Chapter 18 Cerebral Revascularization for Giant Aneurysms of the Transitional Segment of the Internal Carotid Artery
• Giant transitional internal carotid artery (ICA) aneurysms incorporate the cavernous, clinoidal, and supraclinoidal segments of the ICA in their necks and are more than 2.5 cm in diameter.
• The natural history of intracavernous ICA giant aneurysms is relatively benign, compared to transitional segment giant ICA aneurysms. Unruptured giant aneurysms in this location had a rupture rate of 40% in 5 years, according to the International Study of Unruptured Intracranial Aneurysms.
• Elective occlusion of the ICA for treatment of aneurysms carries a significant stroke risk, even after a successful balloon test occlusion of the ICA.
• These aneurysms can be treated by the performance of a high-flow bypass graft (radial artery or saphenous vein) from the external carotid artery or the internal maxillary artery to the M2 divisions of the middle cerebral artery, with simultaneous proximal occlusion or trapping of the giant aneurysm. The procedure carries low mortality and morbidity rates, as shown in the authors’ series of 55 patients with giant ICA transitional aneurysms.
• Flow diversion stents such as the Pipeline stent are being used for the endovascular treatment of unruptured giant ICA aneurysms. Longer follow-up is needed to evaluate the results and complications.
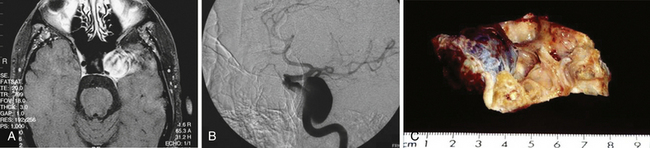
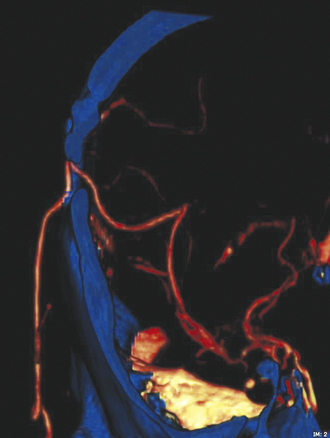
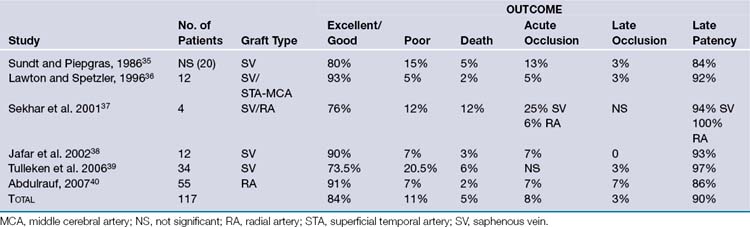
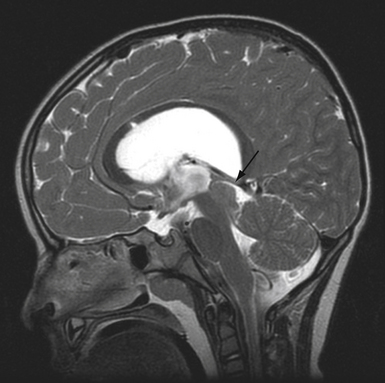
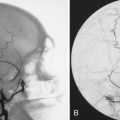
Pure cavernous ICA aneurysms are a separate category of aneurysms that do not extend beyond the dural ring of the ICA. In the neurosurgical literature these aneurysms are considered benign, even when they are larger.1 However, in our experience it is possible that when these aneurysms get significantly large, they can cause rupture through the middle fossa dura, causing hemorrhage and potentially death (Fig. 18.1).
Natural History
Unruptured giant intracranial aneurysms are being diagnosed more often as a result of the availability of better imaging techniques, and these lesions pose management problems for practitioners. The first series of giant aneurysms was presented in 1969 by Morley and Barr, who described giant aneurysms as greater than 25 mm.2 Giant aneurysms are relatively rare, accounting for between 5% and 7% of aneurysms3 in most series. Giant aneurysms have a female predominance with female:male ratios as high as 3:1 in some series4 and most commonly present in the middle decades of life. Giant aneurysms are most frequently located on the ICA, specifically involving the paraclinoid segment 21% of the time.5
