible, and partial or total nephrectomy is often required. The resultant loss of functional renal mass, as well as the morbidity of the operation itself, makes endovascular treatment an attractive approach.
There are now more than 2 decades of experience with percutaneous arterial embolization therapy for congenital and acquired AVFs.110 Because renal arteries are “end arteries,” they are especially amenable to therapeutic occlusion. In earlier reports, autologous clot was used as the embolic material, but recanalization and recurrence of AVFs are possible, and thrombus has been supplanted by other materials, including gelatins, glues, alcohols, silicon, steel and platinum coils, and detachable balloons.* The development of coaxial catheter systems has allowed highly selective embolization, which can preserve renal function; loss of functional renal parenchyma is reportedly between 0% and 30% with modern techniques. In general, smaller AVFs are treated with glues or macroparticles, whereas coils and balloons are used for larger vessel fistulae (Fig. 148-1).113 Because Gelfoam (Pfizer, New York, NY) and autologous clot resorb, recanalization with recurrence of symptoms can occur in up to 50% of cases.114,115 For this reason, and because of the perception that microcoils are associated with less indiscriminate embolization than glues and alcohols, recent trends favor the use of microcoils even for smaller AVFs and AVMs.25,98,103 For very large AVF, covered stent exclusion has been described,116 and others have utilized bare metal stents to provide a barrier to coil extrusion.117 Complications of endovascular exclusion include coil embolization (generally to the pulmonary vasculature), nontarget emboli, and access site complications. Late complication reports are few, but erosion of coils into the colon has been reported.118
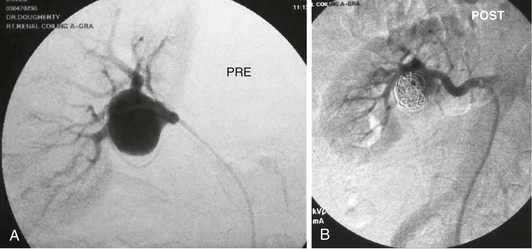
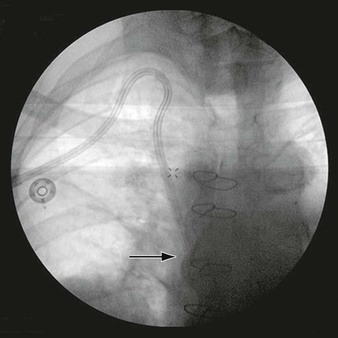
Figure 148-1 A, Arteriogram showing a posttraumatic arteriovenous fistula. This patient experienced a stab wound to the flank and presented with hematuria. B, After Gianturco coil embolization of multiple arterial branches, venous communication is no longer present. Hematuria resolved, and the patient recovered uneventfully.
Very large AVFs may present a technical challenge because of the risk of central embolization; some authors recommend surgery in this setting.107,108,119,120 Others have reported success in this setting using the Amplatz spider device (Cook Medical, Bloomington, Ind) to provide a scaffolding that can then engage other embolic materials.74,83 Staging the procedure—beginning with large coils, followed weeks later by smaller coils and other materials to close off persistent flow channels—may also be an effective strategy.121 Because very large arteriovenous communications tend to be at the renal pedicle rather than intraparenchymal, surgical treatment is feasible and probably preferable for good-risk patients.
Complications of embolization are unusual but not insignificant. In addition to arterial access site morbidity and contrast agent toxicity, pulmonary or peripheral arterial embolization can occur. This is usually related to the improper selection and delivery of embolic materials. Large AVFs require large devices such as coils or detachable balloons, but even these can embolize centrally. Gelfoam, alcohol, and various glues may be more appropriate for very small communications, but the delivery is less precise, and renal parenchymal infarction seems to be greater with these materials.86 It is common for patients to have fever, leukocytosis, and even hypertension after embolization, but these are transient and presumed to be secondary to renal infarction.90 Embolization itself was reported to cause massive collecting system hemorrhage in one case, but this was successfully managed with further embolization.122 With modern techniques, endovascular treatment is successful in more than 80% of patients97 and is clearly the treatment of choice for symptomatic congenital or acquired AVFs.
