[level-membership-for-emergency-medicine-category]
171 Meningitis, Encephalitis, and Brain Abscess
• There is significant overlap among the initial clinical presentations of meningitis, encephalitis, and brain abscess.
• The four most common bacteria responsible for adult bacterial meningitis are Streptococcus pneumoniae, Neisseria meningitides, Haemophilus influenzae type B, and Listeria monocytogenes. Group B Streptococcus remains the predominate cause of meningitis in infants less than 2 months of age.
• The classic constellation of fever, neck stiffness, headache, and change in mental status are seen in less than 50% of cases of acute bacterial meningitis.
• Cranial computed tomography (CT) scan, prior to lumbar puncture, is recommended in patients with a history of immunocompromised state, history of central nervous system (CNS) disease, new-onset seizure, abnormal neurologic examination, papilledema, altered mental status, or altered level of consciousness.
• Empiric therapy in patients with high clinical suspicion for CNS infection should not be delayed for neuroimaging or lumbar puncture.
• Although epidemiologic clues and assessment of risk factors should be sought in all patients with encephalitis, herpes simplex virus and arboviruses remain the most common causes of nonepidemic and epidemic outbreaks of encephalitis, respectively, in the United States.
• Acyclovir should be initiated in all patients with suspected encephalitis, pending the results of diagnostic studies.
• Risk factors for the development of intracranial abscess include inadequately treated subacute or chronic ear, nose, mastoid, and dental infection; endocarditis; congenital heart disease; and having undergone neurosurgical procedures.
• Patients with intracranial abscess often present with mild headache symptoms in the weeks to months prior the emergency department visit. The classic triad of fever, headache, and focal neurologic deficit is seen in less than 20% of patients with brain abscess.
Meningitis
Epidemiology
The combination of routine vaccination against Streptococcus pneumoniae, Haemophilus influenzae type B, and Neisseria meningitides, maternal screening for and intrapartum treatment of group B Streptococcus (Streptococcus agalactiae), and enhanced efforts to reduce the contamination of processed foods by Listeria monocytogenes have all led to a significant decrease in the incidence of acute bacterial meningitis (ABM). Data from the Emerging Infections Programs Network, established by the Centers for Disease Control and Prevention (CDC), have noted a decrease in the incidence of meningitis from 2.0 cases per 100,000 population in 1998 to 1999 to 1.38 cases per 100,000 population in 2006 to 2007.1 During this same time the case fatality rate decreased to 14.3%. Projecting these data on a national level reveals an estimated 4100 cases and 500 deaths from bacterial meningitis annually in the United States.1
Etiology
A 10-year review (1998 to 2007) of 3188 cases of bacterial meningitis noted that S. pneumoniae accounted for the greatest proportion of cases (58%), followed by group B. streptococcus (18.1%), N. meningitides (13.9%), H. influenzae (6.7%), and L. monocytogenes (3.4%).1 Among infants less than 3 months of age, group B Streptococcus and gram-negative rods account for most cases of ABM. After 3 months of age, S. pneumoniae and N. meningitidis become the predominant pathogens. L. monocytogenes is primarily seen in infants less than 1 month of age, in adults more than 50 years old, and in immunocompromised patients. Staphylococcus aureus is acquired mainly nosocomially and occurs predominantly after neurosurgical procedures or following penetrating head trauma. S. aureus may be acquired in the community setting, linked to predisposing conditions such as endocarditis, injection drug use, and compromised immune systems.
Pathophysiology
ABM develops after encapsulated bacteria, which have colonized the nasopharynx and/or oropharynx, penetrate the intravascular space and enter the subarachnoid space through vulnerable sites within the blood-brain barrier.2 Once the pathogens enter the central nervous system (CNS), they replicate rapidly, thus consuming glucose and liberating protein within the cerebrospinal fluid (CSF). The ensuing inflammatory reaction occurs in response to the liberation of bacterial cell wall and cell membrane components (e.g., lipopolysaccharide, peptidoglycan, lipoteichoic acid) and the induction of proinflammatory mediators. These events culminate in injury to the vascular endothelium that results in increased vascular permeability to the blood-brain barrier, meningeal inflammation, and cerebral vasculitis. The accompanying cerebral edema and increase in intracranial pressure (ICP) contribute to CNS hypoperfusion and cell death.
Presenting Signs and Symptoms
Patients with ABM typically appear ill and often present within 24 to 72 hours of symptom onset. Table 171.1 reviews the presenting signs and symptoms of adults with ABM.3 The cardinal symptoms of ABM (i.e., fever, neck stiffness, change in mental status, and headache) are seen in combination in less than half of all patients. Nearly 95% of patients will present with at least two of these cardinal symptoms, which provides the rationale for performing a lumbar puncture in patients who are lethargic or confused and develop a fever. The absence of these four findings typically excludes the diagnosis of ABM.
Table 171.1 Findings in 696 Episodes of Bacterial Meningitis
| SIGN OR SYMPTOM | FREQUENCY (%) |
|---|---|
| Duration of symptoms ≤ 24 hr | 48 |
| Fever (temperature ≥ 38° C) | 77 |
| Headache | 87 |
| Nausea or vomiting | 74 |
| Neck stiffness | 83 |
| GCS ≤ 14 (AMS) | 69 |
| GCS ≤ 8 (coma) | 14 |
| Rash | 26 |
| Focal neurologic deficit | 33 |
| Seizures | 5 |
| Arthritis | 7 |
AMS, altered mental status; GCS, Glasgow Coma Scale.
From Van de Beek D, de Gans J, Spanjaard L, et al. Clinical features and prognostic factors in adults with bacterial meningitis. N Engl J Med 2004;351:1849-59.
The headache described by patients with ABM can be moderate to severe in intensity, generalized, often with an occipital or nuchal component, and unlike “normal” headaches. Photophobia is commonly present, as is nausea. Worsening of the headache while the examiner rapidly turns the patient’s head from side to side (at a rate of two to three times per second), the so-called jolt accentuation test, has been reported to be helpful in identifying patients with ABM,4 but a recent study questioned the utility of this finding.5
Although neck pain may be infrequently reported, the objective finding of neck stiffness is seen in more than 80% of patients. Examining the neck for rigidity, during gentle forward flexion, with the patient in the supine position best assesses neck stiffness, whereas difficulty in lateral motion of the neck is a less reliable finding. Patients with severe meningeal irritation may spontaneously assume the tripod position (also called the Amoss sign or the Hoyne sign) with the knees and hips flexed, the back arched lordotically, the neck extended, and the arms brought back to support the thorax.6 The Kernig sign is performed with the patient lying supine and the hip and knee flexed to 90 degrees. A positive sign is present when extension of the knee from this position elicits resistance or pain in the lower back or posterior thigh. The classic Brudzinski sign refers to spontaneous flexion of the knees and hips during attempted passive flexion of the neck. A separate sign described by Brudzinski, the contralateral reflex, is present if passive flexion of one hip and knee causes flexion of the contralateral leg. The presence or absence of Kernig or Brudzinski signs has been shown to have little positive or negative predictive value in the diagnosis of ABM, unless severe meningeal inflammation is present.7
Atypical presentations of ABM occur in infants, older adults, and immunocompromised patients. Infants with bacterial meningitis may present with fever or hypothermia, hypoglycemia, poor feeding, seizures, or irritability (excessive or abnormal crying). On examination, the findings of jaundice, ill appearance, a bulging fontanelle, meningeal irritation (including neck stiffness, the Kernig sign, and the Brudzinski sign), fever higher than 40° C, and increased general body tone predict bacterial meningitis.8
Older and immunocompromised patients may also present atypically. These populations are associated with a higher rate of misdiagnosis that contributes to an increase in the morbidity and mortality following an episode of acute meningitis. A lower proportion of fever, headache, and nausea or vomiting is present in these subgroups.9 Neck stiffness has a lower sensitivity and specificity for meningitis in older patients. Finally, these populations may present to the emergency department (ED) with altered mental status and/or altered level of consciousness but without a fever.
