Pulmonary Artery Catheter Insertion (Assist) and Pressure Monitoring
PREREQUISITE NURSING KNOWLEDGE
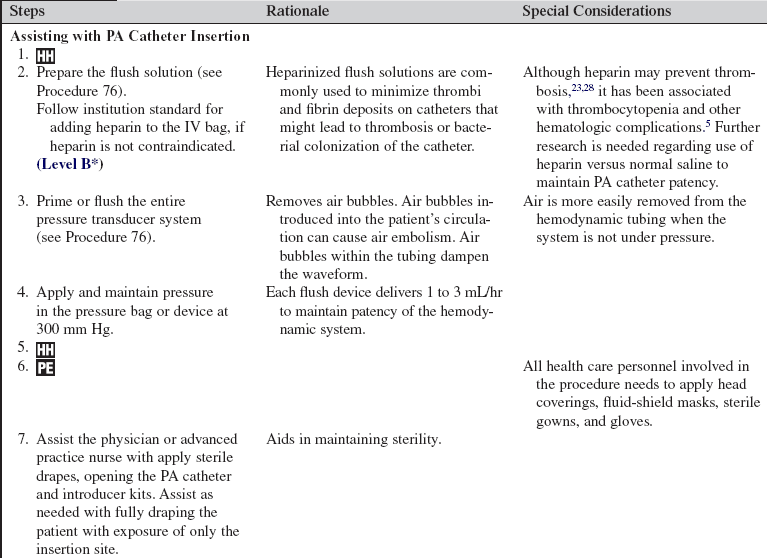
• Knowledge of the normal cardiovascular anatomy and physiology is needed.
• Knowledge of the normal pulmonary anatomy and physiology is necessary.
• Knowledge of principles of aseptic technique is needed.
• Basic dysrhythmia recognition and treatment of life-threatening dysrhythmias should be understood.
• Advanced cardiac life support knowledge and skills are necessary
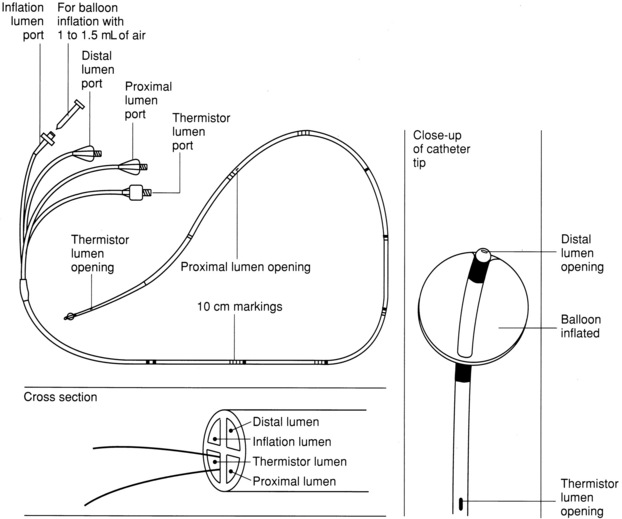
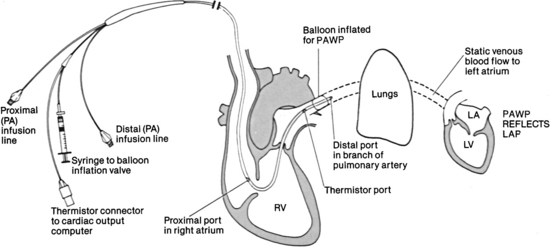
• Knowledge of the anatomy of the pulmonary artery (PA) catheter (Fig. 73-1) and the location of the PA catheter in the heart and pulmonary artery (Fig. 73-2) is important.
• Knowledge of the setup of the hemodynamic monitoring system (see Procedure 76) is essential.
• Understanding of normal hemodynamic values (see Table 67-1) is needed.