Selected Key References
Crotty K, Orihuela E, Warren M. Recent advances in the diagnosis and treatment of renal arteriovenous malformations and fistulas. J Urol. 1993;150:1355–1359.
Good overall review of renal AVMs..
Huppert PE, Duda SH, Erley CM, Roth M, Lauchart W, Dietz K, Claussen CD. Embolization of renal vascular lesions: clinical experience with microcoils and tracker catheters. Cardiovasc Intervent Radiol. 1993;16:361–367.
Good review of endovascular intervention for renal artery aneurysms and AVFs..
Lacombe M. Ex situ repair of complex renal artery lesions. Cardiovasc Surg. 1994;2:767–771.
Good review of surgical technique..
Lumsden AB, Salam TA, Walton KG. Renal artery aneurysm: a report of 28 cases. Cardiovasc Surg. 1996;4:185–189.
Large review of one center’s experience with renal artery aneurysms..
Stanley JC. Natural history of renal artery stenosis and aneurysms. Modern management of renovascular hypertension and renal salvage. Williams & Wilkins: Baltimore; 1996:15–30. Calligaro KD, Dougherty MJ, Dean RH. Modern management of renovascular hypertension and renal salvage. vol 15.
Large series describing the natural history of renal artery aneurysms..
Takebayashi S, Hosaka M, Kubota Y, Ishizuka E, Iwasaki A, Matsubara S. Transarterial embolization and ablation of renal arteriovenous malformations: efficacy and damages in 30 patients with long-term followup. J Urol. 1998;159:696–701.
Large series of endovascular intervention for AVMs..
The reference list can be found on the companion Expert Consult website at www.expertconsult.com.
References
1. Martin RS III, et al. Renal artery aneurysm: selective treatment for hypertension and prevention of rupture. J Vasc Surg. 1989;9:26.
2. Stanley JC. Natural history of renal artery stenosis and aneurysms. Calligaro KD, et al. Modern management of renovascular hypertension and renal salvage. Williams & Wilkins: Baltimore; 1996:15.
3. Charron J, et al. Renal artery aneurysm: polyaneurysmal lesion of kidney. Urology. 1975;5:1.
4. Erdsman G. Angionephrography and suprarenal angiography. Acta Radiol. 1957;155(Suppl):104.
5. Tham G, et al. Renal artery aneurysms: natural history and prognosis. Ann Surg. 1983;197:348.
6. Zhang LJ, et al. Renal artery aneurysm: diagnosis and surveillance with multidetector-row computed tomography. Acta Radiol. 2007;48:274.
7. Cinat M, et al. Management of renal artery aneurysms. Semin Vasc Surg. 1996;9:236.
8. Fry WF. Renal artery aneurysm. Ernst CB, et al. Current therapy in vascular surgery. BC Decker: Philadelphia; 1987:363.
9. Lumsden AB, et al. Renal artery aneurysm: a report of 28 cases. Cardiovasc Surg. 1996;4:185.
10. Stanley JC, et al. Renal artery aneurysms: significance of macroaneurysms exclusive of dissections and fibrodysplastic mural dilations. Arch Surg. 1975;110:1327.
11. Pliskin MJ, et al. A giant renal artery aneurysm diagnosed postpartum. J Urol. 1990;144:1459.
12. Bernhardt J, et al. A major renal artery aneurysm as the cause of a hydronephrosis with renovascular hypertension. Urol Int. 1996;57:237.
13. Dzsinich C, et al. Fibromuscular dysplasia and surgical management of renal artery aneurysm. Cardiovasc Surg. 1993;1:243.
