Chapter 44 Endoscopic Approaches to Skull Base Lesions, Ventricular Tumors, and Cysts
• Become knowledgeable about the equipment and practice on a cadaver or under the guidance of an experienced neuroendoscopist.
• Orientation and anatomy of the cranial skull base and ventricles are everything! Absolutely know where you are, what you are looking at, and your orientation at all times. In this chapter, the most important neurovascular structures related to the cranial skull base approaches are defined. If you are not sure, abort the procedure.
• Use the endoscope for its advantages and don’t fight its disadvantages. Increase your use of angled scopes and increase the size of your visualized field.
• Become comfortable with the endoscopic management of hydrocephalus prior to using the endoscope for neuro-oncological applications.
• Endoscopic ventricular applications are not “all or nothing.” Despite being an experienced endoscopist, some lesions are more safely dealt with from a microscopic surgical approach. In these cases use an endoscope-assisted approach to your advantage.
• The transsellar approach is limited to exposing the sella turcica. Because the sella is the epicenter at the crossroads of the sagittal and coronal planes, the transsellar approach is the starting point for most of the expanded endonasal surgical modules.
• All lateral expanded approaches to the middle and posterior fossae require a transpterygoid approach on the ipsilateral side. The vidian nerve is a key anatomical landmark that must be localized and followed back to the anterior genu of the internal carotid artery (ICA) as its petrous portion turns up to form the vertical paraclival ICA.
During the past two decades there has been increased use of neuroendoscopic surgery. Detailed anatomical studies have improved the understanding of endoscopic ventricular and skull base anatomy. This, along with the use of intraoperative image guidance, has enabled surgeons to approach deeply seated lesions through minimally invasive routes. The feasibility and the safety of such extended approaches have been well established in numerous anatomical and clinical studies.1–11 The endoscope can be used to definitively treat hydrocephalus, to biopsy tumors that are best treated with radiation or chemotherapy, and to confirm adequate resection of tumors when the microscopic view is partially obstructed. On the other hand, the expanded endonasal approach (EEA) can provide access to the anterior, middle, and posterior cranial fossae and is recognized as an important tool in the armamentarium of skull base surgeons.12,13
Many surgical advantages have been attributed to EEA in comparison with traditional transcranial approaches including access to deeply seated lesions, a more direct midline exposure, decreased brain parenchyma injury, absent neurovascular structure manipulation, prompt decompression of visual apparatus when indicated, and early tumor devascularization.4–614 Similar oncological results to those obtained with traditional open approaches have been documented for sinonasal, sellar, and skull base lesions.1,5,8,9,14–16 From the patient’s perspective, decreased surgery time, decreased length of stay, increased patient comfort and lack of external incision are significant advantages to the EEA.11
History
Six major advancements in the use of neuroendoscopy have occurred. A century ago, L’Espinasse, a urologist, attempted to perform a choroid plexus coagulation to treat hydrocephalus.17,18 In the early twentieth century Dandy and Mixter attempted an endoscopic third ventriculostomy (ETV).19 In the 1970s technological advances allowed the production of flexible fiberscopes as well as rigid endoscopes. Throughout the late 1980s and 1990s ETV and decompartmentalization of the ventricular system because popularized and allowed many neurosurgeons to become familiar with the tools of neuroendoscopy. This familiarity resulted in the fifth and sixth advances. The use of the endoscope for extra-axial lesions was popularized by pioneers such as Perneczky, who championed the keyhole approach to aneurysms. The final and most recent wave of endoscopic enthusiasm has been its application to anterior, middle, and posterior skull base surgery, especially transsellar and transpterygoid approaches. The use of the endoscope for intraventricular neuro-oncological applications including lesion removal, tumor biopsy, and cyst management is also becoming more common, so much so that endoscopic colloid cyst surgery has results that are similar to those achieved with microsurgery and with a better risk profile.20–24
Equipment
The flexible fiberscope is maneuverable in three directions with relatively simple controls. The advantage of the flexible scope is that its geometry is not fixed and it may be fashioned to proceed along a curved trajectory such as a trans–foramen of Monro approach to the posterior third ventricle to biopsy a pineal tumor. This allows a single trajectory to treat hydrocephalus via ETV and sample the tumor; the flexible scope may also be used within the working port of the rigid scope in a “scope-in-scope” technique.25 The main drawbacks of the flexible scope over the rigid scope are the poorer optical resolution and the risk that the surgeon may inadvertently withdraw the scope in the “bent” configuration with devastating repercussions.
Frameless neuronavigation is helpful to guide insertion of the sheath, especially in the absence of ventriculomegaly; for localizing lesions for biopsy under an intact ependyma;26 and to localize important neurovascular structures during skull base surgery. In the instance that the surgeon becomes disoriented or confused by distorted anatomy, stereotaxis can assist in reorientation.
Recently, several potentially important advances in instrumentation available to the endoscopic surgeon have been reported. The length of time required to aspirate a tumor using a “biopsy after biopsy” approach may be shortened by the use of traditional ultrasonic aspiration with a specialized handpiece down the working port of the endoscope. It has been used successfully on pituitary tumors, intraventricular clot removal, and craniopharyngioma cyst wall removal.27 Endoneurosonography has been used to supply additional information about the relationship of the tip of the probe and structures orthogonal to it.28 Water-jet dissection has been reported to be a useful adjunct in the safe perforation of a craniopharyngioma cyst wall, septum pellucidum, or floor of the third ventricle.29 This may help to decrease the risk of hemorrhage associated with blindly inserting an instrument through a thin structure such as the third ventricle floor.
Endoscopic Principles
There are two principal forms of endoscopy: coaxial and extra-axial. Coaxial endoscopic approaches, or “pure” endoscopy, are those in which all components of the endoscopic system (lighting, camera, working channels, irrigant channels, and instruments) are all parallel and enclosed in a single sheath. The instruments are introduced through working channels and are aimed by redirecting the endoscope. The impact to the surrounding brain from removing and reintroducing instruments is minimized because the entire working and visualization area is within the endoscopic sheath. Most intraventricular endoscopic procedures are performed in a coaxial manner.30
During “endoscope-controlled” surgery, the endoscope is the sole mode of visualization and surgery is performed using the same techniques and instrumentation as microsurgery, with the addition of curved instruments and suctions that allow the surgeon to operate around corners. In these forms of endoscopy a substantial learning curve exists because of peripheral distortion, the view angle when using non–0 degree endoscopes, and the close proximity of the surgical field to the tip of the endoscope. Once this is overcome, these same “problems” may be used to the surgeon’s advantage, resulting in better outcomes.