• The pulmonary artery catheter contains a proximal injectate lumen port, a PA distal lumen port, a thermistor connector, and a balloon-inflation valve. Some catheters also have two infusion ports, right atrial (RA) and right ventricular (RV) lumens, that can be used for infusion of medications and intravenous fluids.
• The PA distal lumen is used to monitor systolic, diastolic, and mean pressures in the pulmonary artery. This lumen also allows for sampling of mixed venous blood. The proximal injectate lumen is used to monitor the right atrial pressure and inject the solution used to obtain cardiac output (CO). The balloon-inflation valve is used to obtain the pulmonary artery occlusion pressure (PAOP).
• PAOP may be referred to as pulmonary artery wedge pressure.
• The PA diastolic pressure and the PAOP are indirect measures of left ventricular end-diastolic pressure (LVEDP). Usually, the PAOP is approximately 1 to 4 mm Hg less than the pulmonary artery diastolic pressure (PADP). Because these two pressures are similar, the PADP is commonly followed, which minimizes the frequency of balloon inflation, thus decreasing the potential of balloon rupture and pulmonary artery trauma.
• Differences between the PADP and the PAOP may exist for patients with pulmonary hypertension, chronic obstructive lung disease, acute respiratory distress syndrome (ARDS), pulmonary embolus, and tachycardia.
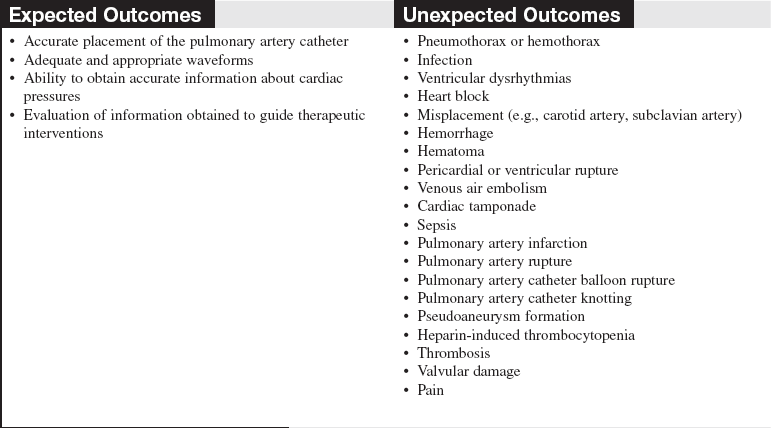
• Indications for PA catheter therapy (see Procedure 72 for additional indications) are as follows:
 Aid in the diagnosis of complications after acute myocardial infarction (MI), which may include heart failure, cardiogenic shock, papillary muscle rupture, mitral regurgitation, ventricular septal rupture, or cardiac rupture with tamponade.
Aid in the diagnosis of complications after acute myocardial infarction (MI), which may include heart failure, cardiogenic shock, papillary muscle rupture, mitral regurgitation, ventricular septal rupture, or cardiac rupture with tamponade.
 Assessment of ventricular function in heart failure.
Assessment of ventricular function in heart failure.
 Management of high-risk cardiac patients undergoing surgical procedures during preoperative, intraoperative, or postoperative periods.
Management of high-risk cardiac patients undergoing surgical procedures during preoperative, intraoperative, or postoperative periods.
 Differentiation of hypotensive states, such as hypovolemia, sepsis, heart failure, and cardiac tamponade.
Differentiation of hypotensive states, such as hypovolemia, sepsis, heart failure, and cardiac tamponade.
 Hemodynamic monitoring and evaluation of patients with major organ dysfunction who need fluid management and infusion of vasoactive medications, such as patients with burns, trauma, ARDS, or gastrointestinal bleeding.
Hemodynamic monitoring and evaluation of patients with major organ dysfunction who need fluid management and infusion of vasoactive medications, such as patients with burns, trauma, ARDS, or gastrointestinal bleeding.