14. Poutasse EF. Renal artery aneurysms: their natural history and surgery. J Urol. 1966;95:297.
15. Poutasse EF. Renal artery aneurysms. J Urol. 1975;43:113.
16. Mattar SG, et al. Vascular complications in Ehlers-Danlos syndrome. Am Surg. 1994;60:827.
17. Gewertz BL, et al. Renal artery dissections. Arch Surg. 1977;112:409.
18. Hare WSC, et al. Dissecting aneurysms of the renal artery. Radiology. 1970;97:255.
19. Kaufman JJ, et al. Primary dissecting aneurysms of renal artery: report of a case causing reversible renal hypertension. Ann Surg. 1973;177:259.
20. McCormack LJ, et al. A pathologic arteriographic correlation of renal arterial disease. Am Heart J. 1966;72:188.
21. Hekali P, et al. Renal complications of polyarteritis nodosa: CT findings. J Comput Assist Tomogr. 1985;9:333.
22. Sellar RJ, et al. The incidence of microaneurysms in polyarteritis nodosa. Cardiovasc Intervent Radiol. 1986;9:123.
23. Henriksson C, et al. Natural history of renal artery aneurysm elucidated by repeated angiography and pathoanatomic studies. Eur Urol. 1985;11:244.
24. Hageman JH, et al. Aneurysms of the renal artery: problems of prognosis and surgical management. Surgery. 1978;84:563.
25. Hubert JP Jr, et al. Solitary renal artery aneurysms. Surgery. 1980;88:557.
26. Harrow BR, et al. Aneurysm of renal artery: report of five cases. J Urol. 1959;81:35.
27. McCarron JP Jr, et al. Indications for surgery on renal artery aneurysms. J Urol. 1975;114:177.
28. Panayiotopoulos YP, et al. Aneurysms of the visceral and renal arteries. Ann R Coll Surg Engl. 1996;78:412.
29. Vaughan TJ, et al. Renal artery aneurysms and hypertension. Radiology. 1971;99:287.
30. Hidai H, et al. Rupture of renal artery aneurysm. Eur Urol. 1985;11:249.
31. Morita K, et al. Long-term outcome of single institutional experience with conservative and surgical management for renal artery aneurysm. Transplant Proc. 2012;44:1795–1799.
32. Cohen JR, et al. Ruptured renal artery aneurysms during pregnancy. J Vasc Surg. 1987;6:51.
33. Sicard GA, et al. Alternatives in renal revascularization. Curr Probl Surg. 1995;32:569.
34. Stanley JC, et al. Splanchnic artery aneurysms. Rutherford RB. Vascular surgery. ed 4. WB Saunders: Philadelphia; 1995:1124.
35. Van Way CW III. Renal artery aneurysms and arteriovenous fistulae. Rutherford RB. Vascular surgery. ed 4. WB Saunders: Philadelphia; 1995:1438.
36. Chivate J, et al. Congenital renal arteriovenous fistula: case report. Br J Urol. 1993;71:358.
37. Arroyo Bielsa A, et al. Extraparenchymal renal artery aneurysms: is hypertension an indication for revascularization surgery? Ann Vasc Surg. 2002;16:339.
38. Turpin S, et al. Radionuclide captopril renography in postpartum renal artery aneurysms. J Nucl Med. 1996;37:1368.
39. Bloemsma GC, et al. Endovascular repair of a ruptured type II renal artery aneurysm using an endograft. Ann Vasc Surg. 2012;7:1011.
40. Routh WD, et al. Transcatheter thrombosis of a leaking saccular aneurysm of the main renal artery with preservation of renal blood flow. AJR Am J Roentgenol. 1990;154:1097.
41. Murray SP, et al. Complex branch renovascular disease: management options and late results. J Vasc Surg. 1994;20:338.
42. Brayman KL, et al. Ex vivo reconstruction of the renal artery for aneurysm and other abnormalities of renal vascular anatomy. Calligaro KD, et al. Modern management of renovascular hypertension and renal salvage. Williams & Wilkins: Baltimore; 1996:269.
43. Dubernard JM, et al. Aneurysms of the renal artery: surgical management with special reference to extracorporeal surgery and autotransplantation. Eur Urol. 1985;11:26.