Endoscopic Intraventricular Surgical Techniques
Cysts
Colloid Cysts
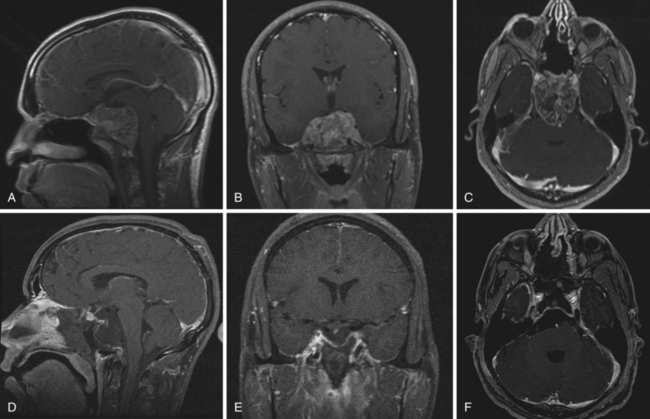
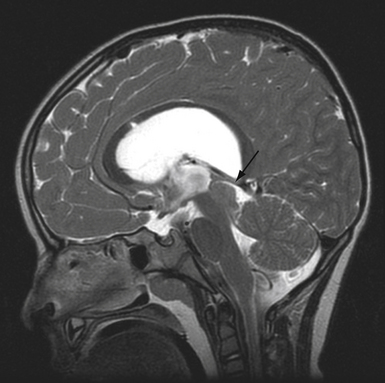
Colloid cysts of the third ventricle are non-neoplastic masses that typically arise from the roof of the third ventricle. They can occlude the foramen of Monro, causing headache, hydrocephalus, memory disturbances, and sudden death. Colloid cysts have a variable consistency, from mucinous, which are easily aspirated, to a hard, cheesy consistency. Cysts 1 cm or larger and those causing symptoms or hydrocephalus are generally recommended for removal. Other options including shunting and stereotactic drainage are possible but not recommended because of their poor durability. Microsurgical removal is effective but more invasive than the endoscopic approach.20,23,31 Therefore, endoscopic removal is recommended in the majority of cases.32–35 However, when imaging predicts the consistency of the cyst contents to be hard and cheesy, then the cyst is better removed microsurgically with bimanual dissection. Additionally, cysts larger than 2 cm may compromise or adhere to the fornix and may be more safely removed using microsurgical bimanual dissection.
A single bur hole, approximately 8 cm behind the nasion and 5 to 7 cm lateral to the midline in the nondominant hemisphere (careful not to injure the caudate head), is sufficient for removal.24,36 Image guidance helps with the initial ventricular entry. A peel-away sheath is optional. The landmarks of the colloid cyst and foramen of Monro are identified and the overlying choroid plexus is coagulated, avoiding the fornix. The cyst is coagulated and opened and the contents are aspirated. A pediatric endotracheal suction catheter with the end cut to 45 degrees is particularly effective if the consistency is favorable. One can twist the catheter and use the cut end as a dissector to “morselize” the contents of the cyst prior to aspiration. If the contents are too dense, forceps may be required to empty the cyst contents. The wall of the cyst is then dissected free of the roof of the third ventricle with generous coagulation. Generally the cyst wall is not densely attached to the fornix and can be removed completely. However, in the case that the wall is so adherent to the internal cerebral veins or the fornix that it cannot be separated using either sharp or blunt dissection it may be prudent to leave a thin “carpet.” Under these circumstances, the recurrence rate appears to be low.21,37,38 Symptomatic relief of obstructive hydrocephalus is generally obtained, though mild ventriculomegaly often persists.37
Neurocysticercotic Cysts
Neurocysticercosis (NCC) is the neurological manifestation of the parasite Taenia solium and is commonly contracted in underdeveloped countries by hand-to-mouth contamination from unclean water or food. NCC most often presents with seizures but may also present with sudden hydrocephalus due to intraventricular cysts blocking normal cerebrospinal fluid (CSF) pathways. There is a growing body of literature suggesting that neuroendoscopic removal of cysts results in improved patient outcomes and lessens or avoids altogether the need for shunting.39–41 Recently, pediatric data regarding neuroendoscopic cyst evacuation has been reported. The shunting rate is lower (22%) in the neuroendoscopic group than in the traditional medical treatment group (70%) and the Karnofsky performance scale was higher in the endoscopic group (90.0% vs. 85.5%, p = 0.003).39 Two other studies show complete resolution of cysts and no need for shunting with minimal transient morbidity.40,41
Miscellaneous Cysts
Other cysts, such as arachnoid cysts, occur within the ventricles. Often fenestration of these cysts to normal CSF spaces is desirable. The ease of working in a fluid-filled space makes the endoscope ideal for fenestration. The anatomy is often distorted and the arachnoid surface is often thick and somewhat opaque. Stereotaxis is very helpful in these cases to know what is behind an opaque membrane prior to fenestration. Blunt perforation should be avoided at all costs as pushing against these flimsy membranes may damage the important neurovascular structures that lie behind. Enlarging interhemispheric cysts can be fenestrated directly to the ventricle or via cystocisternoventricular fenestration.42 Fourth ventricular cysts obstructing outflow are also amenable to fenestration.43
Tumors
Pure Endoscopic Approaches
Endoscopic applications for intraventricular tumors include tumor biopsy, tumor resection, and management of tumor-associated hydrocephalus.44,45 In general, neuroendoscopy for tumors is a step up in technical difficulty from the endoscopic management of hydrocephalus. The ideal conditions for endoscopic resection of an intraventricular tumor are that it should be small, avascular or with relatively low vascularity, partially or totally cystic, and located in enlarged ventricles. Hydrocephalus creates an ideal working space. However, a normal ventricle is adequate to gain access to a tumor and biopsy it and, for smaller tumors, to resect it safely.46 A recent report even suggests that in experienced hands, operating in a normal sized ventricle yields the same success/complication rate as operating in large ventricles.47
A proper working trajectory is one that accomplishes the following:
1. Enters the ventricle with some normal ventricle between the entry point and the mass
2. Allows access to the blood supply, if vascular
3. Allows access to the point of attachment to the ventricular wall or choroid plexus
4. Does not originate in or traverse eloquent structures
5. Allows management of associated hydrocephalus or trapped CSF spaces
Having some normal ventricle and CSF between the entry point into the ventricle and the tumor allows better visualization of the tumor margin and allows visualized normal structures to aid in orientation. Access to the blood supply and point of attachment may turn a tedious piecemeal tumor resection into a disconnection and en bloc removal. As mentioned earlier, the use of transendoscope ultrasonic aspirators may speed up tumor removal if en bloc removal is not possible.27
Image guidance is particularly valuable for approach planning and execution, and is worthwhile even if it is only used for this step of the procedure.48–50 Third ventricle tumor resections are particularly dependent on proper approach angle since often the endoscope must traverse the foramen of Monro, putting the fornix, at the anterior border of the foramen, at some risk. Image guidance allows the surgeon to use trajectory views to draw a line from the anterior border of the tumor to the anteriormost border of the foramen of Monro. This line can then be extended to the surface to choose the appropriate entry point and angle. The fornix will not tolerate anterior “windshield wiping” movements of the endoscope while its tip lies in the third ventricle. Posterior movements are tolerated much better; however, the venous structures coalescing at the posterior margin of the foramen of Monro also limit scope excursion. Fortunately, many intraventricular tumors are associated with ventriculomegaly and an enlarged foramen of Monro that allows for larger excursions of the endoscope.
