CHAPTER 15 Pediatric Distal Radius Fractures
Pediatric distal radius fractures are common. In fact, forearm fractures account for 40% of all pediatric fractures and the distal radius and distal ulna are the most common sites within the forearm.1–3 High-risk activities for these fractures include horseback riding, skateboarding, and snowboarding.4 In addition, overweight adolescents have poorer balance than those of healthy weight, which may explain their propensity for fracture.5 Most distal radius fractures are Salter-Harris II fractures and are treated without surgery. In general, acceptable reduction is angulation less than 20 degrees with 2 years of growth remaining. In the young child, complete bayonet apposition is acceptable. However, operative intervention is indicated in certain fracture types. In this chapter the focus is on those pediatric distal radius fractures that require surgery with an emphasis on indications, technique, and clinical pearls.
Pertinent Anatomy
The pediatric skeleton is unique in multiple ways. The presence of the physis or growth plate provides longitudinal growth. The physis is divided into four distinct zones: germinal, proliferative, hypertrophic, and provisional calcification. The hypertrophic and provisional calcification zones are relatively weaker than the germinal and proliferative layers.6,7 Classically, fracture lines tend to pass through the hypertrophic and provisional calcification zones. However, high-energy injuries may undulate through all four zones of the physis.6–9
The presence of the secondary ossification center is another distinguishing feature that can complicate fracture detection. The distal radius epiphysis is absent at birth and appears at approximately 1 year of age. Specifically, the appearance is between 0.5 and 2.3 years in boys and between 0.4 and 1.7 years in girls.10 The configuration of the epiphysis also changes with age. Initially, the epiphysis is transverse in shape and becomes more triangular with time. The radial physis closes at approximately 16 years of age in girls and 17 years of age in boys.11 The distal radius and ulnar physis contribute 75% to 80% of the forearm growth and 40% of the entire upper extremity.12
Indications
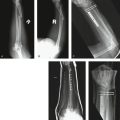
Additional indications for surgery are open fractures and fractures associated with nerve injuries, such as carpal tunnel syndrome (Figs. 15-1 and 15-2). The treating physician must be aware that children and adolescents do not routinely complain of numbness. In addition, sensation is difficult to examine in the pediatric patient and two-point discrimination is unreliable until about 9 years of age. Therefore, the physician must have a high index of suspicion for nerve compression after treatment of markedly displaced fractures. The treatment principles for open fractures and fractures associated with nerve injuries are similar to those for adults and will not be specifically addressed in this chapter.
Surgical Technique
Intra-articular Fractures
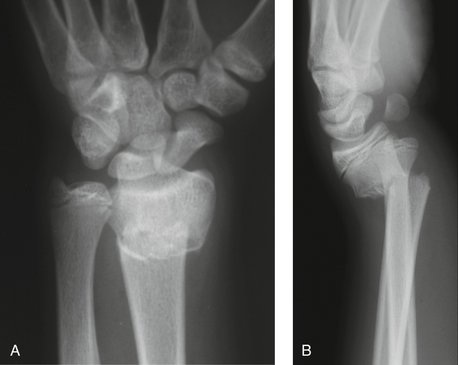
Displaced Salter-Harris III and IV fractures require surgery to restore anatomical alignment of the joint surface.13 The goal of internal fixation is intraepiphyseal fixation with avoidance of the physis. If fixation must cross the physis, smooth pins are preferable to limit further physeal injury. Mini-fluoroscopy is invaluable to assess joint alignment and implant fixation. External fixation may be necessary to augment the fixation and to unload the joint surface until satisfactory healing has occurred.
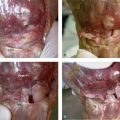
The specific approach depends on the fracture configuration (Fig. 15-3). Computed tomography (CT) can further delineate the fracture pattern. In general, the approach is directed at the fracture fragment. A dorsal fragment requires a dorsal longitudinal incision along Lister’s tubercle. Sharp dissection is performed to the extensor retinaculum. Skin flaps are elevated from this level. The third compartment is opened, and the extensor pollicis longus is transposed in a radial direction. The posterior interosseous nerve is identified and resected to prevent postoperative neuroma formation. The second and fourth compartments are retracted, and a longitudinal capsulotomy is performed.
