Disorders of Prolactin Secretion and Prolactinomas
Nongestational/puerperal prolactin (PRL) hypersecretion is the most prevalent hypothalamic-pituitary dysfunction, and its main cause is PRL-secreting pituitary adenomas (prolactinomas). Prolactinomas are the most common pituitary tumors, with an estimated prevalence of 500 cases/1 million inhabitants.1 These tumors are classified either as microadenomas (diameter <10 mm) or macroadenomas (>10 mm) and can be enclosed, expansive, or invasive.2 Prolactinomas are more common in women, especially microprolactinomas; macroprolactinomas have roughly the same prevalence in both genders. PRL-secreting pituitary carcinomas are exceedingly rare.3 Because hyperprolactinemia usually is associated with menstrual disturbances, anovulation, and sexual impairment in both genders, and prolactinomas have a greater incidence in people in their 20s and 30s, these tumors are an important cause of infertility. To make the correct diagnosis of prolactinoma, other causes of hyperprolactinemia—physiologic, drug-induced, or pathologic—must be ruled out. It also is important to be aware of laboratory and imaging pitfalls that can mislead the diagnosis and treatment.4 This chapter addresses the causes, clinical findings, diagnosis, and therapeutic options for prolactinomas and other causes of hyperprolactinemia.
History
“If a woman is neither pregnant nor has given birth, and produces milk, her menstruation has stopped.” This sentence, attributed to Hippocrates (Aphorisms, Section 5, #39), shows that the association between menstrual disturbances and inappropriate milk secretion has been known since ancient times. It was only in the 20th century, however, that such disturbances were associated with hypersecretion of a pituitary hormone. In 1928, Striker and Grueter5 identified a pituitary factor that was able to induce milk secretion in rabbits. Early in the 18th century, Hunter discovered that “pigeon’s milk,” a substance secreted by male and female parents to feed the young pigeon, is secreted by the crop, and according to Hunter, “the crop behaves like the udder of mammalian females regarding uterine gestation.” In 1933, Riddle and colleagues6 identified the stimulatory effect of a pituitary hormone on pigeons’ crop growth and differentiation, which also controlled milk secretion in mammals, and called it prolactin. The pigeon’s crop model later was used for the PRL bioassay. Coincidentally, the association of amenorrhea, infertility, and galactorrhea were described better around the 1930s.
Afterward, this clinical picture was characterized in three different contexts: (1) postpartum without sellar enlargement (Chiari-Frommel syndrome)7; (2) nonpuerperal period, also without sellar augmentation (Ahumada-Argonz-del Castillo syndrome)8; and (3) associated with a pituitary tumor (Forbes-Albright syndrome).9 The existence of a human PRL, distinct from the growth hormone (GH), remained controversial, however, until the development of a specific radioimmunoassay for PRL in the early 1970s,10 when it was shown that all the above-mentioned syndromes were linked to elevated serum PRL levels. The development of pituitary microsurgery and, later on, of high-resolution imaging techniques showed that most of the cases with normal sella on plain x-rays, described as nontumoral, were small pituitary tumors (microadenomas). When PRL-secreting pituitary adenomas (prolactinomas) were proved to be the main cause of pathologic hyperprolactinemia, linked or not to a previous pregnancy, the division borne by the three eponymic amenorrhea-galactorrhea syndromes became obsolete. There are many other causes of hyperprolactinemia that must be differentiated from prolactinomas so that a correct therapeutic approach can be instituted.
Epidemiology
Hyperprolactinemia is the most prevalent hypothalamic-pituitary dysfunction, with prolactinomas being the main cause. These tumors represent roughly 25% of surgically removed pituitary adenomas and nearly 50% of adenohypophyseal tumors in autopsy series.11 This apparent discrepancy may be due to the excellent results of medical treatment of prolactinomas with dopaminergic agonists. Their prevalence is estimated at 500 cases/1 million inhabitants, with an incidence of 27 cases/1 million/year.1 Microprolactinomas (diameter <10 mm) represent about 60% of PRL-secreting adenomas and are more common in women than in men (20 : 1), whereas macroadenomas have roughly the same prevalence on both genders.12 There are no sex-related differences in autopsy series. Prolactinomas occur in all ages, with the diagnosis made predominantly in the 20s and 30s in both sexes.12 They are the most frequent pituitary adenomas encountered in childhood and adolescence.12 Prolactinomas represent the minority of pituitary tumors diagnosed after age 70, however. Prolactinomas are the most prevalent pituitary adenoma in the multiple endocrine neoplasia type 1 (MEN1) syndrome.12 Isolated familial prolactinomas (not related to MEN1) have been described.13 PRL-secreting pituitary carcinomas are exceedingly rare, with about 50 documented cases in the literature.3
Pathogenesis
Similar to other pituitary adenomas, prolactinomas are monoclonal in origin.14 To date, the exact mechanisms leading to the development of lactotroph adenomas are not well established. Among the candidates are the pituitary tumor transforming gene (PTTG)15 and the heparin-binding secretory transforming gene (HST),16 both of which induce angiogenesis through fibroblast growth factors (FGF-2 and FGF-4). Estrogens seem to play a pivotal role in these mechanisms, because estrogen-modulator drugs (e.g., tamoxifen and raloxifene) and the antiestrogen ICI-182780 inhibit PTTG expression in prolactinomas in vitro and their growth in vivo (Fig. 11-1).17 Reduction in the expression of the cytokine leukemia inhibitory factor18 and nerve growth factor19 and the overexpression of bone morphogenetic protein 420 and the high-mobility group A2 gene (HMGA2)21 are among other events that potentially might be involved in prolactinoma tumorigenesis.
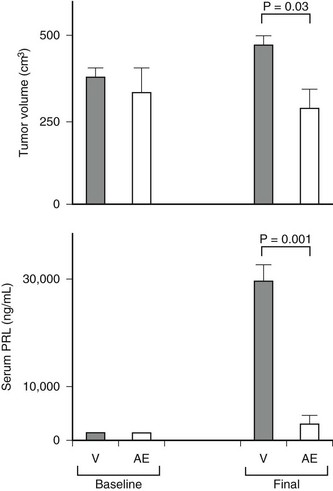
FIGURE 11-1 Selective antiestrogen treatment inhibits pituitary tumor growth in vivo. Pretreatment (baseline) and posttreatment tumor volumes and serum prolactin (PRL) levels after mini osmotic pumps infusion of vehicle or antiestrogen ICI-182780 (0.5 µg/day) was infused in 20 female Wistar-Furth rats harboring subcutaneous pituitary tumors. All animals developed tumors. Each bar represents mean ± SEM for 10 animals per group. *P = 0.03; **P < 0.001. (From Heaney AP, Fernando M, Melmed S: Functional role of estrogen in pituitary tumor pathogenesis. J Clin Invest 109:277–283, 2002.)
Studies on the microvascular density of prolactinomas show conflicting results: One study by electronic microscopy did not disclose differences between vascular density of the normal pituitary and microprolactinomas, but macroprolactinomas exhibited a much lower degree of vascularization. A more recent study using immunohistochemistry with antibodies for different endothelial markers observed that microprolactinomas are less vascular than macroprolactinomas. Additionally, microvascular density was related to tumor invasiveness and malignancy.22
The decrease of dopaminergic inhibition seems to play a role, at least a permissive one, in prolactinoma development. Lesions in the tuberoinfundibular dopaminergic neurons in female rats bearing prolactinomas were described.23 Several studies have shown the development of prolactinomas in mice with D2 receptor knockout, with this phenotype being more severe and presenting a faster evolution in females and in animals treated with estrogens.24,25 To date, no “natural” mutations were found in the D2 receptor gene.26 PRLr(−/−) mice exhibited more intense hyperprolactinemia and larger tumors than did age-matched Drd2(−/−) mice, and there were cumulative effects in compound homozygous mutant male mice. This fact suggests that PRL inhibits lactotrophs not only by the activation of hypothalamic dopamine neurons, but also directly within the pituitary in a dopamine-independent fashion.
Prolactinomas associated with MEN1 present inactivating mutations characterized by loss of heterozygosity in locus 11q13 and mutations in the menin-codifying gene. They tend to be larger and more aggressive than their sporadic counterparts.12 Sporadic prolactinomas may exhibit loss of heterozygosity but without mutations in the menin gene detected to date, suggesting the presence of a tumor suppressor gene located within chromosome 11q13 yet distinct from MEN1.27 The recently described inactivating mutations of the gene encoding aryl hydrocarbon receptor–interacting protein (AIP) on chromosome 11q13.3 are frequently found in isolated familial pituitary adenomas (mainly somatotropinomas but also prolactinomas)28 but rarely in sporadic pituitary tumors.29 Finally, mutations in the proto-oncogene ras and in the tumor suppressor gene TP53 can be linked to the development of the rare PRL-secreting carcinomas.3
Pathology
The terms microadenoma and macroadenoma, coined by Hardy,30 represent pituitary adenomas measuring less or more than 1 cm, respectively. Microprolactinomas are found mainly in young women, usually located in the lateral portions of the pituitary. They are generally enclosed within a pseudocapsule but also can be invasive.31 Macroprolactinomas also can be enclosed but usually expand to the optic chiasmal region or invade local structures such as the cavernous sinus or the sellar floor and sphenoid sinus.31 Histologically, there are no differences between microprolactinomas and macroprolactinomas, which are usually “chromophobic” on hematoxylin-eosin staining. For this reason, many pituitary adenomas previously classified as “functionless” before the development of a radioimmunoassay for PRL were prolactinomas. This fact is explained by the use of electronic microscopy, which characterized most prolactinomas as sparsely granulated: oval and slightly irregular nuclei, complex rough endoplasmic reticulum, large Golgi complexes, and sparse spherical or pleomorphic secretory granules, measuring 130 to 500 nm (Fig. 11-2).32 The hallmark of these tumors is the so-called misplaced exocytosis, the extrusion of secretory granules along the lateral cell border. This phenomenon diagnoses PRL secretion either by normal or by neoplastic lactotrophs. The rare, densely granulated lactotroph adenoma is strongly acidophilic with strong, diffuse immunostaining for PRL in the cytoplasm. The rough endoplasmic reticulum is less prominent, and the secretory granules are bigger (500 to 700 nm) and more numerous than in its sparsely granulated counterpart.32
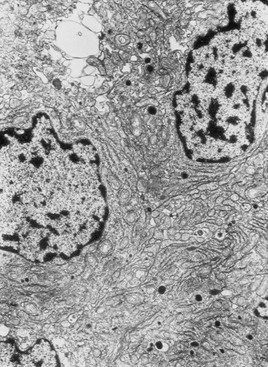
FIGURE 11-2 Electronic microscopy of a sparsely granulated prolactinoma, showing sparse secretory granules, well-developed rough endoplasmic reticulum, and prominent Golgi complexes.
Electron microscopy usually is not required for prolactinoma diagnosis since the immunohistochemical assessment of PRL production became routinely available. This technique directly characterizes PRL-secreting adenomas, ruling out “functionless” macroadenomas associated with hyperprolactinemia due to hypothalamus-pituitary disconnection, the so-called pseudoprolactinomas.33 A study of 120 unselected necropsies showed that 27% of them harbored pituitary microadenomas without clinical expression, 40% of which immunostained for PRL.11 These data indicate that there could be clinically and hormonally non–PRL-secreting pituitary adenomas that are immunohistochemically positive for this hormone. The significance of this finding for the natural history of prolactinomas is unknown.
Acidophil Stem Cell Adenoma
Exceptionally hyperprolactinemic patients might exhibit mild or no clinical features of acromegaly associated with biochemical evidence of slight serum GH elevation. This situation is due to the presence of the rare and aggressive acidophil stem cell pituitary adenoma.34 The acidophilia is attributable to mitochondrial accumulation called oncocytic change. Immunohistochemical analysis is positive for PRL, and occasionally there is a scant positivity for GH. The definitive diagnosis requires electron microscopy, which depicts enlarged mitochondria. Scattered cells containing juxtanuclear fibrous bodies are similar to cells of sparsely granulated somatotroph adenomas. Misplaced exocytosis is present. The secretory granules are sparse and small, measuring 150 to 200 nm.
Nontumoral Lesions Associated With Hyperprolactinemia
Many nontumoral conditions can be mistaken for prolactinomas. Thyrotroph hyperplasia is associated with primary hypothyroidism due to loss of feedback (Fig. 11-3).35 The correct diagnosis is important because this condition regresses with thyroid hormone replacement.35 Idiopathic lactotroph hyperplasia is a rare cause of hyperprolactinemia that mistakenly can be taken for an expanding macroprolactinoma. The pituitary mass shape can help in the differential diagnosis. Inflammatory lesions such as lymphocytic hypophysitis (occurring mainly during pregnancy and puerperium) and sarcoidosis can be misdiagnosed as lactotroph adenoma.36,37
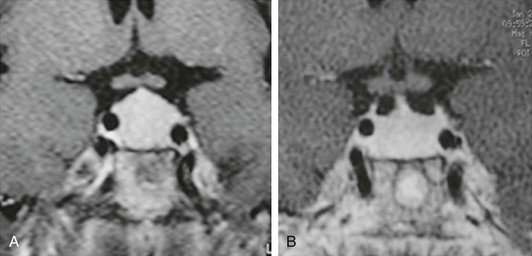
FIGURE 11-3 Magnetic resonance imaging (gadolinium-enhanced T1-weighted coronal views) of a 41-year-old woman with primary hypothyroidism before (A) and after 1 month on levothyroxine replacement therapy (B). (From Bronstein MD: Problems in the differential diagnosis of the hyperprolactinemic patient. Clinical Endocrinology Update 2003, syllabus pp 241–247; with permission of the Endocrine Society.)
Pituitary Prolactin-Secreting Carcinomas
Pituitary PRL-secreting carcinomas are exceedingly rare tumors that exhibit morphologic features indistinguishable from those of PRL-secreting adenomas. In addition to local invasiveness, distant intracranial and extracranial metastases in the nervous system and visceral metastases are the clues for the diagnosis of malignancy. Regarding intracranial lesions, sometimes it is difficult to distinguish between contiguity of an invasive prolactinoma and true metastasis from a PRL-secreting carcinoma.3
Differential Diagnosis of Hyperprolactinemia
The main causes of hyperprolactinemia are listed in Table 11-1.
Table 11-1
PHYSIOLOGIC
DRUG-INDUCED
PATHOLOGIC
Physiologic Hyperprolactinemia
Throughout pregnancy, the size of a normal pituitary increases up to 136%, according to magnetic resonance imaging (MRI) studies.38 This extensive growth is due to estrogen-induced hypertrophy and hyperplasia of lactotrophs, leading to progressive increase in PRL production and its hypersecretion during pregnancy.39 Placental estrogen production stimulates lactotroph mitosis, PRL mRNA levels, and PRL synthesis, leading to a stepwise increase in serum PRL levels, achieving mean levels of 200 ng/mL at the end of pregnancy and up to 450 ng/mL in some cases. Serum PRL levels decline quickly after delivery but are maintained slightly increased in nursing women several months, especially after breastfeeding. At birth, newborn serum PRL concentrations are elevated nearly 10-fold, probably as a result of the stimulatory effect of maternal estrogen levels.40
Because exercise and nonspecific stress are physiologic causes of hyperprolactinemia, there is a concern that the stress-induced PRL increase could lead to hormone elevation during venipuncture, and a period of rest before blood withdrawal is still recommended in many laboratories. A report by Vieira and coworkers,41 which included a large population, provided evidence that rest before blood collection may be needed in only a few patients.
Pharmacologic Hyperprolactinemia
Among medications that increase serum PRL, dopamine receptor blockers are the most potent. Neuroleptics (e.g., sulpiride, haloperidol, chlorpromazine, risperidone) and antiemetic drugs (e.g., metoclopramide, domperidone) can elevate serum PRL to levels that usually are detected with prolactinomas.42 Serotoninergic and antihistaminergic drugs are less potent than antidopaminergic medications. The calcium channel blocker verapamil elevates serum PRL levels probably by decreasing central dopamine generation, possibly through N-type calcium channels.43 It was shown that protease inhibitors used for treatment of acquired immunodeficiency syndrome can cause hyperprolactinemia, but the mechanism is unknown.44 A detailed inquiry about drug use is mandatory for all hyperprolactinemic patients.
Pathologic Hyperprolactinemia
Hyperprolactinemia is present in about 40% of acromegalic patients as a result of GH/PRL co-secretion by the same or by different tumor cells or secondary to hypothalamus-pituitary disconnection.45 Because of the characteristic features of acromegaly, the differential diagnosis is usually not a problem. As already mentioned, in patients harboring the rare acidophil stem cell pituitary adenoma, serum GH is usually low compared with PRL levels, however, and acromegalic features are usually absent or minimally expressed.34 In some cases, PRL resistance to dopamine agonists can be a clue for the differential diagnosis with prolactinomas.
Hyperprolactinemia secondary to impaired hypothalamic/tuberoinfundibular dopamine secretion or to stalk or even intrapituitary disconnection can be caused by tumors, inflammatory diseases, or trauma. In these cases, PRL is produced by normal lactotrophs and rarely exceeds 150 µg/L. The differential diagnosis of macroprolactinomas is mainly with clinically nonfunctioning pituitary adenomas (pseudoprolactinomas) and, to a lesser extent, with craniopharyngiomas.46 Other tumoral lesions, such as meningiomas and chordomas, and nontumoral conditions, such as “empty sella” syndrome and even intrasellar aneurysms, can be associated with hyperprolactinemia, however (Fig. 11-4).47 The differential diagnosis between macroprolactinomas and pseudoprolactinomas is crucial regarding their primary treatment—medical for macroprolactinomas and surgical for pseudoprolactinomas. Patients with nonfunctioning tumors treated with dopamine agonists were considered as having resistant macroprolactinomas, owing to the absence of tumor shrinkage even with the (obvious) PRL decrease to very low or undetectable levels (Fig. 11-5).48 A clue to differentiate pseudoprolactinomas from true prolactinomas is the dramatic early PRL decrease with bromocriptine doses of 1.25 mg/day.