Certain tumors are more amenable than others for endoscopic removal.51 When working within the fluid-filled ventricle, bleeding may be difficult to control and obscures the operative field. Therefore, tumors of low vascularity are preferred for purely endoscopic removal. Examples are subependymomas, ependymomas, the subependymal giant cell astrocytomas associated with tuberous sclerosis, selected neurocytomas, exophytic gliomas (primarily pilocytic or low grade), and hypothalamic hamartomas.52 Some vascular tumors such as choroid plexus and pedunculated tumors can also be approached endoscopically, because the blood supply is often well defined and easy to coagulate and divide.
Most tumors are approached by taking initial biopsy specimens with cup forceps, minimizing coagulation to maintain the quality of the tissue for analysis. Any vessels on the surface of the tumor are then coagulated. Electrocautery (especially monopolar) is capable of generating high CSF temperatures and must be used with caution. Irrigation with warmed lactated Ringer’s solution or a spinal fluid substitute solution is used to dissipate this heat.53 Normal saline is not used because it lacks electrolytes, is acidotic, alters the electrolyte balance in the brain, and leads to postoperative confusion.54 Again, appropriate egress of irrigant will avoid a dangerous rise in intracranial pressure.
Once bleeding is controlled, cautery, blunt dissection, and bites with the forceps or scissors are used to separate the tumor from the normal tissue. The best tumors for neuroendoscopy have a distinct margin and can be gently retracted away from the surrounding tissue. Ideally, a perimeter can be created, the tumor can be isolated as a mass, and it can be removed in one or more large pieces. If the tumor is soft, multiple methods of tumor aspiration are possible. A stainless steel suction cannula or the previously mentioned pediatric endotracheal suction catheter placed down the working channel can be used to morselize and aspirate tumor.47 Shortened endovascular catheters can also be used. They have the advantage of a stiffer, thinner wall that can be shaped. The catheter allows a larger inner lumen diameter for more efficient aspiration of appropriate consistency tumors.55 The gelatinous contents of colloid cysts and some other cystic tumors respond particularly well to this technique. As mentioned earlier, the length of time required to aspirate a tumor using a “biopsy after biopsy” approach may be shortened by the use of ultrasonic aspiration with a specialized handpiece down the working port of the endoscope.27 Regardless of the removal technique, every attempt should be made to avoid dispersion of tumor remnants throughout the ventricles.
In some cases tumor biopsy rather than removal may be the goal. CNS lymphoma is often periventricular and amenable to endoscopic biopsy for diagnosis. “Nonoperative” gliomas may also be appropriate for this biopsy method. Stereotactic guidance is helpful in locating the tumor. However, identification of the tumor tissue through overlying normal ependyma may be problematic. Recently, the use of 5-aminolevulinic acid (5-ALA) fluorescence to identify and biopsy a midbrain glioma through an intact ependymal layer has been reported.56
Complications of tumor biopsy and removal include intraventricular hemorrhage, neurological deficit, tension pneumocephalus, hydrocephalus, and basilar artery injury.57–59 Tension pneumocephalus results from air being exchanged for CSF during the procedure. To avoid this, the ventricles should be refilled with lactated Ringer’s solution. If large quantities of air remain, 100% oxygen administered via facemask will help with dissolution.60
Peratta and associates reported an 8.8% complication rate for neuroendoscopic (nonhydrocephalus) procedures in pediatric patients.61 Hemorrhagic complications of tumor biopsy are reported at 3.5% per patient and 2.4% per procedure.62 To minimize the incidence of hemorrhagic complications one should never cut or pull any structure without being able to visualize the structure completely.
Endoscope-Assisted Microsurgical Approaches
Many intraventricular tumors cannot be completely removed through a purely endoscopic approach, but endoscopy still maintains an important role. The concept of “endoscope-assisted” refers to the traditional microsurgical procedure that uses an endoscope as an adjunct either through the same opening or through a separate bur hole for better overall visualization. The endoscope allows the surgeon to look around corners and to visualize structures that are not visible by microscopic imaging alone, thus expanding the operating field. Utilizing angled endoscopes can maximize the area of the ventricle visualized. Charalampaki and colleagues reported that endoscope-assisted microsurgery has been useful in 35 patients with traditional microsurgical approaches to the ventricular system done through keyhole craniotomies. Thirty-one out of 35 patients had no morbidity at 6 months and 3 of the patients with 6-month morbidity had preexisting Perinaud’s syndrome that persisted. Seventy-eight percent of patients had complete tumor resections. One procedure was aborted due to hemorrhage and was repeated successfully with gross total removal 2 days later.63 Another example of endoscope-assisted microsurgery is the transventricular management of craniopharyngioma including gross total removal of intraventricular components, fenestration of cysts as a stand-alone procedure for symptomatic control, and cyst fenestration followed by collapse and subsequent craniotomy for definitive removal of the solid component.64
Endoscopic Endonasal Skull Base Surgical Technique
Endoscopic endonasal approaches have been organized in modules based on anatomical corridors (Box 44.1).12,13,65,66 The modular approach to learning the various EEAs is based entirely on a thorough understanding of the ventral skull base anatomy as viewed with the endoscope. This is a key principle to avoid complications.