Differential Diagnosis
The differential diagnosis of patients presenting with fever, headache, and altered mental status includes other forms of meningitis (e.g., nosocomial meningitis, aseptic meningitis), encephalitis, and cerebral abscess. The diagnosis of meningitis is challenging in patients who present atypically. In a review of 156 cases of meningitis in patients who presented to a single tertiary care hospital, 66 cases were initially misdiagnosed in the ED as an alternative infection (i.e., sepsis of unclear origin, pneumonia, urinary tract infection), metabolic encephalopathy, or nonspecific conditions (e.g., weakness, malaise, degenerative state). Higher percentages of these patients were more than 65 years of age, and these patients were also noted to have lower proportions of fever, headache, nausea or vomiting, and neck stiffness.10
Aseptic meningitis refers to a disorder in which patients have clinical and laboratory evidence of meningeal irritation with negative results of routine bacterial cultures. Precise epidemiologic data on the incidence of aseptic meningitis are lacking, but aseptic meningitis is associated with an estimated 26,000 to 42,000 hospitalizations per year in the United States. The origin of aseptic meningitis is varied (Box 171.1). Enteroviruses, the leading causes of viral meningitis in adults and children, account for 50% to 75% of all cases of aseptic meningitis. Additional causes include other infections (mycobacteria, fungi, spirochetes), parameningeal infections, medications (especially nonsteroidal antiinflammatory drugs), and malignant disease. The signs and symptoms of bacterial meningitis significantly overlap with those of aseptic meningitis. This overlap led to the development of several decision rules to distinguish the two conditions. The most useful pediatric score appears to be the Bacterial Meningitis Score. This score classifies patients 1 month to 18 years old as being at very low risk of bacterial meningitis if they lack all the following criteria: positive CSF Gram stain, CSF absolute neutrophil count (ANC) of at least 1000 cells/mcL, CSF protein of at least 80 mg/dL, peripheral blood ANC of at least 10,000 cells/mcL, and a history of seizure before or at the time of presentation.11
Medical Decision Making
Routine Laboratory Tests
Routine testing of patients with suspected meningitis should include complete blood cell count (CBC), serum electrolytes, bicarbonate, serum urea nitrogen (BUN), creatinine, and glucose (Table 171.2). Serum lactate determinations and blood cultures are also indicated in patients with suspected meningitis.
Table 171.2 Suggested Laboratory Testing in Suspected Meningitis
| BLOOD TEST | COMMENT |
|---|---|
| Complete blood count | WBC typically elevated with left shift, although normal or low values in infants and immunosuppressed patients |
| Electrolytes | Hyponatremia (Na < 135 mmol/L) seen in 30% of cases of ABM |
| Bicarbonate | Alkalosis seen with excessive vomiting, acidosis seen with poor tissue perfusion |
| BUN, creatinine | Renal function tests essential for antibiotic dosing and timing |
| Glucose | Useful in calculating the CSF/serum glucose ratio and in the initial evaluation of altered mental status or altered level of consciousness |
| Lactate | Has prognostic information (i.e., correlates with mortality) and used to identify candidates for early goal-directed therapy |
| Blood cultures | Positive results in 50% to 75% of patients with ABM when obtained before antibiotic administration |
ABM, Acute bacterial meningitis; BUN, blood urea nitrogen; CSF, cerebrospinal fluid; Na, sodium; WBC, white blood cell count.
Several newer tests have shown potential in distinguishing bacterial meningitis from nonbacterial meningitis. These tests include serum procalcitonin,12 serum C-reactive protein,13 CSF cortisol,14 and CSF lactate.15 Additional tests employing common biochemical laboratory techniques (e.g., latex agglutination, enzyme-linked immunosorbent assay, polymerase chain reaction [PCR] assay, microarrays) have shown significant promise in identifying the specific pathogen responsible for infection.
Neuroimaging Before Lumbar Puncture
Selected patients with meningitis may warrant a computed tomography (CT) scan of the head, to identify patients with lesions that place them at risk for herniation from LP and to diagnose conditions that would make LP unnecessary if the patient’s work-up was limited to the LP (e.g., tumor, cerebral abscess). Unfortunately, cranial CT has inadequate sensitivity for identifying patients at risk for brain herniation. A systematic review on this subject found only a handful of cases of brain herniation that occurred following a normal cranial CT scan.16 Despite this limitation, generally accepted criteria for obtaining a cranial CT scan before LP are listed in Box 171.2. For maximal sensitivity in those patients with suspected or confirmed human immunodeficiency virus infection, contrast-enhanced cranial CT should be performed at the same time as the nonenhanced cranial CT.
Box 171.2
General Recommendations for Computed Tomography Before Lumbar Puncture17
History of immunocompromised state
History of central nervous system disease (e.g., mass lesion, stroke, focal infection)
New-onset seizure (or new-onset seizure within 1 week of presentation)
Papilledema on funduscopic examination (or elevated optic nerve sheath diameter on ultrasound)
Hasbun R, Abrahams J, Jekel J, et al. Computed tomography of the head before lumbar puncture in adults with suspected meningitis. N Engl J Med 2001;345:1727-33.
Lumbar Puncture and Cerebrospinal Fluid Analysis
Although the diagnosis of bacterial meningitis rests on CSF examination, CSF analysis alone cannot reliably distinguish bacterial and aseptic meningitis.13 In addition to measuring the opening pressure, the examiner should obtain four tubes of CSF, each containing 1 to 2 mL of fluid, and send them for analysis. Typically tube 1 (and/or tube 4) is sent for cell count and differential, tube 2 for protein and glucose, tube 3 for Gram stain and culture, and tube 4 for special testing or additional cultures.
The appearance of the CSF can range from clear to cloudy, depending on the presence of significant concentrations of cells, bacteria, and protein. The CSF white blood cell (WBC) count can be significantly elevated, usually in the range of 1000 to 5000 cells/mm3, although this range can be quite broad (<100 to >10,000 cells/mm3). Up to 20% of adults with bacterial meningitis have a CSF WBC count lower than 1000 cells/mm3, and one third of these adults have a CSF WBC count of less than 100 cells/mm3.18 Classically, a CSF neutrophil predominance is present (seen in 80% to 95% of cases). In 10% of cases, such as in neonatal meningitis or patients infected with L. monocytogenes, a CSF lymphocyte predominance can be seen.18 In resource-depleted environments, a urinary reagent strip to determine the presence of leukocyte esterase can be used as a marker for the presence of WBCs in the CSF and a point of care glucose device can be used to rapidly obtain a CSF glucose concentration.