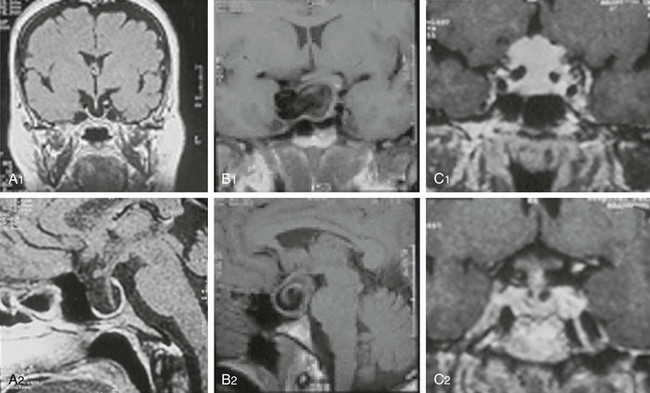
FIGURE 11-4 Magnetic resonance imaging of lesions (1, coronal views; 2, sagittal views) associated with hyperprolactinemia: “empty sella” (A1, A2), intrasellar aneurysm (B1, B2), and meningioma (C1, C2).
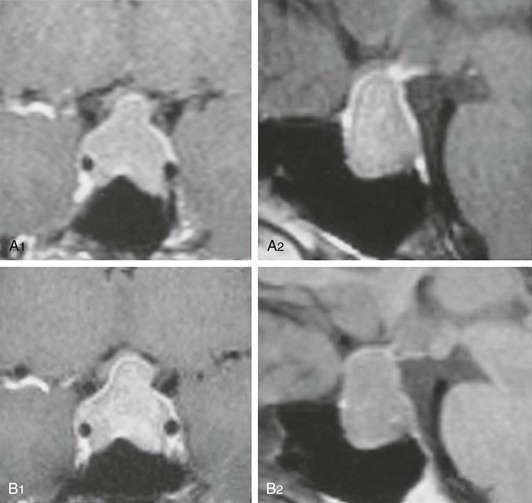
FIGURE 11-5 Magnetic resonance imaging of a 41-year-old woman with a nonfunctioning pituitary adenoma and serum prolactin level of 51 ng/mL before (A1, coronal view; A2, sagittal view) and during the 12th month of cabergoline “treatment” (B1, coronal view; B2, sagittal view). No tumor shrinkage was observed, despite serum prolactin decrease to 1.2 ng/mL. (From Bronstein MD: Problems in the differential diagnosis of the hyperprolactinemic patient. Clinical Endocrinology Update 2003, syllabus pp 241–247; with permission of the Endocrine Society.)
Primary hypothyroidism can be associated with hyperprolactinemia, presumably due to high thyrotropin-releasing hormone levels that stimulate PRL release and presumably reduce prolactin metabolic clearance. Thyrotroph hyperplasia may occur, leading to pituitary enlargement mimicking a pituitary adenoma (see Fig. 11-3).35 Cushing’s disease and adrenal insufficiency can be associated with hyperprolactinemia, which also can be present in Nelson’s syndrome.49,50 Polycystic ovary syndrome also may be associated with hyperprolactinemia.51 Menstrual disturbances are prevalent in patients with polycystic ovary syndrome and in patients with prolactinomas, and sometimes the distinction between the two conditions may be difficult. The presence of mild hyperprolactinemia, negative pituitary imaging, high luteinizing hormone/follicle stimulating–hormone ratio, and clinical features suggestive of polycystic ovary syndrome can help in the differential diagnosis.
Uremia can be associated with hyperprolactinemia, mainly in patients with end-stage renal disease.52 The mechanism probably is related to reduced PRL clearance and to a presumably reduced dopaminergic tonus. Serum PRL elevation is mild but can be considerably increased in uremic patients taking drugs with a dopamine receptor–blocking effect. Hyperprolactinemia is present in up to 20% of patients with liver cirrhosis, probably due to an unbalanced estrogen-to-androgen ratio and to an altered dopaminergic tonus.53 Nipple manipulation and chest-wall lesions, such as herpes zoster and surgical scars, may increase serum PRL via stimulation of neuron pathways going through the spinal cord.54 In contrast to paraneoplastic adrenocorticotropic hormone (ACTH) secretion, ectopic production of PRL has rarely been described.55 Finally, when all the above-mentioned causes are ruled out, hyperprolactinemia is considered idiopathic or functional. It is likely, however, that most patients with this condition harbor small microprolactinomas that went undetected with less-sensitive imaging tools used in the past, such as hypocycloidal polytomography and computed tomography (CT) and even with MRI. If there is no radiologic evidence of a prolactinoma at initial diagnosis of hyperprolactinemia, however, an identifiable adenoma is unlikely to develop in the long-term follow-up.56 Nevertheless, a recent study by De Bellis et al.57 points out the presence of anti-pituitary antibodies in 25.7% of patients with idiopathic hyperprolactinemia versus 0% in those with microprolactinoma, suggesting an autoimmune etiology for a subset of cases of idiopathic hyperprolactinemia.
Diagnosis of Prolactinomas
Hyperprolactinemia and its impact on the gonadotropic axis is the hallmark of microprolactinomas and macroprolactinomas and their clinical manifestations. Macroprolactinomas also may cause neurologic and visual disturbances and impairment of other pituitary functions owing to the tumor-mass effect (Table 11-2).58 Loss of visual fields and impairment of visual acuity are the main ophthalmologic manifestations. Headache is the most common neurologic presentation; it is attributed to dural stretch or cavernous sinus invasion and less frequently as part of the clinical symptoms of pituitary apoplexy.59 There are a few reports of special types of headache, such as the SUNCT (short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing) syndrome secondary to prolactinomas, but the role of hyperprolactinemia as causative agent is obscure.60 Other rare presentations of invasive macroprolactinomas are hydrocephalus,61 neuropsychiatric manifestations, and otoneurologic manifestations.62,63
Table 11-2
Clinical Manifestations of Prolactinomas
RELATED TO HYPERPROLACTINEMIA
RELATED TO TUMOR MASS EFFECT
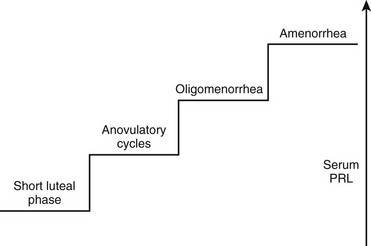
Women generally present with a correlation between the degree of serum PRL elevation and gonadal impairment, ranging from a short luteal phase to amenorrhea (Fig. 11-6). Most PRL-secreting pituitary adenomas are associated with amenorrhea, because hyperprolactinemia due to prolactinomas usually shows serum PRL levels greater than 100 ng/mL. Because hyperprolactinemia is present in about 20% of women with amenorrhea,64 most patients with such menstrual disturbance and high serum PRL levels may harbor a prolactinoma. Because microprolactinomas can be associated with serum PRL levels between 40 and 100 ng/mL, and because PRL biological activity does not always correspond to routine laboratory assay levels,65 some women with prolactinomas may exhibit milder forms of gonadotropic impairment such as anovulatory cycles and oligomenorrhea.66 In addition to menstrual disturbances, hyperprolactinemic women complain of loss of libido, vaginal dryness, dyspareunia, and psychological distress.67 Similar to other disorders associated with hypogonadism, osteoporosis is a frequent finding in women with hyperprolactinemia/prolactinomas, mainly in prolactinomas with long-standing hypogonadism. The relative risk of developing osteoporosis in premenopausal women with prolactinomas can be 4.5%.68 Although a direct effect of PRL on bone has been considered, there is evidence that hyperprolactinemia is not a risk factor in itself for the development of osteoporosis, with the associated hypoestrogenism being the major determinant.69
Galactorrhea is a frequent finding in women with hyperprolactinemia, but it can be absent, mainly in patients with macroprolactinomas associated with severe hypogonadism. Breast examination, if not adequate, also may fail to disclose mild galactorrhea. These facts may explain the discrepancy among series, which show a prevalence of galactorrhea ranging from 30% to 84%.64,70 Galactorrhea also may occur in normoprolactinemic women. A study including 235 patients with galactorrhea associated with diverse conditions showed that serum PRL was normal in 86% of women with idiopathic galactorrhea without amenorrhea.71 The mechanism is unknown, being attributed to different causes such as mammary hypersensitivity to PRL or “occult” transient hyperprolactinemia.
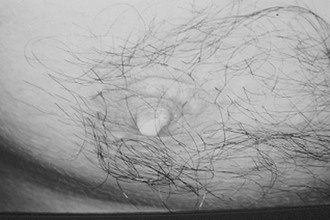
Hyperprolactinemia in men is predominantly associated with macroprolactinomas. The main clinical manifestations are hypogonadism, which is usually severe, along with loss of libido and reduced body hair growth, visual impairment, and headache.72 Only 22.5% of 80 patients with macroprolactinomas in our series sought medical assistance due to sexual complaints, whereas most patients requested appointments because of visual or neurologic disturbances. During the interview, 85% of the patients admitted, however, that loss of libido was of utmost importance.73 In patients with microprolactinomas (n = 12), sexual dysfunction was the main complaint in 67% of the cases, but that number increased to 92% after the interview.73 Testosterone replacement without serum PRL normalization seldom restores libido, an observation that points to a direct effect of PRL on sexual behavior, as previously suggested by animal models.74 Galactorrhea is far less frequent in men than in women with prolactinomas.75 It was disclosed in 15% and 25% of our patients with macroprolactinomas and microprolactinomas.73 When present in men harboring pituitary tumors, however, it strongly suggests the presence of a prolactinoma (Fig. 11-7).75 Osteoporosis also is present in hyperprolactinemic men.76
Although the prevalence of prolactinomas in both genders is higher in the 20s and 30s, prolactinomas can occur in elderly and in younger individuals. Data on 44 young patients (12 males and 32 females, aged 16.3 ± 1.9 years at diagnosis) with pituitary adenomas showed a predominance of macroadenomas (61%) over microadenomas (39%). Of those, prolactinomas were the most prevalent (68% of cases).77 Other series on prolactinomas diagnosed in childhood or adolescence showed that the prevalence of macroadenomas was also higher (15 versus 11 cases)78 or similar (24 versus 23 cases) compared with microadenomas.79 The predominance of larger tumors in children and adolescents points to molecular mechanisms influencing proliferation rather than the time course of the disease influencing the progression of prolactinoma size and invasiveness.
Laboratory Evaluation
Basal serum PRL evaluation usually confirms the clinical suspicion of a prolactinoma. Serum PRL usually ranges from 50 to 300 ng/mL in the presence of microprolactinomas and from 200 to 5000 ng/mL in the presence of macroprolactinomas (normal values range from 2 to 15 ng/mL). Values of 30 ng/mL have been associated with microprolactinomas, however, and values of 35,000 ng/mL have been found in patients harboring large and invasive macroprolactinomas. Stimulation tests with thyrotropin-releasing hormone and metoclopramide or suppression tests using levodopa, previously popular mainly for the differential diagnosis of microprolactinomas and so-called idiopathic hyperprolactinemia, give nonspecific results and have been largely abandoned.80
Hyperprolactinemia secondary to hypothalamus-pituitary disconnection rarely exceeds 150 ng/mL, and lesions that mimic macroprolactinomas, especially nonfunctioning pituitary adenomas, generally exhibit serum PRL less than this value (pseudoprolactinomas).46 A laboratory artifact may lead to an erroneous differential diagnosis between macroprolactinomas and pseudoprolactinomas, however. When serum PRL is evaluated by two-site immunometric assays, large amounts of antigen—PRL in this case—saturate capture and signal antibodies, impairing their binding and causing serum PRL to be underestimated (the so-called high-dose hook effect). Patients bearing macroprolactinomas with extremely high serum PRL levels (generally >10,000 ng/mL, depending on the assay measuring range) may present with falsely lower levels, within the 30 to 150 ng/mL range, causing the patient to be misdiagnosed as harboring a nonfunctioning pituitary adenoma. To avoid unnecessary surgeries (treatment of choice for nonfunctioning tumors), PRL assays with serum dilution or using two-step incubation are recommended in patients with macroadenomas who may harbor a prolactinoma.81
If such assays are not readily available, clinical clues pointing to prolactinomas are patient age younger than 50 years, presence of galactorrhea in male patients, and tumor shrinkage under dopamine agonist drugs, as shown by fast visual improvement in cases with chiasmal compression or rapid tumor reduction evidenced by MRI.75
Another laboratory pitfall concerns the presence of high serum PRL levels in subjects with few or no symptoms related to PRL excess. Human PRL in circulation manifests as marked size heterogeneity, with three forms (23 kD, 50 kD, and 150 to 170 kD) that are indistinguishable by routine assays.82 The 23-kD form (little PRL) is the most common form, but serum PRL can be elevated secondary to the presence of 150- to 170-kD aggregates with low biological activity (big-big PRL), leading to macroprolactinemia, a term coined by Jackson and colleagues83 in the 1980s. Less frequently, the 50-kD form (big PRL) can be the prevalent circulating form.84,85 The presence of molecular aggregates with low biological activity, such as big-big PRL, should be suspected when high serum PRL levels are detected in patients without or with scarce signs and symptoms related to hyperprolactinemia.4,86 Precipitation with polyethylene glycol is an excellent screening method.87 The predominant molecular form recovered (i.e., assayed after the precipitation) is the highly biologically active little PRL. The gel filtration chromatography confirms the presence of big-big PRL (Fig. 11-8), but being a costly and time-consuming method, it is performed for practical clinical purposes only when polyethylene glycol precipitation results are inconclusive. Macroprolactinemia is a common finding, occurring in 8% to 42% of all cases of hyperprolactinemia.4 The pathogenesis of macroprolactinemia is still unknown. It could be in part a complex of monomeric PRL with immunoglobulin G89; anti-PRL autoantibodies were identified in patients with idiopathic hyperprolactinemia.89
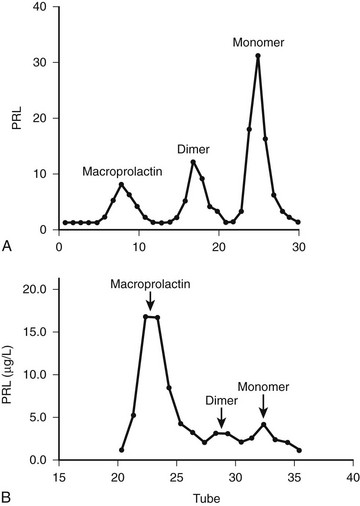
FIGURE 11-8 Gel filtration chromatography of a patient with symptomatic prolactinoma (A) and of an asymptomatic woman with macroprolactinemia (B). PRL, Prolactin.
Big-big PRL biological activity is still controversial in the literature. Studies in vitro with rat Nb2 cell bioassays show either the presence or the absence of biological activity.4 To explain the dissociation of presence of activity in vitro but not in vivo, we can speculate that because of its large molecular weight, macroprolactin does not cross the capillary barrier, and it is unable to reach target cells. Additionally, the PRL receptor forms of rat cells are different from those of humans, so a bioassay using cells harboring human PRL receptors which addresses the biological activity of macroprolactin should be of interest. As a matter of fact, a study by Glezer et al.90 showed that sera of individuals with macroprolactinemia presented lower biological activity in a bioassay using a mouse cell transfected with the long form of human PRL receptor as compared to the rat Nb2 bioassay (Fig. 11-9). Moreover, in a recent publication, Hattori et al.89 claim that the level of bioactivity of macroprolactin in the Nb2 bioassay is present due to the dissociation of monomeric PRL from the autoantibodies, as a result of the longer incubation and more dilute assay conditions. Despite these controversies in the literature concerning the biological activity of PRL aggregates, most patients with macroprolactinemia do not manifest clinical features related to hyperprolactinemia and do not need any treatment. To avoid unnecessary medical or surgical procedures, macroprolactin screening is mandatory when clinical features and serum PRL assay results are conflicting.
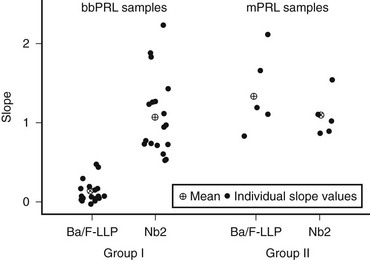
FIGURE 11-9 Individual slopes of bioactivity/immunoactivity (BA/IA) of macroprolactinemic individuals (group I) and hyperprolactinemic patients without macroprolactinemia (group II). The mean slope obtained in Ba/F-LLP assay for group I was significantly lower, compared to that obtained in the Nb2 assay for the same group. On the contrary, no significant differences between the mean slopes of both assays were found in group II. (From Glezer et al: Human macroprolactin displays low biological activity via its homologous receptor in a new sensitive bioassay. J Clin Endocrinol Metab 91:1048–1055, 2006.)
Assessment of other pituitary functions is mandatory, mainly in the presence of macroprolactinomas. Insulin-like growth factor 1 must be followed either to verify GH deficiency or to disclose the rare cases of prolactinomas that progress with PRL/GH co-secretion.91 Gonadotropin assessment may explain cases with persistent amenorrhea or sexual impairment despite PRL normalization. Although less frequently affected, thyrotropic and adrenocorticotropic axes also must be evaluated. Thyrotropin assessment also may disclose primary hypothyroidism as the cause of hyperprolactinemia.35 Because restoration of pituitary function may occur after adenoma removal by surgery or tumor shrinkage attained by medical therapy,92,93 these assessments have to be repeated after such procedures to avoid unnecessary hormone replacement.