Fluoroscopic imaging is used to confirm joint reduction and wire position—preferably intraepiphyseal fixation with avoidance of the physis. Direct joint inspection avoids intra-articular wire placement. In sequential fashion, the pins are removed and replaced with compression screws across the fracture (Fig. 15-4). The placement of screws across the physis is avoided. At this point, the fixation is assessed and a determination made regarding the necessity of an external fixator to unload the joint surface until healing has occurred. In children, I have a low threshold to apply an external fixator because compliance is unlikely. The fixator is placed using standard open incisions. Distraction is applied until the scaphoid and lunate are elevated from the articular surface to eliminate compressive forces until union. Standard closure is performed with absorbable suture, and the external fixator is securely tightened. The tourniquet is deflated to ensure capillary refill. A long arm splint is applied. The external fixator is removed 5 to 6 weeks after surgery. Active and active-assisted range of motion is instituted. Formal therapy is usually not required.
Arthroscopically assisted reduction has been described to directly visualize the articular surface.14,15 However, this technique is relatively demanding and requires additional equipment. I prefer direct visualization of the joint surfaces.
Pediatric Galeazzi Fractures
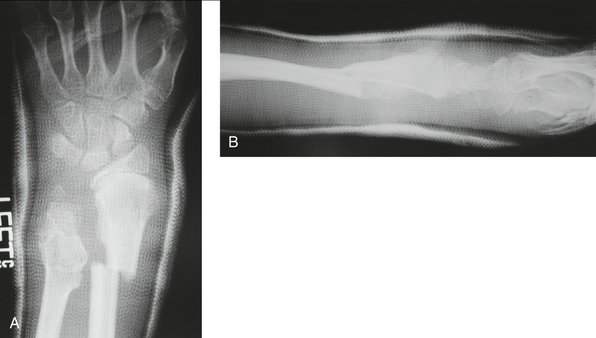
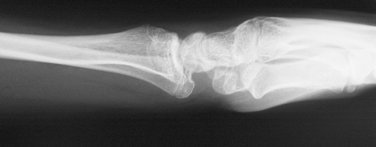
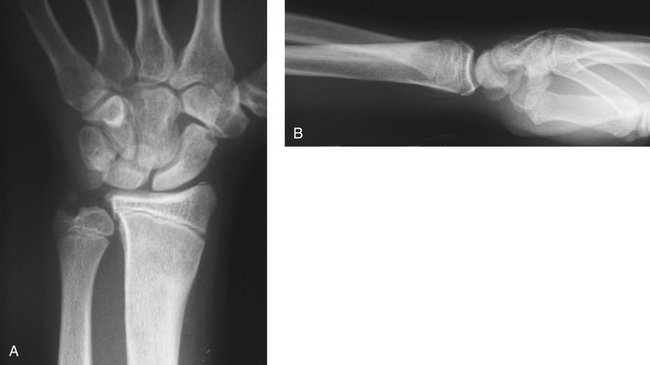
The most common pattern of pediatric Galeazzi fracture is when there is dorsal displacement of the distal radius fracture and volar displacement of the distal ulna (Fig. 15-5). A less common type is when there is volar displacement of the distal radius and dorsal displacement of the distal ulna.16 Regardless of the pattern, recognition of the disrupted DRUJ is mandatory. A true lateral radiograph is necessary to assess the status of the DRUJ. The injury pattern can also involve the distal ulnar physis and then is called a Galeazzi-equivalent fracture.