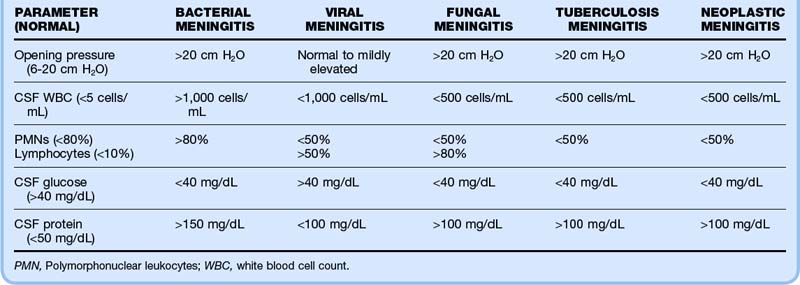
Despite the classic teachings on CSF findings (Table 171.3), the absence of one or more typical findings is commonly seen in patients with confirmed ABM. For example, in a review of 296 episodes of ABM, 50% of patients had a CSF glucose concentration of approximately 40 mg/dL, 44% had a CSF protein level lower than 200 mg/dL, and 13% had a CSF WBC count lower than 100 cells/mm3.19 In another series of 696 episodes of ABM, 12% had none of the characteristic CSF findings of ABM.3
Overall, the sensitivity of CSF Gram stain in bacterial meningitis ranges from 60% to 90%, depending on the concentration of the bacteria in the CSF.20 Sterilization of bacteria can begin to occur as soon as 15 minutes after the initiation of antibiotic therapy. A positive CSF Gram stain result is highly specific for bacterial meningitis. The following patterns are important to recognize: gram-positive diplococci suggest pneumococci; gram-negative diplococci suggest meningococci; small pleomorphic gram-negative coccobacilli suggest H. influenzae; gram-positive rods and coccobacilli suggest L. monocytogenes.
Treatment
A reasonable approach to immunocompetent patients with highly suspected meningitis or meningoencephalitis consists of empiric treatment with ceftriaxone (or cefotaxime), vancomycin, and acyclovir, along with dexamethasone (Table 171.4). Acyclovir is given to cover herpes simplex virus (HSV) encephalitis, the most common cause of nonepidemic encephalitis in the United States, whose presentation can significantly overlap with that of suspected meningitis. A more conservative approach to pharmacotherapy is reasonable for stable, immunocompetent patients with normal mentation and alertness in whom CNS infection is less strongly suspected.
Table 171.4 Empiric Antimicrobial Therapy for Suspected Meningitis
| PREDISPOSING FACTOR | ANTIMICROBIAL REGIMEN |
|---|---|
| Age < 1 mo | Cefotaxime 50 mg/kg IV q6h or |
| and | |
| Ampicillin 50 mg/kg IV q8h | |
| Age 1 mo-50 yr | Ceftriaxone 50 mg/kg (maximum dose, 2 g) IV q12h or Cefotaxime 50 mg/kg (maximum dose, 2 g) IV q6h |
| and | |
| Vancomycin 15 mg/kg IV q12h | |
| and | |
| Acyclovir 10 mg/kg IV q8h | |
| and | |
| Dexamethasone 0.15 mg/kg (maximum dose, 10 mg) IV q6h | |
| Age > 50 yr | Ceftriaxone 50 mg/kg (maximum dose, 2 g) IV q12h or Cefotaxime 50 mg/kg (maximum dose, 2 g) IV q6h |
| and | |
| Vancomycin 15 mg/kg IV q12h | |
| and | |
| Ampicillin 50 mg/kg (maximum dose, 2 g) IV q4h | |
| and | |
| Acyclovir 10 mg/kg IV q8h | |
| and | |
| Dexamethasone 0.15 mg/kg (maximum dose, 10 mg) IV q6h | |
| Postoperative neurosurgical patients | Ceftazidime 50 mg/kg (maximum dose, 2 g) IV q8h or Cefepime 50 mg/kg (maximum dose, 2 g) IV q8h |
| and | |
| Vancomycin 15 mg/kg IV q12h | |
| Patients with penetrating skull trauma | Ceftriaxone 50 mg/kg IV (maximum dose, 2 g) IV q12h or Cefotaxime 50 mg/kg IV (maximum dose, 2 g) IV q6h |
| and | |
| Vancomycin 15 mg/kg IV q12h |
IV, Intravenously; q4h, q6h, q8h, q12h, every 4, 6, 8, and 12 hours, respectively.
Treatment with high-dose dexamethasone (0.15 mg/kg intravenously, maximum dose, 10 mg, every 6 hours) before or concurrent with the first dose of antibiotics is thought to attenuate the inflammatory response and to lead to better outcome in children (excluding neonates) and adults with meningitis.20 The rationale for this approach is provided by animal studies showing that hearing loss is temporally associated with the severe inflammatory changes induced by bacterial meningitis and that dexamethasone reduces CSF synthesis of cytokines, CSF inflammation, and cerebral edema.
Antibiotics should not be delayed for CT or LP when the clinical suspicion of ABM is high. Although no prospective clinical data are available on the relationship of the timing of antibiotics with clinical outcome in patients with bacterial meningitis, several retrospective reviews examined this issue and concluded that an association may exist between delayed administration of antibiotics and worse overall outcome.18
Follow-Up, Next Steps of Care, and Patient Education
During hospitalization, focal neurologic deficits are seen in 50% of patients, and seizures occur in 15% of patients.3 Cardiopulmonary failure occurs in nearly 30% of patients, and mechanical ventilation is required in almost 25% of patients. Two thirds of patients with ABM have mild or no disability using a Glasgow outcome scale. Approximately 15% of patients have moderate to severe disability following infection. The most common neurologic findings on discharge are as follows: eighth nerve cranial palsy, which occurs in nearly 15% of survivors; hemiparesis, occurring in 4% of survivors; and sixth nerve cranial palsy, occurring in 3% of survivors.3 Aphasia, quadriparesis, third nerve cranial palsy, and seventh nerve cranial palsy are all rare.
Acute Encephalitis
Epidemiology
Encephalitis refers to inflammation of brain parenchyma that may coexist with inflammation of the meninges (meningoencephalitis) or spinal cord (encephalomyelitis). The overall incidence of encephalitis is reported to be 3 to 4 cases per 100,000 population. Most cases of encephalitis are often not recognized or misdiagnosed because routine care and time are sufficient for many cases to improve. Children less than 1 year of age, patients more than 65 years of age, and immunocompromised patients are at greatest risk for acute encephalitis.21 Viral infection is the most common identifiable cause of encephalitis, although infections with other pathogens and noninfectious causes (i.e., immune-mediated encephalitis) have been described (Table 171.5).
Table 171.5 Causes of Encephalitis Among Hospitalized Patients
| DIAGNOSIS | FREQUENCY (%) |
|---|---|
| Encephalitis, unspecified cause | 72 |
| Herpetic encephalitis | 14 |
| Immune-mediated encephalitis | 8 |
| Other viral encephalitis with identified cause | 4 |
| Bacterial encephalitis | 1 |
| Fungal, parasitic, or protozoal encephalitis | <1 |
Etiology
Many of the viruses that cause meningitis can also cause encephalitis, but certain viruses are more likely to cause encephalitis and are responsible for most cases. These include the herpes family viruses (e.g., HSV, human herpesvirus-6 [HHV-6], varicella-zoster virus [VZV], cytomegalovirus [CMV]), arboviruses (e.g., La Crosse virus, St. Louis virus, West Nile virus [WNV], Western Equine virus, Eastern Equine virus), and enteroviruses.22 Although HSV is the most common cause of nonepidemic, acute focal encephalitis in the United States, the arboviruses can account for as many as 50% of cases during epidemics.