Imaging
MRI is currently the gold-standard imaging method for diagnosis and treatment follow-up of micro- and macroprolactinomas, although CT scan still have a place for sellar region assessment. MRI is superior to CT for tumor boundary delineation, especially regarding its associations with the optic chiasm and cavernous sinus (Fig. 11-10). Additionally, tumor consistency, presence of hemorrhage, and presence of cystic lesions are better shown by MRI, especially comparing T1-weighted and T2-weighted images. CT has the advantage of showing bone erosion and calcifications and may help surgical planning.94 Although MRI is currently the most sensitive imaging method for microprolactinomas, tumors measuring less than 2 mm may remain undetected by this technique. This fact is well known for the usually tiny ACTH-secreting adenomas leading to Cushing’s disease, with about 50% of them not identified by MRI.95 It is probable that many of the so-called idiopathic hyperprolactinemic patients harbor a small microprolactinoma. Procedures such as dynamic imaging with gadolinium enhancement (Fig. 11-11) and spoiled gradient recalled acquisition technique (SPGR)96 may contribute to improve MRI sensitivity for such tiny adenomas.
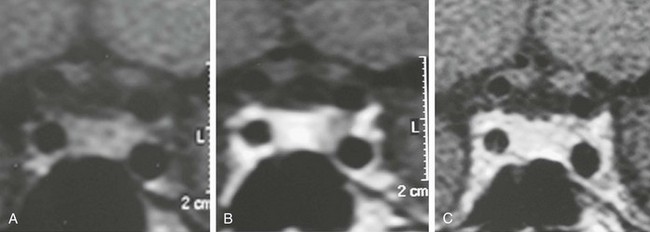
FIGURE 11-10 Magnetic resonance imaging (coronal views) of a microprolactinoma disclosed only during dynamic gadolinium-enhanced sequence. A, Before contrast injection. B, Lack of contrast enhancement on the left side of the pituitary gland suggestive of microadenoma. C, Whole gland contrasted.
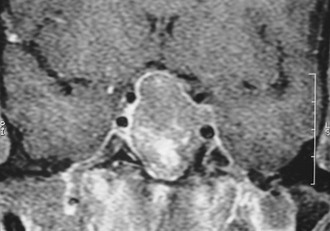
FIGURE 11-11 Magnetic resonance imaging (gadolinium-enhanced coronal view) showing optic chiasm compression and left cavernous sinus invasion by a macroadenoma.
MRI can disclose the so-called pituitary incidentaloma, a term coined by Reincke in 1990 to describe incidentally discovered pituitary masses by imaging performed to evaluate conditions not linked to pituitary disease, including head trauma and sinusitis. Controversies exist in the literature concerning the definition of pituitary incidentalomas.97 Molitch98 included patients who presented with symptomatic pituitary disease and who were diagnosed only when a sellar mass was incidentally disclosed. In our opinion, the term incidentaloma should be reserved for cases with no endocrine or mass effects of a pituitary adenoma.4 Incidental imaging findings of pituitary adenomas, mainly microadenomas, are present in up to 27% of autopsy findings in the general population.11 Other imaging pitfalls include normal anatomic variations such as asymmetric sphenoid septum with bulging of the sellar floor or displacement of the pituitary stalk,99 artifacts such as clips and prostheses, and global pituitary enlargement—either physiologic, as shown in puberty or even in young adults100 and pregnancy,101 or pathologic, as seen in primary hypothyroidism 35 and mental depression.102
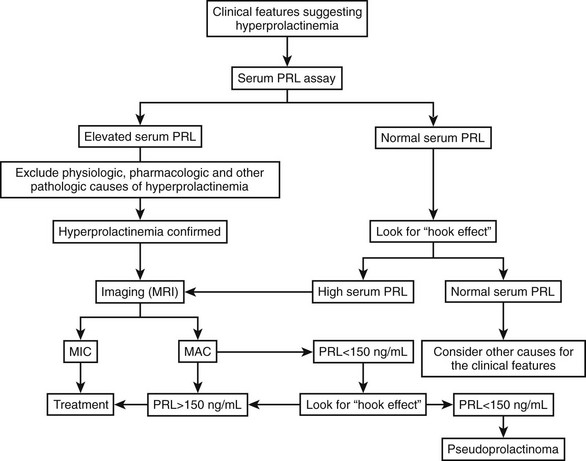
Regarding the functional evaluation of prolactinomas by imaging, in-vivo imaging by single-photon emission computed tomography using radiolabeled high-affinity benzamine derivatives to D2 receptors, such as iodine 123–methoxybenzamide or iodine 123–epidepride, could serve as a response predictor to dopamine agonist treatment.103,104 Positron emission tomography using 18-fluorodeoxyglucose, carbon 11–methionine, or dopamine D2 receptor ligands also has been used for in-vivo assessment of the metabolic rate and D2 receptor density of macroprolactinomas and other pituitary tumors.105 These methods are expensive and time consuming, however, and are better reserved for the study of tumors without a hormone marker, such as clinically nonfunctioning pituitary adenomas. Regarding prolactinomas, such techniques currently should be reserved for investigational purposes or for special cases, such as searching for metastases in the rare malignant prolactinomas.106 Fig. 11-12 is a flow diagram for the diagnostic evaluation of hyperprolactinemia.
Treatment
As far as prolactinoma treatment is concerned, the 1970s brought almost concomitantly two powerful therapeutic advances: the improvement of pituitary microsurgery2 and the development of ergot derivatives with potent dopaminergic agonistic activity.107 Additionally, radiation therapy, including more recent stereotactic techniques, has a place, albeit restricted, in prolactinoma treatment (Table 11-3). Therapeutic strategy must consider several aspects, such as the patient’s clinical presentation, the differences between microadenomas and macroadenomas concerning their natural history, the desire for pregnancy, and the patient’s treatment preference, if applicable.
Table 11-3
MEDICAL
SURGICAL
RADIOTHERAPY
Surgical Therapy
Pituitary surgery by the transsphenoidal approach was used at the beginning of the 20th century and was reintroduced in the early 1960s and greatly improved when the surgical microscope was introduced.2,108 More recently, endonasal endoscopic surgery has become available. Compared to the classic transsphenoidal approach, endoscopic pituitary surgery seems to reduce the time of hospitalization, but improvement of surgical results remains to be demonstrated.109,110 These developments made the selective removal of the pituitary adenoma possible, sparing the normal gland, along with low complication and mortality rates, mainly for surgeons with more than 500 operations.111 Besides serum PRL normalization, this surgical modality aims at reducing or eliminating the mass effect of expanding macroadenomas, often leading to the resolution of neurologic and visual manifestations. The transcranial surgical approach is reserved solely for tumors with a predominance of extrasellar location expanding out of the midline.112
The surgical success in normalizing serum PRL levels depends on the experience and ability of the surgeon and on the tumor size and invasiveness. Preoperative serum PRL levels, usually associated with tumor dimension and location, were found to be paramount in predicting surgical remission, being the only predictive factor in multivariate analysis in some reports.113,114 Consequently, the best results were achieved in microprolactinomas with a preoperative serum PRL less than or equal to 100 ng/mL. A study on the initial outcome of 219 women with prolactinomas operated on by transsphenoidal microsurgery showed a remission rate of 92% of cases with preoperative PRL less than or equal to 100 ng/mL and 91% with intrasellar microadenomas, but only 59% in women with microadenomas with cavernous sinus extension, leading to an overall remission rate of 82% for microadenomas. Of the women with macroprolactinomas, 88% with intrasellar adenomas, 86% with moderate suprasellar extension, and 80% with focal sphenoid sinus invasion achieved remission. Surgical remission in patients with diffuse sphenoid sinus invasion, cavernous sinus invasion, and major suprasellar extension was poor, however, ranging from 0% to 44%.113 In another large series of 120 patients with prolactinomas (93 women and 27 men) who underwent pituitary surgery by the transsphenoidal route, PRL normalization occurred in 78% of patients with microadenomas, in 87.5% of patients with intrasellar macroadenomas, and in 27% of patients with extrasellar macroadenomas.114
A compilation of 50 published series showed that postsurgical serum PRL normalization was achieved in 73.7% of 2137 microprolactinomas and 33.9% of 2226 macroprolactinomas.115 Comparison among the series is difficult, however, because many authors do not mention preoperative serum PRL levels, the tumor size, and degree of invasiveness. The grade of clinical improvement in cured patients is high, mainly in smaller tumors. Menses and fertility restoration is high, and pregnancy rates ranged from 75% to 90% in different series. Some patients can achieve menstrual regulation and even become pregnant without full PRL normalization.114,116 Sexual improvement in men is less likely to occur, probably owing to irreversible gonadotropic damage caused by the higher frequency of extrasellar and invasive prolactinomas compared with women. Nonetheless, pituitary function generally is preserved in intrasellar adenomas and is restored by the removal of suprasellar adenomas causing hypothalamus-pituitary disconnection.114 In addition, neurologic and visual amelioration often is achieved after pituitary surgery.
Many patients bearing prolactinomas have undergone treatment with dopamine agonist drugs before surgery. The effect of previous medical therapy on surgical outcome is still a matter of debate. Some reports point to poorer results compared with nontreated patients117,118; others do not show significant differences.113,114 There is also evidence, however, of improvement of surgical results in the medically pretreated group.119,120 Because the issue of dopamine agonist–induced fibrosis remains unresolved, it is unclear whether the negative outcome was caused directly by the drug or whether it was biased by a tendency to treat medically large and invasive tumors, which present with the poorest surgical results.
An important caveat of surgical treatment is prolactinoma recurrence. This issue was raised in the early 1980s when an article from the Montreal group reported recurrence rates in surgically “cured” patients to be 50% for microprolactinomas and 80% for macroprolactinomas.121 Subsequently, several reports on this issue were published but with less impressive figures. A literature compilation reported recurrence rates of 18.2% of 809 microprolactinomas and 22.8% of 465 macroprolactinomas.115 In our series, the recurrence of hyperprolactinemia in surgically cured patients was 27% for microprolactinomas and 17% for macroprolactinomas.116 Median time to recurrence varied among the different surgical series, from 1 to 7 years. Concerning recurrence predictors, many studies pointed to higher postoperative serum PRL levels in patients who recurred compared with patients without recurrence,113,121,122 whereas others did not find serum PRL levels to be a predictor of late outcome.123 Another marker of recurrence is the absence of PRL response to dynamic tests, especially to thyrotropin-releasing hormone.114,123 These findings raise the question whether, in prolactinomas, serum PRL increase after surgical normalization represents true recurrence or important but incomplete tumor removal.
Although the occurrence of relapses contributes to the decrease of long-term surgical cure rate of prolactinomas, many patients with recurrent hyperprolactinemia remain asymptomatic and have no evidence of tumor regrowth.113,124 Relapse can be transient. From a cohort of 44 patients with surgically cured microprolactinomas, 8 (18.2%) experienced recurrence and were followed up. Only two of the eight patients experienced permanent relapse.125 Additional therapy is reserved only for patients with symptomatic recurrence.
Surgical treatment for prolactinomas is indicated for cases with persistent intolerance or hormonal or tumor resistance to dopamine agonists (see later); pituitary apoplexy126; tumor growth during medical therapy127; cerebrospinal fluid (CSF) leakage due to dopamine agonist–induced tumor shrinkage of invasive macroprolactinomas128,129; and (rarely) visual loss on medical therapy, secondary to optic chiasm herniation resulting from tumor retraction.130 Additionally, surgery is an excellent alternative for patients harboring microprolactinomas, especially patients with serum PRL less than or equal to 100 ng/mL, with poor compliance to medical therapy.113,131
Medical Therapy
Bromocriptine
At the end of the 1960s, an ergot derivative, 2-bromo-α-ergocryptine (bromocriptine) was developed, and shortly thereafter its use in clinical trials was initiated.107,132 Bromocriptine binds and stimulates the seven-membrane–spanning dopamine D2 receptors in normal and in adenomatous lactotrope cells, inducing activation of the Gi receptor (negatively coupled to adenylate cyclase) and, consequently, leading to postreceptor events that ultimately cause the inhibition of PRL synthesis and secretion.133 Although many other dopamine agonist drugs were developed thereafter, bromocriptine is still the one that presents the largest and longest worldwide experience (Table 11-4).
Pharmacokinetic studies with bromocriptine show that after a single oral dose of 2.5 mg, serum levels peak, and maximal suppressive action occurs between 1 and 3 hours. The drug has a relatively short mean elimination half-life (about 6.2 hours) and generally is administered twice a day.134 Owing to a considerable interindividual variability in the PRL-lowering effects of a given dose of bromocriptine, however, some patients need to take the drug three times a day and others just once a day, mainly when higher doses are required or normoprolactinemia already has been achieved.135
Bromocriptine treatment results in serum PRL normalization and clinical improvement in most patients with hyperprolactinemia/prolactinomas. A compilation of 13 series from the literature, encompassing 286 women treated with bromocriptine, showed PRL normalization and return of menses from 64% to 100% and 57% to 100% of cases.134 Our data showed evidence of normoprolactinemia in 55% of patients with microprolactinoma treated primarily or postsurgically with bromocriptine (mean dose 3.8 mg/day), with menses return in 98% of the patients. Of women without PRL normalization, 81% also recovered menses, making unnecessary the increase of bromocriptine dose, optimizing drug tolerance and reducing costs.116 Although less frequent, microprolactinoma in men also can be treated successfully with dopamine agonists. Of 12 patients predominantly treated with bromocriptine, 83% achieved normal PRL level and clinical improvement.135 Eleven men with microprolactinoma have been treated by us, with serum PRL and serum testosterone normalization in 73% and 86% of them.73
Because macroprolactinomas often present with mass effect, they were first surgically treated and, if not cured, then treated with bromocriptine. The observation by Corenblum and colleagues136 in the mid-1970s that bromocriptine use reduces tumor size in addition to its PRL-lowering effect, consequently relieving neurologic and visual complaints, also allowed primary treatment for macroprolactinomas. In a prospective multicenter trial, normal PRL levels were achieved in 18 of 27 patients (67%) followed up for at least 12 months while receiving variable doses of bromocriptine.137 All patients exhibited some degree of tumor shrinkage: 9, less than 25%; 5, between 25% and 50%; and 13, greater than 50% tumor size reduction. Another study with macroprolactinomas with extrasellar extension showed serum PRL normalization in all of the 10 men and 17 of 19 women, with mean bromocriptine doses of 13 mg/day and 8 mg/day. Tumor size was reduced by more than 50% in 18 of 29 patients (62%) with a secondary empty sella in 5 cases and by less than 50% in 11 patients. Visual field improved in most of the patients who initially presented with such abnormalities.138 Patients with giant prolactinomas (diameter >4 cm) also achieved disease control with primary bromocriptine therapy.139 In our series, serum PRL was normalized in 60% of women with macroprolactinomas (mean bromocriptine dose 7 mg/day); 72% of these women had menses recovery. Menses also were restored in 44% of patients who remained hyperprolactinemic.116 The lower rate of menses normalization compared with women harboring microprolactinomas is probably due to the higher prevalence of gonadotropic impairment in the macroprolactinoma group. Regarding macroprolactinoma in men, serum PRL and testosterone levels normalized in 67% and 85% of 66 patients bearing macroprolactinomas, showing that in men also, the gonadotropic axis may normalize even when PRL is not fully normalized.73 Other series show different figures, with PRL returning to normal levels in 83% of patients but with testosterone normalization in only 62% of them, probably reflecting different degrees of gonadotropic impairment. We obtained 80% of tumor reduction in primarily treated macroprolactinoma patients.
The mechanism of prolactinoma shrinkage by dopamine agonists is not yet fully understood. Some reports in the early 1980s suggested that tumor reduction was due to lactotrope cell size reduction.140 Bromocriptine decreases mRNA and PRL synthesis within days and cell multiplication (antiproliferative and proapoptotic mechanisms have been suggested) and tumor growth.141 These events are evidenced quickly microscopically by a decrease in the number of PRL secretory granules, involution of the rough endoplasmic reticulum and Golgi apparatus, and decrease of cytoplasmic volume. With longer periods of treatment (e.g., 6 months), there is evidence of cell vacuolization and fragmentation with collagen deposition.142 These early and late morphologic changes of the tumor lactotropes may explain the rapid regrowth of macroprolactinomas when bromocriptine is discontinued after a short period of therapy,143 whereas often no tumor expansion is observed when the drug is withdrawn after being used for a longer period.144 Serum PRL levels may be suppressed in some patients without tumor shrinkage, although the converse does not occur.
The degree of macroprolactinoma reduction by bromocriptine and its time course have been carefully analyzed.145 Data on 271 patients included from prospective series on primary therapy of true macroprolactinomas showed that 79% of patients presented with more than 25% shrinkage and that 89% of them had shrunk to some degree. Pretreatment serum PRL concentration and gender difference did not predict the degree of tumor reduction. Of 102 prolactinomas large enough to produce chiasmal compression, 85% showed tumor shrinkage of more than 25%. Another compilation, including eight series from the literature totaling 112 patients with macroprolactinomas,146 showed that 40.2% had a greater than 50% reduction in tumor size; in 28.6%, the reduction was 25% to 50%; in 12.5%, the reduction was less than 25%; and 18.7% had no evidence of tumor size reduction (Table 11-5). The time course of macroprolactinoma shrinkage is highly variable. Some patients experience a dramatic improvement of visual acuity 12 hours after bromocriptine has been introduced, with tumor shrinkage being documented within 1 week. In others, significant tumor reduction is observed only 1 year after medical therapy has been started. In the U.S. multicenter study cited previously,137 19 patients had tumor shrinkage in 6 weeks, but in 8 others, the reduction was not observed until the 6-month imaging reassessment was performed. General data from most series show that rapid shrinkage occurs during the first 6 months in most cases, with slower reduction thereafter, and additional decreases in tumor size observed after 1 year for several years in some cases. Visual improvement generally parallels and often precedes tumor shrinkage unless the optic tract has been chronically and severely damaged. Fig. 11-13 illustrates an unusual case of long-term macroprolactinoma shrinkage, showing that minor tumor reduction, even when not depicted by imaging, is sufficient for visual field improvement. In such situations, the clinical amelioration signified that the primary medical therapy was effective even without clear tumor shrinkage, and pituitary surgery could be postponed or, as it was in this case, needless.