The natural history of giant aneurysms is one of a poor prognosis. Some have compared the fate of unruptured giant aneurysms to that of subarachnoid hemorrhage (SAH).6 Patients often present with symptoms consistent with SAH or mass effect. The Italian cooperative study has shown a 28% mortality rate for untreated giant aneurysms compared to a 14% mortality rate for patients who received treatment.7 In the same series, morbidity was seen in 48% of cases owing to aneurysm expansion. Giant aneurysms, not unlike other aneurysms, continue to enlarge over time. Expansion is thought to be from growth of the lumen or thrombus formation within the sac. This progressive enlargement often causes symptoms of mass effect.8
Historically, it was thought that giant aneurysms were less prone to rupture; however, contemporary evidence suggests that this is inaccurate. Drake found that 62 of 174 cases did rupture.9 Aneurysms that were 25 mm or more had a relative risk of rupture of 59.0 compared to aneurysms less than 10 mm.10 Giant aneurysms had a 6% rupture rate in the first year compared to less than 1% for aneurysms less than 10 mm.10 Patients with a giant aneurysm, but without previous SAH, were at a significantly increased risk for rupture based on size. The rupture rate was not dependent on the location of the aneurysm; all anterior circulation giant aneurysms were at increased risk of rupture.3 Risk factors for aneurysm rupture include hypertension, current or former tobacco use, alcohol use greater than five drinks per day, and oral contraceptives.10 It was thought that partially or completely thrombosed aneurysms would be less likely to rupture, thus not requiring treatment; however, in Drake’s large series this was not the case.9
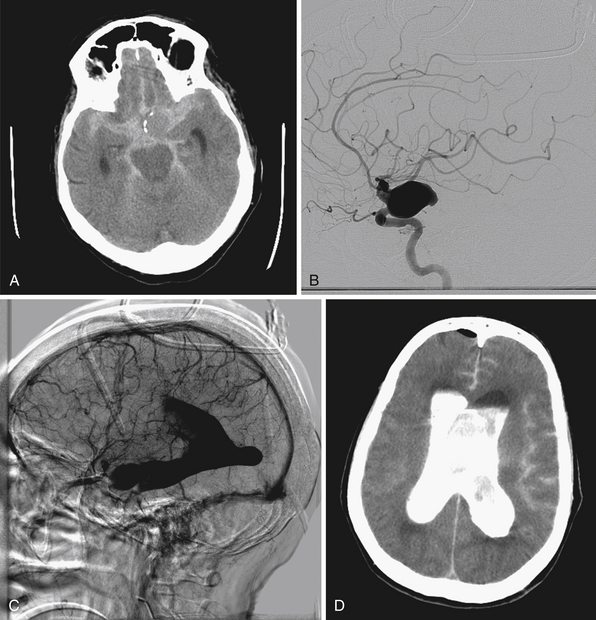
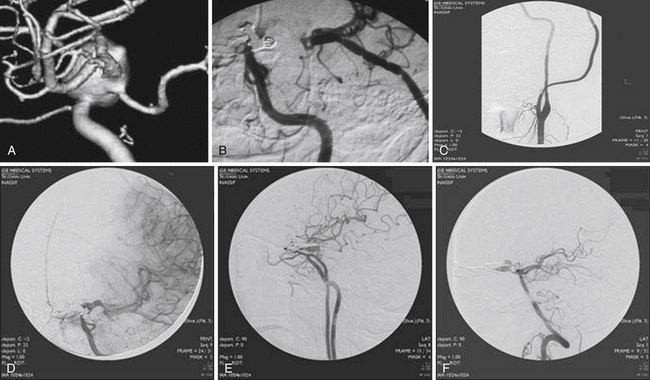
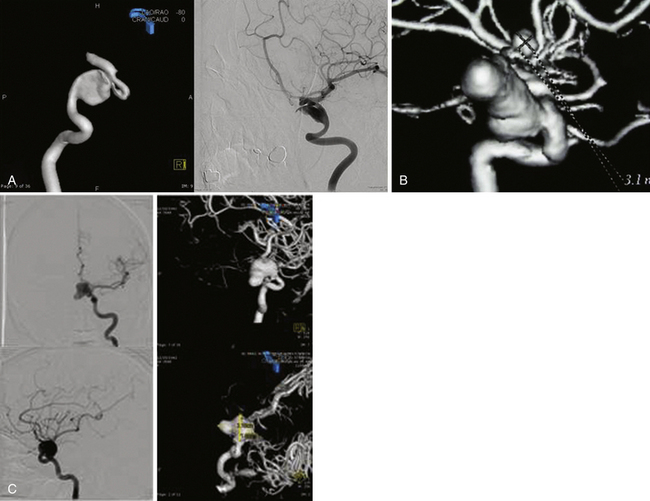
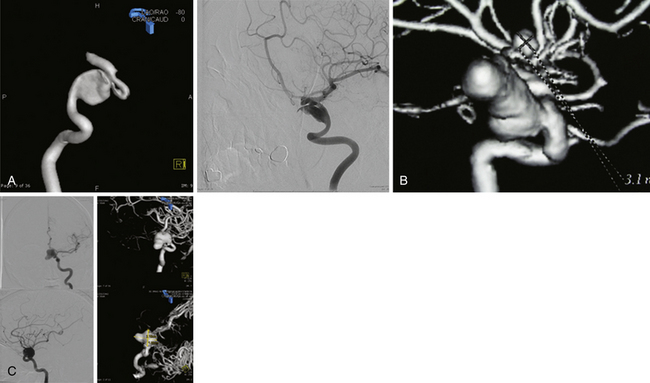
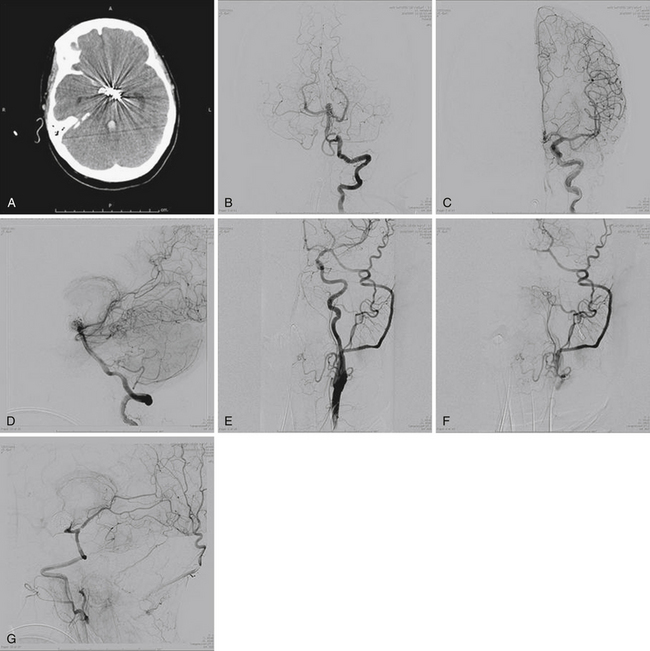
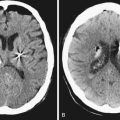
The International Study of Unruptured Intracranial Aneurysms (ISUIA) trial has shown that unruptured aneurysms overall do not pose as high a risk as once thought; and that treating aneurysms, specifically small ones, puts the patient at greater risk than watchful waiting. However, this advice should not be applied to the special case of giant aneurysms, in which “the natural history is ominous.”6 Morley and Barr’s original series demonstrated similar findings, with an 80% mortality rate for untreated aneurysms. Other studies have also shown high 1-year mortality rates.11 Peerless and associates found that 85% of patients with giant intracranial aneurysms were dead at 5 years.12 Additionally, the ISUIA II trial found that the cumulative rupture rates for anterior circulation giant aneurysms with no previous history of SAH were 40%. Patients who do present with SAH are at increased risk of death compared to patients without SAH.13 Figure 18.2 presents a case of a ruptured giant aneurysm without previous history of SAH 6 months after initial diagnosis.