Pathophysiology
Access of viruses to the CNS can occur by either hematogenous or neuronal routes.23 For example, after an insect bite with local arboviral replication in the skin, transient viremia ensues, followed by penetration of the blood-brain barrier and the development of encephalitis. Other agents can enter through the respiratory or gastrointestinal tract or through blood transfusion or organ transplantation. Several herpes family viruses (e.g., HSV, VZV) and the rabies virus reach the CNS through retrograde travel along neuronal axons where they have gained access to nerve endings.
Presenting Signs and Symptoms
Clinically distinguishing among the various infectious encephalitides is difficult because of the large degree of overlap in symptoms. Epidemiologic clues that may help in directing the investigation into a cause include the following: the season of the year; the geographic locale; the prevalence of the disease in the local community; and the patient’s travel history, recreational activities, occupational exposure, insect contact, animal contact, vaccination history, and immune status. Viral affinity for certain CNS locations can provide clues to the diagnosis (Table 171.6).22
| VIRUS | PRIMARY SITE OF CNS INFECTION | CLINICAL MANIFESTATIONS |
|---|---|---|
| Herpes simplex | Frontal and temporal lobe | Fever, hemicranial headache, taste and smell hallucinations, language and behavior abnormalities, memory impairment, seizures; SIADH |
| West Nile | Anterior horn cells | Abrupt onset of fever, headache, stiff neck, and vomiting; other clinical features including tremors, myoclonus, parkinsonism, and poliomyelitis-like flaccid paralysis |
| La Crosse | Cortical areas | Seizures, disorientation, focal neurologic signs; seen in late spring to early fall; primarily in school-age children |
| St. Louis | Substantia nigra, pons, thalamus, cerebellum | Tremor, myoclonus, opsoclonus, nystagmus, ataxia, stupor, disorientation; SIADH and urinary symptoms (dysuria, urgency, incontinence) |
| Eastern Equine | Basal ganglia, thalamus, brainstem | Headache, altered mental status, seizures; primary seen in summer |
CNS, Central nervous system; SIADH, syndrome of inappropriate antidiuretic hormone.
Medical Decision Making
The findings on CSF analysis of patients with encephalitis may be close to normal or similar to those seen in viral infections causing aseptic meningitis (i.e., increased CSF WBC count, usually < 250 cells/mm3, normal or mildly elevated CSF protein, and normal or mildly reduced CSF glucose).21 Red blood cells in an atraumatic LP suggest HSV encephalitis, but they can be present in other conditions (e.g., other viral encephalitides, amebic encephalitis, acute necrotizing hemorrhagic leukoencephalitis) (Box 171.3). CSF analysis is essential in patients with suspected acute encephalitis, and a CSF sample should be sent for routine analysis to exclude ABM. An additional CSF sample should be sent for nucleic acid amplification tests (e.g., PCR). A positive CSF PCR result is very helpful for documenting infection caused by a specific pathogen, but a negative PCR result cannot exclude this diagnosis. Viral cultures of CSF specimens are of limited value in patients with encephalitis and are not routinely recommended.
Treatment
The treatment of acute encephalitis is mainly supportive.22 This includes optimization of fluid balance and electrolytes, symptomatic treatment of fever, headache, and nausea, airway protection, management of ICP, and management of seizures. Effective therapy exists for HSV and VZV infection (i.e., acyclovir, 10 mg/kg intravenously every 8 hours). Anecdotal reports of improvement have also been described with the combination of ganciclovir (5 mg/kg intravenously every 12 hours) and foscarnet (90 mg/kg intravenously every 12 hours) for CMV or HHV-6 infection and with pleconaril (5 mg/kg orally every 8 hours) for severe enteroviral disease.22
A reasonable approach to immunocompetent patients with a high suspicion of meningitis, encephalitis, or meningoencephalitis consists of empiric treatment with ceftriaxone, vancomycin, and acyclovir, along with dexamethasone (Box 171.4). These therapies should be initiated as soon as possible after blood cultures are obtained, but before CT or LP are performed. A more conservative approach to pharmacotherapy is reasonable for immunocompetent patients with normal mentation and alertness who are less likely to have CNS infection.
Box 171.4 Recommended Empiric Treatment for Suspected Acute Encephalitis
Acyclovir 10 mg/kg IV every 8 hours
Ceftriaxone 50 mg/kg IV (maximum dose, 2 g) every 12 hours
Vancomycin 15 mg/kg IV (maximum dose, 500 mg) every 6 hours
Dexamethasone 0.15 mg/kg IV (maximum dose, 10 mg) every 6 hours
A key decision point in the continuation of therapy rests with the results of the Gram stain. Whereas acyclovir can be discontinued or omitted in patients with a positive Gram stain result, acyclovir should be continued or initiated in patients with a negative Gram stain result. Currently, acyclovir is given to less than one in three patients in the ED who ultimately have the diagnosis of encephalitis.24 Corticosteroids should be continued regardless of the results of the Gram stain. The use of corticosteroids in patients with HSV encephalitis is associated with improved outcome.
Follow-Up, Next Steps in Care, and Patient Care
Patients with all but the mildest cases of encephalitis should be admitted to the hospital. The overall risks of death and morbidity from encephalitis are 3% to 4% and 7% to 10%, respectively. These rates are greatly influenced by the infectious pathogen and by the immune response elicited by the host. Infections with HSV, rabies virus, and Eastern Equine virus and infections in immunocompromised, pediatric, and geriatric patients are all associated with a worse outcome. Before the advent of routine treatment with antiviral therapy, the mortality rate of untreated HSV encephalitis was greater than 70%, with less than 5% of the survivors returning to a normal lifestyle. The current mortality rate of HSV encephalitis in patients treated with acyclovir is less than 30%.25,26 Mortality rates of Eastern Equine, St. Louis, and La Crosse, and WNV encephalitis are greater than 30%, 20%, 10% to 15%, and 7.5%, respectively.
As with overall mortality from encephalitis, the prognosis depends on the specific etiologic agent and host factors. Significant lifelong morbidity may result from acute encephalitis. In one series that examined outcome after acyclovir-treated HSV encephalitis, 40% of surviving patients at 1 month had moderate to severe disability. Nearly 75% of the long-term survivors reported memory impairment, and approximately 50% had personality or behavioral abnormalities.22
Intracranial Abscess
Epidemiology
A brain abscess is a focal, intracerebral infection that begins as a localized area of cerebral inflammation and develops into a collection of pus surrounded by a well-vascularized capsule. With an incidence of 0.9 cases per 100,000 population, approximately 2500 cases of brain abscess are diagnosed each year in the United States.26 In the general population, brain abscess is a disease of young male patients. Case series have reported male-to-female ratios of 2 : 1 to 3 : 1.27 Although brain abscess can occur in any stage of life, most cases occur during the third and fourth decades. Additional risk factors for development of brain abscess are listed in Box 171.5.