Table 11-5
Comparison of Efficacy of Dopamine Agonists in Effecting Tumor Size Reduction
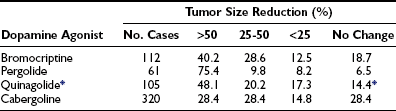
*Not approved in the United States.
Data from Molitch ME: Medical management of prolactin-secreting pituitary adenomas. Pituitary 5:55–65, 2002.
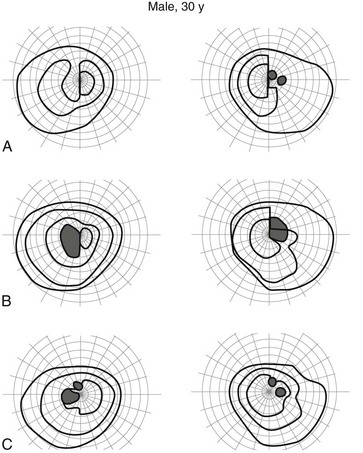
FIGURE 11-13 Visual fields (Goldman perimetry) of a 30-year-old man harboring a macroprolactinoma. Impaired visual fields before treatment (A) showed amelioration 3 days after the beginning of bromocriptine treatment (B) and were almost normalized after 28 days (C) of drug therapy.
Macroprolactinoma reexpansion during successful medical therapy, albeit uncommon, may occur and can lead to visual impairment or headache recurrence.127 Another rare situation is visual deterioration despite initial improvement, in parallel with tumor shrinkage during medical treatment, provoked by chiasm herniation, which may result from traction on the optic chiasm that is pulled down into the now partially empty sella. The two above-mentioned situations generally require surgical intervention, although drug reduction or discontinuation may repair the chiasm herniation.130 In addition, pituitary surgery often is needed for a third rare situation, CSF leakage, occurring in macroprolactinomas invading the sphenoid sinus in cases treated with dopamine agonists (Fig. 11-14). In such cases, the tumor shrinkage behaves as the cork removed from a wine bottle, opening a pathway for CSF flow. CSF leakage, which brings a risk for meningitis, often occurs shortly after the beginning of medical therapy129 but may manifest several months later.128
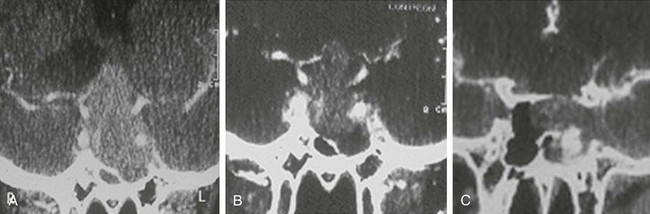
FIGURE 11-14 Computed tomography (coronal views) of a man harboring a macroprolactinoma invading the sphenoid sinus before (A) and 6 days after bromocriptine treatment, showing shrinkage mainly of the sphenoid sinus mass (B), with cerebrospinal fluid leakage confirmed by metrizamide injection (C).
Besides its effect on visual and neurologic symptoms, bromocriptine-induced shrinkage of macroprolactinomas can recover other impaired anterior pituitary functions as a result of the restoration of hypothalamus-pituitary connection, which has been compromised by the tumor-mass effect.92,93 To avoid the maintenance of unnecessary hormonal replacement, pituitary function must be reassessed in patients with macroprolactinoma and previous hypopituitarism successfully treated by dopamine agonists.
Despite the effectiveness of medical therapy for prolactinomas, one of the drawbacks is the need for long-term therapy. Treatment with bromocriptine and other dopamine agonists generally is considered as “symptomatic” because bromocriptine discontinuation leads to recurrence of hyperprolactinemia in most patients and, as previously mentioned, to tumor regrowth, at least after short-term use. Concerning long-term therapy with bromocriptine, a retrospective study from our group showed that 25.8% of 62 patients with microprolactinomas and 15.9% of 69 patients with macroprolactinomas treated with bromocriptine for a median time of 47 months continued to be normoprolactinemic after a median time of 44 months of drug withdrawal.147 There were no statistically significant differences regarding age, gender, bromocriptine initial dose and length of use, tumor size, pregnancy during treatment, and previous pituitary surgery or radiotherapy among patients who continued to be normoprolactinemic and patients who did not. Other reports from the literature point to a percentage of patients who remained normoprolactinemic after bromocriptine interruption ranging from 6.6% to 44%.147 Biswas et al.148 recently reported that 50% of 22 patients with microprolactinomas on bromocriptine therapy for a mean period of 3.1 years persisted normoprolactinemic for more than 1 year after drug withdrawal (Table 11-6).The question regarding why long-term findings differ from short-term findings may be answered by the formerly described microscopic alterations of the lactotrope during bromocriptine administration, suggesting a cytostatic effect related to short-term therapy and a cytocidal effect related to long-term treatment, which could explain the maintenance of normoprolactinemia after drug withdrawal.149
Table 11-6
Overview of Studies on Normoprolactinemia After Bromocriptine (BRC) Withdrawal
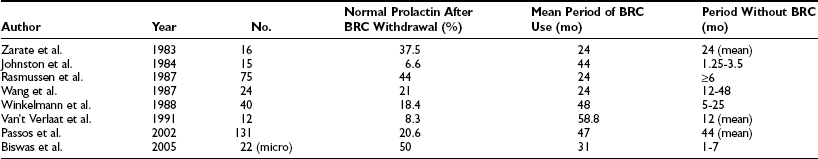
Data from Passos VQ, Souza JJS, Musolino NRC et al: Long-term follow-up of prolactinomas: Normoprolactinemia after bromocriptine withdrawal. J Clin Endocrinol Metab 87:3578–3582, 2002.
Another factor that may influence remission of prolactinomas is their natural history. A study including 25 women with untreated hyperprolactinemia (18 microadenomas, 7 macroadenomas) for a mean period of 11.3 years (mean initial PRL levels 225 ng/mL) showed that 7 of 22 patients with amenorrhea resumed menses spontaneously. Galactorrhea resolved completely in 8 of 19 patients. Only one patient had a slight progression of sellar abnormality. At the reevaluation, mean PRL levels had decreased to 155 ng/mL.150 Another report was concerned with 30 women with hyperprolactinemia (18 with normal pituitary imaging or empty sella) without treatment, followed for an average time of 5.2 years. Of the women, 35% showed improvement in clinical symptoms. Six of 30 women had increased PRL levels, 14 showed no changes, and 10 had a decrease with PRL normalization in 6.151 A study of 41 patients with “idiopathic” hyperprolactinemia followed for 1 year found that 83% of patients had unchanged PRL levels or even showed a decrease, and 34% showed normalization of PRL levels.56
During a mean period of 31 months, 38 patients with microprolactinomas were followed without treatment. Nearly 55% of patients had normalization of PRL levels, and there was no evidence of tumor growth.152 Finally, there is evidence that women with hyperprolactinemia who experience menopause have a significant chance of normalizing their PRL levels, pointing to estrogen influence.153 These data on untreated patients indicate that natural history has an important role in the outcome of prolactinomas and PRL normalization. The mechanisms involved are yet to be clarified, however. Although bromocriptine use has been associated with pituitary apoplexy,154 the evolution of one of our patients who became normoprolactinemic, shown in Fig. 11-15, which shows a bright T1-weighted image without contrast on MRI highly indicative of hemorrhage before medical treatment, suggested that subclinical pituitary apoplexy may play a role in the natural history of prolactinomas and PRL normalization.
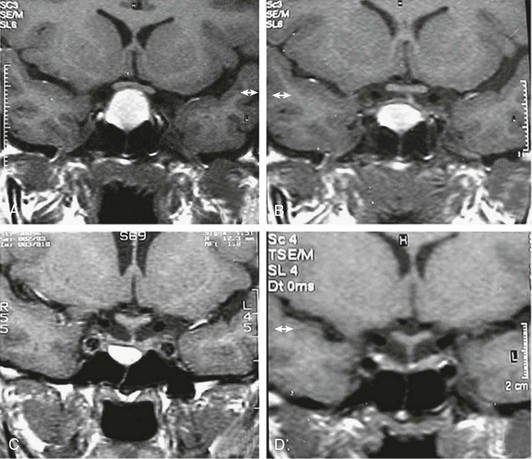
FIGURE 11-15 Magnetic resonance imaging (T1-weighted coronal views without gadolinium enhancement) of a 14-year-old girl harboring a macroadenoma. A, Before treatment (prolactin, 108 ng/mL). B, 2 months on bromocriptine (2.5 mg/day). C, 8 months on bromocriptine (5 mg/day) (prolactin, 18 ng/mL). D, 16 months after bromocriptine withdrawal (prolactin, 13 ng/mL). (From Passos VQ, Souza JJ, Musolino NR et al: Long-term follow-up of prolactinomas: normoprolactinemia after bromocriptine withdrawal. J Clin Endocrinol Metab 87:3578–3582.)
Among the problems associated with medical treatment of prolactinomas, drug side effects are the most prevalent. Bromocriptine is generally well tolerated with doses between 2.5 and 20 mg/day. The most frequent side effects are nausea, vomiting, and orthostatic hypotension and, to a lesser extent, nasal congestion, headache, constipation, and psychotic events, such as auditory hallucinations, delusional ideas, and mood changes. The drug should be given carefully to psychiatric patients.155 There are isolated reports of leukopenia, hepatitis, headaches, and cardiac arrhythmias. Higher doses (20 to 140 mg daily), used for patients with Parkinson’s disease, may lead to pleural effusions, thickening and parenchymal lung changes, and retroperitoneal fibrosis.156
Bromocriptine side effects can be minimized by starting therapy at 1.25 mg at bedtime after food intake and progressively increasing it to include 1.25 mg a day after breakfast and at bedtime, according to individual tolerance, until the therapeutic dose is achieved. When normoprolactinemia is obtained, the dose can be reduced in many cases.157 A subset of patients remain intolerant, however, even when all such recommendations are followed. Our data show persistent intolerance to bromocriptine in 24% and 12% of women harboring microprolactinomas and macroprolactinomas and in 5% of men with macroprolactinomas. Intolerance to bromocriptine may be overcome in some cases with a slow-releasing oral formulation (bromocriptine SRO), which also can be used as a once-a-day formulation.158 We did not find better tolerance of this extended oral form, however, compared with the regular formulation. A single long-lasting injectable form (bromocriptine LA), with 50 mg of the drug, peaking after 2 hours and lasting for 28 days, which was developed for lactation inhibition, was efficacious for prolactinoma treatment, mainly in patients with intracranial hypertension and vomiting.159 Thereafter, an injectable repeatable form was developed (bromocriptine-LAR), with good local and systemic tolerance even in patients who were previously intolerant to the oral formulation.63,160 The injectable forms of bromocriptine are not commercially available, however. Additionally, the use of bromocriptine by the intravaginal route offered better tolerance for some patients, probably as a result of a slower drug absorption, but the absence of a local upper gastrointestinal effect of oral bromocriptine cannot be excluded.161 The administration is bothersome, however, and there are local side effects, such as vaginal irritation. With the development of better-tolerated new oral drugs, the intravaginal administration of bromocriptine for prolactinoma treatment is now seldom employed.
An even more vexing problem in the medical treatment of prolactinomas is their partial or complete lack of responsiveness to bromocriptine. The definition of bromocriptine resistance is arbitrary, with the failure of the drug in normalizing serum PRL levels or in reducing the tumor size using a dose equal to or greater than 15 mg/day for at least 3 months being the most currently accepted. According to this principle, about 10% of prolactinomas are resistant to bromocriptine.162 This criterion has limitations regarding clinical practice, however. Patients who are intolerant to bromocriptine would not attain the dose established as limit, and although they are different conceptually, bromocriptine intolerance and resistance overlap. Patients harboring expanding tumors with visual impairment should not wait for 3 months without an improvement. As previously mentioned, clinical amelioration and tumor reduction may be achieved even if serum PRL is still above the normal range.116 Pragmatically, bromocriptine resistance can be defined as the failure to obtain adequate clinical results in patients using the highest tolerable drug dose.
Mechanisms implicated for bromocriptine resistance of prolactinomas have not yet been fully elucidated. Studies in vivo and in vitro using radiolabeled dopamine antagonists as markers103,163 or assessing D2 receptor expression by RT-PCR164 show that dopamine D2 receptor density is reduced in resistant prolactinomas compared with tumors that are responsive to bromocriptine. When the D2 receptor density in lactotrope cell membranes is extremely low, the dopamine agonist drug paradoxically may lead to tumor growth.163 The paucity of dopamine D2 receptors seems to be the main mechanism of bromocriptine resistance in prolactinomas. Additionally, postreceptor events have been described, including decreased ratio between the short and long isoforms of the dopamine D2 receptor, derived from alternative splicing.165 No mutations in the dopamine D2 receptor that could be ascribed to bromocriptine resistance have been described to date.26
Regarding dopamine agonist resistance and tumor invasiveness, Delgrange et al.166 recently showed that parasellar extension of macroprolactinomas, assessed on the basis of strict MRI criteria, may predict a negative response to DA. Additionally, some prolactinomas that are initially responsive to bromocriptine become resistant during therapy.167 In such situations, especially in aggressive tumors, the diagnosis of the rare PRL-secreting carcinoma must be considered.3
Several other dopamine agonist drugs have been employed for the treatment of hyperprolactinemia/prolactinomas (see Table 11-3). Many show therapeutic efficacy, but three of them in particular merit detailed description, aiming mainly at overcoming intolerance and resistance to bromocriptine.
Pergolide
Pergolide is an ergot derivative, with an estimated potency 100 times that of bromocriptine, that has been approved by the U.S. Food and Drug Administration only for Parkinson’s disease therapy. Considerable data concerning prolactinoma treatment have been collected, however. The drug can be given once daily in a dose ranging from 0.025 to 0.5 mg. When pergolide is compared with bromocriptine, some studies point to similar efficacy and tolerance of both drugs,168,169 whereas others favor pergolide.170,171 Regarding tumor reduction, a compilation of 61 patients with macroprolactinoma shows 75.4% of cases with more than 50% shrinkage and 9.8% of patients with adenoma reduction between 25% and 50% (see Table 11-5).146 Heart valvulopathy occurring in patients taking pergolide for Parkinson’s disease have been reported (see cabergoline).
Quinagolide (CV 205-502)
Quinagolide is a non-ergot dopamine agonist drug that has specific affinity to the dopamine D2 receptor. Its efficacy in normalizing serum PRL levels is similar to that of bromocriptine, ranging from 45% to 100%, with most studies with macroprolactinomas showing PRL normalization between 58% and 75%, paralleled by clinical improvement.172–176 Quinagolide is administered once a day in doses ranging from 0.075 mg to 0.6 mg. Some studies point to better tolerance compared with bromocriptine.175,176 Additionally, there is evidence that about 50% of patients with bromocriptine-resistant prolactinomas attained normoprolactinemia when switched to quinagolide.177,178 Such tumors were partially responsive to bromocriptine, however, with the further decrease attributed to better patient tolerance and a higher affinity to the already reduced but still present dopamine D2 receptors. Prolactinoma patients who are severely resistant to bromocriptine are not expected to respond to quinagolide either. Tumor size reduction evaluated in 105 patients showed more than 50% shrinkage and reduction between 25% and 50% in 48.1% and 20.2% of cases (see Table 11-5).146 Quinagolide is commercially available only in Europe.
Cabergoline
Cabergoline is a synthetic ergoline that shows high specificity and affinity for the dopamine D2 receptor. It is a potent and long-acting inhibitor of PRL secretion, with an elimination half-life ranging between 63 and 109 hours.179 PRL-lowering effects occur rapidly within 3 hours and were evident after a single-dose administration at the end of follow up (21 days) in puerperal women and at 14 days in patients with hyperprolactinemia. This pharmacologic profile allows cabergoline to be administrated once or twice a week in most patients, usually at a weekly dose of 0.5 to 2 mg. In our experience, normoprolactinemia was maintained in some patients taking one tablet (0.5 mg) of cabergoline every 10 days and even every 2 weeks. Many studies have shown the efficacy of cabergoline in normalizing serum PRL levels and reducing tumor size, with consequent improvement of clinical and visual manifestations (Fig. 11-16). In 127 hyperprolactinemic patients (71 microprolactinomas, 19 macroprolactinomas, 37 idiopathic), cabergoline was administered at a dose between 0.25 and 3.5 mg/wk, given once or twice weekly in 114 patients and three times weekly or daily in 13 cases. Serum PRL levels were normalized in 114 patients (90%). Of 56 women with amenorrhea, 52 resumed menses; 17 women became pregnant, and sexual potency was restored in the three men. A total of 48 mild to moderate adverse events were reported by 29 patients (23%).180
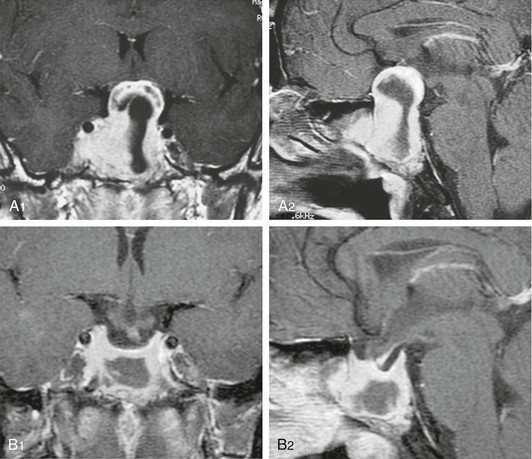
FIGURE 11-16 Magnetic resonance imaging (T1-weighted with gadolinium enhancement) of a man bearing a large macroprolactinoma. A1 (coronal view) and A2 (sagittal view), before treatment. B1 (coronal view) and B2 (sagittal view), after 1 month of cabergoline treatment, showing significant tumor shrinkage that was paralleled by visual improvement.