Deliberate Internal Carotid Artery Occlusion
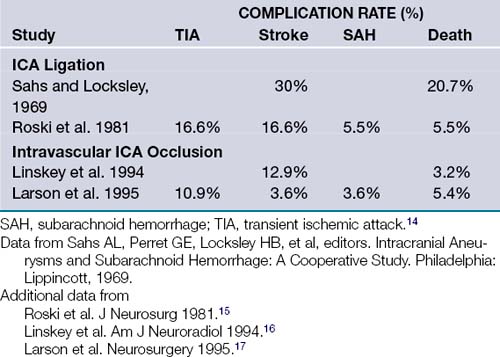
Traditional neurosurgical literature and practice have held a liberal view of ICA occlusion for the treatment of giant ICA aneurysms. Historically this has been accompanied by a relatively high risk of stroke following ICA occlusion (Table 18.1). With more recent improvements in the balloon test occlusion (BTO) paradigms, the risk is smaller but clearly not trivial.
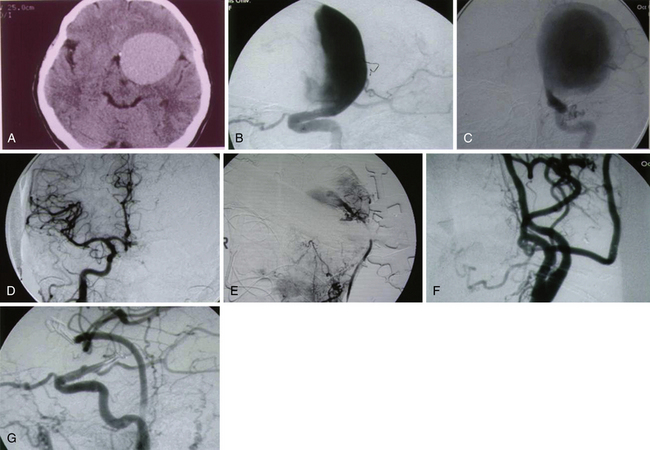
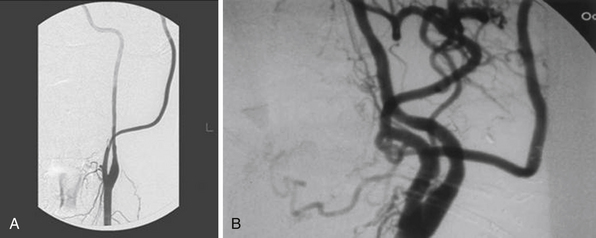
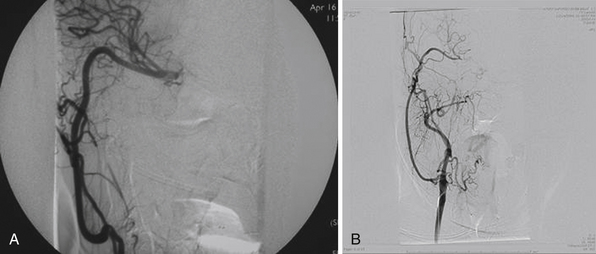
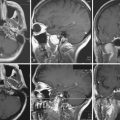
Patients with giant ICA aneurysms are usually younger and have relatively fewer medical comorbid conditions when compared to patients with cervical carotid occlusive disease, and they face an insignificant risk after planned carotid sacrifice. Long-term effects of deliberate carotid occlusion are not well studied. The risks of forming additional aneurysms, developing hemodynamic ischemic disease, and potential cognitive disorders have to be taken into account when this decision is made. Therefore, it has been the practice of the senior author (SIA) to preserve ipsilateral hemispheric flow in the treatment of giant aneurysms, especially in younger patients, by performing a bypass procedure (Fig. 18.3).
Treatment of Giant Internal Carotid Artery Aneurysms
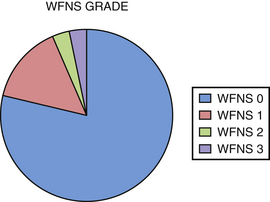
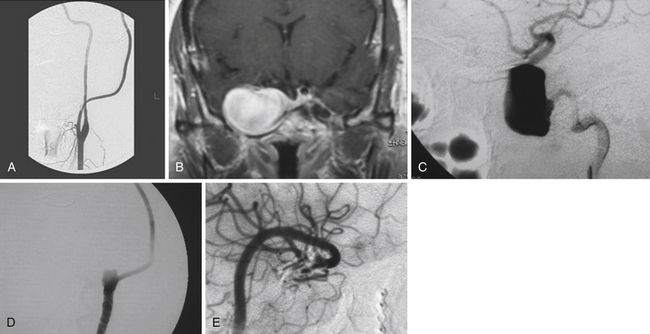
We performed a retrospective review of 55 consecutive patients treated at Saint Louis University Hospital from June 1999 to June 2007. The objectives of this study were to assess the use of high-flow extracranial-intracranial (EC-IC) bypass for the treatment of giant transitional ICA aneurysms, delineate the surgical mortality and morbidity rates, and determine the long-term efficacy of the treatment and the functional outcomes of these patients. All 55 patients had giant transitional ICA aneurysms. The mean aneurysm size was 34.7 mm (range 25 to 70 mm). The mean age was 46 years (range 30-64 years). The mean follow-up period was 34.7 months (range 1-76 months). The most common presenting symptoms were cranial neuropathy (42%), presence of headaches or incidental finding (40%), and subarachnoid hemorrhage (18%) (Fig. 18.4). The World Federation of Neurosurgical Societies (WFNS) grade was 0 in 81%, 1 in 15%, 2 in 2%, and 3 in 2% (Fig. 18.5). All 55 patients underwent a high-flow EC-IC bypass procedure prior to ICA ligation.
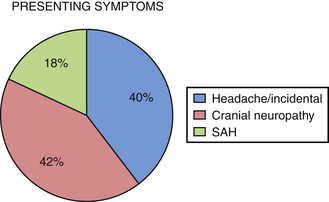
FIGURE 18.4 Percentage of patients initially presenting with headache, cranial neuropathy, and subarachnoid hemorrhage.
Surgical Techniques for Extracranial-Intracranial Bypass Procedure
Minimally Invasive Low-Flow Bypass
We rarely utilize low-flow bypass for giant aneurysms in which primary occlusion of the internal carotid artery is planned. In certain situations, such as elderly patients who pass balloon test occlusion and SPECT (single-photon emission computed tomography) scanning, it may be reasonable to perform low-flow bypass to maximally minimize surgical morbidity.