• Hemodynamic monitoring with a PA catheter has no absolute contraindications, but an assessment of risk versus benefit to the patient should be considered. Relative contraindications to pulmonary artery catheter insertion include presence of fever, presence of a mechanical tricuspid valve, and a coagulopathic state. A patient with left bundle-branch block may have a right bundle-branch block develop during PA catheter insertion, resulting in complete heart block. In these patients, a temporary pacemaker should be readily available.
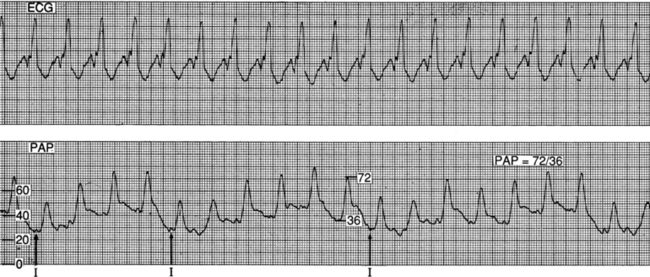
• Pulmonary artery pressures may be elevated as a result of pulmonary artery hypertension, pulmonary disease, mitral valve disease, left ventricular failure, atrial or ventricular left-to-right shunt, pulmonary emboli, or hypervolemia.
• Pulmonary artery pressures may be decreased due to hypovolemia or vasodilation.
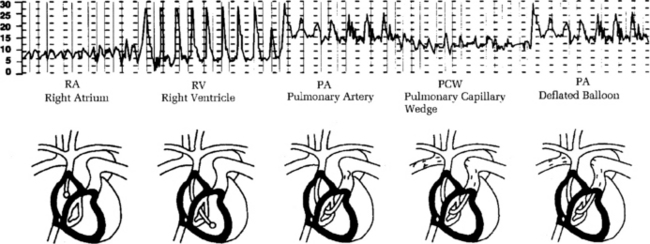
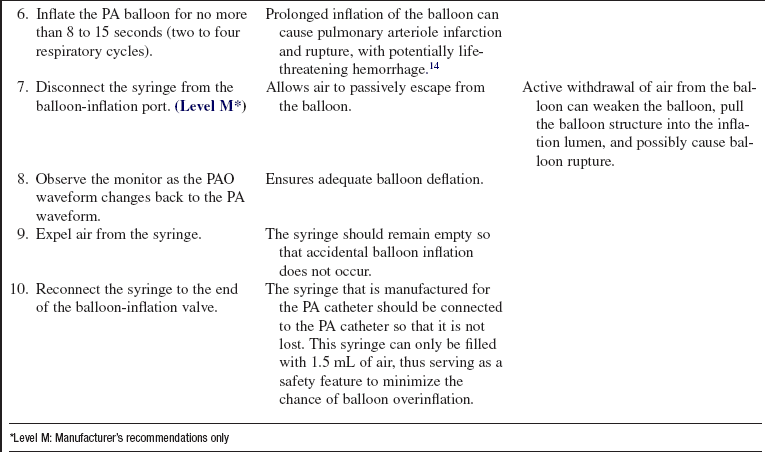
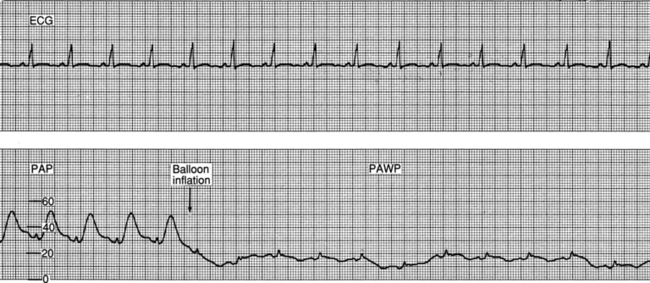
• Waveforms that occur during insertion include RA, RV, PA, and pulmonary artery occlusion (PAO; Fig. 73-3).