In a study comprising 37 new patients, cabergoline normalized PRL levels in 88% of 26 microprolactinomas and in 100% of 11 macroprolactinomas. Regular menses were restored in 7 of 10 macroprolactinomas and in all oligomenorrheic patients with microadenoma; serum testosterone levels normalized in 2 of 3 hypogonadal men. Side effects developed in only three cases.181 Another study with cabergoline (0.5 to 3 mg/wk) administered once per week was conducted in 15 patients (8 women) with macroprolactinomas. Normal PRL levels were attained in 73% of cases. Gonadal function was restored in all hypogonadal men and in 75% of premenopausal women with amenorrhea. Side effects were minimal.182 A study addressing long-term cabergoline treatment in men showed that after 24 months of therapy, PRL levels normalized in 31 patients with macroprolactinoma (75.6%) and in eight with microprolactinoma (80%). Galactorrhea disappeared in all patients, and sperm volume and count normalized in all patients who normalized testosterone levels, whereas motility normalized in more than 80%.183
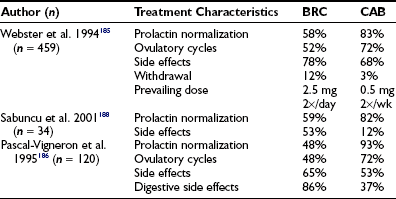
Many studies pointed to better efficacy and tolerance of cabergoline compared with bromocriptine (Table 11-7).184–188 In a recent Brazilian multicenter study, data collected from 388 patients (320 women and 68 men) bearing prolactinomas (220 microadenomas and 68 macroadenomas) showed that cabergoline was significantly more effective than BCR in inducing normoprolactinemia (81.9% vs 67.1%), without any difference according to the gender.184 In a large, multicenter European comparative study encompassing 459 women with hyperprolactinemic amenorrhea, 0.5 to 1 mg of cabergoline twice weekly was more effective than 2.5 to 5 mg of bromocriptine twice daily in the treatment of hyperprolactinemic amenorrhea, restoring ovulatory cycles in 72% of women and normalizing plasma PRL levels in 83%, compared with 52% and 59% for bromocriptine. Adverse effects were recorded in 68% of women taking cabergoline and 78% of women taking bromocriptine, but only 3% discontinued cabergoline, whereas 12% stopped taking bromocriptine because of drug intolerance.185 Regarding cabergoline effect on macroprolactinoma size, in a compilation of 12 series including 320 patients, 28.4% had a greater than 50% tumor shrinkage, 28.4% had a reduction between 25% and 50%, and 43% had less than 25% or no reduction at all (see Table 11-5).146 Because many of these patients had been previously intolerant or resistant to other dopamine agonists, the poorer cabergoline results for macroprolactinoma shrinkage compared with other drugs may have been biased. This bias can be illustrated by a study showing that the prevalence of macroprolactinoma shrinkage greater than 80% after cabergoline treatment was higher in naive patients (92.3%) than in patients who were previously intolerant (42.1%), resistant (30.3%), or responsive (38.4%) to bromocriptine or quinagolide.189 As far as dopamine-agonist resistance in prolactinomas is concerned, some studies indicate that cabergoline normalized serum PRL levels in patients who were resistant to bromocriptine and even to quinagolide. In two series dealing with bromocriptine-treated prolactinomas without full PRL normalization, normoprolactinemia was achieved in 70% and 85% of patients when they were switched to cabergoline.187,190 As previously pointed out (see section on quinagolide), such tumors did respond partially to bromocriptine, however, and the additional decrease brought up by cabergoline is probably related to its better tolerability and higher affinity to the reduced but still present dopamine D2 receptors. Consequently, severely resistant prolactinomas are not expected to respond to any dopamine agonist drug and have to be treated by a different means.
Table 11-7
Treatment of Hyperprolactinemia: Comparison Between Bromocriptine (BRC) and Cabergoline (CAB) Efficacy and Tolerability
Some studies pointed out maintenance of normoprolactinemia in 23% of patients after cabergoline withdrawal for 12 months, on average.180,181 A long-term prospective study including 200 hyperprolactinemic patients showed that after 2 to 5 years after cabergoline withdrawal, normoprolactinemia still persisted in 76% of patients with “nontumoral” hyperprolactinemia, in 70% of patients with microprolactinomas, and in 65% of patients with macroprolactinomas.191 These results suggest that cabergoline compares favorably with bromocriptine also in terms of maintenance of normal PRL levels after drug withdrawal.147 Nevertheless, a recent study dealing only with microprolactinomas did not show difference in remission rates between subjects treated with cabergoline and bromocriptine.148
Recently, two studies pointed to a high prevalence of cardiac valve dysfunction, compared to the general population, in patients treated for Parkinson’s disease with cabergoline and also with pergolide but not with other dopamine agonist drugs.192,193 The supposed mechanism is related to a serotoninergic effect presented by the drugs in addition to dopaminergic agonism. The risk of valvular regurgitation appears to be greatest in patients who have received at least 3 mg of cabergoline a day. Although this dose is much higher than that used for prolactinomas, the cumulative dose derived from the potential long-term use of cabergoline by patients harboring prolactinomas brought concerns about harmful cardiac valve effects of this drug also in this disease.
Three recently published studies addressing this issue did not show increased prevalence of clinically relevant valvular heart disease in patients with hyperprolactinemia/prolactinomas on long-term treatment with cabergoline.194–196 However, additional findings of uncertain clinical relevance, like a higher prevalence of mild regurgitation of the tricuspid valve and of calcifications of the aortic valve, did occur in one study.196 There was no relation between the cumulative dose of cabergoline and the presence of mild, moderate, or severe valve regurgitation.196 Therefore, until more data are available that focus on the safety of cabergoline on the heart during long-term treatment of macroprolactinomas, it is prudent to recommend that all patients starting on this dopamine agonist receive cardiac echography and periodic reassessment during therapy.
Medical therapy seems to be the first option for prolactinoma therapy, being more effective than surgery, especially for macroprolactinomas.197 To date, cabergoline seems to be the first-choice drug for prolactinoma treatment in view of its remarkable tolerance, capacity to normalize serum PRL levels, reduce tumor size, and induce high rates of normoprolactinemia persistence after drug withdrawal.
Radiotherapy
The efficacy of conventional radiation therapy for prolactinomas is lower than medical or surgical treatment. Our results of 19 patients submitted to radiotherapy after medical/surgical failure showed that only 3 (16%) achieved serum PRL normalization 5, 6, and 15 years after the procedure.116 A study with 63 patients treated with radiation after noncurative surgery showed that only 30% had normal PRL levels by 10 years.198 A compilation from the literature shows serum PRL normalization in less than one third of patients 5 to 15 years after conventional radiotherapy, with hypopituitarism occurring in 5.5% to 93.3% of cases.199 This prevalence is probably greater because many series have underestimated GH deficiency in adults. Other complications associated with conventional radiotherapy include optic nerve lesions, neurologic dysfunctions, cerebrovascular disease, and the development of other intracranial tumors.
There are emerging data on gamma-knife radiosurgery for prolactinomas. In one retrospective investigation, the authors examined the results of gamma-knife radiosurgery for tumor remnants after unsuccessful open surgery and medical treatment in 20 patients with prolactinomas. Serum PRL levels decreased into the normal range in five cases (25%). Patients treated with dopamine agonists during gamma-knife radiosurgery did significantly less well compared with the untreated group, suggesting a “radioprotective” effect of the drug.200 This event is also suggested by a recent study201 showing serum PRL normalization in 26% of 23 patients in an average time of 24.5 months, with new pituitary hormone deficiencies in 28% of patients and cranial nerve palsy in two cases (7%). Best results were achieved in patients with tumor volume less than 3.0 cm3 and who are not receiving dopamine agonist at the time of treatment. Another study including 128 patients estimated the efficacy of gamma-knife radiosurgery as the primary therapy for prolactinomas. The mean follow-up time was 33.2 months (range, 6 to 72 months). Tumor control was observed in all but two patients who underwent surgery 18 and 36 months after gamma-knife radiosurgery. Clinical cure was achieved in 67 cases (52%). Nine infertile women became pregnant 2 to 13 months after irradiation, and all gave birth to normal infants. There was no visual deterioration related to gamma-knife radiosurgery. This study points to better results in terms of PRL normalization but does not mention the impact of treatment on pituitary function, besides mentioning five women who experienced “premature menopause.”202 More data and follow-up time are required to assess the superiority of gamma-knife radiosurgery compared with conventional radiotherapy. To date, in the author’s opinion, radiation therapy for prolactinomas is reserved for cases with medical and surgical failures, especially for invasive tumors.
Therapeutic Perspectives
Many studies did not find tumor enlargement in patients with “idiopathic” hyperprolactinemia or microprolactinomas, before or after menopause.153,203 When pregnancy is not a concern, and galactorrhea is not disturbing, sexual steroid replacement can be considered for those patients, who must be followed carefully. Additionally, testosterone may be given to men harboring resistant microprolactinomas and even partially resistant macroprolactinomas, with monitoring of PRL levels and with imaging. The introduction of an aromatase-inhibitor drug may be considered if there is evidence of tumor growth or serum PRL increase.204
Prolactinomas usually do not respond to somatostatin.205 It has been shown, however, that a selective analogue of the somatostatin receptor subtype 5 (SSTR5) inhibited PRL secretion in human prolactinoma cell culture.206 Nevertheless, another in-vitro study did not show better response of this selective analogue compared with quinagolide.207 A recent publication by Fusco et al.208 pointed to an efficacy comparable to cabergoline of a selective analogue of SSTR5, BIM-23206, and the chimeric somatostatin/dopamine D2 receptor ligant, BIM-23A760, in responsive prolactinomas but not in resistant ones. Also the “universal” somatostatin ligand SOM230 (pasireotide) was ineffective in cabergoline-resistant adenomas.208 In summary, although the SSTRs are expressed in prolactinomas, the somatostatinergic ligands do not appear to be highly effective in suppressing PRL in tumors resistant to dopamine agonists.
Studies show that the human PTTG induces angiogenesis via basic fibroblast growth factor induction209 and that estrogen is involved in paracrine regulation of pituitary tumorigenesis by PTTG. Selective estrogen receptor modulators, such as tamoxifen and raloxifene, and inhibitors, such as ICI 182780, abolished estrogen-induced pituitary PTTG expression in vivo, suppressed serum PRL concentrations by 88%, and attenuated PRL-secreting pituitary tumor growth by 41% in rats. Antiestrogen treatment of primary human pituitary tumor cultures reduced PTTG expression approximately 65%.17 These findings may indicate a role for selective antiestrogens in prolactinoma treatment, including resistant ones.
The development of PRL receptor antagonists could also represent an option for medical treatment of resistant macroprolactinomas without mass effect, because they should be able to block the actions of PRL, if not its production.210 One therapeutic approach would be the association of dopamine agonists and PRL receptor–antagonist drugs in cases of macroprolactinomas exhibiting tumor shrinkage but not PRL normalization when on dopamine agonist monotherapy.
The alkylating agent temozolomide has been experimentally used in many tumors. Its product of hydrolyzation, 5-(3-methyltriazen-1-yl) imidazole-4-carboxamide, is thought to exert a cytotoxic effect by DNA alkylation. Temozolomide can be given orally and is currently approved in the United States for the treatment of patients with refractory anaplastic astrocytoma and for the treatment of newly diagnosed patients with glioblastoma multiforme, as an adjunct to radiotherapy. Three case reports recently published describe patients with invasive prolactinoma or PRL-secreting carcinoma resistant to conventional therapy who responded to this drug211–213 (Fig. 11-17). More data is needed to assess the efficacy and tolerability of this promising drug in a larger cohort of aggressive, resistant prolactinomas.
FIGURE 11-17 Left: Serum PRL levels over the course of several treatment modalities for an invasive prolactinoma resistant to dopamine agonists, showing a remarkable decrease when temozolomide was introduced. Right: Magnetic resonance imaging (sagittal views) before (A) and after (B) temozolomide therapy of the patient harboring the invasive prolactinoma. A marked reduction in size is depicted. (From Neff LM, Weil M, Cole A et al: Temozolomide in the treatment of an invasive prolactinoma resistant to dopamine agonists. Pituitary 10:81–86, 2007.)
Finally, there is evidence that nerve growth factor expression is reduced in resistant prolactinomas and that this growth factor restores p53 function in pituitary cell lines.214 Nerve growth factor administration to athymic mice with transplanted human bromocriptine-resistant prolactinomas results in the expression of dopamine D2 receptors in the tumor and restores sensitivity to subsequent treatment with bromocriptine. This could be a promising therapy for patients who are refractory to dopamine-agonist treatment.215
Prolactinomas and Pregnancy
Regarding microprolactinomas, a study including 91 pregnancies mostly induced by bromocriptine, without previous surgery or radiotherapy, indicated symptomatic tumor growth in 5.5% of cases.216 In a compilation of pregnancies in 246 women with microprolactinomas treated with bromocriptine only, tumor growth symptoms were reported in 1.6% of patients, although an asymptomatic increase of the tumor was shown in 4.5% of the cases. None of the patients needed surgical intervention during pregnancy.217 We followed 71 term pregnancies, and the results were similar.218,219 Of the 22 patients with previous surgery, none presented symptoms of tumor growth; of the 41 pregnant patients who underwent treatment with bromocriptine alone, only 1 (2.4%) presented with headaches in the third month of pregnancy, which regressed with drug reintroduction. Seven patients got pregnant without treatment and did not develop any complications. There was an asymptomatic increase of the tumor in one case with previous surgery and in two cases with bromocriptine alone, as assessed by scanning postpartum. Because of the low risk of tumor growth during pregnancy in patients with microprolactinomas, there is no need to perform periodic imaging or ophthalmologic examinations. These assessments should be reserved for cases with clinical complaints suggesting tumor growth, such as headache or visual field changes.
The risk of complications during pregnancy is much greater with macroprolactinomas. One study revealed symptomatic tumor growth in 41.3% of 56 pregnancies occurring in 46 patients who were medically treated only, compared with 7.1% of 70 pregnancies that occurred in 67 women who previously had been submitted to surgery or radiotherapy.216 A review from the literature pointed to symptoms related to tumor growth in 15.5% of 45 patients with macroprolactinomas treated with bromocriptine only. The incidence of complications was only 4.3% in the 46 patients who underwent surgery or radiotherapy before pregnancy. Additionally, asymptomatic tumor growth was seen in 8.9% of the patients without prior surgery or radiotherapy.217 We followed 51 term pregnancies in patients with macroprolactinomas.218,219 Of those, 21 were in patients with previous surgery, and none of the patients presented with symptoms or signs of tumor growth. Of the 30 patients treated with pregestational bromocriptine only, 11 (37%) manifested complaints related to tumor growth: all of them presented with headaches, and seven had visual alterations (Fig. 11-18). Pituitary imaging after delivery was performed in 23 other patients, and an asymptomatic growth of the tumor was observed in 4 more cases.
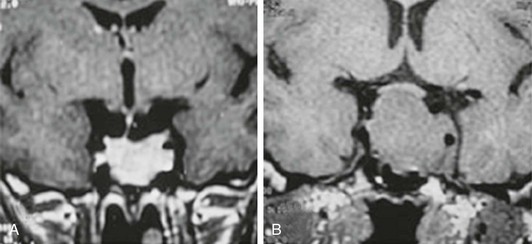
FIGURE 11-18 Magnetic resonance imaging (coronal views) of a patient with macroprolactinoma. A, Tumor is limited to sellar boundaries during bromocriptine treatment, before pregnancy. B, Tumor growth during the 4th month of pregnancy without bromocriptine use. (From Musolino NRC, Bronstein MD: Prolactinomas and pregnancy. In Bronstein MD [ed]: Pituitary Tumors in Pregnancy. Boston: Kluwer Academic Publishers, 2001, pp 91–108.)
These data show the higher risk of tumor growth in macroprolactinomas during pregnancy, necessitating a stricter follow-up (Fig. 11-19). The first recommendation should be the use of a nonhormonal contraceptive along with a dopamine agonist until tumor shrinkage has been shown within sellar boundaries. The duration of previous treatment with dopamine also might be important. A follow-up study of 37 pregnancies showed signs of tumor growth in 7, all of them treated with bromocriptine for less than 1 year. No tumor enlargement was found in the 14 macroprolactinoma patients treated for a longer time, suggesting that the duration of bromocriptine use before conception might be a good prognostic factor in pregnancy.220
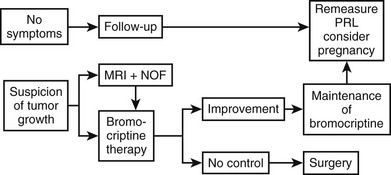
FIGURE 11-19 Suggested algorithm for the follow-up of patients with macroprolactinoma during pregnancy. NOF, Neuroophthalmologic examination; PRL, prolactin. (From Musolino NRC, Bronstein MD: Prolactinomas and pregnancy. In Bronstein MD [ed]: Pituitary Tumors in Pregnancy. Boston: Kluwer Academic Publishers, 2001, pp 91–108.)