44. Dean RH, et al. Ex vivo renal artery reconstructions: indications and techniques. J Vasc Surg. 1986;4:546.
45. Crutchley TA, et al. Branch renal artery repair with cold perfusion protection. J Vasc Surg. 2007;46:405–412.
46. Lacombe M. Ex situ repair of complex renal artery lesions. Cardiovasc Surg. 1994;2:767.
47. Seki T, et al. Experience with revascularizing renal artery aneurysms: is it feasible, safe, and worth attempting? J Urol. 1997;158:357.
48. English WP, et al. Surgical management of renal artery aneurysms. J Vasc Surg. 2004;40:53.
49. Pfeiffer T, et al. Reconstruction for renal artery aneurysm: operative techniques and long-term results. J Vasc Surg. 2003;37:293.
50. Robinson WP, et al. Favorable outcomes with in situ techniques for surgical repair of complex renal artery aneurysms. J Vasc Surg. 2011;53:684–691.
51. Toshino A, et al. Ex vivo surgery for renal artery aneurysms. Int J Urol. 1996;3:421.
52. Gill IS, et al. Laparoscopic repair of renal artery aneurysm. J Urol. 2001;166:202.
53. Luke P, et al. Robot-assisted laparoscopic renal artery aneurysm reconstruction. J Vasc Surg. 2006;44:651.
54. Giulianotti PC, et al. Robot-assisted laparoscopic repair of renal artery aneurysms. J Vasc Surg. 2012;51:842–849.
55. Bui BT, et al. Renal artery aneurysm: treatment with percutaneous placement of a stent-graft. Radiology. 1995;195:181.
56. Klein GE, et al. Endovascular treatment of renal artery aneurysm with conventional non-detachable microcoils and Guglielmi detachable coils. Br J Urol. 1997;79:852.
57. Tateno T, et al. Successful embolization of a renal artery aneurysm with preservation of renal blood flow. Int Urol Nephrol. 1996;28:283.
58. Tshomba Y, et al. Renal artery aneurysm: improved renal function after coil embolization. J Endovasc Ther. 2002;9:54.
59. Pall AA, et al. Renal artery aneurysm six years after percutaneous renal biopsy: successful treatment by embolization. Nephrol Dial Transplant. 1992;7:883.
60. Elaassar O, et al. Endovascular techniques for the treatment of renal artery aneurysms. Cardiovasc Intervent Radiol. 2011;34:926–935.
61. Sedat J, et al. Endovascular treatment of renal aneurysms: a series of 18 cases. Eur J Radiol. 2012.
63. Lupattelli T, et al. Embolization of a renal artery aneurysm using ethylene vinyl alcohol copolymer (Onyx). J Endovasc Ther. 2003;10:366.
64. Sfyroeras GS, et al. Flow-diverting stents for the treatment of arterial aneurysms. J Vasc Surg. 2012;56:839–846.
65. Antoniou G, et al. Endovascular stent graft repair of renal artery aneurysms. Int Angiol. 2011;30:481–487.
66. Gandini R, et al. Bilateral renal artery aneurysm: percutaneous treatment with stent-graft placement. Cardiovasc Intervent Radiol. 2006;29:875.
67. Andersen PE, et al. Endovascular exclusion of renal artery aneurysm. Cardiovasc Intervent Radiol. 2005;28:665.
68. Klonaris C, et al. Renal artery aneurysm endovascular repair. Int Angiol. 2007;26:189.
69. Bruce M, et al. Endoluminal stent-graft repair of a renal artery aneurysm. J Endovasc Ther. 2002;9:359.
70. Sahin S, et al. Wide-necked renal artery aneurysm: endovascular treatment with stent-graft. Diagn Interv Radiol. 2007;13:42.
71. Sachs D, et al. Polyarteritis nodosa and familial Mediterranean fever. Br J Rheumatol. 1987;26:139.
72. Motta J, et al. Congenital renal arteriovenous malformation in pregnancy presenting with hypertension: case report. Urology. 1994;44:911.