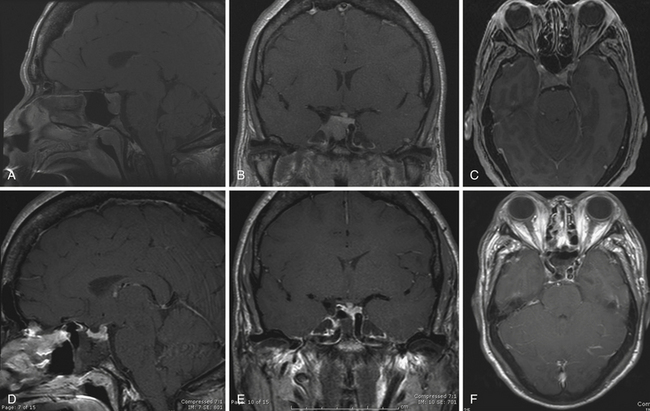
Transsellar Approach12 (Figs. 44.1 to 44.3)
Exposure
In itself, the transsellar approach is not considered an expanded approach because it is limited to exposing the sella turcica. It is appropriate for the removal of pituitary adenomas as well as other intrasellar entities such as Rathke’s cleft cysts and craniopharyngiomas with minimal suprasellar extension. However, because the sella is the epicenter at the crossroads of the sagittal and coronal planes, it is the starting point for most of the expanded surgical modules. The sphenoidotomies are widened by exposing the sphenoid lateral recesses and the posterior ethmoidal cells. The planum-tuberculum junction and bilateral lateral opticocarotid recesses (OCR) should be visualized. The floor of the sphenoid is drilled back to the level of the clivus, giving a greater caudorostral trajectory into the suprasellar space. Intrasphenoidal septations are to be drilled down carefully because they may lead directly toward the vertical canal of the internal carotid artery (ICA).67 The sphenoid mucosa is removed in the areas the bone will be drilled. Once the posterior wall of the sphenoid sinus is completely exposed the following structures are viewed: the sellar prominence in the center, the ventral aspect of the tuberculum sellae covering the superior intercavernous sinus (SIS) above, the clival recess below. The carotid prominences are seen laterally and the optic canals superolaterally and between them the medial OCR and the lateral OCR. Bone removal over the sellar face should extend laterally beyond the medial portions of the cavernous sinus (CS) and expose both superior and inferior intercavernous sinuses (see Fig. 44.2). The medial OCR does not need to be opened unless the lesion has suprasellar and lateral extensions toward the opticocarotid cistern. The most important structures related to this module are the CS, which contains both ICAs and limits the area laterally.
Expanded Endonasal Approaches—Sagittal Plane
The sagittal plane modules extend from the frontal sinus to the second cervical vertebra, enabling access through the crista galli, planum, tuberculum, dorsum sella, clivus, and odontoid process (see Box 44.1).
Transtuberculum/Transplanum Approach12
Exposure
The transtuberculum/transplanum approach is indicated for lesions involving both the posterior aspect of the anterior skull base and the suprasellar region. Bony exposure builds on that obtained with the transsellar approach. It is extended rostrally by completing wide bilateral posterior ethmoidectomies. Ethmoidal septations are drilled flush with the anterior cranial base. Anteriorly, bony resection should not continue anterior to the posterior ethmoidal arteries (PEAs) and the rostral margin of the nasal septum is left attached to the skull base. These precautions prevent injury to the olfactory neuroepithelium, preserving olfaction. The planum sphenoidale is drilled eggshell thin. The bone over the sella and the tuberculum sellae over the SIS are removed completely, exposing the dura under the medial OCR (see Fig. 44.2). This enables access to the suprasellar extensions of tumors in the prechiasmatic cisterns with optic nerve and carotid control. The paraclinoid carotid canals can also be opened to allow lateral retraction of soft tissue at that level. Arterial feeders arising from the distal portion of the paraclinoid carotid artery at the level of the medial OCR as well as from the PEAs can be hypertrophic in meningiomas and should be identified and coagulated. The SIS is not transected in pure suprasellar tumors such as tuberculum sellae meningiomas. Craniopharyngiomas can occupy sellar and suprasellar compartments and, in these cases, the SIS is coagulated and transected to allow exposure of both locations.
Transcribriform Approach12
Exposure
This module extends the rostral extension of the previous approach to the level of the crista galli or even the frontal sinus if required. Indeed, the transcribriform approach is commonly combined with a transplanum approach for resection of anterior fossa meningiomas. The attachment of the anterior portion of the nasal septum to the skull base is resected posteriorly, olfaction being most likely already compromised. Complete ethmoidectomies are performed with exposure of the medial orbital walls. The skull base is drilled rostrocaudally starting at the frontoethmoidal recess. The ethmoidal arteries (AEA and PEA) are identified, coagulated, and transected medially, contributing to tumor devascularization. The lamina papyracea can be removed in order to gain lateral exposure, although the periorbital fascia should not be disrupted. After bilateral removal of the cribriform plate, the crista galli is drilled until eggshell thin, fractured, and removed. The ethmoidal arteries represent a risk for retrobulbar hematoma if not well coagulated or clipped before being incised during this approach.
Transclival Approach65 (see Fig. 44.3)
Upper Third of the Clivus
Transsellar exposure (intradural)
A transtuberculum/transplanum approach is first performed. Rostral exposure only needs to reach the tuberculum/planum junction. Bone covering the sellar face is removed to expose the SIS, IIS, and the sella-clival junction. Cruciform dural openings are performed over the parachiasmatic cistern and the pituitary gland, taking care not to transgress the pituitary capsule. After ligation and transection of the SIS, both dural openings communicate. If present, the IIS must also be transected. The diaphragma sella is cut in the midline to expose the stalk. The diaphragma sella is then cut in a paramedian direction to release the stalk circumferentially. The ligaments connecting the pituitary capsule to the medial cavernous sinus wall are systematically cut along the lateral contour of the gland. The gland may be mobilized superiorly, enabling exposure of the posterior sellar dura, which is coagulated and the posterior intercavernous sinus (PIS) is transected, exposing the dorsum sellae and posterior clinoid laterally.68 These bony structures are then drilled until eggshell thin and are carefully removed, avoiding injury to the ICA and abducent nerve located laterally and posteriorly. Once these structures are drilled, the retroclival dura harboring the basilar plexus is visualized. Transgressing the basilar plexus can generate intense venous bleeding. The surgeons must be prepared to control such bleeding, and hemostatic agents as such as microfibrillar collagen and absorbable gelatin powder with thrombin work very well in this location. Gland transposition enables unobstructed visualization of the posterior wall of the sella.68
Panclival Approaches (see Fig. 44.3)
The panclival approach can extend from the dorsum sellae and posterior clinoids to the anterior aspect of the foramen magnum. Modifications of the initial bilateral sphenoid exposure are needed to gain such a caudal access. To gain progressively caudal access, the nasal septum needs to be completely detached from the sphenoid rostrum. Wide sphenoidotomies must be performed as they provide deeper positioning of the scope and enable a direct caudal view. The basopharyngeal fascia is removed from the sphenoid rostrum and clivus face. The sphenoid sinus floor is drilled flush with the clivus. Before drilling the clivus, it is important to identify the vidian nerve because it marks the petrous ICA level. The clivus bone is removed in the midline, between the carotid canals above the level of the vidian nerve. Inferiorly, drilling of the jugular tubercle and petrous bone can be performed laterally bellow the petrous ICA using the vidian canal as the superior limit.69
After meticulous coagulation of the underlying dura and basilar venous plexus, the dura is opened segmentally in the midline. Neurophysiological and sixth cranial nerve stimulation should be used to avoid opening on the sixth cranial nerve. Image guidance should also be used under computed tomography angiography (CTA) visualization to determine the vertebrobasilar junction (VBJ). The dural opening should be below the VBJ because the sixth cranial nerve will be above that level.70
Transodontoid and Foramen Magnum/Craniovertebral Approach65,71,72
This approach can be used for resection of the odontoid process in degenerative or inflammatory disease, or to allow for exposure of the ventral medulla and upper cervical spinal cord. The transodontoid approach is an extension of the transclival approach. However, it can be performed independently with clival preservation because in most cases the disease is isolated in the cervical spine. Furthermore, the sphenoid sinus doesn’t need to be exposed in most of these cases. After removing the nasopharyngeal mucosa, the paraspinal muscles and the atlanto-occipital membrane are exposed and partially resected. At this point, the lower clivus as well as the anterior arch of C1 are exposed. Bone removal is guided by the pathological entity and concerns for stability.73 For foramen magnum exposure, only the superior aspect of the C1 ring needs to be drilled to expose the tip of the odontoid. For transodontoid exposure, the anterior arch of C1 is resected and the odontoid process of C2 is exposed. The anterior cortex of the dens and the trabecular bone are drilled and the posterior cortical shell is removed preferentially by sharp dissection incising the ligaments. After removal of the dens, the normal dura covering the brainstem is exposed or an underlying pannus if surgery is performed in the setting of rheumatoid arthritis.