After pregnancy has been confirmed, the dopamine agonist can be withdrawn, and the patient must be monitored closely for symptoms related to tumor growth. If there is a suspected tumor expansion, the confirmation can be made through MRI, after the fourth month of gestation, and by visual field testing (see Fig. 11-19). Monitoring serum PRL levels during pregnancy does not seem to be useful, because they are not always related to tumor behavior during gestation. The reintroduction of bromocriptine in such cases often leads to clinical amelioration and tumor reduction. In 9 of 11 patients who exhibited complications during pregnancy, bromocriptine reintroduction brought complete resolution of the symptoms related to tumor growth.219 Surgery also can be employed as treatment for symptomatic tumor growth in pregnancy. Several authors have reported good results, although the increased risk of spontaneous abortion in patients who undergo surgery is well known.219
The safety of bromocriptine reintroduction or even maintenance during pregnancy is supported by a large experience with this dopamine agonist reported in the literature. A large review221 consisted of 2587 pregnancies and did not show an increase of maternal or fetal morbidity or mortality. Since then, some authors have favored the maintenance of bromocriptine in pregnancy to prevent complications in patients with macroprolactinomas without previous surgery or radiotherapy.222 It is our policy to indicate such an approach only when the patient gets pregnant after a short treatment period, mainly without confirmation of tumor shrinkage or when the tumor is outside sellar boundaries. Surgery is indicated before pregnancy in cases without tumor reduction during treatment with dopamine agonists or in patients who developed tumor growth in previous gestations.219
Pregnancy and Other Dopamine Agonist Drugs
In recent years, the use of new dopamine agonists such as quinagolide, cabergoline, and pergolide for the treatment of hyperprolactinemia has increased, and pregnancies have been described. Data were obtained on 176 pregnancies in which quinagolide was used, on average, for 37 days. Miscarriages occurred in 14% of the cases, with one ectopic pregnancy. Fetal malformation was described in nine cases, although other drugs had been used in three patients.223 Quinagolide was used successfully during pregnancy in two bromocriptine-resistant patients who presented symptoms of tumor growth.178 Cabergoline has been the most used medication among the more recent dopamine agonist drugs, and reports on pregnancies during cabergoline therapy are emerging. We followed six full-term gestations in patients who withdrew cabergoline as soon as pregnancy was confirmed and did not observe malformations, but two premature births occurred.219 A review from 1996, with 204 gestations, did not observe an increase of spontaneous abortion (12%) or malformations (four cases).224 A recent multicenter study encompassing 329 pregnancies in hyperprolactinemic women also suggests that fetal exposure to cabergoline through early pregnancy does not induce any increase in the risk of miscarriage or fetal malformation.225 Nevertheless, the drug’s long action, which persists 3 weeks after its withdrawal, associated with fewer data compared with bromocriptine (around 700 versus >6000 pregnancies) (Table 11-8), still limits (albeit not so strictly) its indication for patients who wish to conceive or its use during pregnancy. Regarding pergolide, animal data point to its safety in pregnancy.226 No human data are available to date.
Table 11-8
Effect of Bromocriptine on Pregnancies
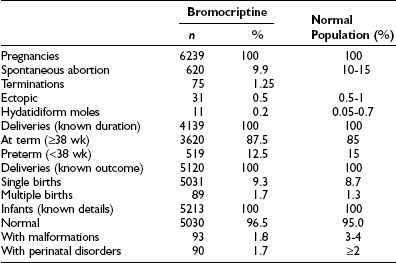
Data from Krupp P, Monka C, Richter K: Program of the Second World Congress of Gynecology and Obstetrics, Rio de Janeiro, 1988, p 9.
Follow-Up After Delivery
Breastfeeding does not increase the risk of tumor growth in patients who progressed well during pregnancy.219 Breastfeeding is contraindicated only when patients need to maintain the dopamine agonist after delivery, owing to tumor-growth signs.
There have been several reports in the literature regarding reduction or normalization of serum PRL levels after delivery.219,227 In our hands, 60% and 72% of patients with microprolactinomas and macroprolactinomas showed a decrease in PRL levels after delivery compared with pregestational levels. In 11% of all patients who conceived, PRL levels normalized after pregnancy, some with a new gestation without therapy. On average, PRL levels decreased from 336 ± 105 ng/mL to 133 ± 20 ng/mL in 62 patients who were available for comparison.218 These results are similar to ones reported by other authors. A study reported PRL normalization after pregnancy in 29% of women.227 Tumor reduction after pregnancy also has been described. A study with 16 patients harboring prolactinomas found 27% of tumor reduction or disappearance after delivery.228 We also observed tumor reduction in 8 of 23 patients with macroprolactinomas, assessed by imaging before and after delivery. Two other patients developed asymptomatic apoplexy. The explanation for this “curative” effect of pregnancy is to be clarified. It may be related partly to modifications in the vasculature of the adenoma due to the estrogen stimulation, resulting in necrosis or microinfarctions of the adenomatous tissue. Hemorrhagic zones in prolactinoma patients receiving estrogen therapy already have been described.229
Another issue of concern is the outcome of the children whose mothers took dopamine agonist drugs during pregnancy. One study reported the follow-up spanning 4 months to 9 years in 546 children exposed to intrauterine bromocriptine.221 The authors did not find any developmental impairment in the children. We followed 70 children born to mothers who conceived on bromocriptine.219 At a mean follow-up of 67 months (range, 12 to 240 months), only two children presented with disorders of neuropsychomotor development: one case of idiopathic hydrocephaly and another of tuberous sclerosis. We did not find any similar reports in the literature. Fifteen of these children already had started puberty, one of them precociously.
Treatment Planning and Follow-Up
Based on the previously described evidence, to date the gold-standard therapy for either microprolactinomas or macroprolactinomas is medical treatment with dopamine agonist drugs (Fig. 11-20). Physicians must motivate patients to embark on such long-term treatment based on the overall better results compared with surgery and, mainly based on more recent data,147,191 the possibility of drug withdrawal for a substantial number of patients with maintenance of normoprolactinemia. Surgical treatment of prolactinomas is indicated for patients with persistent intolerance or hormonal or tumor resistance to more than one dopamine agonist drug—in particular if pregnancy is desired—or if the tumor has grown during medical treatment. In cases of resistance, rare conditions, such as acidophil stem cell adenoma or PRL-secreting carcinomas, must be considered. Malignant prolactinomas respond only temporarily, if they respond at all, when patients are switched to another dopamine agonist drug or undergo surgery. Surgical therapy also is indicated frequently in pituitary apoplexy, in dopamine agonist–induced CSF leakage occurring in invasive macroprolactinomas, and in the exceedingly rare occurrence of visual loss secondary to optic chiasm herniation during medical therapy. Additionally, surgery in skilled hands may be considered for patients not willing to be submitted to long-term medical therapy, especially patients harboring microprolactinomas and serum PRL levels less than 100 ng/mL. Radiotherapy, either conventional or stereotactic, is reserved for prolactinomas not responsive to medical or surgical treatment, particularly regarding the invasive ones.
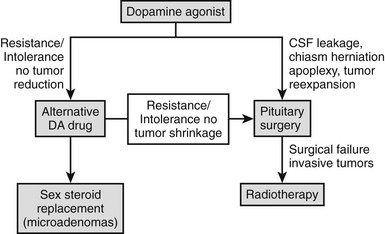
FIGURE 11-20 Suggested algorithm for treatment of prolactinomas. CSF, Cerebrospinal fluid; DA, dopamine.
There is evidence that, in general, estrogen replacement is not harmful for women harboring microprolactinomas. This approach may be used when fertility is not an issue for patients with intolerance or resistance to medical therapy and not cured by or not willing to undergo pituitary surgery. In addition, women successfully treated medically may use hormonal contraceptives if they have not adapted to barrier methods. Menopausal women bearing microprolactinomas can interrupt dopamine agonist drug use and are allowed to start hormone replacement therapy if it is indicated.203 Although there is evidence of a PRL role in carcinogenesis in animal models, mainly carcinoma of the mammary gland, human data are still highly controversial.230–233 It is now apparent that human mammary epithelial cells can synthesize PRL endogenously, permitting autocrine/paracrine actions within the mammary gland that are independent of pituitary PRL and probably not affected by dopamine agonist drugs. To date, the maintenance of high serum PRL levels in premenopausal and postmenopausal women is not a concern with regard to carcinogenesis. Finally, for macroprolactinomas that are not adequately controlled, the use of estrogens is generally discouraged, given their potential for inducing growth.
References
1. Miyai, K, Ichibara, K, Kondo, L, et al. Asymptomatic hyperprolactinemia and prolactinoma in the general population mass screening by paired assays of serum prolactin. Clin Endocrinol (Oxf). 1986;25:549–554.
2. Hardy, J. Transsphenoidal microsurgery of the normal and pathological pituitary. Clin Neurosurg. 1969;16:185–217.
3. Kars, M, Roelfsema, F, Romijn, JA, et al. Malignant prolactinoma: case report and review of the literature. Eur J Endocrinol. 2006;155:523–534.
4. Glezer, A, D’Alva, CB, Salgado, LR, et al. Pitfalls in pituitary diagnosis: peculiarities of three cases. Clin Endocrinol (Oxf). 2002;57:135–139.
5. Striker, P, Grueter, F. Action du lobe antérieur de l’hypophyse sur la montée laiteuse. C R Soc Biol Paris. 1929;99:1978–1980.
6. Riddle, O, Bartes, RW, Dykshorn, DW. The preparation, identification and assay of prolactin—a hormone of the anterior pituitary. Am J Physiol. 1933;105:191–216.
7. Mendel, EB. Chiari-Frommel syndrome. Am J Obstet Gynecol. 1946;51:889–892.
8. Argonz, J, del Castillo, EB. A syndrome characterized by estrogenic insufficiency, galactorrhea and decreased urinary gonadotropin. J Clin Endocrinol Metab. 1953;13:79–87.
9. Forbes, AP, Henneman, PH, Griswold, GC, et al. Syndrome characterized by galactorrhea, amenorrhea and low urinary FSH: comparison with acromegaly and normal lactation. J Clin Endocrinol Metab. 1954;14:264–271.
10. Hwang, P, Guyda, H, Friesen, H. A radioimmunoassay for human prolactin. Proc Natl Acad Sci U S A. 1971;68:1902–1906.
11. Burrow, GN, Wortzman, G, Rewcastle, NB, et al. Microadenomas of the pituitary and abnormal sellar tomograms in an unselected autopsy series. N Engl J Med. 1981;304:156–158.
12. Ciccarelli, A, Daly, AF, Beckers, A. The epidemiology of prolactinomas. Pituitary. 2005;8:3–6.
13. Berezin, M, Karasik, A. Familial prolactinoma. Clin Endocrinol (Oxf). 1995;42:483–486.
14. Herman, V, Fagin, J, Gonsky, R, et al. Clonal origin of pituitary adenomas. J Clin Endocrinol Metab. 1990;71:1427–1433.
15. Zhang, X, Horwitz, GA, Heaney, AP, et al. Pituitary tumor transforming gene (PTTG) expression in pituitary adenomas. J Clin Endocrinol Metab. 1999;84:761–767.
16. Shimon, I, Hinton, DR, Weiss, MH, et al. Prolactinomas express human heparin-binding secretory transforming gene (HST) protein product: marker of tumour invasiveness. Clin Endocrinol (Oxf). 1998;48:23–29.
17. Heaney, AP, Fernando, M, Melmed, S. Functional role of estrogen in pituitary tumor pathogenesis. J Clin Invest. 2002;109:277–283.
18. Ben-Shlomo, A, Miklovsky, I, Ren, SG, et al. Leukemia inhibitory factor regulates prolactin secretion in prolactinoma and lactotroph cells. J Clin Endocrinol Metab. 2003;88:858–863.
19. Fiorentini, C, Guerra, N, Facchetti, M, et al. Nerve growth factor regulates dopamine D(2) receptor expression in prolactinoma cell lines via p75(NGFR)-mediated activation of nuclear factor-kappaB. Mol Endocrinol. 2002;16:353–366.
20. Paez-Pereda, M, Giacomini, D, Refojo, D, et al. Involvement of bone morphogenetic protein 4 (BMP-4) in pituitary prolactinoma pathogenesis through a Smad/estrogen receptor crosstalk. Proc Natl Acad Sci U S A. 2003;100:1034–1039.
21. Finelli, P, Pierantoni, GM, Giardino, D, et al. The high-mobility group A2 gene is amplified and overexpressed in human prolactinomas. Cancer Res. 2002;62:2398–2405.
22. Turner, HE, Harris, AL, Melmed, S, et al. Angiogenesis in endocrine tumors. Endocr Rev. 2003;24:600–632.
23. Sarkar, DK, Gottschall, PE, Meites, J. Damage to hypothalamic dopaminergic neurons is associated with development of prolactin-secreting pituitary tumors. Science. 1982;218:684–686.
24. Schuff, KG, Hentges, ST, Kelly, MA, et al. Lack of prolactin receptor signaling in mice results in lactotroph proliferation and prolactinomas by dopamine-dependent and -independent mechanisms. J Clin Invest. 2002;110:973–981.
25. Cruz-Soto, ME, Scheiber, MD, Gregerson, KA, et al. Pituitary tumorigenesis in prolactin gene-disrupted mice. Endocrinology. 2002;143:4429–4436.
26. Friedman, E, Adams, EF, Hoog, A, et al. Normal structural dopamine type 2 receptor gene in prolactin-secreting and other pituitary tumors. J Clin Endocrinol Metab. 1994;78:568–574.
27. Prezant, TR, Levine, J, Melmed, S. Molecular characterization of the MEN1 tumor suppressor gene in sporadic pituitary tumors. J Clin Endocrinol Metab. 1998;83:1388–1391.
28. Daly, AF, Vanbellinghen, JF, Khoo, SK, et al. Aryl hydrocarbon receptor-interacting protein gene mutations in familial isolated pituitary adenomas: analysis in 73 families. J Clin Endocrinol Metab. 2007;92:1891–1896.
29. Yu, R, Bonert, V, Saporta, I, Raffel, LJ, et al. Aryl hydrocarbon receptor interacting protein variants in sporadic pituitary adenomas. J Clin Endocrinol Metab. 2006;91:5126–5129.
30. Hardy, J. Transsphenoidal surgery of hypersecreting pituitary tumors. In: Kolhler G, Ross GT, eds. Diagnosis and Treatment of Pituitary Tumors. New York: Elsevier; 1973:179–194.
31. Scheithauer, BW, Kovacs, KT, Laws, ER, Jr., et al. Pathology of invasive pituitary tumors with special reference to functional classification. J Neurosurg. 1986;65:733–744.
32. Horvath, E, Kovacs, K. Pathology of prolactin cell adenomas of the human pituitary. Semin Diagn Pathol. 1986;3:4–17.
33. Bevan, JS, Burke, CW, Esiri, MM, et al. Misinterpretation of prolactin levels leading to management errors in patients with sellar enlargement. Am J Med. 1987;82:29–32.
34. Horvath, E, Kovacs, K, Singer, W, et al. Acidophil stem cell adenoma of the human pituitary: clinicopathologic analysis of 15 cases. Cancer. 1981;47:761–771.
35. Ozbey, N, Sariyildiz, E, Yilmaz, L, et al. Primary hypothyroidism with hyperprolactinaemia and pituitary enlargement mimicking a pituitary macroadenoma. Int J Clin Pract. 1997;51:409–411.
36. Bellastella, A, Bizzarro, A, Coronella, C, et al. Lymphocytic hypophysitis: a rare or underestimated disease? Eur J Endocrinol. 2003;149:363–376.
37. Bihan, H, Christozova, V, Dumas, JL, et al. Sarcoidosis: clinical, hormonal, and magnetic resonance imaging (MRI) manifestations of hypothalamic-pituitary disease in 9 patients and review of the literature. Medicine (Baltimore). 2007;86:259–268.
38. Gonzalez, JG, Elizondo, G, Saldriar, D, et al. Pituitary gland growth during normal pregnancy: an in-vivo study using magnetic resonance imaging. Am J Med. 1988;85:217–220.
39. Scheithauer, BW, Sano, T, Kovacs, KT, et al. The pituitary gland in pregnancy: a clinic pathologic and immunohistochemical study of 69 cases. Mayo Clin Proc. 1990;65:461–474.
40. Morris, LF, Braunstein, GD. Impact of pregnancy on normal pituitary function. In: Bronstein MD, ed. Pituitary Tumors in Pregnancy. Boston: Kluwer Academic Publishers; 2001:1–32.
41. Vieira, JG, Oliveira, JH, Tachibana, T, et al. Evaluation of plasma prolactin levels: is it necessary to rest before the collection? Arq Bras Endocrinol Metabol. 2006;50:569–702.
42. Molitch, ME. Drugs and prolactin. Pituitary. 2008;11:209–218.
43. Kelley, SR, Kamal, TJ, Molitch, ME. Mechanism of verapamil calcium channel blockade-induced hyperprolactinemia. Am J Physiol. 1996;270:96–100.
44. Hutchinson, J, Murphy, M, Harries, R, et al. Galactorrhoea and hyperprolactinaemia associated with protease inhibitors. Lancet. 2000;356:1003–1004.
45. Drange, MR, Fram, NR, Herman-Bonert, V, et al. Pituitary tumor registry: a novel clinical resource. J Clin Endocrinol Metab. 2000;85:168–174.
46. Karavitaki, N, Thanabalasingham, G, Shore, HC, et al. Do the limits of serum prolactin in disconnection hyperprolactinaemia need re-definition? A study of 226 patients with histologically verified non-functioning pituitary macroadenoma. Clin Endocrinol (Oxf). 2006;65:524–529.
47. Glezer, A, Paraiba, DB, Bronstein, MD. Rare sellar lesions. Endocrinol Metab Clin North Am. 2008;37:195–211.
48. Greenman, Y. Dopaminergic treatment of nonfunctioning pituitary adenomas. Nat Clin Pract Endocrinol Metab. 2007;3:554–555.
49. Barbetta, L, Dall’Asta, C, Ambrosi, B. Hyperprolactinemia preceding Cushing’s disease. J Endocrinol Invest. 2000;23:491–492.
50. Stryker, TD, Molitch, ME. Reversible hyperthyrotropinemia, hyperthyroxinemia, and hyperprolactinemia due to adrenal insufficiency. Am J Med. 1985;79:271–276.
51. Estopinan Garcia, V, Martinez Burgui, JA, Ballester Ferrer, A, et al. Prolactin and polycystic ovary síndrome. Med Clin (Barc). 2001;116:759.
52. Saha, MT, Saha, HH, Niskanen, LK, et al. Time course of serum prolactin and sex hormones following successful renal transplantation. Nephron. 2002;92:735–737.
53. Zietz, B, Lock, G, Plach, B, et al. Dysfunction of the hypothalamic-pituitary-glandular axes and relation to Child-Pugh classification in male patients with alcoholic and virus-related cirrhosis. Eur J Gastroenterol Hepatol. 2003;15:495–501.
54. Leung, AK, Pacaud, D. Diagnosis and management of galactorrhea. Am Fam Physician. 2004;70:543–550.
55. Molitch, ME, Schwartz, S, Mukherji, B. Is prolactin secreted ectopically? Am J Med. 1981;70:803–807.
56. Martin, TL, Kim, M, Malarkey, WB. The natural history of idiopathic hyperprolactinemia. J Clin Endocrinol Metab. 1985;60:855–858.