73. Cho KJ, et al. Non-neoplastic congenital and acquired renal arteriovenous malformations and fistulas. Radiology. 1978;129:333.
74. Kearse W Jr, et al. Transcatheter embolization of large idiopathic renal arteriovenous fistula: case report. J Urol. 1994;151:967.
75. McAlhany JC Jr, et al. Renal arteriovenous fistula as a cause of hypertension. Am J Surg. 1971;122:117.
76. Piquet P, et al. Aneurysmes des arteres renales et fistules arterioveineuses renales. Kieffer E. Chirurgie des Arteres Renales. AERCV: Paris; 1993:237.
77. Gopalakrishnan G, et al. Renal arteriovenous malformation presenting as haematuria in pregnancy: case report. Br J Urol. 1995;75:110.
78. Fogazzi G, et al. Spontaneous renal arteriovenous fistula as a cause of haematuria. Nephrol Dial Transplant. 1997;12:350.
79. Morimoto A, et al. A unique case of renovascular hypertension caused by combined renal artery disease: case report. Hypertens Res. 1995;18:255.
80. Imray TJ, et al. Renal arteriovenous fistula associated with fibromuscular dysplasia. Urology. 1989;23:378.
81. Vasavada S, et al. Renal arteriovenous malformations masquerading as renal cell carcinoma. Urology. 1995;46:716.
82. Crotty K, et al. Recent advances in the diagnosis and treatment of renal arteriovenous malformations and fistulas. J Urol. 1993;150:1355.
83. Robinson D, et al. Transcatheter embolization of an aortocaval fistula caused by residual renal artery stump from previous nephrectomy: a case report. J Vasc Surg. 1993;17:794.
84. Steffens J, et al. Selective transcatheter embolization of a pediatric postnephrectomy arteriovenous fistula: case report. Urol Int. 1994;53:99.
85. van der Zee J, et al. Traumatic renal arteriovenous fistula in a 3-year-old girl, successfully treated by percutaneous transluminal embolization. J Pediatr Surg. 1995;30:1513.
86. Huppert P, et al. Embolization of renal vascular lesions: clinical experience with microcoils and tracker catheters. Cardiovasc Intervent Radiol. 1993;16:361.
87. Lawen JD, et al. Arteriovenous fistulas after renal allograft biopsy: a serious complication in patients beyond one year. Clin Transplant. 1990;4:357.
88. Thistlethwaite JR Jr, et al. Aggressive needle biopsy protocol prevents loss of renal allografts to undetected rejection during early post-transplant dysfunction. Transplant Proc. 1989;21:1890.
89. Merkus J, et al. High incidence of arteriovenous fistula after biopsy of kidney allografts. Br J Surg. 1993;80:310.
90. Reilly K, et al. Angiographic embolization of a penetrating traumatic renal arteriovenous fistula. J Trauma. 1996;41:763.
91. Ozbek S, et al. Image-directed and color Doppler ultrasonography in the diagnosis of postbiopsy arteriovenous fistulas of native kidneys. J Clin Ultrasound. 1995;23:239.
92. Rollino C, et al. Colour-coded Doppler sonography in monitoring native kidney biopsies. Nephrol Dial Transplant. 1994;9:1260.
93. Kolb L, et al. Percutaneous renal allograft biopsy: a comparison of two needle types and analysis of risk factors. Transplantation. 1994;57:1742.
94. Riehl J, et al. Percutaneous renal biopsy: comparison of manual and automated puncture techniques with native and transplant kidneys. Nephrol Dial Transplant. 1994;9:1568.
95. Mori K, et al. Spontaneous rupture of an aneurysmal intrarenal arteriovenous fistula. Urol Int. 2003;70:83.
96. Schmid T, et al. Vascular lesions after percutaneous biopsies of renal allografts. Transplant Int. 1989;2:56.
97. Harrison K, et al. Renal dysfunction due to an arteriovenous fistula in a transplant recipient. J Am Soc Nephrol. 1994;5:1300.