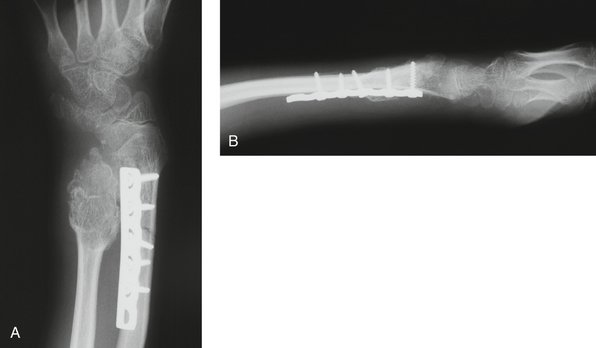
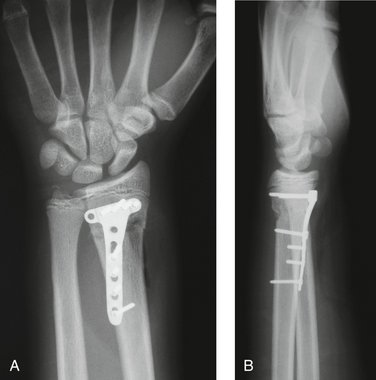
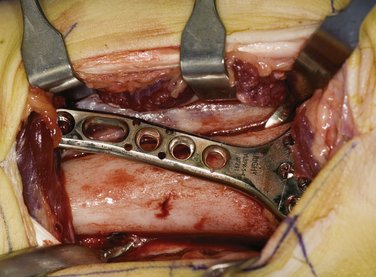
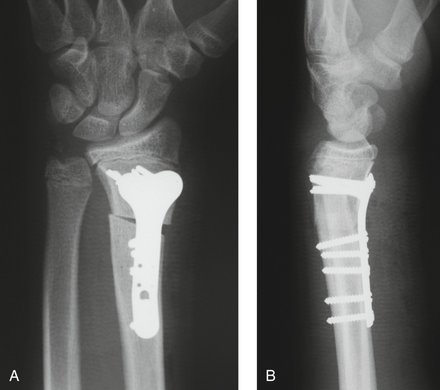
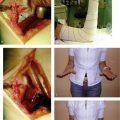
The pronator quadratus is elevated from the radius in an extraperiosteal manner leaving a 2-mm sleeve along the radial border. The radius and fracture site are clearly exposed (Fig. 15-6). Hematoma is removed from the fracture site. The fracture is reduced under direct visualization (Fig. 15-7). Fixation can be accomplished using a variety of plates. The fixation device selected should consider the close proximity of the growth plate. Fixed-angle devices provide rigid fixation distal to the fracture site and can avoid the physis. Mini-fluoroscopy is used to assess fracture reduction and internal fixation placement (Fig. 15-8).
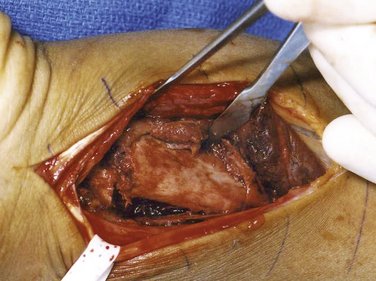
FIGURE 15-6 Exposure of the fracture site reveals considerable displacement.
(Courtesy of Shriners Hospital for Children, Philadelphia, PA.)
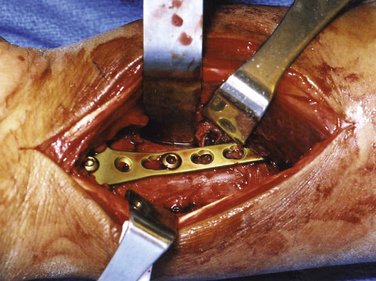
FIGURE 15-7 Open reduction and internal fixation with T-plate.
(Courtesy of Shriners Hospital for Children, Philadelphia, PA.)
At this point, the status of the DRUJ is assessed. Usually, anatomical restoration of the radius results in DRUJ reduction. On rare occasion, the DRUJ is irreducible because of interposed tissues.17–19 Periosteum or tendon (e.g., extensor carpi ulnaris or extensor digiti quinti) can be blocking reduction. Exploration of the DRUJ is necessary to remove the offending agent. A separate ulnar approach to the DRUJ is necessary.
