Immunoglobulins and Their Production by B Cells
I Immunoglobulin Structure and Functions
• Immunoglobulins, synthesized by B cells, are antigen-binding glycoproteins (antibodies) that function in the recognition of and defense against antigens (Table 3-1).
TABLE 3-1
Antigen and Antibody Terminology
| Term | Definition |
| Adjuvant | Substance that enhances immune response to an antigen when administered with it; used to improve response to vaccines |
| Affinity | Binding strength of a single variable region of an antibody for a corresponding epitope on the larger antigen structure |
| Antigen | Substance that binds to antibodies and T cell receptors. Although most antigens are also immunogens, some small molecules are antigenic but not immunogenic. |
| Avidity | Combined binding strength of the multiple interactions between a multivalent antibody molecule and all the corresponding epitopes on an antigen |
| Epitope (antigenic determinant) | Region on an antigen molecule to which a single antibody molecule or T cell receptor binds. An antigen usually has multiple epitopes and thus can react with antibodies of different specificities. |
| Fab fragment | Portion of antibody molecule, produced by papain digestion, that contains a single antigen-binding site. All antibodies have two or more Fab regions and thus are bivalent or multivalent. |
| Fc fragment | Portion of antibody molecule, produced by papain digestion, that fixes complement and binds to Fc receptors; varies among immunoglobulin isotypes |
| Hinge region | Flexible portion of antibody heavy chains located between the Fab and Fc regions and containing intrachain disulfide bonds; present in IgG, IgA, and IgD |
| Immunogen | Substance capable of eliciting a specific immune response |
| Monoclonal antibody | Homogeneous antibody that recognizes only one epitope; produced by a single clone of plasma cells |
| Polyclonal antibody | Mixture of antibodies that recognize different epitopes on an antigen; produced by multiple clones of plasma cells in response to an antigen containing different epitopes. Natural antiserum to a microbial antigen is polyclonal. |
| Thymus-dependent antigens | Antigens that require helper T cells to induce antibody production (humoral response); most protein antigens |
| Thymus-independent antigens | Antigens possessing many repetitive structures (e.g., flagellin, polysaccharide, and LPS) that can induce antibody production (humoral response) without helper T cells |
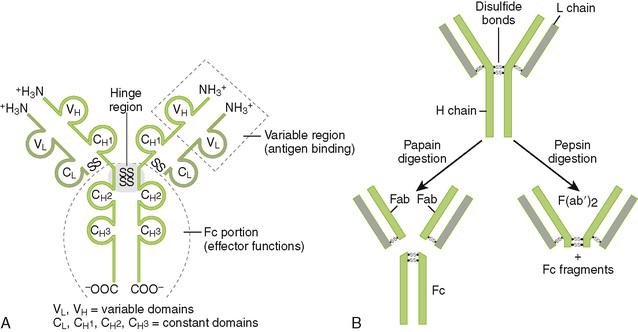
A Chain structure of immunoglobulins (Fig. 3-1A)
1. Each monomeric antibody molecule comprises two identical heavy (H) chains and two identical light (L) chains (κ or λ).
2. Antigenic specificity is determined by the amino acid sequence of the variable domains near the amino-terminal end of each chain.
B Functional regions of antibody molecules
1. Papain digestion cleaves the antibody molecule into two Fab fragments and one Fc fragment (Fig. 3-1B).
2. The Fab portion contains variable region (VL/VH) domains, which bind antigen.
3. The Fc portion mediates antigen clearance by binding to complement and to Fc receptors on immune system cells (Table 3-2).
TABLE 3-2
Functions Mediated by Interactions with Antibody Fc Region
| Function | Fc Region Interacts with |
| Opsonization | Fc receptors on macrophages and neutrophils |
| Killing by means of ADCC | Fc receptors on neutrophils, macrophages, NK cells, eosinophils |
| Degranulation leading to allergic and antiparasitic responses | Fc receptors for IgE on mast cells |
| Activation of cells | Fc receptors on lymphocytes |
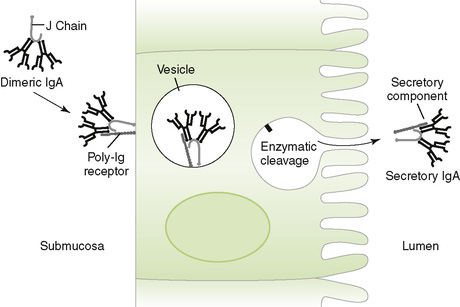
| Transmucosal movement | Fc receptors for dimeric IgA on epithelial cells |
| Activation of classical complement pathway leading to cell lysis (especially of bacteria), opsonization, and inflammatory response | Initial component of pathway (C1) |
ADCC, antibody-dependent cellular cytotoxicity; NK, natural killer.