98. Beaujeux R, et al. Superselective endovascular treatment of renal vascular lesions. J Urol. 1995;153:14.
99. Yeo E, et al. Intrarenal arteriovenous fistula simulating a hypervascular renal tumor on radionuclide renal imaging. Clin Nucl Med. 1995;20:549.
100. Peces R, et al. Giant saccular aneurysm of the renal artery presenting as malignant hypertension. J Hum Hypertens. 1991;5:465.
101. Bagga H, et al. Contrast-enhanced MR angiography in the assessment of arteriovenous fistula after renal transplant biopsy. AJR Am J Roentgenol. 1999;172:1509.
102. Gainza F, et al. Evaluation of complications due to percutaneous renal biopsy in allografts and native kidneys with color-coded Doppler sonography. Clin Nephrol. 1995;43:303.
103. Beaujeux R, et al. Endovascular treatment of renal allograft postbiopsy arteriovenous fistula with platinum microcoils. Transplantation. 1994;57:311.
104. Messing E, et al. Renal arteriovenous fistulas. Urology. 1976;8:101.
105. Inoue T, et al. Spontaneous regression of a renal arteriovenous malformation. J Urol. 2000;163:232–233.
106. Kubota H, et al. Spontaneous disappearance of a renal arteriovenous malformation. Int J Urol. 2003;10:547–549.
107. Kumar U, et al. Perioperative use of a balloon-occluding arterial catheter in renal arteriovenous malformation. Br J Urol. 1996;77:312.
108. Takatera H, et al. Renal arteriovenous fistula associated with a huge renal vein dilatation. J Urol. 1987;137:722.
109. Torres G, et al. Ex vivo repair of a large renal artery aneurysm and associated arteriovenous fistula. Ann Vasc Surg. 2002;16:141.
110. Takebayashi S, et al. Transarterial embolization and ablation of renal arteriovenous malformations: efficacy and damages in 30 patients with long-term followup. J Urol. 1998;159:696.
111. Cursio R, et al. Successful transarterial embolization of idiopathic renal arteriovenous fistula. Minerva Chir. 2001;56:321.
112. Wikholm G, et al. Superselective transarterial embolization of renal arteriovenous malformations of cryptogenic origin. Scand J Urol Nephrol. 1994;28:29.
113. Saliou C, et al. Idiopathic renal arteriovenous fistula: treatment with embolization. Ann Vasc Surg. 1998;12:75.
114. Defrayne L, et al. Cirsoid renal arteriovenous malformation treated by endovascular embolization with N-butyl 2-cyanoacrylate. Eur Radiol. 2000;10:772.
115. Nakamura H, et al. Renal aorto-venous malformations: transcatheter embolization and follow-up. AJR Am J Roentgenol. 1981;137:113.
116. Ginat DT, et al. Transcatheter renal artery embolization: clinical applications and techniques. Tech Vasc Interv Radiol. 2009;12(4):224–239.
117. Resnick S, et al. Transcatheter embolization of a high-flow renal arteriovenous fistula with use of a constrained wallstent to prevent coil migration. J Vasc Interv Radiol. 2006;17(2 Pt 1):363–367.
119. Lord RSA, et al. Massive renal arteriovenous fistula presenting as a pulsatile abdominal mass. Cardiovasc Surg. 2000;8:164.
120. Ziani M, et al. Renal arteriovenous malformation requiring surgery in Rendu-Osler-Weber disease (hereditary hemorrhagic telangiectasia). J Urol. 2000;164:1292.
121. Gralino BJ, et al. Staged endovascular occlusion of giant idiopathic renal arteriovenous fistula with platinum microcoils and silk suture threads. J Vasc Interv Radiol. 2002;13:747.
122. Kamai T, et al. A case of gross hematuria arising during embolization for renal arteriovenous malformation. Urol Int. 1997;58:55.