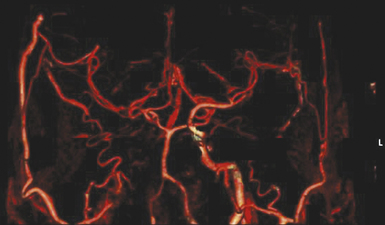
Using a preoperative computed tomography (CT) angiogram that provides clear visualization of the superficial temporal artery (STA), including its frontal and parietal branches, we measure the diameters of these branches and identify the optimal vessel for use as a donor (Fig. 18.6). This enables us to plan a linear skin incision overlying the chosen donor vessel.
The bur hole/craniotomy can be planned preoperatively with the help of the stereotactically reconstructed model based on the CT angiogram. The recipient vessel is chosen based on its caliber and superficial location in the sylvian fissure (Fig. 18.7). The ideal recipient vessel is identified based on the review of the CT angiogram. Using CT angiography–based neuronavigation, the exact location of the bur hole/craniotomy is planned to overlie the selected recipient vessel and to be in the immediate proximity of the selected donor vessel (Fig. 18.8).
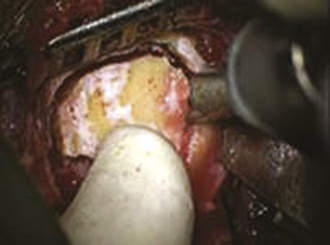
FIGURE 18.7 Intraoperative photograph obtained through the microscope while the bur hole/craniotomy was being performed.
(From Coppens JR, Cantando JD, Abdulrauf SI. Minimally invasive superficial temporal artery to middle cerebral artery bypass through an enlarged bur hole: the use of computed tomography angiography neuronavigation in surgical planning, J Neurosurg 2009;109(3):553-558, used with permission.)
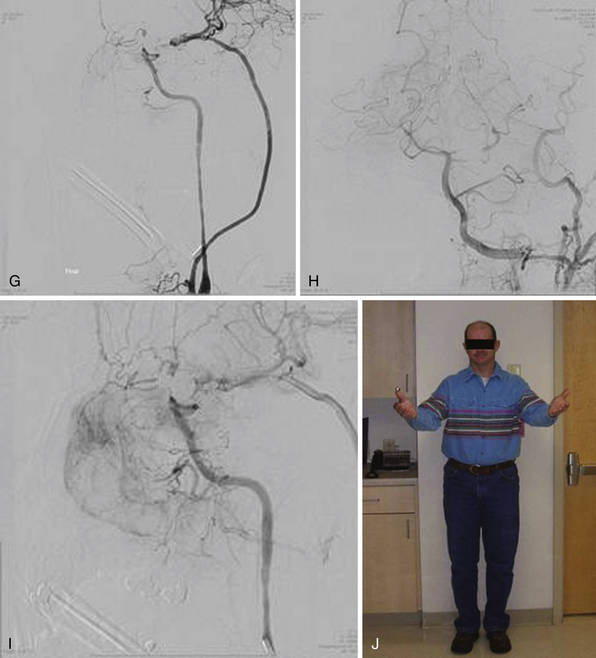
A bur hole is made and enlarged to the size of a very small craniotomy (~ 2-2.5 cm) under the microscope (Figs. 18.9 and 18.10), and the recipient vessel is exposed in the distal sylvian fissure over a length of 1 cm. A rubber dam is applied and the anastomosis is performed with a 9-0 nylon suture in a running fashion. The back wall is anastomosed first, followed by the front wall. Temporary clips are applied on the M4 recipient vessel during the anastomosis. Postoperatively, the patients are followed up with angiography (Fig. 18.11) or CT angiography (Fig. 18.12).18
Standard High-Flow Extracranial-Intracranial Bypass Technique
Craniotomy
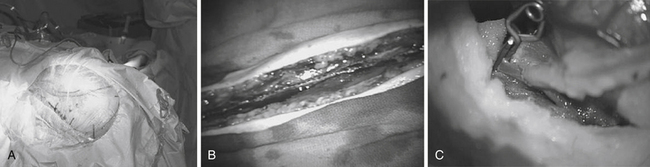
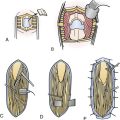
Patients are started on acetylsalicylic acid (ASA) 81 mg daily for 3 days prior to the surgery. Intraoperative electroencephalogram (EEG), somatosensory evoked potentials (SSEPs), and motor evoked potentials (MEPs) are continuously monitored. A skull base approach, usually a cranio-orbital-zygomatic approach, is the senior author’s preference to facilitate a wide access to the sylvian fissure and better exposure of the proximal supraclinoid ICA for possible proximal occlusion or trapping of the giant aneurysm. We prefer a single-piece craniotomy that entails an osteotomy of the superior and lateral orbital roofs. A high-speed drill is used to thin the zygomatic arch posteriorly which allows the radial artery to lie over the zygomatic arch without risk of kinking (Fig. 18.13A).
Radial Artery Graft Harvest and Tunneling
The radial artery is then harvested in the contralateral arm to serve as an interposition graft. It is the preference of the senior author to use radial artery as opposed to saphenous vein in order to minimize diameter mismatch between donor and recipient vessels and the graft. Prior to radial artery harvesting, an Allen’s test is performed to assure proper palmar arch perfusion by the ulnar artery alone. The critical aspect of radial artery harvest is the length of the graft, which can be limiting. Obtaining maximal length is critical during this harvest. The superficial wall of the radial artery is marked with a marking pen prior to removal of the graft. This step is crucial to avoid graft rotation during the tunneling (Fig. 18.13B). Once the graft is harvested, it is placed in a heparinized saline solution. The graft is tunneled using a pediatric chest tube. The marked surface of the graft is kept most superficial.
Cervical Anastomosis
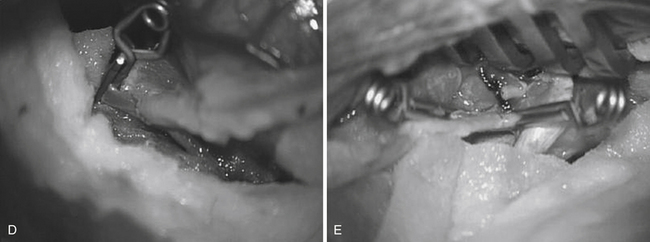
The proximal cervical anastomosis is performed first to minimize time of temporary occlusion of the ICA. The radial artery graft is placed opposite an area of the ECA where the proximal anastomosis is planned. Long temporary aneurysm clips are used for the ECA anastomosis. An end-to-side or end-to-end anastomosis can be performed between the radial artery and the ECA after temporary occlusion of this vessel. We prefer a running suture technique using 7-0 nylon or prolene stitches. We perform the “back” wall (i.e., the more difficult anastomosis) first followed by the front wall (Fig. 18.13E). When the anastomosis is completed, antegrade flow is confirmed in the graft followed by the placement of a temporary clip on the graft just distal to the anastomosis.