57. De Bellis, A, Colao, A, Pivonello, R, et al. Antipituitary antibodies in idiopathic hyperprolactinemic patients. Ann N Y Acad Sc. 2007;1107:129–135.
58. Mah, PM, Webster, J. Hyperprolactinemia: etiology, diagnosis, and management. Semin Reprod Med. 2002;20:365–374.
59. Abe, T, Matsumoto, K, Kuwazawa, J, et al. Headache associated with pituitary adenomas. Headache. 1998;38:782–786.
60. Matharu, MS, Levy, MJ, Merry, RT, et al. SUNCT syndrome secondary to prolactinoma. J Neurol Neurosurg Psychiatry. 2003;74:1590–1592.
61. Zikel, OM, Atkinson, JL, Hurley, DL. Prolactinoma manifesting with symptomatic hydrocephalus. Mayo Clin Proc. 1999;74:475–477.
62. Minniti, G, Jaffrain-Rea, ML, Santoro, A, et al. Giant prolactinomas presenting as skull-base tumors. Surg Neurol. 2002;57:99–103.
63. Bronstein, MD, Musolino, NR, Cardim, CS, et al. Treatment of macroprolactinomas with a long-acting, parenteral and repeatable new form of bromocriptine. In: Landolt AM, Heitz PU, Zapf J, et al, eds. Advances in Pituitary Adenoma Research. Oxford: Pergamon Press, 1988.
64. Franks, S, Murray, MA, Jequier, AM, et al. Incidence and significance of hyperprolactinaemia in women with amenorrhea. Clin Endocrinol (Oxf). 1975;4:597–607.
65. Hattori, N. Macroprolactinemia: a new cause of hyperprolactinemia. J Pharmacol Sci. 2003;92:171–177.
66. Serri, O, Chik, CL, Ur, E, Ezzat, S. Diagnosis and management of hyperprolactinemia. Can Med Assoc J. 2003;169:575–581.
67. Kars, M, van der Klaauw, AA, Onstein, CS, et al. Quality of life is decreased in female patients treated for microprolactinoma. Eur J Endocrinol. 2007;157:133–139.
68. Vartej, P, Poiana, C, Vartej, I. Effects of hyperprolactinemia on osteoporotic fracture risk in premenopausal women. Gynecol Endocrinol. 2001;15:3–7.
69. Ciccarelli, E, Savino, L, Carlevatto, V. Vertebral bone density in non-amenorrhoeic hyperprolactinaemic women. Clin Endocrinol (Oxf). 1988;28:1–6.
70. Bronstein, MD, Marino, R, Jr., Pereira, DHM. Therapeutic alternatives for hyperprolactinemia: the role of bromoergocriptine. Rev Bras Ginecol Obstet. 1983;5:193.
71. Kleinberg, DL, Noel, GL, Frantz, AG. Galactorrhea: a study of 235 cases, including 48 with pituitary tumors. N Engl J Med. 1977;296:589–600.
72. Asano, S, Ueki, K, Suzuki, I, et al. Clinical features and medical treatment of male prolactinomas. Acta Neurochir (Wien). 2001;143:465–470.
73. Bronstein, MD. Prolactinoma in men. Arq Bras Endocrinol Metab. 1999;43:338.
74. Drago, F, Scapagnini, U. Side effects of drugs stimulating prolactin secretion on the behavior of male rats. Arch Int Pharmacodyn Ther. 1985;276:271–278.
75. Molitch, ME, Thorner, MO, Wilson, C. Management of prolactinomas. J Clin Endocrinol Metab. 1997;82:996–1000.
76. Naliato, EC, Farias, ML, Braucks, GR, et al. Prevalence of osteopenia in men with prolactinoma. J Endocrinol Invest. 2005;28:12–17.
77. Cannavo, S, Venturino, M, Curto, L, et al. Clinical presentation and outcome of pituitary adenomas in teenagers. Clin Endocrinol (Oxf). 2003;58:519–527.
78. Colao, A, Loche, S, Cappa, M, et al. Prolactinomas in children and adolescents: clinical presentation and long-term follow-up. J Clin Endocrinol Metab. 1998;83:2777–2780.
79. Jallad, RS, Goic, MSZ, Musolino, NR, et al. Prolactinomas in children and adolescents: retrospective study on 47 patients. Arq Bras Endocrinol Metabol. 2000;44:300.
80. Di Sarno, A, Rota, F, Auriemma, R, et al. An evaluation of patients with hyperprolactinemia: have dynamic tests had their day? J Endocrinol Invest. 2003;26:39–47.
81. Frieze, TW, Mong, DP, Koops, MK. “Hook effect” in prolactinomas: case report and review of literature. Endocr Pract. 2003;8:296–303.
82. Sinha, YN. Structural variants of prolactin: occurrence and physiological significance. Endocr Rev. 1995;16:354–369.
83. Jackson, RD, Wortsman, J, Malarkey, WB. Macroprolactinemia presenting like a pituitary tumor. Am J Med. 1985;78:346–350.
84. Tritos, NA, Guay, AT, Malarkey, WB. Asymptomatic “big” hyperprolactinemia in two men with pituitary adenomas. Eur J Endocrinol. 1998;138:82–85.
85. Glezer A, Vieira JG, Giannella-Neto D, et al: Clinical asymptomatic hyperprolactinemia is not always linked to macroprolactinemia: peculiarities of two cases. ENDO Society 85th Annual Meeting, Philadelphia, 2003, P3-649, p 630.
86. Vallette-Kasic, S, Morange-Ramos, I, Selim, A, et al. Macroprolactinemia revisited: a study on 106 patients. J Clin Endocrinol Metab. 2002;87:581–588.
87. Vieira, JG, Tachibana, TT, Obara, LH, et al. Extensive experience and validation of polyethylene glycol precipitation as a screening method for macroprolactinemia. Clin Chem. 1998;44:1758–1759.
88. Cavaco, B, Leite, V, Santos, MA, et al. Some forms of big-big prolactin behave as a complex of monomeric prolactin with an immunoglobulin G in patients with macroprolactinemia or prolactinoma. J Clin Endocrinol Metab. 1995;80:2342–2346.
89. Hattori, N, Nakayama, Y, Kitagawa, K, et al. Anti-prolactin (PRL) autoantibodies suppress PRL bioactivity in patients with macroprolactinaemia. Clin Endocrinol (Oxf). 2008;68:72–76.
90. Glezer, A, Soares, CR, Vieira, JG, et al. Human macroprolactin displays low biological activity via its homologous receptor in a new sensitive bioassay. J Clin Endocrinol Metab. 2006;91:1048–1055.
91. Andersen, M, Hagen, C, Frystyk, J, et al. Development of acromegaly in patients with prolactinomas. Eur J Endocrinol. 2003;149:17–22.
92. George, LD, Nicolau, N, Scanlon, MF, et al. Recovery of growth hormone secretion following cabergoline treatment of macroprolactinomas. Clin Endocrinol (Oxf). 2000;53:595–599.
93. Sibal, L, Ugwu, P, Kendall-Taylor, P, et al. Medical therapy of macroprolactinomas in males: I. Prevalence of hypopituitarism at diagnosis. II. Proportion of cases exhibiting recovery of pituitary function. Pituitary. 2002;5:243–246.
94. Rennert, J, Doerfler, A. Imaging of sellar and parasellar lesions. Clin Neurol Neurosurg. 2007;109:111–124.
95. Buchfelder, M, Nistor, R, Fahlbusch, R, et al. The accuracy of CT and MR evaluation of the sella turcica for detection of adrenocorticotropic hormone-secreting adenomas in Cushing disease. Am J Neuroradiol. 1993;14:1183–1190.
96. Patronas, N, Bulakbasi, N, Stratakis, CA, et al. Spoiled gradient recalled acquisition in the steady state technique is superior to conventional postcontrast spin echo technique for magnetic resonance imaging detection of adrenocorticotropin-secreting pituitary tumors. J Clin Endocrinol Metab. 2003;88:1565–1569.
97. Aron, DC, Howlett, TA. Pituitary incidentalomas. Endocrinol Metab Clin North Am. 2000;29:205–221.
98. Molitch, ME. Pituitary incidentalomas. Endocrinol Metab Clin North Am. 1997;26:725–740.
99. Hall, WA, Luciano, MG, Doppman, JL, et al. Pituitary magnetic resonance imaging in normal human volunteers: occult adenomas in the general population. Ann Intern Med. 1994;120:817–820.
100. Chanson, P, Daujat, F, Young, J, et al. Normal pituitary hypertrophy as a frequent cause of pituitary incidentaloma: a follow-up study. J Clin Endocrinol Metab. 2001;86:3009–3015.
101. Dinç, H, Esen, F, Demirci, A, et al. Pituitary dimensions and volume measurements in pregnancy and post partum. MR assessment. Acta Radiol. 1998;39:64–69.
102. Krishnan, KRR, Doraiswamy, PM, Lurie, SN, et al. Pituitary size in depression. J Clin Endocrinol Metab. 1991;72:256–259.
103. Scillitani, A, Dicembrino, F, Di Fazio, P, et al. In-vivo visualization of pituitary dopaminergic receptors by 123iodine methoxybenzamide (IBZM) correlates with sensitivity to dopamine agonists in two patients with macroprolactinomas. J Clin Endocrinol Metab. 1995;80:2523–2525.
104. de Herder, WW, Reijs, AE, de Swart, J, et al. Comparison of iodine-123 epidepride and iodine-123 IBZM for dopamine D2 receptor imaging in clinically non-functioning pituitary macroadenomas and macroprolactinomas. Eur J Nucl Med. 1999;26:46–50.
105. Muhr, C. Positron emission tomography in acromegaly and other pituitary adenoma patients. Neuroendocrinology. 2006;83:205–210.
106. Petrossians, P, de Herder, W, Kwekkeboom, D, et al. Malignant prolactinoma discovered by D2 receptor imaging. J Clin Endocrinol Metab. 2000;85:398–401.
107. Fluckiger, E, Wagner, HR. 2-Br-alpha-ergokryptin: influence on fertility and lactation in the rat. Experientia. 1968;24:1130–1131.
108. Grosvenor, AE, Laws, ER. The evolution of extracranial approaches to the pituitary and anterior skull base. Pituitary. 2008;11(4):337–345.
109. Cho, DY, Liau, WR. Comparison of endonasal endoscopic surgery and sublabial microsurgery for prolactinomas. Surg Neurol. 2002;58:371–375.
110. Prevedello, DM, Doglietto, F, Jane, JA, Jr., et al. History of endoscopic skull base surgery: its evolution and current reality. J Neurosurg. 2007;107:206–213.
111. Ciric, I, Ragin, A, Baumgartner, C, et al. Complications of transsphenoidal surgery: results of a national survey, review of the literature, and personal experience. Neurosurgery. 1997;40:225–236.
112. Chandler, WF, Barkan, AL. Treatment of pituitary tumors: a surgical perspective. Endocrinol Metab Clin North Am. 2008;37:51–66.
113. Tyrrell, JB, Lamborn, KR, Hannegan, LT, et al. Transsphenoidal microsurgical therapy of prolactinomas: initial outcomes and long-term results. Neurosurgery. 1999;44:254–261.
114. Losa, M, Mortini, P, Barzaghi, R, et al. Surgical treatment of prolactin-secreting pituitary adenomas: early results and long-term outcome. J Clin Endocrinol Metab. 2002;87:3180–3186.
115. Gillam, MP, Molitch, ME, Lombardi, G, et al. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27:485–534.
116. Bronstein, M, Musolino, N, Cunha-Neto, M, et al. Hyperprolactinemia therapy: lessons learned from long-term follow-up. Eur J Endocrinol. 1994;130:116.
117. Landolt, AM, Keller, PJ, Froesch, ER, et al. Bromocriptine: does it jeopardise the result of later surgery for prolactinomas? Lancet. 1982;2:657–658.
118. Soule, SG, Farhi, J, Conway, GS, et al. The outcome of hypophysectomy for prolactinomas in the era of dopamine agonist therapy. Clin Endocrinol (Oxf). 1996;44:711–716.
119. Perrin, G, Treluyer, C, Trouillas, J, et al. Surgical outcome and pathological effects of bromocriptine preoperative treatment in prolactinomas. Pathol Res Pract. 1991;187:587–592.
120. Sughrue, ME, Chang, EF, Tyrell, JB, et al. Pre-operative dopamine agonist therapy improves post-operative tumor control following prolactinoma resection. Pituitary. 2008 Jul 24.
121. Serri, O, Rasio, E, Beauregard, H, et al. Recurrence of hyperprolactinemia after selective transsphenoidal adenomectomy in women with prolactinoma. N Engl J Med. 1983;309:280–283.
122. Amar, AP, Couldwell, WT, Chen, JC, et al. Predictive value of serum prolactin levels measured immediately after transsphenoidal surgery. J Neurosurg. 2002;97:307–314.
123. Maira, G, Anile, C, De Marinis, L, et al. Prolactin-secreting adenomas: surgical results and long-term follow-up. Neurosurgery. 1989;24:736–743.
124. Massoud, F, Serri, O, Hardy, J, et al. Transsphenoidal adenomectomy for microprolactinomas: 10 to 20 years of follow-up. Surg Neurol. 1996;45:341–346.
125. Thomson, JA, Gray, CE, Teasdale, GM. Relapse of hyperprolactinemia after transsphenoidal surgery for microprolactinoma: lessons from long-term follow-up. Neurosurgery. 2002;50:36–39.
126. Nawar, RN, AbdelMannan, D, Selman, WR, et al. Pituitary tumor apoplexy: a review. Intensive Care Med. 2008;23:75–90.
127. Adler, I, Barsi, P, Czirják, S, et al. Rapid re-enlargement of a macroprolactinoma after initial shrinkage in a young woman treated with bromocriptine. Gynecol Endocrinol. 2005;20:317–321.
128. Bronstein, MD, Musolino, NR, Benabou, S, et al. Cerebrospinal fluid rhinorrhea occurring in long-term bromocriptine treatment for macroprolactinomas. Surg Neurol. 1989;32:346–349.
129. Suliman, SG, Gurlek, A, Byrne, JV, et al. Nonsurgical cerebrospinal fluid rhinorrhea in invasive macroprolactinoma: incidence, radiological, and clinicopathological features. J Clin Endocrinol Metab. 2007;92:3829–3835.
130. Jones, SE, James, RA, Hall, K, et al. Optic chiasmal herniation—an under-recognized complication of dopamine agonist therapy for macroprolactinoma. Clin Endocrinol (Oxf). 2000;53:529–534.
131. Kreutzer, J, Buslei, R, Wallaschofski, H, et al. Operative treatment of prolactinomas: indications and results in a current consecutive series of 212 patients. Eur J Endocrinol. 2008;158(1):11–18.
132. Besser, GM, Parke, L, Edwards, CR, et al. Galactorrhoea: successful treatment with reduction of plasma prolactin levels by brom-ergocryptine. Br Med J. 1972;3:669–672.
133. Ben-Jonathan, N. Regulation of prolactin secretion. In: Imura H, ed. The Pituitary Gland. New York: Raven Press; 1994:261.
134. Vance, ML, Evans, WS, Thorner, MO. Bromocriptine. Ann Intern Med. 1984;100:78–91.
135. Pinzone, JJ, Katznelson, L, Danila, DC, et al. Primary medical therapy of micro- and macroprolactinomas in men. J Clin Endocrinol Metab. 2000;85:3053–3057.
136. Corenblum, B, Webster, BR, Mortimer, CB, et al. Possible antitumour effect of bromoergocryptine in 2 patients with large prolactin-secreting pituitary adenomas. Clin Res. 1975;23:614A.
137. Molitch, ME, Elton, RL, Blackwell, RE, et al. Bromocriptine as primary therapy for prolactin-secreting macroadenomas: results of a prospective multicenter study. J Clin Endocrinol Metab. 1985;60:698–705.
138. Essais, O, Bouguerra, R, Hamzaoui, J, et al. Efficacy and safety of bromocriptine in the treatment of macroprolactinomas. Ann Endocrinol (Paris). 2002;63:524–531.
139. Wu, ZB, Yu, CJ, Su, ZP, et al. Bromocriptine treatment of invasive giant prolactinomas involving the cavernous sinus: results of a long-term follow up. J Neurosurg. 2006;104:54–61.
140. Tindall, GT, Kovacs, K, Horvath, E, et al. Human prolactin-producing adenomas and bromocriptine: a histological, immunocytochemical, ultrastructural, and morphometric study. J Clin Endocrinol Metab. 1982;55:178–183.
141. Gruszka, A, Pawlikowski, M, Kunert-Radek, J. Anti-tumoral action of octreotide and bromocriptine on the experimental rat prolactinoma: anti-proliferative and pro-apoptotic effects. Neuroendocrinol Lett. 2001;22:343–348.
142. Kovacs, K, Stefaneanu, L, Horvath, E, et al. Effect of dopamine agonist medication on prolactin producing pituitary adenomas: a morphological study including immunocytochemistry, electron microscopy and in-situ hybridization. Virchows Arch. 1991;418:439–446.
143. Orrego, JJ, Chandler, WF, Barkan, AL. Rapid re-expansion of a macroprolactinoma after early discontinuation of bromocriptine. Pituitary. 2000;3:189–192.
144. van’t Verlaat, JW, Croughs, RJ. Withdrawal of bromocriptine after long-term therapy for macroprolactinomas: effect on plasma prolactin and tumour size. Clin Endocrinol (Oxf). 1991;34:175–178.
145. Bevan, JS, Webster, J, Burke, CW, et al. Dopamine agonists and pituitary tumor shrinkage. Endocr Rev. 1992;13:220–240.