Results
The actual results of pediatric and adolescent distal radius fractures are difficult to decipher based on the literature. In general, most children do well with closed or open treatment as long as the treatment approach adheres to the standard guidelines previously discussed. However, a study that shows consistently good outcomes is often not worthy of formal publication. Instead, most reports discuss complications of distal radius fractures, such as growth arrest, missed fracture-dislocations, and instability.20–24
Complications
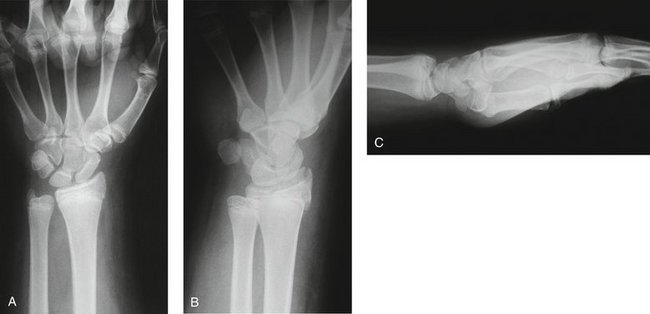
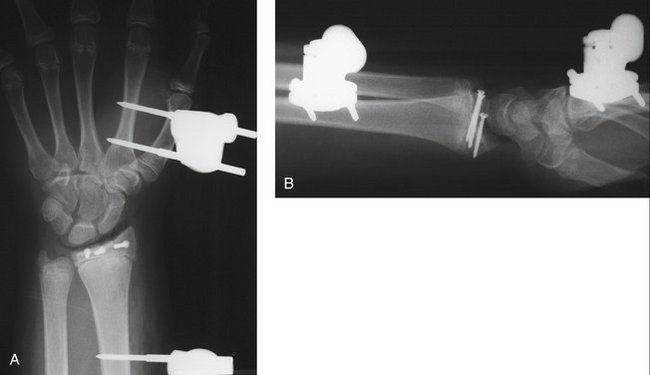
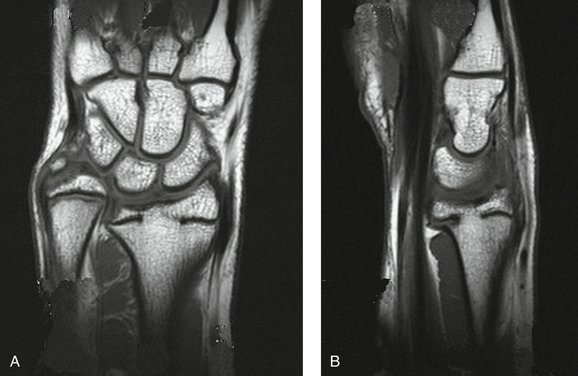
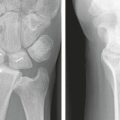
Growth plate closure occurs in 4% to 5% of all Salter-Harris distal radius fractures.20,21 All growth plate fractures require a radiograph 3 to 6 months after healing to ensure continued growth. Failure to recognize a growth plate arrest can lead quickly to deformity (Figs. 15-9 and 15-10). The deformity varies according to the location and extent of the physeal bar. Peripheral bars lead to angular deformity as unequal growth occurs. In contrast, small central bars cause tenting of the articular surface and larger bars prevent any longitudinal growth, which results in shortening of the radius relative to the ulna. Advanced imaging studies better delineate the size and location of the physeal bar (Fig. 15-11). Management depends on the location of the bar, size of the bar, and the amount of remaining growth. Radioulnar discrepancy of less than 1 cm is well tolerated at long-term follow-up.21 Options include resection of the bar, formal epiphysiodesis, corrective osteotomy, and distraction osteogenesis. In addition, the ulna may be addressed by epiphysiodesis and/or shortening (Fig. 15-12).
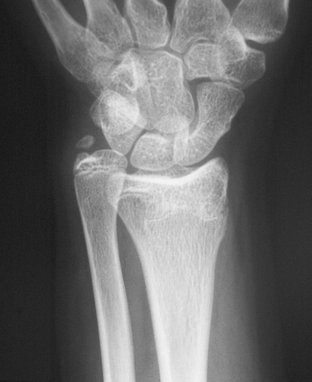
FIGURE 15-10 Anteroposterior radiograph shows shortening of the radius with markedly positive ulnar variance.
(Courtesy of Shriners Hospital for Children, Philadelphia, PA.)
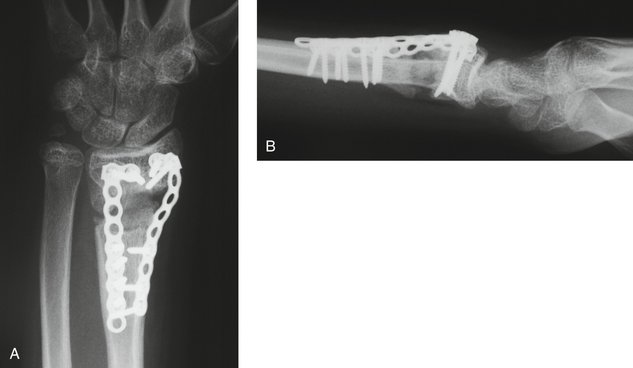
FIGURE 15-12 Patient shown in Figure 15-17 after a corrective osteotomy, bone grafting, and ulnar epiphysiodesis. A, Anteroposterior radiograph shows restoration of radial length. B, Lateral radiograph demonstrates reestablishment of volar tilt.