Expanded Endonasal Approaches—Coronal Plane
Middle and Posterior Coronal Plane13
Transpterygoid Approach (Fig. 44.4)
All the modules in the middle and posterior coronal planes begin with a transpterygoid approach. Initially a maxillary antrostomy is performed, providing access to the back wall of the maxillary sinus. The sphenopalatine and posterior nasal arteries are identified and ligated. The soft tissues of the pterygopalatine fossa are mobilized in a medial to lateral direction to expose the base and the medial wedge of the pterygoids. The vidian canal (pterygoid canal) should be identified early because it represents, along with the middle pterygoid plates, critical surgical landmarks for endoscopic approaches to the petrous apex.69,74 The vidian canal leads directly to the anterior genu of the ICA because its petrous portion turns up to form the vertical paraclival ICA. The medial pterygoid plate (MPP) is drilled medial and inferior to the vidian canal while following it posteriorly, toward the foramen lacerum.
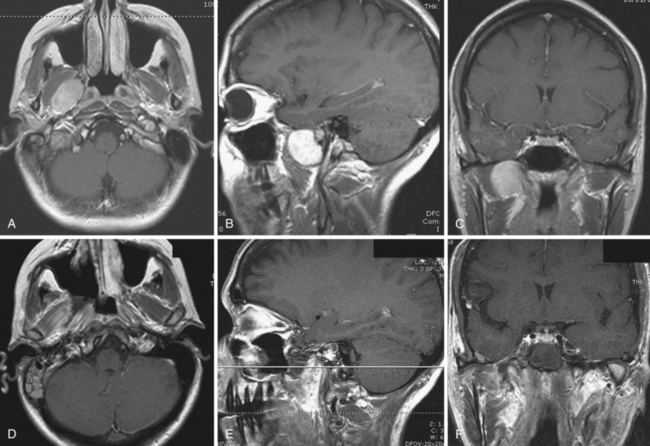
Medial Petrous Apex (Zone 1: Posterior Fossa)
This module adds on the transpterygoid approach and can be conceived as a lateral extension of a transclival approach in its middle third. It is indicated to access lesions in the medial petrous apex such as chondrosarcomas and cholesterol granulomas. Following the transpterygoid approach, drilling of the bone covering the paraclival carotid may be required if the ICA needs to be mobilized laterally.75 This enables direct access to the petrous apex. Greater access can also be provided by drilling a portion of the lateral clivus at its junction with the petrous apex.
The most relevant structures are the ICAs and the sixth cranial nerve at Dorello’s canal.
Petroclival Approaches (Zone 2: Posterior Fossa)
The important structures related to this module are the vidian nerves and the ICAs.
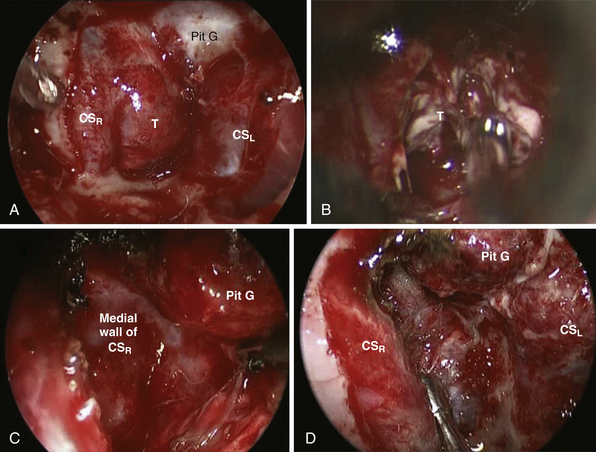
Inferior Cavernous Sinus/Quadrangular Space (Zone 3: Middle Fossa)76
This module is used for resection of Meckel’s cave lesions. Pathological entities commonly encountered in this region are invasive adenoid cystic carcinomas, meningiomas, schwannomas, and invasive pituitary adenomas. Removal of the posterior wall of the maxillary antrum is extended laterally until the maxillary branch (V2) of the trigeminal nerve is identified traveling superiorly toward the foramen rotundum. The MPP is drilled inferior and medial to the vidian canal. Next, the bone between the vidian canal and V2 is drilled away, knowing that this bony corridor narrows progressively as it deepens. Removal of this bone gives access to the quadrangular space, which is delimited by the parasellar ICA medially, the V2 and dura of middle fossa laterally, horizontal petrous ICA inferiorly, and the sixth cranial nerve superiorly. Bone covering the horizontal petrous ICA, the anterior genu, and parasellar ICA may need to be removed if mobilization of the carotid is required. The periosteum is opened lateral to the vertical paraclival ICA and medial to V2.
The structures at risk for this approach are exactly its boundaries: ICA, sixth cranial nerve, and trigeminal nerve.76
Superior Cavernous Sinus (Zone 4: Middle Fossa)
This module requires the exact same bone removal and ICA exposure as performed for the inferior cavernous sinus module. However, it is rarely used due to the high risk of cranial nerve injury. This approach has mostly served for patients with already established cranial nerve deficits (CN III, IV, VI) such as in apoplectic pituitary adenomas that invade the cavernous sinus or for tumors refractory to medical treatment or radiosurgery.13 Prior to dural incision, it is advised to identify the medial margin of the ICA in the sella so that it can be protected during dural opening. The dura incision is initiated directly over the superior lateral portion of the cavernous sinus and performed in a medial-to-lateral direction. Often, the cavernous sinus has already thrombosed and little venous bleeding occurs during initial opening. However, profuse bleeding may be encountered once the tumor is removed.