146. Molitch, ME. Medical management of prolactin-secreting pituitary adenomas. Pituitary. 2002;5:55–65.
147. Passos, VQ, Souza, JJ, Musolino, NR, et al. Long-term follow-up of prolactinomas: normoprolactinemia after bromocriptine withdrawal. J Clin Endocrinol Metab. 2002;87:3578–3582.
148. Biswas, M, Smith, J, Jadon, D, et al. Long-term remission following withdrawal of dopamine agonist therapy in subjects with microprolactinomas. Clin Endocrinol (Oxf). 2005;63:26–31.
149. Gen, M, Uozumi, T, Ohta, M, et al. Necrotic changes in prolactinomas after long term administration of bromocriptine. J Clin Endocrinol Metab. 1984;59:463–470.
150. Koppelman, MC, Jaffe, MJ, Rieth, KG, et al. Hyperprolactinemia, amenorrhea, and galactorrhea: a retrospective assessment of 25 cases. Ann Intern Med. 1984;100:115–121.
151. Schlechte, J, Dolan, K, Sherman, B, et al. The natural history of untreated hyperprolactinemia: a prospective analysis. J Clin Endocrinol Metab. 1989;68:412–418.
152. Sisam, DA, Sheehan, JP, Sheeler, LR. The natural history of untreated microprolactinomas. Fertil Steril. 1987;48:67–71.
153. Karunakaran, S, Page, RC, Wass, JA. The effect of the menopause on prolactin levels in patients with hyperprolactinaemia. Clin Endocrinol (Oxf). 2001;54:295–300.
154. Pinto, G, Zerah, M, Trivin, C, et al. Pituitary apoplexy in an adolescent with prolactin-secreting adenoma. Horm Res. 1998;50:38–41.
155. Turner, TH, Cookson, JC, Wass, JA, et al. Psychotic reactions during treatment of pituitary tumours with dopamine agonists. Br Med J. 1984;289:1101–1103.
156. Ciubotaru, V, Poinsignon, Y, Brunet-Bourgin, F, et al. Severe pleuropericarditis induced by long-term bromocriptine therapy, report of a case and review of the literature. Rev Med Interne. 2004;25:310–314.
157. Liuzzi, A, Dallabonzana, D, Oppizzi, G, et al. Low doses of dopamine agonists in the long-term treatment of macroprolactinomas. N Engl J Med. 1985;313:656–659.
158. Merola, B, Colao, A, Caruso, E, et al. Effectiveness and long-term tolerability of the slow release oral form of bromocriptine on tumoral and non-tumoral hyperprolactinemia. J Endocrinol Invest. 1992;15:173–176.
159. Bronstein, MD, Cardim, CS, Marino, R, Jr. Short-term management of macroprolactinomas with a new injectable form of bromocriptine. Surg Neurol. 1987;28:31–37.
160. Beckers, A, Petrossians, P, Abs, R, et al. Treatment of macroprolactinomas with the long-acting and repeatable form of bromocriptine: a report on 29 cases. J Clin Endocrinol Metab. 1992;75:275–280.
161. Darwish, AM, Farah, E, Gadallah, WA, et al. Superiority of newly developed vaginal suppositories over vaginal use of commercial bromocriptine tablets: a randomized controlled clinical trial. Reprod Sci. 2007;14:280–285.
162. Brue, T, Pellegrini, I, Priou, A, et al. Prolactinomas and resistance to dopamine agonists. Horm Res. 1992;38:84–89.
163. Pellegrini, I, Rasolonjanahary, R, Gunz, G, et al. Resistance to bromocriptine in prolactinomas. J Clin Endocrinol Metab. 1989;69:500–509.
164. Passos, VQ, Fortes, MA, Giannella-Neto, D, et al. Genes differentially expressed in prolactinomas responsive and resistant to dopaminergic agonists. Neuroendocrinology. 2009;89(2):163–170.
165. Caccavelli, L, Feron, F, Morange, I, et al. Decreased expression of the two D2 dopamine receptor isoforms in bromocriptine-resistant prolactinomas. Neuroendocrinology. 1994;60:314–322.
166. Delgrange, E, Duprez, T, Maiter, D. Influence of parasellar extension of macroprolactinomas defined by magnetic resonance imaging on their responsiveness to dopamine agonist therapy. Clin Endocrinol (Oxf). 2006;64:456–462.
167. Delgrange, E, Crabbe, J, Donckier, J. Late development of resistance to bromocriptine in a patient with macroprolactinoma. Horm Res. 1998;49:250–253.
168. Lamberts, SW, Quik, RF. A comparison of the efficacy and safety of pergolide and bromocriptine in the treatment of hyperprolactinemia. J Clin Endocrinol Metab. 1991;72:635.
169. Berezin, M, Avidan, D, Baron, E. Long-term pergolide treatment of hyperprolactinemic patients previously unsuccessfully treated with dopaminergic drugs. Isr J Med Sci. 1991;27:375–379.
170. Freda, PU, Andreadis, CI, Khandji, AG, et al. Long-term treatment of prolactin-secreting macroadenomas with pergolide. J Clin Endocrinol Metab. 2000;85:8–13.
171. Orrego, JJ, Chandler, WF, Barkan, AL. Pergolide as primary therapy for macroprolactinomas. Pituitary. 2000;3:251–256.
172. Vance, ML, Lipper, M, Klibanski, A, et al. Treatment of prolactin-secreting pituitary macroadenomas with the long-acting non-ergot dopamine agonist CV 205-502. Ann Intern Med. 1990;112:668–673.
173. Serri, O, Beauregard, H, Lesage, J, et al. Long term treatment with CV 205-502 in patients with prolactin-secreting pituitary macroadenomas. J Clin Endocrinol Metab. 1990;71:682–687.
174. van der Lely, AJ, Brownell, J, Lamberts, SW. The efficacy and tolerability of CV 205-502 (a nonergot dopaminergic drug) in macroprolactinoma patients and in prolactinoma patients intolerant to bromocriptine. J Clin Endocrinol Metab. 1991;72:1136–1141.
175. Kvistborg, A, Halse, J, Bakke, S, et al. Long-term treatment of macroprolactinomas with CV 205-502. Acta Endocrinol (Copenh). 1993;128:301–307.
176. Glaser, B, Nesher, Y, Barziliai, S. Long-term treatment of bromocriptine-intolerant prolactinoma patients with CV 205-502. J Reprod Med. 1994;39:449–454.
177. Brue, T, Pellegrini, I, Gunz, G, et al. Effects of the dopamine agonist CV 205-502 in human prolactinomas resistant to bromocriptine. J Clin Endocrinol Metab. 1992;74:577–584.
178. Morange, I, Barlier, A, Pellegrini, I, et al. Prolactinomas resistant to bromocriptine: long-term efficacy of quinagolide and outcome of pregnancy. Eur J Endocrinol. 1996;135:413–420.
179. Del Dotto, P, Bonuccelli, U. Clinical pharmacokinetics of cabergoline. Clin Pharmacokinet. 2003;42:633–645.
180. Ferrari, C, Paracchi, A, Mattei, AM, et al. Cabergoline in the long-term therapy of hyperprolactinemic disorders. Acta Endocrinol (Copenh). 1992;126:489–494.
181. Cannavo, S, Curto, L, Squadrito, S, et al. Cabergoline: a first-choice treatment in patients with previously untreated prolactin-secreting pituitary adenoma. J Endocrinol Invest. 1999;22:354–359.
182. Biller, BM, Molitch, ME, Vance, ML, et al. Treatment of prolactin-secreting macroadenomas with the once-weekly dopamine agonist cabergoline. J Clin Endocrinol Metab. 1996;81:2338–2343.
183. Colao, A, Vitale, G, Cappabianca, P, et al. Outcome of cabergoline treatment in men with prolactinoma: effects of a 24-month treatment on prolactin levels, tumor mass, recovery of pituitary function, and semen analysis. J Clin Endocrinol Metab. 2004;89:1704–1711.
184. Vilar, L, Freitas, MC, Naves, LA, et al. Diagnosis and management of hyperprolactinemia: results of a Brazilian multicenter study with 1234 patients—intolerance/resistance. J Endocrinol Invest. 2008;31:436–444.
185. Webster, J, Piscitelli, G, Polli, A, et al. Cabergoline Comparative Study Group: A comparison of cabergoline and bromocriptine in the treatment of hyperprolactinemic amenorrhea. N Engl J Med. 1994;331:904–909.
186. Pascal-Vigneron, V, Weryha, G, Bosc, M, et al. Hyperprolactinemic amenorrhea: treatment with cabergoline versus bromocriptine: results of a national multicenter randomized double-blind study. Presse Med. 1995;24:753–757.
187. Verhelst, J, Abs, R, Maiter, D, et al. Cabergoline in the treatment of hyperprolactinemia: A study in 455 patients. J Clin Endocrinol Metab. 1999;84:2518–2522.
188. Sabuncu, T, Arikan, E, Tasan, E, et al. Comparison of the effects of cabergoline and bromocriptine on prolactin levels in hyperprolactinemic patients. Intern Med. 2001;40:857–861.
189. Colao, A, Di Sarno, A, Landi, ML, et al. Macroprolactinoma shrinkage during cabergoline treatment is greater in naive patients than in patients pretreated with other dopamine agonists: a prospective study in 110 patients. J Clin Endocrinol Metab. 2000;85:2247–2252.
190. Colao, A, Di Sarno, A, Sarnacchiaro, F, et al. Prolactinomas resistant to standard dopamine agonists respond to chronic cabergoline treatment. J Clin Endocrinol Metab. 1997;82:876–883.
191. Colao, A, Di Sarno, A, Cappabianca, P, et al. Withdrawal of long-term cabergoline therapy for tumoral and nontumoral hyperprolactinemia. N Engl J Med. 2003;349:2023–2033.
192. Schade, R, Andersohn, F, Suissa, S, et al. Dopamine agonists and the risk of cardiac valve regurgitation. N Engl J Med. 2007;356:29–38.
193. Zanettini, R, Antonini, A, Gatto, G, et al. Valvular heart disease and the use of dopamine agonists for Parkinson’s disease. N Engl J Med. 2007;356:39–46.
194. Bogazzi, F, Buralli, S, Manetti, L, et al. Treatment with low doses of cabergoline is not associated with increased prevalence of cardiac valve regurgitation in patients with hyperprolactinaemia. Int J Clin Pract. 2008;62:1865–1869.
195. Vallette, S, Serri, K, Rivera, J, et al. Long-term cabergoline therapy is not associated with valvular heart disease in patients with prolactinomas. Pituitary. Jul 2, 2008.
196. Kars, M, Delgado, V, Holman, ER, et al. Aortic valve calcification and mild tricuspid regurgitation, but no clinical heart disease after 8 years of dopamine agonist therapy for prolactinoma. J Clin Endocrinol Metab. Jun 17, 2008.
197. Acquati, S, Pizzocaro, A, Tomei, G, et al. A comparative evaluation of effectiveness of medical and surgical therapy in patients with macroprolactinoma. J Neurosurg Sci. 2001;45:65–69.
198. Tsang, RW, Brierley, JD, Panzarella, T, et al. Role of radiation therapy in clinical hormonally active pituitary adenomas. Radiother Oncol. 1996;41:45–53.
199. Molitch, ME. Pathologic hyperprolactinemia. Endocrinol Metab Clin North Am. 1992;21:877–901.
200. Landolt, AM, Lomax, N. Gamma-knife radiosurgery for prolactinomas. J Neurosurg. 2000;93:14–18.
201. Pouratian, N, Sheehan, J, Jagannathan, J, et al. Gamma-knife radiosurgery for medically and surgically refractory prolactinomas. Neurosurgery. 2006;59:255–266.
202. Pan, L, Zhang, N, Wang, EM, et al. Gamma-knife radiosurgery as a primary treatment for prolactinomas. J Neurosurg. 2000;93:10–13.
203. Testa, G, Vegetti, W, Motta, T, et al. Two-year treatment with oral contraceptives in hyperprolactinemic patients. Contraception. 1998;58:69–73.
204. Gillam, MP, Middler, S, Freed, DJ, et al. The novel use of very high doses of cabergoline and a combination of testosterone and an aromatase inhibitor in the treatment of a giant prolactinoma. J Clin Endocrinol Metab. 2002;87:4447–4451.
205. Bronstein, MD, Knoepfelmacher, M, Liberman, B, et al. Absence of suppressive effect of somatostatin on prolactin levels in patients with hyperprolactinemia. Horm Metab Res. 1987;19:271–274.
206. Shimon, I, Yan, X, Taylor, JE, et al. Somatostatin receptor (SSTR) subtype-selective analogues differentially suppress in vitro growth hormone and prolactin in human pituitary adenomas: Novel potential therapy for functional pituitary tumors. J Clin Invest. 1997;100:2386–2392.
207. Jaquet, P, Ouafik, L, Saveanu, A, et al. Quantitative and functional expression of somatostatin receptor subtypes in human prolactinomas. J Clin Endocrinol Metab. 1999;84:3268–3276.
208. Fusco, A, Gunz, G, Jaquet, P, et al. Somatostatinergic ligands in dopamine-sensitive and resistant prolactinomas. Eur J Endocrinol. 2008;158:595–603.
209. Ishikawa, H, Heaney, AP, Yu, R, et al. Human pituitary tumor-transforming gene induces angiogenesis. J Clin Endocrinol Metab. 2001;86:867–874.
210. Goffin, V, Bernichtein, S, Touraine, P, et al. Development and Potential Clinical Uses of Human Prolactin Receptor Antagonists. Endocrine Reviews. 2005;26:400–422.
211. Neff, LM, Weil, M, Cole, A, et al. Temozolomide in the treatment of an invasive prolactinoma resistant to dopamine agonists. Pituitary. 2007;10:81–86.
212. Kovacs, K, Horvath, E, Syro, LV, et al. Temozolomide therapy in a man with an aggressive prolactin-secreting pituitary neoplasm: Morphological findings. Hum Pathol. 2007;38:185–189.
213. Fadul, CE, Kominsky, AL, Meyer, LP, et al. Long-term response of pituitary carcinoma to temozolomide. Report of two cases. J Neurosurg. 2006;105:621–626.
214. Facchetti, M, Uberti, D, Memo, M, et al. Nerve growth factor restores p53 function in pituitary tumor cell lines via trkA-mediated activation of phosphatidylinositol 3-kinase. Mol Endocrinol. 2004;18:162–172.
215. Missale, C, Losa, M, Boroni, F, et al. Nerve growth factor and bromocriptine: a sequential therapy for human bromocriptine-resistant prolactinomas. Br J Cancer. 1995;72:1397–1399.
216. Gemzell, C, Wang, CF. Outcome of pregnancy in women with pituitary adenoma. Fertil Steril. 1979;31:363–372.
217. Molitch, ME. Pregnancy and the hyperprolactinemic woman. N Engl J Med. 1985;312:1364–1370.
218. Musolino, NRC, Bronstein, MD. Prolactinomas and pregnancy. In: Bronstein MD, ed. Pituitary Tumors in Pregnancy. Boston: Kluwer Academic Publishers; 2001:91–108.
219. Bronstein, MD. Prolactinomas and pregnancy. Pituitary. 2005;8:31–38.
220. Holmgren, U, Bergstrand, G, Hagenfeldt, K, et al. Women with prolactinoma-effect of pregnancy and lactation on serum prolactin and on tumour growth. Acta Endocrinol (Copenh). 1986;111:452–459.
221. Krupp, P, Monka, C. Bromocriptine in pregnancy: safety aspects. Klin Wochenschr. 1987;65:823–827.
222. Konopka, P, Raymond, JP, Merceron, RE, et al. Continuous administration of bromocriptine in the prevention of neurological complications in pregnant women with prolactinomas. Am J Obstet Gynecol. 1983;146:935–938.
223. Webster, J. A comparative review of the tolerability profiles of dopamine agonists in the treatment of hyperprolactinaemia and inhibition of lactation. Drug Saf. 1996;14:228–238.
224. Robert, E, Musatti, L, Piscitelli, G, et al. Pregnancy outcome after treatment with the ergot derivative, cabergoline. Reprod Toxicol. 1996;10:333–337.
225. Colao, A, Abs, R, Bárcena, DG, et al. Pregnancy outcomes following cabergoline treatment: extended results from a 12-year observational study. Clin Endocrinol (Oxf). 2008;68:66–71.
226. Buelke-Sam, J, Cohen, IR, Tizzano, JP, et al. Developmental toxicity of the dopamine agonist pergolide mesylate in CD-1 mice: II. Perinatal and postnatal exposure. Neurotoxicol Teratol. 1991;13:297–306.
227. Crosignani, PG, Mattei, AM, Severini, V, et al. Long-term effects of time, medical treatment and pregnancy in 176 hyperprolactinemic women. Eur J Obstet Gynaecol Reprod Biol. 1992;44:175–180.
228. Badawy, SZ, Marziale, JC, Rosenbaum, AE, et al. The long-term effects of pregnancy and bromocriptine treatment on prolactinomas—the value of radiologic studies. Early Pregnancy. 1997;3:306–311.
229. Peillon, F, Racadot, J, Moussy, D, et al. Prolactin-secreting adenomas: a correlative study of morphological and clinical data. In: Fahlbuch R, von Werder K, eds. Treatment of Pituitary Adenomas. Stuttgart: Thieme; 1978:114.
230. Wang, PS, Walker, AM, Tsuang, MT, et al. Dopamine antagonists and the development of breast cancer. Arch Gen Psychiatry. 2002;59:1147–1154.
231. Manjer, J, Johansson, R, Berglund, G, et al. Postmenopausal breast cancer risk in relation to sex steroid hormones, prolactin and SHBG (Sweden). Cancer Causes Control. 2003;14:599–607.
232. Rose-Hellekant, TA, Arendt, LM, Schroeder, MD, et al. Prolactin induces ERα-positive and ERα-negative mammary cancer in transgenic mice. Oncogene. 2003;22:4664–4674.
233. Liby, K, Neltner, B, Mohamet, L, et al. Prolactin overexpression by MDA-MB-435 human breast cancer cells accelerates tumor growth. Breast Cancer Res Treat. 2003;79:241–252.