4. Membrane-spanning region is a heavy-chain carboxyl-terminal domain present only in immunoglobulins expressed on the surface of B cells.
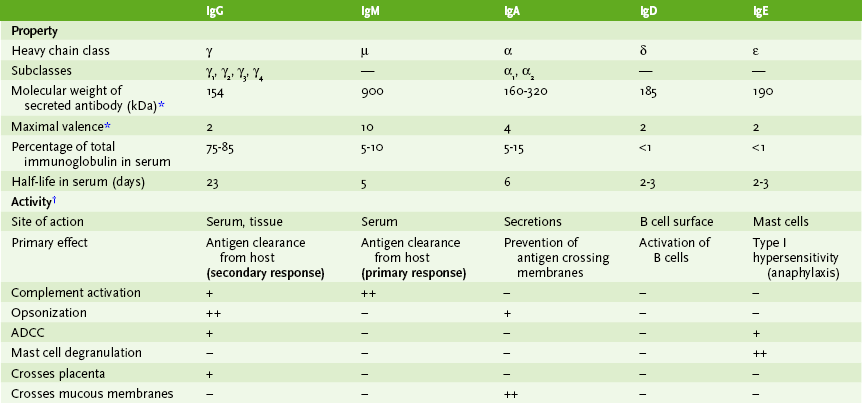
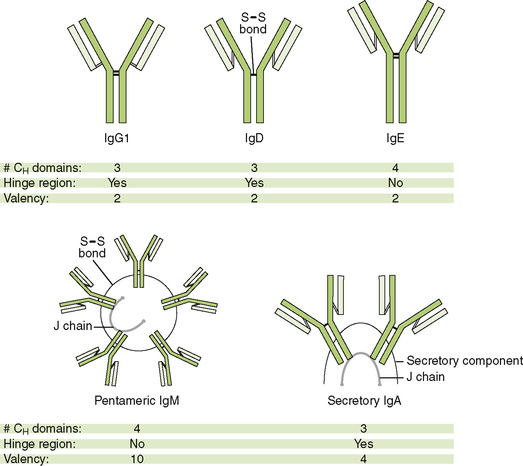
C Properties of immunoglobulin isotypes
• The five major immunoglobulin classes, or isotypes, exhibit different functions and roles in immunity (Table 3-3; Fig. 3-2).
b. Too large to spread into tissue from serum
c. Held in multimeric form by J chain and disulfide bonds
d. Effective antibacterial, complement-binding antibody
e. Major component of rheumatoid factor (an autoantibody against the Fc portion of IgG)
• Monomeric IgM: present in membrane form on surface of B cells
• Fixes complement; acts as opsonin; stimulates chemotaxis
• Occurs as several subtypes, which have slightly different structures and vary slightly in their functional activity
• Predominant antibody isotype in external secretions (e.g., saliva, mucus, breast milk)
• Found mostly as monomer in serum and as dimer in secretions (secretory IgA) held together by J chain
D Antigenic determinants on antibodies
1. Immunoglobulins, like other proteins, can induce an immune response.
2. Three major groups of immunoglobulin epitopes—isotypic, allotypic, and idiotypic—differ in their location within antibody molecules and/or distribution among individuals (Table 3-4).
TABLE 3-4
Antigenic Determinants on Antibodies
| Epitope Class | Location | Comment |
| Isotype | Constant region | These epitopes, which define each class of Ig heavy chains, are identical in all members of a species. The five human isotypes are IgA, IgD, IgE, IgG, and IgM. (Iso = same.) |
| Allotype | Constant region | These epitopes vary among individuals. IgG exhibits the most allotypic differences. (Allo = different.) |
| Idiotype | Variable region | These epitopes differ among antibodies because of different antigen-binding specificities. Monoclonal antibodies have the same idiotype. (There are many “idiot” types.) |
II Development and Activation of B Cells
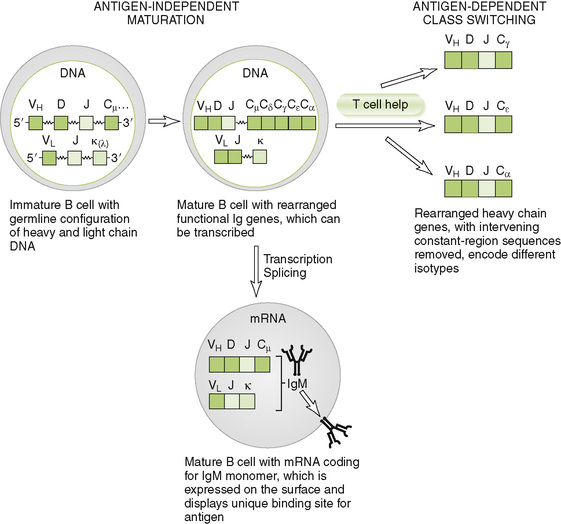
A Antigen-independent maturation of B cells
• Naive, immunocompetent B cells are generated in the bone marrow from hematopoietic precursors (see Fig. 1-3).