Box 171.5 Identified Risk Factors for the Development of Brain Abscess
Immunocompromised patients (acquired immunodeficiency syndrome, transplantation, neutropenia)
Contiguous source of subacute or chronic infection (e.g., sinusitis, otitis media, mastoiditis, odontogenic infection, meningitis)
Chronic pulmonary infection (e.g., lung abscess, empyema)
Other infection (e.g., intraabdominal, pelvic, skin, bone)
Earlier neurosurgical procedure
Intrapulmonary right-to-left shunt in patients with pulmonary arteriovenous malformation
Pathophysiology
Bacteria can invade the brain by contiguous spread from nearby structures, by hematogenous invasion, or from direct implantation during surgery or penetrating head trauma. The most common contiguous infections include sinusitis, otitis media, and mastoiditis.28 Odontogenic infections (particularly involving those involving the molar teeth) account for up to 10% of cases. Hematogenous spread from distant sites of infection has been implicated in approximately 25% of brain abscesses. Endocarditis and pulmonary infections are among the most commonly reported distant foci of infection, but other sites of infection (e.g., intraabdominal, pelvic, skin, bone) can lead to the development of brain abscess. Direct implantation from invasive neurosurgical procedures or from penetrating head trauma, especially injury associated with a gunshot wound or retained foreign bone fragments, can also lead to the development of a brain abscess.29 No primary site or underlying condition can be identified in approximately one third of patients with brain abscess.
In pediatric patients, congenital heart disease is a significant risk factor for the development of a brain abscess. It accounts for 25% to 50% of the cases of brain abscesses in some pediatric series.30 Patients with cyanotic congenital heart disease have low-perfusion regions in their brain as a result of chronic severe hypoxemia and metabolic acidosis, as well as increased viscosity of the blood from secondary polycythemia, which may serve as a focus of infection. Furthermore, right-to-left shunting of the venous blood in the heart bypasses the pulmonary circulation, where phagocytes normally filter bacteria in the bloodstream.
Presenting Signs and Symptoms
The presenting symptoms of a brain abscess are often nonspecific and vary according to several factors, including the location and size of the abscess, the underlying immune status of the host, and the virulence of the infecting organism. Common presenting signs and symptoms in brain abscess are shown in Table 171.7. Patients with intracranial abscess often have a subacute onset of illness and rarely appear toxic. Because the initial presentation is often nonspecific, the diagnosis may be initially missed in the ED, and the patient may return for subsequent visits when symptoms persist. On average, the diagnosis is made 13 to 14 days after the onset of symptoms, although symptoms can last from a few hours to several months. The most common signs and symptoms of brain abscess are headache, mental status change, focal neurologic deficit, and fever.31 The clinical triad of headache, fever, and focal neurologic deficit is present in less than 20% of cases.
Table 171.7 Common Presenting Signs and Symptoms in Brain Abscess
| SIGN OR SYMPTOM | FREQUENCY (%) |
|---|---|
| Fever | 37-75 |
| Headache | 56-94 |
| Nausea or vomiting | 31-77 |
| Neck stiffness | 11 |
| Altered level of consciousness | 10-100 |
| Focal neurologic deficit | 49-75 |
| Seizure | 12-47 |
| Papilledema | 6-50 |
Medical Decision Making
The diagnosis of brain abscess is made by contrast-enhanced cranial CT or MRI. Nonenhanced cranial CT can identify approximately 90% of mature brain abscesses,32 but it is considered inadequate to exclude the diagnosis. The classic contrast-enhanced cranial CT appearance of a mature brain abscess is that of a ring-enhancing mass lesion with a hypodense center that is frequently surrounded by a substantial amount of edema. Contrast-enhanced cranial CT is highly sensitive (>95%) for identifying this type of lesion.29 Unfortunately, ring-enhancing lesions seen on CT images are not specific for brain abscess; cystic and necrotic neoplastic lesions, hematomas, demyelinating diseases, thrombosed giant aneurysm, and infarcted brain tissue may have the same CT characteristics. Both nonenhanced and enhanced cranial CT may miss lesions that are early in their maturity (i.e., during the “cerebritis” stage of brain abscess formation), small lesions, and lesions of the posterior circulation. Gadolinium-enhanced MRI is considered the gold standard for diagnostic imaging for this disease.
Treatment
Successful management of confirmed intracranial abscess involves a combination of broad-spectrum antibiotics and radiologically guided surgical drainage. Once the diagnosis is established, a neurosurgeon and an infectious disease specialist should be consulted. A sample of the infected tissue or fluid (pus) must be obtained quickly, to guide the initial therapy. The initial antibiotic regimen is based on the presumptive source of the abscess and on the Gram stain results, if available. Additional consultation with an oral-maxillofacial or ear, nose, and throat specialist may be required, depending on the extent of the primary infection. The principles of effective treatment of brain abscess are outlined in Box 171.6. Effective empiric antibiotic regimens for brain abscess are listed in Table 171.8.
Box 171.6 Principles of Effective Treatment for Brain Abscess
| LIKELY SOURCE OF INFECTION | ANTIBIOTIC REGIMEN |
|---|---|
| Contiguous site: odontogenic source | Penicillin G 3 to 4 million units IV q4h |
| and | |
| Metronidazole 15 mg/kg IV loading dose, followed by 7.5 mg/kg IV q8h | |
| Contiguous site: nonodontogenic source (e.g., sinusitis, otitis media, mastoiditis) | Ceftriaxone 50 mg/kg IV (maximum dose, 2 g) IV q12h or |
| Cefotaxime 50 mg/kg IV (maximum dose, 2 g) IV q6h | |
| and | |
| Metronidazole 15 mg/kg IV loading dose, followed by 7.5 mg/kg IV q8h | |
| Hematogenous spread of infection (e.g., bacteremia, endocarditis) | Ceftriaxone 50 mg/kg IV (maximum dose, 2 g) IV q12h or |
| Cefotaxime 50 mg/kg IV (maximum dose, 2 g) IV q6h | |
| and | |
| Metronidazole 15 mg/kg IV loading dose, followed by 7.5 mg/kg IV q8h | |
| and | |
| Vancomycin 15 mg/kg IV q12h | |
| Postoperative neurosurgical patients | Ceftazidime 50 mg/kg IV (maximum dose, 2 g) IV q8h or |
| Cefepime 50 mg/kg IV (maximum dose, 2 g) IV q8h | |
| and | |
| Vancomycin 15 mg/kg IV q12h | |
| Patients with penetrating skull trauma | Ceftriaxone 50 mg/kg IV (maximum dose, 2 g) IV q12h or |
| Cefotaxime 50 mg/kg IV (maximum dose, 2 g) IV q6h | |
| and | |
| Vancomycin 15 mg/kg IV q12h |
IV, Intravenous(ly); q4h, q6h, q8h, q12h, every 4, 6, 8, and 12 hours, respectively.