Transcondylar and Supracondylar (Transjugular Tubercle) (Zone 6: Posterior Fossa)13
Complication Avoidance During the Expanded Endonasal Approach
The following general principles should prevail during each EEA.
1. The critical neurovascular structures must be located on the perimeter of the lesion, allowing access to the lesion with minimal manipulation of normal neurovascular structures.
2. EEAs must be performed using bimanual and binarial access to allow for a two-surgeon, three/four-hand technique. The bony and soft tissue removal required for dynamic and free movement as well as optimal visualization has been described for each module. A wide surgical corridor enables exposure of key anatomical landmarks, prevents crowding of instruments, minimizes soiling of the lens, and helps maintain an unobstructed view of the surgical field. This step becomes more critical if there is a bleeding complication, so that the surgeons can maintain visualization while controlling the hemorrhage and avoiding injury to adjacent structures.
3. Endoneurosurgical tumor resection uses the same techniques and respects the same principles as microneurosurgery. Specifically, capsular bipolar coagulation, internal debulking, capsular mobilization, extracapsular dissection of neurovascular structures, coagulation, and removal of the capsule are sequentially performed in a bimanual fashion. Grasping and pulling must be avoided.
4. Skull base defect reconstruction is of major importance. The use of vascularized flaps for large dural defects has significantly reduced the incidence of postoperative CSF leaks. Although other materials have been used for closure, we believe the use of a vascularized flap offers the best chance at effective reconstruction following EEAs.
5. EEAs should be performed by an integrated team composed of a neurosurgeon and ear, nose, and throat (ENT) surgeon, both with substantial knowledge in ventral skull base endoscopic anatomy and thorough experience in conventional skull base surgery and expanded endoscopic surgery.
6. Endoscopic cranial base surgery must be learned in an incremental and modular fashion which applies to all endonasal surgeons, irrespective of their specialty (Box 44.2).66 A level must be fully mastered prior to proceeding to the next because a higher level translates into increased anatomical complexity, technical difficulty, and potential risk of neurovascular injury.
Outcome After the Expanded Endonasal Approach
Numerous series have demonstrated that endoscopic resection of sinonasal, sellar, and various skull base lesions is associated with at least equivalent oncological results to standard transcranial procedures in well-trained hands.1,5,8,9,14–16 In addition, EEAs have been associated with decreased length of hospital stay, decreased morbidity, increased patient comfort, and lack of external incision.1,5,8,9,14–16
Overall, endoscopic skull base surgery shares the same primary goal of oncological surgery as open procedures: to perform the most complete tumor excision. The endoscope provides visualization of tumor limits and margins and enables better assessment of the extent of the tumor. Planning the specific margins of resection is therefore facilitated.66 Because endoscopic procedures attack lesions from a ventral perspective, the resection of infiltrative skull base tumors may be more extensive considering that bony and dural invasion is more easily addressed through EEAs than through traditional skull base approaches.
Conclusion and Future Directions
The choice of a specific surgical route should be guided by lesion characteristics, patient comorbidities, and skill and experience of the operating team. Each patient should be evaluated with a 360-degree approach, considering the least destructive route with the fewest complications to achieve the most complete lesion resection. Modular approaches should be combined as mandated by the lesion and its location.12,13,65 However, when a lesion cannot be completely removed through an EEA, an open route may be considered or a combination of endonasal and open approaches may be used.1,5,6
Kassam A., Snyderman C.H., Mintz A., et al. Expanded endonasal approach: the rostrocaudal axis. Part I. Crista galli to the sella turcica. Neurosurg Focus. 2005;19:E3.
Kassam A., Snyderman C.H., Mintz A., et al. Expanded endonasal approach: the rostrocaudal axis. Part II. Posterior clinoids to the foramen magnum. Neurosurg Focus. 2005;19:E4.
Kassam A.B., Gardner P., Snyderman C., et al. Expanded endonasal approach: fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg Focus. 2005;19:E6.
Kassam A.B., Prevedello D.M., Carrau R.L., et al. The front door to Meckel’s cave: an anteromedial corridor via expanded endoscopic endonasal approach—technical considerations and clinical series. Neurosurgery. 2009;64:71-82.
Kassam A.B., Prevedello D.M., Thomas A., et al. Endoscopic endonasal pituitary transposition for a transdorsum sellae approach to the interpeduncular cistern. Neurosurgery. 2008;62:57-72.
Kassam A.B., Vescan A.D., Carrau R.L., et al. Expanded endonasal approach: vidian canal as a landmark to the petrous internal carotid artery. J Neurosurg. 2008;108:177-183.
Please go to expertconsult.com to view the complete list of references.
1. Carrabba G., Dehdashti A.R., Gentili F. Surgery for clival lesions: open resection versus the expanded endoscopic endonasal approach. Neurosurg Focus. 2008;25:E7.
2. de Divitiis E., Cappabianca P., Cavallo L.M., et al. Extended endoscopic transsphenoidal approach for extrasellar craniopharyngiomas. Neurosurgery. 2007;61:219-227. discussion 228
3. de Divitiis E., Cavallo L.M., Cappabianca P., Esposito F. Extended endoscopic endonasal transsphenoidal approach for the removal of suprasellar tumors: part 2. Neurosurgery. 2007;60:46-58.
4. de Divitiis E., Esposito F., Cappabianca P., et al. Endoscopic transnasal resection of anterior cranial fossa meningiomas. Neurosurg Focus. 2008;25:E8.
5. Dehdashti A.R., Ganna A., Witterick I., Gentili F. Expanded endoscopic endonasal approach for anterior cranial base and suprasellar lesions: indications and limitations. Neurosurgery. 2009;64:677-687.
6. Fatemi N., Dusick J.R., de Paiva Neto M.A., et al. Endonasal versus supraorbital keyhole removal of craniopharyngiomas and tuberculum sellae meningiomas. Neurosurgery. 2009;64:269-284.
7. Frank G., Pasquini E., Doglietto F., et al. The endoscopic extended transsphenoidal approach for craniopharyngiomas. Neurosurgery. 2006;59:ONS75-ONS83. discussion ONS75-ONS83
8. Fraser J.F., Nyquist G.G., Moore N., et al. Endoscopic endonasal transclival resection of chordomas: operative technique, clinical outcome, and review of the literature. J Neurosurg. 2010;112(5):1061-1069.
9. Gardner P.A., Kassam A.B., Snyderman C.H., et al. Outcomes following endoscopic, expanded endonasal resection of suprasellar craniopharyngiomas: a case series. J Neurosurg. 2008;109:6-16.
10. Laufer I., Anand V.K., Schwartz T.H. Endoscopic, endonasal extended transsphenoidal, transplanum transtuberculum approach for resection of suprasellar lesions. J Neurosurg. 2007;106:400-406.