Intracranial Anastomosis
The site of the intracranial anastomosis is determined based on several factors: the intended revascularization territory, the size of donor and recipient vessels, the length of the graft, and the location of the aneurysm. Mild hypothermia is utilized. EEG burst suppression using pentobarbital is maintained during the temporary clipping period. A color background is placed underneath the recipient vessel. Temporary clips are applied (Fig. 18.13C). An arteriotomy is made using an arachnoid knife. The arteriotomy is extended using microscissors to the width of the interposition graft lumen. The lumen of the interposition graft is expanded by “fish-mouthing” the opening. We used 9-0 nylon for M2 and M3 vessels and 8-0 nylon for the supraclinoidal ICA. A similar technique of “back” wall and “front” wall can be utilized as described previously for the cervical anastomosis (Fig. 18-13D).
Parent Vessel Occlusion
Once the preceding steps, including intraoperative angiography, have documented flow in the graft, we then advocate parent vessel occlusion during the same surgical procedure, rather than delayed (staged) occlusion. In our opinion, the risk of graft occlusion would be high if competitive flow in the parent vessel were allowed to continue. We occlude the ICA at the level of the bifurcation in the cervical area and just distal to the giant aneurysm in the supraclinoidal portion of the ICA. The distal occlusion must be performed proximal to the anterior choroidal and dominant posterior communicating arteries.19
Minimally Invasive High-Flow Extracranial-Intracranial Bypass Technique
Once within the pterygopalatine fossa, the fibers of the infratemporal head of the lateral pterygoid muscle are visualized and dissected until the IMAX is found surrounded by adipose tissue. The IMAX is usually located inferior to the maxillary nerve and lying on the posterior wall of the maxillary sinus. This technique provides very good exposure of the IMAX and its branches, which can then be mobilized successfully for an end-to-side anastomosis with the radial artery graft. The radial artery is then trimmed to reach the desired M3 recipient vessel within the sylvian fissure, where another end-to-side anastomosis is performed using the same technique already described for the standard bypass. (Figs. 18.14 and 18.15). Intraoperative indocyanine green angiography is used to demonstrate graft patency (Fig. 18.16).20
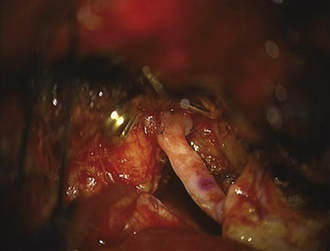
FIGURE 18.14 Proximal radial artery graft to internal maxillary artery anastomosis.
(From Abdulrauf SI, Sweeney JM, Mohan YS, Palejwala SK. Short segment internal maxillary artery to middle cerebral artery bypass: a novel technique for extracranial-to-intracranial bypass. Neurosurgery 2011;68(3):804-809, used with permission.)
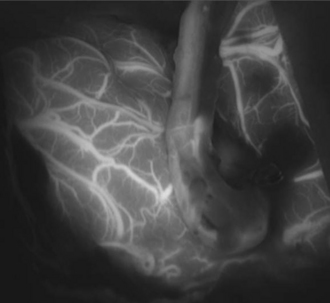
FIGURE 18.16 Intraoperative indocyanine green (ICG) angiogram showing patency of the radial artery graft.
(From Abdulrauf SI, Sweeney JM, Mohan YS, Palejwala SK. Short segment internal maxillary artery to middle cerebral artery bypass: a novel technique for extracranial-to-intracranial bypass. Neurosurgery 2011;68(3):804-809, used with permission.)
Results of Giant Aneurysm Series
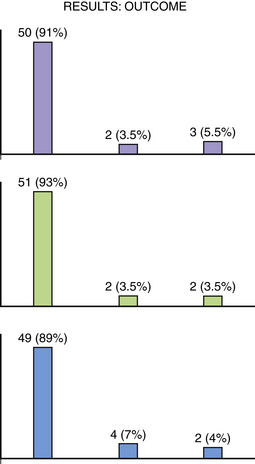
The outcome of this series shows that 50 patients (91%) had a Modified Rankin Scale (MRS) score of 0 to 1, 2 patients (3.5%) had an MRS score of 2 to 3, and 3 patients (5.5%) had an MRS score of 4 to 5. Fifty-one patients (93%) had a Glasgow Outcome Scale (GOS) score of 5, 2 patients (3.5%) had a GOS score 3 to 4, and 2 patients (3.5%) had a GOS score of 1 to 2. At discharge from the hospital 49 (81%) patients went home, 4 patients (7%) went to rehabilitation facilities, and 2 patients (4%) went to nursing homes (Fig. 18.17).
Acute graft occlusion occurred in 4 patients (7.3%), and late graft occlusion in 4 patients (7.3%). The main complication/morbidity was cerebrovascular accident (CVA), which occurred in 4 patients (7.3%). Two patients had CVA secondary to acute graft occlusion, and 2 patients had CVA secondary to microsurgical manipulation of calcified aneurysms. Aneurysm recurrence was seen in only 1 patient (Fig. 18.18). One patient died from a pulmonary embolus at home 5 weeks after operation.
Treatment Options
Traditional treatment of giant aneurysms was direct microsurgical clipping, which continues to be a feasible option in certain cases. However, the risk of direct clipping of giant aneurysms is not low. Our review of major series of direct clipping of giant ICA aneurysms revealed a fair to poor outcome in approximately 18% of the patients. In these same series, the mortality rate was 11% (Table 18.2). Endovascular treatment of giant aneurysms also poses certain challenges. Our review of major series of coiling or stenting plus coiling of giant aneurysms reveals that approximately 42% of the aneurysms are completely occluded by the treatment. Thirty-seven percent of the aneurysms recanalize. Major morbidity rate was approximately 21% in these patients, and mortality rate was 15% (Table 18.3).