Follow-Up, Next Steps in Care, and Patient Care
Intracranial abscesses are associated with significant mortality and permanent neurologic morbidity. With the development of rapid diagnostic imaging, effective antibiotics, and improved surgical technique, reports from contemporary case series place the overall mortality from brain abscess at less than 10%. However up to 20% of the survivors will have severe neurologic disability or end up in a vegetative state. Neurologic morbidity and overall mortality are related to the initial level of consciousness at the time of diagnosis, host immune status, and response to initial therapy. Seizures occur in approximately 25% of patients with brain abscess.33 A frontoparietal location of the brain abscess or underlying valvular heart disease predicts seizure development in the presence of brain abscess.
![]() Documentation
Documentation
• The most important procedure to document in a patient presenting with suspected brain abscess is a good neurologic examination.
• Consider intracranial abscess in patients who have had recent, subacute, or chronic sinus, ear, mastoid, dental, or cardiac infection and new headache or focal neurologic deficit.
• Although this disease has nonspecific symptoms early in its course, litigation often focuses on the failure to diagnose brain abscess. Consistent documentation of return precautions in patients with headache and fever may limit time to diagnosis and your exposure if the case becomes litigated.
1 Thigpen MC, Whitney CG, Messonnier NE, et al. Bacterial meningitis in the United States, 1998-2007. N Engl J Med. 2011;364:2016–2025.
2 van de Beek D, Drake J, Tunkel AR. Nosocomial bacterial meningitis. N Engl J Med. 2010;362:146–154.
3 van de Beek D, de Gans J, Spanjaard L, et al. Clinical features and prognostic factors in adults with bacterial meningitis. N Engl J Med. 2004;351:1849–1859.
4 Uchihara T, Tsukagoshi H. Jolt accentuation of headache: The most sensitive sign of CSF pleocytosis. Headache. 1991;31:167–171.
5 Waghdhare S, Kalantri A, Joshi R, et al. Accuracy of physical signs for detecting meningitis: a hospital-based diagnostic accuracy study. Clin Neurol Neurosurg. 2010;112:752–757.
6 Attia J, Hatala R, Cook DJ, et al. Does this adult patient have acute meningitis? JAMA. 1999;282:175–181.
7 Thomas KE, Hasbun R, Jekel J, et al. The diagnostic accuracy of Kernig’s sign, Brudzinski’s sign, and nuchal rigidity in adults with suspected meningitis. Clin Infect Dis. 2002;35:46–52.
8 Curtis S, Stobart K, Vandermeer B. Clinical features suggestive of meningitis in children: a systematic review of prospective data. Pediatr. 2010;126:952–960.
9 Delerme S, Castro S, Viallon A, et al. Meningitis in elderly patients. Eur J Emerg Med. 2009;16:273–276.
10 Chern CH, Yee YL, Wei CH, et al. The misdiagnosis of meningitis in the Emergency Department. Ann Emerg Med. 38, 2001. abstract 110
11 Nigrovic LE, Kupperman N, Macias CG, et al. Clinical prediction rule for identifying children with cerebrospinal fluid pleocytosis at very low risk of bacterial meningitis. JAMA. 2007;297:52–60.
12 Dubos F, Korczowski B, Aygun DA, et al. Serum procalcitonin level and other biological markers to distinguis between bacterial and aseptic meningitis in children. Arch Pediatr Adolesc Med. 2008;162:1157–1163.
13 Ray P, Badarou-Acossi G, Viallon A. Accuracy of the cerebrospinal fluid results to differentiate bacterial from non bacterial meningitis in case of negative gram-stained smear. Am J Emerg Med. 2007;25:179–184.
14 Holub M, Beran O, Dzupova O, et al. Cortisol levels in cerebrosppinal fluid correlate with severity and bacterial origin of meningitis. Crit Care. 2007;11:R41.
15 Huy NT, Thao NTH, Diep DTN, et al. Cerebrospinal fluid lactate concentration to distinguish bacterial from aseptic meningitis: A systematic review and meta-analysis. Crit Care. 2010;14:R240.
16 Joffe AR. Lumbar puncture and brain herniation in acute bacterial meningitis: A review. J Intensiv Care Med. 2007;22:194–207.
17 Hasbun R, Abrahams J, Jekel J, et al. Computed tomography of the head before lumbar puncture in adults with suspected meningitis. N Engl J Med. 2001;345:1727–1733.
18 Sadoun T, Singh A. Adult acute bacterial meningitis in the United States: 2009 Update. Emerg Med Prac. 2009;11:1–30.
19 Durand MI, Calderwood SB, Weber DJ, et al. Acute bacterial meningitis in adults. A review of 493 episodes. N Engl J Med. 2003;328:21–28.
20 Tunkel AR, Hartman BJ, Kaplan SL, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis. 2004;39:1267–1284.
21 Trevejo RT. Acute encephalitis hospitalizations, California 1990-1999: unrecognized arboviral encephalitis. Emerg Infect Dis. 2004;10:1442–1449.
22 Trunkel AR, Glaser CA, Bloch KC, et al. The management of encephalitis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2008;47:303–327.
23 Somamd D, Meurer W. Central nervous system infection. Emerg Med Clin N Am. 2009:89–100.
24 Benson PC, Swadron SP. Empiric acyclovir is infrequently initiated in the emergency department to patients ultimately diagnosed with encephalitis. Ann Emerg Med. 2006;47:100–105.
25 Whitley RJ, Alford CA, Hirsch MS, et al. Vidarabine versus acyclovir therapy in herpes simplex encephalitis. N Engl J Med. 1986;314:144–149.
26 McGrath N, Anderson NE, Croxson MC, et al. Herpes simplex encephalitis treated with acyclovir: diagnosis and long term outcome. J Neurol Neurosurg Psychiatry. 1997;63:321–326.
27 Sharma R, Mohandas K, Cooke RP. Intracranial abscesses: changes in epidemiology and management over five decades in Merseyside. Infection. 2009;37:39–43.
28 Mathisen GE, Johnson JP. Brain abscess. Clin Infect Dis. 1997;25:763–781.
29 Honda H, Warren DK. Central nervous system infections: meningitis and brain abscess. Infect Dis Clin North Am. 2009;23:609–623.
30 Goodkin HP, Harper MB, Pomeroy SL. Intracerebral abscess in children: histroical trends at Children’s Hospital Boston. Pediatr. 2004;113:1765–1770.
31 Tunkel AR. Brain abscess. In: Mandel GL, Bennett JE, Dolin R. Principles and Practice of infectious disease. 6th ed. Philadelphia: Churchill Livingstone; 2005:1154.
32 Fitzpatrick MO, Gan P. Lesson of the week: contrast enhanced tomography in the early diagnosis of cerebral abscess. BMJ. 1999;319:239–240.
33 Xiao F, Tseng MY, Teng LJ, et al. Brain abscess: clinical experience and analysis of prognostic factors. Semin Neurol. 2005;63:442–450.
[/level-membership-for-emergency-medicine-category][not-level-membership-for-emergency-medicine-category]
171 Meningitis, Encephalitis, and Brain Abscess
• There is significant overlap among the initial clinical presentations of meningitis, encephalitis, and brain abscess.
• The four most common bacteria responsible for adult bacterial meningitis are Streptococcus pneumoniae, Neisseria meningitides, Haemophilus influenzae type B, and Listeria monocytogenes. Group B Streptococcus remains the predominate cause of meningitis in infants less than 2 months of age.
• The classic constellation of fever, neck stiffness, headache, and change in mental status are seen in less than 50% of cases of acute bacterial meningitis.