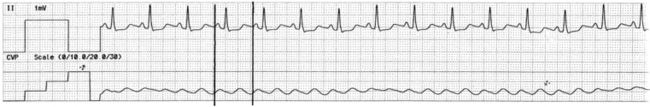
 The a wave reflects atrial contraction, the c wave reflects closure of the atrioventricular valve, and the v wave reflects passive filling of the atria during ventricular systole (Figs. 73-4 and 73-5).
The a wave reflects atrial contraction, the c wave reflects closure of the atrioventricular valve, and the v wave reflects passive filling of the atria during ventricular systole (Figs. 73-4 and 73-5).
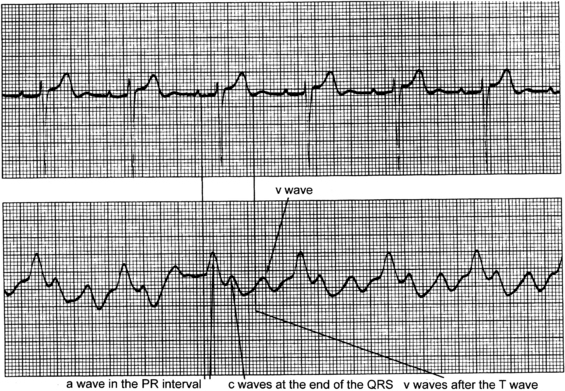
Figure 73-4 Identification of a, c, and v waves in the waveform for right atrial and central venous pressure (RA/CVP). Atrial waveforms are characterized by three components: a, c, and v waves. The a wave reflects atrial contraction, the c wave reflects closure of the tricuspid valve, and the v wave reflects passive filling of the atria. (From Ahrens TS, Taylor LK: Hemodynamic waveform analysis, Philadelphia, 1992, Saunders.)
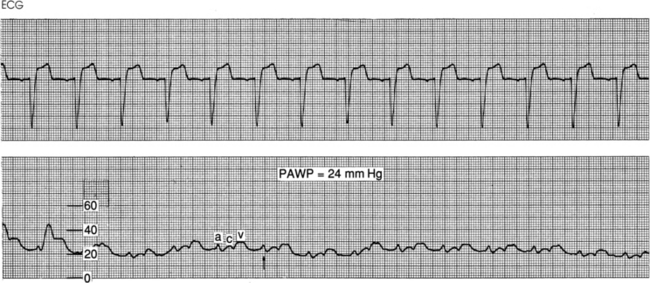
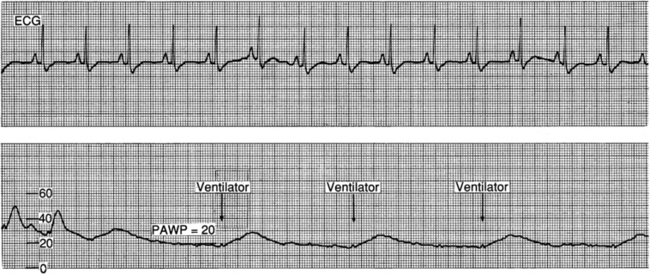
Figure 73-5 Normal pulmonary artery occlusion pressure (PAOP) waveform. Note the delay in the a, c, and v waves because of the time needed for the mechanical events to show a pressure change. This waveform is from a spontaneously breathing patient. The arrow indicates end expiration, where the mean of a wave pressure is measured. Pulmonary artery occlusion pressure (PAOP) is also referred to as pulmonary artery wedge pressure (PAWP).
 The a wave reflects right ventricular filling at end diastole. The mean of the a wave is determined by averaging the top and bottom values of the a wave.
The a wave reflects right ventricular filling at end diastole. The mean of the a wave is determined by averaging the top and bottom values of the a wave.
 Elevated a and v waves may be evident in right atrial pressure (RAP/central venous pressure [CVP]) and in PAOP waveforms. These elevations may occur in patients with cardiac tamponade, constrictive pericardial disease, and hypervolemia.