TABLE 18.2 Outcomes with Direct Clipping of Giant Internal Carotid Artery (ICA) Aneurysms: Summary of Clinical Studies
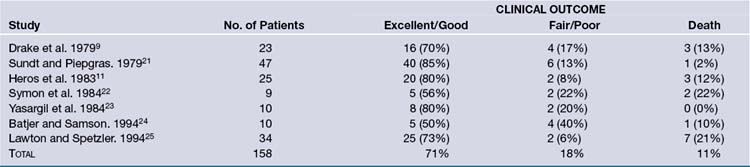
TABLE 18.3 Outcomes with Endovascular Treatment of Giant Internal Carotid Artery (ICA) Aneurysms: Summary of Clinical Studies
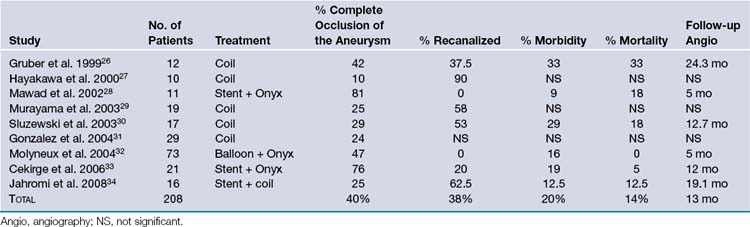
EC-IC bypass for giant aneurysms in the major published series shows relatively better outcomes than direct microsurgical clipping or endovascular treatment. Based on our review of these major series, excellent to good outcome was achieved on average in 84% of the patients. Significant morbidity was seen on average in 11% of the patients. On average, 5% of the patients died (Table 18.4).
Technical Aspects in High-Flow Extracranial-Intracranial Bypass Surgery
Atypical Appearance of Giant Aneurysms
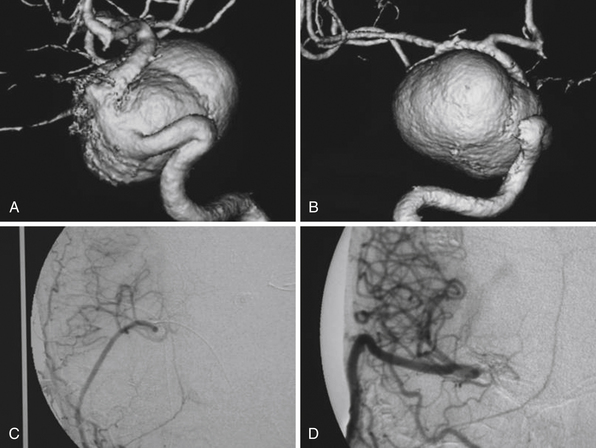
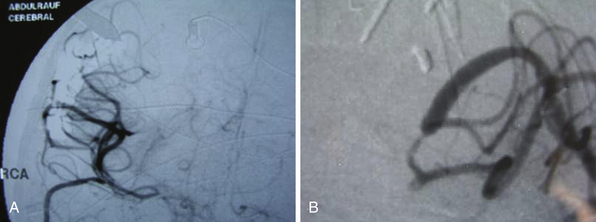
Irregular appearance of the wall, or the pattern of more than one aneurysmal sac, ought to indicate partial calcification or thrombosis. The appearance of the aneurysm on cerebral angiography is a critical factor in decision making regarding treatment. In the experience of the senior author, complex aneurysms, including those with irregular appearance of the wall, outpouching within the dome, or the presence of calcification, may carry a higher risk with direct clipping. Partial thrombosis of the aneurysm may show a smaller aneurysm on angiography when compared to magnetic resonance imaging (MRI). Partially thrombosed aneurysms also carry a risk of embolic complications during microsurgical clipping or endovascular coiling (Fig. 18.19).
Aneurysm Size
The size of the aneurysm, although a critical factor when considering direct microsurgical clipping or endovascular coiling, is not an equally important factor when considering EC-IC bypass. In our opinion, for significantly large aneurysms, EC-IC bypass would be the least risky procedure (Fig. 18.20).
Giant Cavernous Segment Internal Carotid Artery Aneurysm
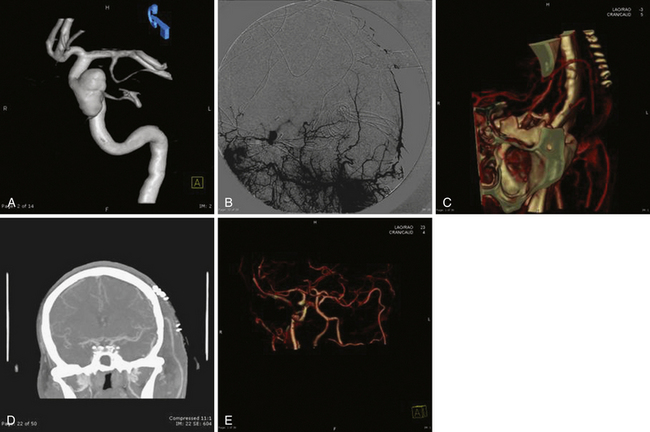
The location of the aneurysm is an important factor when making decisions regarding treatment. Endovascular stenting plus coiling or EC-IC bypass are potential treatments of a giant cavernous segment aneurysm. Direct surgical clipping should not be considered in these cases, as it would almost certainly lead to ophthalmoplegia (Fig. 18.21).
End-to-Side versus End-to-End Proximal Anastomosis
For the proximal anastomosis, end-to-end or end-to-side are both reasonable options. The senior author prefers the use of the ECA as the donor vessel. This avoids temporary occlusion of the ICA during the procedure. The advantage of end-to-end anastomosis is, presumably, more vigorous flow into the graft. The disadvantage of end-to-end anastomosis is potential occlusion of ophthalmic vessel collaterals into the surpraclinoidal carotid artery. End-to-side anastomosis, therefore, is a reasonable option and an easier procedure in comparison to end-to-end anastomosis (end-to-end may create a size mismatch, which may in certain situations be difficult to compensate for) (Fig. 18.22).
Competitive Flow
It is important to recognize that during intraoperative angiography the graft may only supply a single division of the recipient territory (i.e., a single M2 division). This should not be a concerning sign. In general, this is due to the fact that there is significant competitive flow coming through the anterior communicating artery from the contralateral side, which is protective, and the surgeon ought not to change the strategy during the case (Fig. 18.23).