(Courtesy of Shriners Hospital for Children, Philadelphia, PA.)
Malunion after distal radius fractures is common, but remodeling with growth results in gradual correction. Angulation less than 20 degrees will remodel over 2 years; greater angulation requires additional growth. If the patient is close to skeletal maturity, little remodeling can be expected. Corrective osteotomy with bone grafting is indicated in patients with pain or limited motion. The goal is to restore alignment to alleviate pain, enhance motion, correct midcarpal instability, or prevent degenerative arthritis.22 The surgery should correct both the sagittal and coronal alignment (Fig. 15-13). A distal radius corrective osteotomy can be addressed from a dorsal or volar approach. Similar to adults, the volar approach and volar locking systems have gained popularity to avoid prominent hardware. A trans-FCR exposure is performed. The malunion site is identified. The distal portion of a fixed-angle plate is contoured and applied parallel to the physis and articular surface (Fig. 15-14). Mini-fluoroscopy is used to avoid the physis. The plate protrudes from the proximal radius but acts as a guide to correction. The radius is cut parallel to the physis and articular surface at the level of malunion. The osteotomy is opened using a laminar spreader, and the plate is used as a guide to correction. The proximal plate is affixed to the radius using a bone reduction clamp. Correction is assessed via mini-fluoroscopy, and adjustments made accordingly. The plate is then firmly secured to the radius using bicortical screw fixation (Fig. 15-15). Cancellous bone graft is placed within the osteotomy site. The graft can be harvested from the ulna or iliac crest depending on the size of the defect. I do not use bone graft substitutes because ample autologous bone is available. Radiographs are taken to verify correction of the coronal and sagittal alignment (Fig. 15-16).
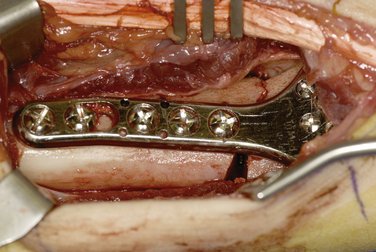
FIGURE 15-15 Plate was reduced to the proximal radius to correct the volar tilt and maintain radial inclination.
(Courtesy of Shriners Hospital for Children, Philadelphia, PA.)
A missed Galeazzi fracture-dislocation is a formidable problem. The ulna is subluxated and forearm rotation is limited. Treatment depends on time from the injury and the status of the DRUJ. Early recognition can be treated with corrective osteotomy of the radius and reduction of the DRUJ. Later recognition requires assessment of the articular surfaces of the distal ulna and sigmoid notch. Articular degeneration is a contraindication for reduction. In these cases, a salvage procedure, such as a Darrach or Sauvé-Kapandji, is required (Fig. 15-17). Fortunately, this injury occurs in children close to skeletal maturity and progressive ulnar-negative variance is not a concern because ulnar-negative variance up to 1 cm is usually asymptomatic.21 However, in the young child the length of the ulna needs to be addressed by distraction osteogenesis.
Persistent pain after distal radius fracture may be related to an associated chondral injury, triangular fibrocartilage (TFC) tear, or scapholunate ligament injury. These injuries are rare in children. Most TFC and scapholunate ligament tears are partial and can be treated by arthroscopic inspection and débridement. Arthroscopic treatment can result in long-term improvement.23,24 In cases of peripheral TFC tears, open or arthroscopic repair is warranted.24
Volar fixation with plate and screws can result in prominent hardware along the dorsum of the distal forearm and wrist area. The child presents with extensor tenosynovitis of the irritated tendons. This finding is a forewarning of future problems, including tendon rupture. Treatment requires plate and screw removal, which can be difficult with titanium implants. Another option is dorsal exposure and burring the prominent screw head(s).