Elevated a and v waves may be evident in right atrial pressure (RAP/central venous pressure [CVP]) and in PAOP waveforms. These elevations may occur in patients with cardiac tamponade, constrictive pericardial disease, and hypervolemia.
 Elevated a waves in the RAP/CVP waveform may occur in patients with pulmonic or tricuspid stenosis, right ventricular ischemia or infarction, RV failure, pulmonary artery hypertension, and atrioventricular (AV) dissociation.
Elevated a waves in the RAP/CVP waveform may occur in patients with pulmonic or tricuspid stenosis, right ventricular ischemia or infarction, RV failure, pulmonary artery hypertension, and atrioventricular (AV) dissociation.
 Elevated a waves in the PAOP waveform may occur in patients with mitral stenosis, acute left ventricular ischemia or infarction, left ventricular failure, and AV dissociation.
Elevated a waves in the PAOP waveform may occur in patients with mitral stenosis, acute left ventricular ischemia or infarction, left ventricular failure, and AV dissociation.
 Elevated v waves in the RAP/CVP waveform may occur in patients with tricuspid insufficiency.
Elevated v waves in the RAP/CVP waveform may occur in patients with tricuspid insufficiency.
 Elevated v waves in the PAOP waveform may occur in patients with mitral insufficiency or a ruptured papillary muscle.
Elevated v waves in the PAOP waveform may occur in patients with mitral insufficiency or a ruptured papillary muscle.
• Insertion and placement verification should occur as follows:
 The PA catheter may be inserted through the subclavian, internal jugular, femoral, external jugular, or antecubital veins.
The PA catheter may be inserted through the subclavian, internal jugular, femoral, external jugular, or antecubital veins.
 The standard 7.5 Fr PA catheter is 110 cm long and has black markings at 10-cm increments and wide black markings at 50-cm increments for facilitation of insertion and positioning (see Fig. 73-1). The catheter should reach the PA after being advanced 40 to 55 cm from the internal jugular vein, 35 to 50 cm from the subclavian vein, 60 cm from the femoral vein, 70 cm from the right antecubital fossa, and 80 cm from the left antecubital fossa.
The standard 7.5 Fr PA catheter is 110 cm long and has black markings at 10-cm increments and wide black markings at 50-cm increments for facilitation of insertion and positioning (see Fig. 73-1). The catheter should reach the PA after being advanced 40 to 55 cm from the internal jugular vein, 35 to 50 cm from the subclavian vein, 60 cm from the femoral vein, 70 cm from the right antecubital fossa, and 80 cm from the left antecubital fossa.
 Verification of PA catheter placement is validated with waveform analysis. Correct catheter placement shows a PAO tracing when the balloon is inflated and a PA tracing when the balloon is deflated.
Verification of PA catheter placement is validated with waveform analysis. Correct catheter placement shows a PAO tracing when the balloon is inflated and a PA tracing when the balloon is deflated.
 Catheter placement is also verified with chest radiography.
Catheter placement is also verified with chest radiography.
 The PA catheter balloon contains latex, which may cause allergic reactions. Latex-free catheters are available.
The PA catheter balloon contains latex, which may cause allergic reactions. Latex-free catheters are available.
EQUIPMENT
• PA catheter (non–heparin-coated PA catheters and latex-free PA catheters are available)
• Percutaneous sheath introducer kit and sterile catheter sleeve
• Pressure modules and cables for interface with the monitor
• Cardiac output cable with a thermistor/injectate sensor
• Pressure transducer system, including flush solution recommended according to institution standard, a pressure bag or device, pressure tubing with transducers, and flush device
• Sterile normal saline intravenous (IV) solution for flushing of the introducer and catheter infusion ports
• Antiseptic solution (e.g., 2% chlorhexidine-based preparation)
• Head covers, fluid-shield masks, sterile gowns, sterile gloves, nonsterile gloves, and sterile drapes
• 1% lidocaine without epinephrine
• Sterile water or normal saline solution
• Chlorhexidine-impregnated sponge
• Stopcocks (may be included in some pressure tubing systems)
PATIENT AND FAMILY EDUCATION
• Provide the patient and family with information about the PA catheter, reason for the PA catheter, and explanation of the equipment.  Rationale: The patient and family understand the procedure, why it is needed, and how it will help manage care. Patient and family anxiety may decrease.