Radial Artery Graft Enlargement
The diameter size of the graft as seen during intraoperative catheter or ICG angiography ought not be concerning as long as there is no occlusion or specific area of stenosis. In our experience, the graft will expand in diameter over time to accommodate for blood flow demand (Fig. 18.24).
Intraoperative Failure of Graft
In most situations in which the graft is not visible during intraoperative angiography, technical failure is assumed and the anastomosis, as well as the tunneling, ought to be reinvestigated. In patients who have passed the BTO, it is possible to have such significant competitive flow that the resistance in the graft cannot match the higher contralateral flow and the graft is not necessary. It is important to look at every step of the procedure to make sure that there is no technical failure before making any other decisions. If no technical failures are found and no changes in the patient’s motor and sensory evoked potentials are detected, it is reasonable to leave the graft in place. In certain circumstances the graft may enlarge over time if demand is placed on it, while in other situations, the graft would be occluded without any neurological symptoms based on the fact that competitive flow was significantly strong and the graft was not needed to start with (Fig. 18.25).
Creation of a New Posterior Communicating Artery
EC-IC bypass surgery is an intellectual exercise given the complexity of the aneurysm. In certain situations an intraposition graft may be needed. In some cases an intracranial-to-intracranial bypass may be more appropriate than an EC-IC bypass. In other cases the creation of a new anterior communicating or posterior communicating artery (using an interposition graft) may aid in the treatment of the specific of aneurysm, whether it is microsurgical or endovascular (Fig. 18.26).
The Length of the Graft and Risk of Occlusion
The length of the graft is also an important aspect. Additional unneeded length may lead to kinking in the cervical or cranial areas and could be a risk factor for graft occlusion. This aspect has to be inspected well during the operation, as this is an important variable in achieving success in EC-IC bypass surgery (Fig. 18.27).
Distal Anastomosis Size Mismatch
From a technical standpoint of the distal anastomosis, it is important to match the size of the donor and recipient arteries. In our opinion a significant size mismatch could have a higher risk of turbulent flow and subsequent thrombosis at the anastomosis site (Fig. 18.28).
Abdulrauf S.I. Extracranial-to-intracranial bypass using radial artery grafting for complex skull base tumors: technical note. Skull Base. 2005;15(3):207-213.
Abdulrauf S.I., Sweeney J.M., Mohan Y.S., Palejwala S.K. Short segment internal maxillary artery to middle cerebral artery bypass: a novel technique for extracranial-to-intracranial bypass. Neurosurgery. 2011;68(3):804-809.
Awad A.I., Barrow D.L., editors. Giant Intracranial Aneurysms. Park Ridge, IL: American Association of Neurological Surgeons, 1995.
International Study of Unruptured Intracranial Aneurysms Investigators. Unruptured intracranial aneurysms—risk of rupture and risks of surgical intervention. N Engl J Med. 1998;339(24):1725-1733.
Linskey M.E., Sekhar L.N., Hirsch W.L.Jr., et al. Aneurysms of the intracavernous carotid artery: natural history and indications for treatment. Neurosurgery. 1990;26(6):933-937. discussion 937-938
Please go to expertconsult.com to view the complete list of references.
1. Linskey M.E., Sekhar L.N., Hirsch W.L.Jr., et al. Aneurysms of the intracavernous carotid artery: natural history and indications for treatment. Neurosurgery. 1990;26(6):933-937. discussion 937-938
2. Morley T.P., Barr H.W. Giant intracranial aneurysms: diagnosis, course, and management. Clin Neurosurg. 1969;16:73-94.
3. Wiebers D.O., Whisnant J.P., Huston J.3rd, et al. Unruptured intracranial aneurysms: natural history, clinical outcome, and risks of surgical and endovascular treatment. Lancet. 2003;362(9378):103-110.
4. Whittle I.R., Dorsch N.W., Besser M. Giant intracranial aneurysms: diagnosis, management, and outcome. Surg Neurol. 1984;21(3):218-230.
5. Nukui H., Imai S., Fukamachi A., Aiba T. [Bilaterally symmetrical giant aneurysms of the internal carotid artery within the cavernous sinus, associated with a aneurysm of the basilar artery] (author’s translation). No Shinkei Geka. 1977;5(5):479-484.
6. Awad I.A., Barrow D.L., editors. Giant Intracranial Aneurysms. Park Ridge, IL: American Association of Neurological Surgeons, 1995.
7. Pasqualin A., Battaglia R., Scienza R., et al. Italian cooperative study on giant intracranial aneurysms: 4. Results of treatment. Acta Neurochir Suppl (Wien). 1988;42:65-70.
8. Sonntag V.K., Yuan R.H., Stein B.M. Giant intracranial aneurysms: a review of 13 cases. Surg Neurol. 1977;8(2):81-84.
9. Drake C.G. Giant intracranial aneurysms: experience with surgical treatment in 174 patients. Clin Neurosurg. 1979;26:12-95.
10. International Study of Unruptured Intracranial Aneurysms Investigators. Unruptured intracranial aneurysms—risk of rupture and risks of surgical intervention. N Engl J Med. 1998;339(24):1725-1733.
11. Heros R.C., Kistler J.P. Intracranial arterial aneurysm—an update. Stroke. 1983;14(4):628-631.
12. Peerless S.J., Drake C.G. Giant intracranial aneurysms. In: Youmans J.R., editor. Neurological Surgery: A Comprehensive Reference Guide to Diagnosis and Management of Neurological Problems. Philadelphia: WB Saunders, 1990.
13. Hamburger C., Schonberger J., Lange M. Management and prognosis of intracranial giant aneurysms. A report on 58 cases. Neurosurg Rev. 1992;15(2):97-103.
14. Sahs A.L., Perret G.E., Locksley H.B., et al. Intracranial Aneurysms and Subarachnoid Hemorrhage: A Cooperative Study. Philadelphia: J.B. Lippincott; 1969.
15. Roski R.A., Spetzler R.F., Nulsen F.E. Late complications of carotid ligation in the treatment of intracranial aneurysms. J Neurosurg. 1981 May;54(5):583-587.