• Cranial computed tomography (CT) scan, prior to lumbar puncture, is recommended in patients with a history of immunocompromised state, history of central nervous system (CNS) disease, new-onset seizure, abnormal neurologic examination, papilledema, altered mental status, or altered level of consciousness.
• Empiric therapy in patients with high clinical suspicion for CNS infection should not be delayed for neuroimaging or lumbar puncture.
• Although epidemiologic clues and assessment of risk factors should be sought in all patients with encephalitis, herpes simplex virus and arboviruses remain the most common causes of nonepidemic and epidemic outbreaks of encephalitis, respectively, in the United States.
• Acyclovir should be initiated in all patients with suspected encephalitis, pending the results of diagnostic studies.
• Risk factors for the development of intracranial abscess include inadequately treated subacute or chronic ear, nose, mastoid, and dental infection; endocarditis; congenital heart disease; and having undergone neurosurgical procedures.
• Patients with intracranial abscess often present with mild headache symptoms in the weeks to months prior the emergency department visit. The classic triad of fever, headache, and focal neurologic deficit is seen in less than 20% of patients with brain abscess.
Meningitis
Epidemiology
The combination of routine vaccination against Streptococcus pneumoniae, Haemophilus influenzae type B, and Neisseria meningitides, maternal screening for and intrapartum treatment of group B Streptococcus (Streptococcus agalactiae), and enhanced efforts to reduce the contamination of processed foods by Listeria monocytogenes have all led to a significant decrease in the incidence of acute bacterial meningitis (ABM). Data from the Emerging Infections Programs Network, established by the Centers for Disease Control and Prevention (CDC), have noted a decrease in the incidence of meningitis from 2.0 cases per 100,000 population in 1998 to 1999 to 1.38 cases per 100,000 population in 2006 to 2007.1 During this same time the case fatality rate decreased to 14.3%. Projecting these data on a national level reveals an estimated 4100 cases and 500 deaths from bacterial meningitis annually in the United States.1
Etiology
A 10-year review (1998 to 2007) of 3188 cases of bacterial meningitis noted that S. pneumoniae accounted for the greatest proportion of cases (58%), followed by group B. streptococcus (18.1%), N. meningitides (13.9%), H. influenzae (6.7%), and L. monocytogenes (3.4%).1 Among infants less than 3 months of age, group B Streptococcus and gram-negative rods account for most cases of ABM. After 3 months of age, S. pneumoniae and N. meningitidis become the predominant pathogens. L. monocytogenes is primarily seen in infants less than 1 month of age, in adults more than 50 years old, and in immunocompromised patients. Staphylococcus aureus is acquired mainly nosocomially and occurs predominantly after neurosurgical procedures or following penetrating head trauma. S. aureus may be acquired in the community setting, linked to predisposing conditions such as endocarditis, injection drug use, and compromised immune systems.
Pathophysiology
ABM develops after encapsulated bacteria, which have colonized the nasopharynx and/or oropharynx, penetrate the intravascular space and enter the subarachnoid space through vulnerable sites within the blood-brain barrier.2 Once the pathogens enter the central nervous system (CNS), they replicate rapidly, thus consuming glucose and liberating protein within the cerebrospinal fluid (CSF). The ensuing inflammatory reaction occurs in response to the liberation of bacterial cell wall and cell membrane components (e.g., lipopolysaccharide, peptidoglycan, lipoteichoic acid) and the induction of proinflammatory mediators. These events culminate in injury to the vascular endothelium that results in increased vascular permeability to the blood-brain barrier, meningeal inflammation, and cerebral vasculitis. The accompanying cerebral edema and increase in intracranial pressure (ICP) contribute to CNS hypoperfusion and cell death.
Presenting Signs and Symptoms
Patients with ABM typically appear ill and often present within 24 to 72 hours of symptom onset. Table 171.1 reviews the presenting signs and symptoms of adults with ABM.3 The cardinal symptoms of ABM (i.e., fever, neck stiffness, change in mental status, and headache) are seen in combination in less than half of all patients. Nearly 95% of patients will present with at least two of these cardinal symptoms, which provides the rationale for performing a lumbar puncture in patients who are lethargic or confused and develop a fever. The absence of these four findings typically excludes the diagnosis of ABM.
Table 171.1 Findings in 696 Episodes of Bacterial Meningitis
| SIGN OR SYMPTOM | FREQUENCY (%) |
|---|---|
| Duration of symptoms ≤ 24 hr | 48 |
| Fever (temperature ≥ 38° C) | 77 |
| Headache | 87 |
| Nausea or vomiting | 74 |
| Neck stiffness | 83 |
| GCS ≤ 14 (AMS) | 69 |
| GCS ≤ 8 (coma) | 14 |
| Rash | 26 |
| Focal neurologic deficit | 33 |
| Seizures | 5 |
| Arthritis | 7 |
AMS, altered mental status; GCS, Glasgow Coma Scale.
From Van de Beek D, de Gans J, Spanjaard L, et al. Clinical features and prognostic factors in adults with bacterial meningitis. N Engl J Med 2004;351:1849-59.
The headache described by patients with ABM can be moderate to severe in intensity, generalized, often with an occipital or nuchal component, and unlike “normal” headaches. Photophobia is commonly present, as is nausea. Worsening of the headache while the examiner rapidly turns the patient’s head from side to side (at a rate of two to three times per second), the so-called jolt accentuation test, has been reported to be helpful in identifying patients with ABM,4 but a recent study questioned the utility of this finding.5
Although neck pain may be infrequently reported, the objective finding of neck stiffness is seen in more than 80% of patients. Examining the neck for rigidity, during gentle forward flexion, with the patient in the supine position best assesses neck stiffness, whereas difficulty in lateral motion of the neck is a less reliable finding. Patients with severe meningeal irritation may spontaneously assume the tripod position (also called the Amoss sign or the Hoyne sign) with the knees and hips flexed, the back arched lordotically, the neck extended, and the arms brought back to support the thorax.6 The Kernig sign is performed with the patient lying supine and the hip and knee flexed to 90 degrees. A positive sign is present when extension of the knee from this position elicits resistance or pain in the lower back or posterior thigh. The classic Brudzinski sign refers to spontaneous flexion of the knees and hips during attempted passive flexion of the neck. A separate sign described by Brudzinski, the contralateral reflex, is present if passive flexion of one hip and knee causes flexion of the contralateral leg. The presence or absence of Kernig or Brudzinski signs has been shown to have little positive or negative predictive value in the diagnosis of ABM, unless severe meningeal inflammation is present.7
Atypical presentations of ABM occur in infants, older adults, and immunocompromised patients. Infants with bacterial meningitis may present with fever or hypothermia, hypoglycemia, poor feeding, seizures, or irritability (excessive or abnormal crying). On examination, the findings of jaundice, ill appearance, a bulging fontanelle, meningeal irritation (including neck stiffness, the Kernig sign, and the Brudzinski sign), fever higher than 40° C, and increased general body tone predict bacterial meningitis.8
Older and immunocompromised patients may also present atypically. These populations are associated with a higher rate of misdiagnosis that contributes to an increase in the morbidity and mortality following an episode of acute meningitis. A lower proportion of fever, headache, and nausea or vomiting is present in these subgroups.9 Neck stiffness has a lower sensitivity and specificity for meningitis in older patients. Finally, these populations may present to the emergency department (ED) with altered mental status and/or altered level of consciousness but without a fever.