11. Zada G., Kelly D.F., Cohan P., et al. Endonasal transsphenoidal approach for pituitary adenomas and other sellar lesions: an assessment of efficacy, safety, and patient impressions. J Neurosurg. 2003;98:350-358.
12. Kassam A., Snyderman C.H., Mintz A., et al. Expanded endonasal approach: the rostrocaudal axis. Part I. Crista galli to the sella turcica. Neurosurg Focus. 2005;19:E3.
13. Kassam A.B., Gardner P., Snyderman C., et al. Expanded endonasal approach: fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg Focus. 2005;19:E6.
14. O’Malley B.W.Jr., Grady M.S., Gabel B.C., et al. Comparison of endoscopic and microscopic removal of pituitary adenomas: single-surgeon experience and the learning curve. Neurosurg Focus. 2008;25:E10.
15. Gardner P.A., Kassam A.B., Thomas A., et al. Endoscopic endonasal resection of anterior cranial base meningiomas. Neurosurgery. 2008;63:36-52.
16. Tabaee A., Anand V.K., Barron Y., et al. Endoscopic pituitary surgery: a systematic review and meta-analysis. J Neurosurg. 2009;111:545-554.
17. Doglietto F., Prevedello D.M., Jane J.A.Jr., et al. Brief history of endoscopic transsphenoidal surgery—from Philipp Bozzini to the First World Congress of Endoscopic Skull Base Surgery. Neurosurg Focus. 2005;19(6):E3.
18. Grant J.A. Victor Darwin L’Espinasse: a biographical sketch. Neurosurgery. 1996;39:1232-1233.
19. Dandy W.E. Cerebral ventriculoscopy. Johns Hopkins Hosp Bull. 1922;33:189.
20. Lewis A.I., Crone K.R., Taha J., et al. Surgical resection of third ventricle colloid cysts. Preliminary results comparing transcallosal microsurgery with endoscopy. J Neurosurg. 1994;81(2):174-178.
21. Levine N.B., Miller M.N., Crone K.R. Endoscopic resection of colloid cysts: indications, technique, and results during a 13-year period. Minim Invasive Neurosurg. 2007;50(6):313-317.
22. Schroeder H.W., Gaab M.R. Endoscopic resection of colloid cysts. Neurosurgery. 2002;51(6):1441-1444. discussion 1444-1445
23. Horn E.M., Feiz-Erfan I., Bristol R.E., et al. Treatment options for third ventricular colloid cysts: comparison of open microsurgical versus endoscopic resection. Neurosurgery. 2007;60(4):613-618. discussion 618-620
24. Greenlee J.D., Teo C., Ghahreman A., Kwok B. Purely endoscopic resection of colloid cysts. Neurosurgery. 2008;62(3 Suppl 1):51-55. discussion 55-56
25. Suri A., Goel R.K., Ahmad F.U., et al. Transventricular, transaqueductal scope-in-scope endoscopic excision of fourth ventricular neurocysticercosis: a series of 13 cases and a review. J Neurosurg Pediatr. 2008;1(1):35-39.
26. Prat R., Galeano I. Endoscopic biopsy of foramen of Monro and third ventricle lesions guided by frameless neuronavigation: usefulness and limitations. Clin Neurol Neurosurg. 2009;111(7):579-582. (Epub 2009 May 26)
27. Oertel J., Krauss J.K., Gaab M.R. Ultrasonic aspiration in neuroendoscopy: first results with a new tool. J Neurosurg. 2008;109(5):908-911.
28. Resch K.D. Transendoscopic ultrasound in ventricular lesions. Surg Neurol. 2008;69(4):375-382. discussion 382 (Epub 2008 Feb 20)
29. Oertel J., Gen M., Krauss J.K., et al. The use of water jet dissection in endoscopic neurosurgery. Technical note. J Neurosurg. 2006;105(6):928-931.
30. Gore P.A., Gonzalez L.F., Rekate H.L., Nakaji P. Endoscopic supracerebellar infratentorial approach for pineal cyst resection: technical case report. Neurosurgery. 2008;62(3 Suppl 1):108-109. discussion 109
31. Grondin R.T., Hader W., MacRae M.E., Hamilton M.G. Endoscopic versus microsurgical resection of third ventricle colloid cysts. Can J Neurol Sci. 2007;34(2):197-207.
32. Teo C. Complete endoscopic removal of colloid cysts: issues of safety and efficacy. Neurosurg Focus. 1999;6(4):e9.
33. Abdou M.S., Cohen A.R. Endoscopic treatment of colloid cysts of the third ventricle. Technical note and review of the literature. J Neurosurg. 1998;89(6):1062-1068.
34. King W.A., Ullman J.S., Frazee J.G., et al. Endoscopic resection of colloid cysts: surgical considerations using the rigid endoscope. Neurosurgery. 1999;44(5):1103-1109. discussion 1109-1111
35. Decq P., Le Guerinel C., Brugières P., et al. Endoscopic management of colloid cysts. Neurosurgery. 1998;42(6):1288-1294. discussion 1294-1296
36. Bristol R.E., Nakaji P., Smith K.A. Endoscopic management of colloid cysts. Oper Tech Neurosurg. 2005;8(4):176-178.
37. Hellwig D., Bauer B.L., Schulte M., et al. Neuroendoscopic treatment for colloid cysts of the third ventricle: the experience of a decade. Neurosurgery. 2003;52(3):525-533. discussion 532-533
38. Longatti P., Godano U., Gangemi M., et al. Cooperative study by the Italian neuroendoscopy group on the treatment of 61 colloid cysts. Childs Nerv Syst. 2006;22(10):1263-1267. (Epub 2006 Apr 29). Erratum in Childs Nerv Syst2006;22(10):1375
39. Proana J.V., Torres-Corzo J., Guizar-Sahagun G., et al. Intraventricular and subarachnoid basal cisterns neurocysticercosis: a comparative study between traditional treatment versus neuroendoscopic surgery. Childs Nerv Syst. 2009;25(11):1467-1475. (Epub 2009 Jun 26)
40. Goel R.K., Ahmad F.U., Vellimana A.K., et al. Endoscopic management of intraventricular neurocysticercosis. J Clin Neurosci. 2008;15(10):1096-1101. (Epub 2008 Jul 23)
41. Suri A., Goel R.K., Ahmad F.U., et al. Endoscopic excision of intraventricular neurocysticercosis in children: a series of six cases and review. Childs Nerv Syst. 2008;24(2):281-285. (Epub 2007 Nov 10)
42. Cinalli G., Peretta P., Spennato P., et al. Neuroendoscopic management of interhemispheric cysts in children. J Neurosurg. 2006;105(Suppl 3):194-202.