1. Lawton L. Fractures of the distal radius and ulna in management of pediatric fractures. In: Letts M, editor. Management of Pediatric Fractures. New York: Churchill-Livingstone, 1994.
2. Landin LA. Fracture patterns in children: analysis of 8682 fractures with special reference to incidence, etiology, and secular changes in a Swedish urban population, 1950-79. Acta Chir Scand Suppl. 1983;202:1-109.
3. Thomas EM, Tuson KW, Brownie PS. Fractures of the radius and ulna in children. Injury. 1975;7:120-124.
4. Matsumoto K, Sumi H, Sumi Y, Shimizu K. Wrist fractures from snowboarding: a prospective study for 3 seasons from 1998 to 2001. Clin J Sport Med. 2004;14:64-71.
5. Goulding A, Jones IE, Taylor RW, et al. Dynamic and static tests of balance and postural sway in boys: effects of previous wrist bone fractures and high adiposity. Gait Posture. 2003;17:136-141.
6. Bright RW, Bursein AH, Elmore SM. Epiphyseal plate cartilage: a biomechanical and histological analysis of failure modes. J Bone Joint Surg Am. 1974;56:688-703.
7. Moen CT, Pelker RR. Biomechanical and histological correlations in growth plate failure. J Pediatr Orthop. 1984;4:1180-1184.
8. Smith DG, Geist RW, Cooperman DR. Microscopic examination of naturally occurring epiphyseal plate fracture. J Pediatr Orthop. 1985;5:306-308.
9. Wattenbarger JM, Gruber HE, Phieffer LS. Physeal fractures: I. Histologic features of bone, cartilage, and bar formation in a small animal model. J Bone Joint Orthop. 2002;2:703-709.
10. Garn SM, Rohmann CG, Silverman FN. Radiographic standards for postnatal ossification and tooth calcification. Med Radiogr Photogr. 1967;43:45-66.
11. Greulich W, Pyle SI. Radiographic Atlas of Skeletal Development of the Hand and Wrist. Stanford, CA: Stanford University Press; 1959.
12. Ogden JA, Beall JK, Conlogue GJ, et al. Radiology of postnatal skeletal development: IV. Distal radius and ulna. J Skeletal Radiol. 1981;6:255-266.
13. Peterson HA. Triplane fracture of the distal radius: case report. J Pediatr Orthop. 1996;16:192-194.
14. Geissler W, Freeland A, Weiss AP, et al. Techniques of wrist arthroscopy. Instructional course lecture. J Bone Joint Surg Am. 1999;81:1184-1197.
15. Doi K, Hattori Y, et al. Intra-articular fractures of the distal aspect of the radius: arthroscopically assisted reduction compared with open reduction and internal fixation. J Bone Joint Surg Am. 1999;81:1093-1110.
16. Walsh HP, McLaren CA, Owen R. Galeazzi fractures in children. J Bone Joint Surg Am. 1987;69:730-733.
17. Kozin SH, Wood MB. Early soft tissue complications after fractures of the distal part of the radius. J Bone Joint Surg [Am]. 1993;75:144-153.
18. Letts RM. Monteggia and Galeazzi fractures. In: Letts RM, editor. Management of Pediatric Fractures. New York: Churchill-Livingstone, 1994.
19. Kraus B, Horne G. Galeazzi fractures. J Trauma. 1985;25:1093-1095.
20. Lee BS, Esterhai JLJr, Das M. Fracture of the distal radial epiphysis: characteristics and surgical treatment of premature, post-traumatic epiphyseal closure. Clin Orthop Relat Res. 1984;185:90-96.
21. Cannata G, De Maio F, Mancini F, et al. Physeal fractures of the distal radius and ulna: long-term prognosis. J Orthop Trauma. 2003;17:172-179.
22. Watson K, Taleisnik J. Midcarpal instability caused by malunited fracture of the distal radius. J Hand Surg [Am]. 1984;9:350-357.
23. Earp BE, Waters PM, Wyzykowski RJ. Arthroscopic treatment of partial scapholunate ligament tears in children with chronic wrist pain. J Bone Joint Surg [Am]. 2006;88:2448-2455.
24. Bae DS, Waters PM. Pediatric distal radius fractures and triangular fibrocartilage complex injuries. Hand Clin. 2006;22:43-53.