Differential Diagnosis
The differential diagnosis of patients presenting with fever, headache, and altered mental status includes other forms of meningitis (e.g., nosocomial meningitis, aseptic meningitis), encephalitis, and cerebral abscess. The diagnosis of meningitis is challenging in patients who present atypically. In a review of 156 cases of meningitis in patients who presented to a single tertiary care hospital, 66 cases were initially misdiagnosed in the ED as an alternative infection (i.e., sepsis of unclear origin, pneumonia, urinary tract infection), metabolic encephalopathy, or nonspecific conditions (e.g., weakness, malaise, degenerative state). Higher percentages of these patients were more than 65 years of age, and these patients were also noted to have lower proportions of fever, headache, nausea or vomiting, and neck stiffness.10
Aseptic meningitis refers to a disorder in which patients have clinical and laboratory evidence of meningeal irritation with negative results of routine bacterial cultures. Precise epidemiologic data on the incidence of aseptic meningitis are lacking, but aseptic meningitis is associated with an estimated 26,000 to 42,000 hospitalizations per year in the United States. The origin of aseptic meningitis is varied (Box 171.1). Enteroviruses, the leading causes of viral meningitis in adults and children, account for 50% to 75% of all cases of aseptic meningitis. Additional causes include other infections (mycobacteria, fungi, spirochetes), parameningeal infections, medications (especially nonsteroidal antiinflammatory drugs), and malignant disease. The signs and symptoms of bacterial meningitis significantly overlap with those of aseptic meningitis. This overlap led to the development of several decision rules to distinguish the two conditions. The most useful pediatric score appears to be the Bacterial Meningitis Score. This score classifies patients 1 month to 18 years old as being at very low risk of bacterial meningitis if they lack all the following criteria: positive CSF Gram stain, CSF absolute neutrophil count (ANC) of at least 1000 cells/mcL, CSF protein of at least 80 mg/dL, peripheral blood ANC of at least 10,000 cells/mcL, and a history of seizure before or at the time of presentation.11
Medical Decision Making
Routine Laboratory Tests
Routine testing of patients with suspected meningitis should include complete blood cell count (CBC), serum electrolytes, bicarbonate, serum urea nitrogen (BUN), creatinine, and glucose (Table 171.2). Serum lactate determinations and blood cultures are also indicated in patients with suspected meningitis.
Table 171.2 Suggested Laboratory Testing in Suspected Meningitis
| BLOOD TEST | COMMENT |
|---|---|
| Complete blood count | WBC typically elevated with left shift, although normal or low values in infants and immunosuppressed patients |
| Electrolytes | Hyponatremia (Na < 135 mmol/L) seen in 30% of cases of ABM |
| Bicarbonate | Alkalosis seen with excessive vomiting, acidosis seen with poor tissue perfusion |
| BUN, creatinine | Renal function tests essential for antibiotic dosing and timing |
| Glucose | Useful in calculating the CSF/serum glucose ratio and in the initial evaluation of altered mental status or altered level of consciousness |
| Lactate | Has prognostic information (i.e., correlates with mortality) and used to identify candidates for early goal-directed therapy |
| Blood cultures | Positive results in 50% to 75% of patients with ABM when obtained before antibiotic administration |
ABM, Acute bacterial meningitis; BUN, blood urea nitrogen; CSF, cerebrospinal fluid; Na, sodium; WBC, white blood cell count.
Several newer tests have shown potential in distinguishing bacterial meningitis from nonbacterial meningitis. These tests include serum procalcitonin,12 serum C-reactive protein,13 CSF cortisol,14 and CSF lactate.15 Additional tests employing common biochemical laboratory techniques (e.g., latex agglutination, enzyme-linked immunosorbent assay, polymerase chain reaction [PCR] assay, microarrays) have shown significant promise in identifying the specific pathogen responsible for infection.
Neuroimaging Before Lumbar Puncture
Selected patients with meningitis may warrant a computed tomography (CT) scan of the head, to identify patients with lesions that place them at risk for herniation from LP and to diagnose conditions that would make LP unnecessary if the patient’s work-up was limited to the LP (e.g., tumor, cerebral abscess). Unfortunately, cranial CT has inadequate sensitivity for identifying patients at risk for brain herniation. A systematic review on this subject found only a handful of cases of brain herniation that occurred following a normal cranial CT scan.16 Despite this limitation, generally accepted criteria for obtaining a cranial CT scan before LP are listed in Box 171.2. For maximal sensitivity in those patients with suspected or confirmed human immunodeficiency virus infection, contrast-enhanced cranial CT should be performed at the same time as the nonenhanced cranial CT.
Box 171.2
General Recommendations for Computed Tomography Before Lumbar Puncture17
History of immunocompromised state
History of central nervous system disease (e.g., mass lesion, stroke, focal infection)
New-onset seizure (or new-onset seizure within 1 week of presentation)
Papilledema on funduscopic examination (or elevated optic nerve sheath diameter on ultrasound)
Hasbun R, Abrahams J, Jekel J, et al. Computed tomography of the head before lumbar puncture in adults with suspected meningitis. N Engl J Med 2001;345:1727-33.
Lumbar Puncture and Cerebrospinal Fluid Analysis
Although the diagnosis of bacterial meningitis rests on CSF examination, CSF analysis alone cannot reliably distinguish bacterial and aseptic meningitis.13 In addition to measuring the opening pressure, the examiner should obtain four tubes of CSF, each containing 1 to 2 mL of fluid, and send them for analysis. Typically tube 1 (and/or tube 4) is sent for cell count and differential, tube 2 for protein and glucose, tube 3 for Gram stain and culture, and tube 4 for special testing or additional cultures.
The appearance of the CSF can range from clear to cloudy, depending on the presence of significant concentrations of cells, bacteria, and protein. The CSF white blood cell (WBC) count can be significantly elevated, usually in the range of 1000 to 5000 cells/mm3, although this range can be quite broad (<100 to >10,000 cells/mm3). Up to 20% of adults with bacterial meningitis have a CSF WBC count lower than 1000 cells/mm3, and one third of these adults have a CSF WBC count of less than 100 cells/mm3.18 Classically, a CSF neutrophil predominance is present (seen in 80% to 95% of cases). In 10% of cases, such as in neonatal meningitis or patients infected with L. monocytogenes, a CSF lymphocyte predominance can be seen.18 In resource-depleted environments, a urinary reagent strip to determine the presence of leukocyte esterase can be used as a marker for the presence of WBCs in the CSF and a point of care glucose device can be used to rapidly obtain a CSF glucose concentration.
Despite the classic teachings on CSF findings (Table 171.3), the absence of one or more typical findings is commonly seen in patients with confirmed ABM. For example, in a review of 296 episodes of ABM, 50% of patients had a CSF glucose concentration of approximately 40 mg/dL, 44% had a CSF protein level lower than 200 mg/dL, and 13% had a CSF WBC count lower than 100 cells/mm3.19 In another series of 696 episodes of ABM, 12% had none of the characteristic CSF findings of ABM.3
[/not-level-membership-for-emergency-medicine-category]