1. Germline Ig DNA consists of multiple coding segments separated by noncoding regions.
• Light chain germline DNA contains many V, 5 J, and 1 C segments in humans.
• Heavy chain germline DNA contains many V, many D, 6 J, and multiple C segments in humans.
2. Random recombination of gene segments forms VLJ (light chain) or VHDJ (heavy chain) units, which encode the variable region of each chain and determine the antigenic specificity of mature B cells (Fig. 3-4).
3. Splicing of primary RNA transcripts formed by mature B cells yields messenger RNAs (mRNAs) with a single variable region and single constant region.
B Stimulation by T-independent (TI) antigens
2. Repetitive, polymeric antigens (e.g., lipopolysaccharide, dextran, capsular polysaccharides, and flagellin) activate B cells in the absence of TH cells.
3. B cell response to TI antigens does not exhibit isotype switching, affinity maturation, or production of memory cells.
C Stimulation by T-dependent (TD) antigens (Box 3-1)
• Activation, proliferation, and differentiation of naive B cells in response to most protein antigens is driven by direct interaction with CD4 TH cells and the action of various cytokines.
1. Three types of signals are required for response to TD antigens.
• Antigen-triggered signal: antigen binds to immunoglobulin and triggers tyrosine kinase activation cascade.
a. Coreceptors (e.g., CD21 [C3d receptor] and CD19) intensify initial signal triggered by cross-linkage of membrane immunoglobulin molecules by antigen.
b. Increased expression of class II major histocompatibility complex (MHC) molecules is induced.
c. Antigen is endocytosed and degraded, and peptides are displayed on B cell surface associated with class II MHC molecules.
a. Binding of cytokines secreted primarily by activated TH cells stimulates subsequent proliferation and differentiation of B cells.
b. Plasma cells are terminally differentiated B cells (do not divide) that secrete antibody.
c. Memory B cells, which express membrane-bound antibody of any isotype, respond faster than naive B cells to second exposure to antigen.
2. Affinity maturation (clonal expansion) results from selective expansion of B cell clones that make the best antibody.
• The immunoglobulins produced by these cells have increased average binding strength improving the antibody mixture to the antigen.
a. Somatic mutation occurs randomly within the variable region of heavy and light chain genes during the course of the B cell response.
• As a result, some B cells begin to produce higher-affinity immunoglobulin.
3. Class (isotype) switching occurs as the immune response progresses.
III Antibody Effector Mechanisms
A Neutralization of viruses and toxins
• Occurs when these agents become coated with antibody, which interferes with their binding to their receptors and prevents the infectious or toxic process from proceeding
C Complement activation (IgG and IgM)
D Antibody-mediated agglutination (IgM) of bacteria
E Antibody-dependent cellular cytotoxicity
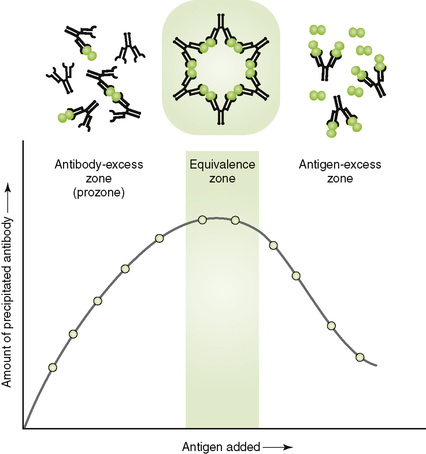
• Antigen-antibody reactions provide the basis for qualitative and quantitative tests for both antigen (Ag) and antibody (Ab) (see Chapter 5).