43. Longatti P., Fiorindi A., Martinuzzi A., Feletti A. Primary obstruction of the fourth ventricle outlets: neuroendoscopic approach and anatomic description. Neurosurgery. 2009;65(6):1078-1085. discussion 1085-1086
44. Gaab M.R., Schroeder H.W. Neuroendoscopic approach to intraventricular lesions. J Neurosurg. 1998;88(3):496-505.
45. Macarthur D.C., Buxton N., Punt J., et al. The role of neuroendoscopy in the management of brain tumors. Br J Neurosurg. 2002;16(5):465-470.
46. Souweidane M.M. Endoscopic surgery for intraventricular brain tumors in patients without hydrocephalus. Neurosurgery. 2008;62(6 Suppl 3):1042-1048.
47. Souweidane M.M., Luther N. Endoscopic resection of solid intraventricular brain tumors. J Neurosurg. 2006;105(2):271-278.
48. Harris A.E., Hadjipanayis C.G., Lunsford L.D., et al. Microsurgical removal of intraventricular lesions using endoscopic visualization and stereotactic guidance. Neurosurgery. 2008;62(Suppl 2):622-629.
49. Tirakotai W., Bozinov O., Sure U., et al. The evolution of stereotactic guidance in neuroendoscopy. Childs Nerv Syst. 2004;20(11-12):790-795. (Epub 2004 Jul 17)
50. Schroeder H.W., Wagner W., Tschiltschke W., Gaab M.R. Frameless neuronavigation in intracranial endoscopic neurosurgery. J Neurosurg. 2001;94(1):72-79.
51. Cappabianca P., Cinalli G., Gangemi M., et al. Application of neuroendoscopy to intraventricular lesions. Neurosurgery. 2008;62(Suppl 2):575-597. discussion 597-598
52. Ng Y.T., Rekate H.L., Prenger E.C., et al. Endoscopic resection of hypothalamic hamartomas for refractory symptomatic epilepsy. Neurology. 2008;70(17):1543-1548.
53. Oka K., Yamamoto M., Nonaka T., Tomonaga M. The significance of artificial cerebrospinal fluid as perfusate and endoneurosurgery. Neurosurgery. 1996;38(4):733-736.
54. Salvador L., Valero R., Carrero E., et al. Cerebrospinal fluid composition modifications after neuroendoscopic procedures. Minim Invasive Neurosurg. 2007;50(1):51-55.
55. Husain M., Jha D.K., Rastogi M. Angiographic catheter: unique tool for neuroendoscopic surgery. Surg Neurol. 2005;64(6):546-549.
56. Tamura Y., Kuroiwa T., Kajimoto Y., et al. Endoscopic identification and biopsy sampling of an intraventricular malignant glioma using a 5-aminolevulinic acid-induced protoporphyrin IX fluorescence imaging system. J Neurosurg. 2007;106(3):507-510.
57. Hamada H., Hayashi N., Kurimoto M., et al. Tension pneumocephalus after a neuroendoscopic procedure—case report. Neurol Med Chir (Tokyo). 2004;44(4):205-208.
58. Saxena S., Ambesh S.P., Saxena H.N., Kumar R. Pneumoencephalus and convulsions after ventriculoscopy: a potentially catastrophic complication. J Neurosurg Anesthesiol. 1999;11(3):200-202.
59. Abtin K., Thompson B.G., Walker M.L. Basilar artery perforation as a complication of endoscopic third ventriculostomy. Pediatr Neurosurg. 1998;28(1):35-41.
60. Gore P.A., Maan H., Chang S., et al. Normobaric oxygen therapy strategies in the treatment of postcraniotomy pneumocephalus. J Neurosurg. 2008;108(5):926-929.
61. Peretta P., Ragazzi P., Galarza M., et al. Complications and pitfalls of neuroendoscopic surgery in children. J Neurosurg. 2006;105(Suppl 3):187-193.
62. Luther N., Cohen A., Souweidane M.M. Hemorrhagic sequelae from intracranial neuroendoscopic procedures for intraventricular tumors. Neurosurg Focus. 2005;19(1):E9.
63. Charalampaki P., Filippi R., Welschehold S., et al. Tumors of the lateral and third ventricle: removal under endoscope-assisted keyhole conditions. Neurosurgery. 2005;57(Suppl 4):302-311. discussion 302-311
64. Cinalli G., Spennato P., Cianciulli E., et al. The role of transventricular neuroendoscopy in the management of craniopharyngiomas: three patient reports and review of the literature. J Pediatr Endocrinol Metab. 2006;19(Suppl 1):341-354.
65. Kassam A., Snyderman C.H., Mintz A., et al. Expanded endonasal approach: the rostrocaudal axis. Part II. Posterior clinoids to the foramen magnum. Neurosurg Focus. 2005;19:E4.
66. Snyderman C.H., Carrau R.L., Kassam A.B., et al. Endoscopic skull base surgery: principles of endonasal oncological surgery. J Surg Oncol. 2008;97:658-664.
67. Fernandez-Miranda J.C., Prevedello D.M., Madhok R., et al. Sphenoid septations and their relationship with internal carotid arteries: anatomical and radiological study. Laryngoscope. 2009;119:1893-1896.
68. Kassam A.B., Prevedello D.M., Thomas A., et al. Endoscopic endonasal pituitary transposition for a transdorsum sellae approach to the interpeduncular cistern. Neurosurgery. 2008;62:57-72.
69. Kassam A.B., Vescan A.D., Carrau R.L., et al. Expanded endonasal approach: vidian canal as a landmark to the petrous internal carotid artery. J Neurosurg. 2008;108:177-183.
70. Stippler M., Gardner P.A., Snyderman C.H., et al. Endoscopic endonasal approach for clival chordomas. Neurosurgery. 2009;64:268-277.
71. Kassam A.B., Snyderman C., Gardner P., et al. The expanded endonasal approach: a fully endoscopic transnasal approach and resection of the odontoid process: technical case report. Neurosurgery. 2005;57:E213. discussion E213
72. Messina A., Bruno M.C., Decq P., et al. Pure endoscopic endonasal odontoidectomy: anatomical study. Neurosurg Rev. 2007;30:189-194. discussion 194
73. Wu J.C., Huang W.C., Cheng H., et al. Endoscopic transnasal transclival odontoidectomy: a new approach to decompression: technical case report. Neurosurgery. 2008;63:ONSE92-ONSE94. discussion ONSE94
74. Osawa S., Rhoton A.L.Jr., Seker A., et al. Microsurgical and endoscopic anatomy of the vidian canal. Neurosurgery. 2009;64:385-411.
75. Zanation A.M., Snyderman C.H., Carrau R.L., et al. Endoscopic endonasal surgery for petrous apex lesions. Laryngoscope. 2009;119:19-25.
76. Kassam A.B., Prevedello D.M., Carrau R.L., et al. The front door to Meckel’s cave: an anteromedial corridor via expanded endoscopic endonasal approach—technical considerations and clinical series. Neurosurgery. 2009;64:71-82.