16. Linskey M.E., Jungreis C.A., Yonas H., Hirsch W.L.Jr., Sekhar L.N., Horton J.A., Janosky J.E. Stroke risk after abrupt internal carotid artery sacrifice: accuracy of preoperative assessment with balloon test occlusion and stable xenon-enhanced CT. AJNR Am J Neuroradiol. 1994 May;15(5):829-843.
17. Larson J.J., Tew J.M.Jr., Tomsick T.A., van Loveren H.R. Treatment of aneurysms of the internal carotid artery by intravascular balloon occlusion: long-term follow-up of 58 patients. Neurosurgery. 1995 Jan;36(1):26-30. discussion 30
18. Coppens J.R., Cantando J.D., Abdulrauf S.I. Minimally invasive superficial temporal artery to middle cerebral artery bypass through an enlarged bur hole: the use of computed tomography angiography neuronavigation in surgical planning. J Neurosurg. 2008;109(3):553-558.
19. Abdulrauf S.I. Extracranial-to-intracranial bypass using radial artery grafting for complex skull base tumors: technical note. Skull Base. 2005;15(3):207-213.
20. Abdulrauf S.I., Sweeney J.M., Mohan Y.S., Palejwala S.K. Short segment internal maxillary artery to middle cerebral artery bypass: a novel technique for extracranial-to-intracranial bypass. Neurosurgery. 2011;68(3):804-809.
21. Sundt T.M.Jr., Piepgras D.G. Surgical approach to giant intracranial aneurysms. Operative experience with 80 cases. J Neurosurg. 1979 Dec;51(6):731-742.
22. Symon L., Vajda J. Surgical experiences with giant intracranial aneurysms. J Neurosurg. 1984 Dec;61(6):1009-1028.
23. Yasargil M.G. MICRONEUROSUERGERY II. Clinical considerations, surgery of the intracranial aneurysm and results. Stuttgart: Georg Thieme Verlag; 1984. ppt I 386
24. Batjer H.H., Kopitnik T.A.Jr., Giller C.A., Samson D.S. Surgical management of proximal carotid artery aneurysms. Clin Neurosurg. 1994;41:21-38.
25. Lawton M.T., Spetzler R.F. Management strategies for giant intracranial aneurysms. Contemp Neurosurgery. 1994;16:1-6.
26. Gruber A., Killer M., et al. Clinical and angiographic results of endovascular coiling treatment of giant and very large intracranial aneurys: A 7-year, single-center experience. Neurosurgery. 1999-45:793-804.
27. Hayakawa M., Murayama Y., Duckwiler G.R., Gobin Y.P., Guglielmi G., Viñuela F. Natural history of the neck remnant of a cerebral aneurysm treated with the Guglielmi detachable coil system. J Neurosurg. 2000 Oct;93(4):561-568.
28. Mawad M.E., Cekirge S., Ciceri E., Saatci I. Endovascular treatment of giant and large intracranial aneurysms by using a combination of stent placement and liquid polymer injection. J Neurosurg. 2002 Mar;96(3):474-482.
29. Murayama Y., Nien Y.L., Duckwiler G., Gobin Y.P., Jahan R., Frazee J., Martin N., Viñuela F. Guglielmi detachable coil embolization of cerebral aneurysms: 11 years’ experience. J Neurosurg. 2003 May;98(5):959-966.
30. Sluzewski M., Menovsky T., van Rooij W.J., Wijnalda D. Coiling of very large or giant cerebral aneurysms: long-term clinical and serial angiographic results. AJNR Am J Neuroradiol. 2003 Feb;24(2):257-262.
31. Gonzalez N.R., Dusick J.R., Duckwiler G., Tateshima S., Jahan R., Martin N.A., Viñuela F. Endovascular coiling of intracranial aneurysms in elderly patients: report of 205 treated aneurysms. Neurosurgery. 2010 Apr;66(4):714-720. discussion 720-1
32. Molyneux A.J., Cekirge S., Saatci I., et al. Cerebral Aneurysm Multicenter European Onyx (CAMEO) trial: results of a prospective observational study in 20 European centers. AJNR Am J Neuroradiol. 2004;25:39-51.
33. Cekirge H.S., Saatci I., Geyik S., Yavuz K., Oztürk H., Pamuk G. Intrasaccular combination of metallic coils and onyx liquid embolic agent for the endovascular treatment of cerebral aneurysms. J Neurosurg. 2006 Nov;105(5):706-712.
34. Jahromi B.S., Mocco J., Bang J.A., Gologorsky Y., Siddiqui A.H., Horowitz M.B., Hopkins L.N., Levy E.I. Clinical and angiographic outcome after endovascular management of giant intracranial aneurysms. Neurosurgery. 2008 Oct;63(4):662-674. discussion 674-5
35. Sundt T.M.Jr., Piepgras D.G., Marsh W.R., Fode N.C. Saphenous vein bypass grafts for giant aneurysms and intracranial occlusive disease. J Neurosurg. 1986 Oct;65(4):439-450.
36. Lawton MT, Hamilton MG, Morcos JJ, Spetzler RF. Revascularization and aneurysm surgery: current techniques, indications, and outcome. Lawton MT, Hamilton MG, Morcos JJ, Spetzler RF.
37. Sekhar L.N., Duff J.M., Kalavakonda C., Olding M. Cerebral revascularization using radial artery grafts for the treatment of complex intracranial aneurysms: techniques and outcomes for 17 patients. Neurosurgery. 2001 Sep;49(3):646-658. discussion 658-9
38. Jafar J.J., Russell S.M., Woo H.H. Treatment of giant intracranial aneurysms with saphenous vein extracranial-to-intracranial bypass grafting: indications, operative technique, and results in 29 patients. Neurosurgery. 2002 Jul;51(1):138-144. discussion 144-6
39. van Doormaal T.P., van der Zwan A., Verweij B.H., Langer D.J., Tulleken C.A. Treatment of giant and large internal carotid artery aneurysms with a high-flow replacement bypass using the excimer laser-assisted nonocclusive anastomosis technique. Neurosurgery. 2006 Oct;59(4 Suppl. 2):ONS328-ONS334. discussion ONS334-5
40. Abdulrauf S.I. Extracranial-to-intracranial bypass using radial artery grafting for complex skull base tumors: technical note. Skull Base. 2005 Aug;15(3):207-213.