Rationale: The patient and family understand the procedure, why it is needed, and how it will help manage care. Patient and family anxiety may decrease.
• Explain the patient’s expected participation during the procedure.  Rationale: This explanation will encourage patient assistance.
Rationale: This explanation will encourage patient assistance.
PATIENT ASSESSMENT AND PREPARATION
Patient Assessment
• Determine baseline hemodynamic, cardiovascular, peripheral vascular, and neurovascular status.  Rationale: Assessment provides data that can be used for comparison with postinsertion assessment data and hemodynamic values.
Rationale: Assessment provides data that can be used for comparison with postinsertion assessment data and hemodynamic values.
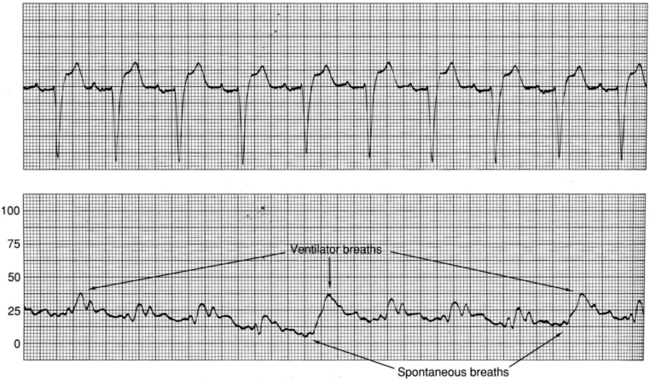
• Determine the patient’s baseline pulmonary status. If the patient is mechanically ventilated, note the type of support, ventilator mode, and presence or absence of positive end-expiratory pressure (PEEP) or continuous positive airway pressure (CPAP).  Rationale: The presence of mechanical ventilation alters hemodynamic waveforms and pressures.
Rationale: The presence of mechanical ventilation alters hemodynamic waveforms and pressures.
• Assess the patient’s medical history specifically related to problems with venous access sites, cardiac anatomy, and pulmonary anatomy.  Rationale: Identification of obstructions or disease should be made before the insertion attempt.
Rationale: Identification of obstructions or disease should be made before the insertion attempt.
• Assess the patient’s current laboratory profile, including electrolyte, coagulation, and arterial blood gas results.  Rationale: Laboratory abnormalities are identified. Baseline coagulation studies are helpful in determination of the risk for bleeding. Electrolyte and arterial blood gas imbalances may increase cardiac irritability.
Rationale: Laboratory abnormalities are identified. Baseline coagulation studies are helpful in determination of the risk for bleeding. Electrolyte and arterial blood gas imbalances may increase cardiac irritability.
Patient Preparation
• Verify correct patient with two identifiers.  Rationale: Prior to performing a procedure, the nurse should ensure the correct identification of the patient for the intended intervention.
Rationale: Prior to performing a procedure, the nurse should ensure the correct identification of the patient for the intended intervention.
• Ensure that the patient and family understand preprocedural teaching. Answer questions as they arise, and reinforce information as needed.  Rationale: Understanding of previously taught information is evaluated and reinforced.
Rationale: Understanding of previously taught information is evaluated and reinforced.
• Ensure that informed consent has been obtained.  Rationale: Informed consent protects the rights of the patient and makes a competent decision possible for the patient.
Rationale: Informed consent protects the rights of the patient and makes a competent decision possible for the patient.
• Perform a pre-procedure verification and time out, if non-emergent.  Rationale: Ensures patient safety.
Rationale: Ensures patient safety.
• Validate the patency of the peripheral IV line.  Rationale: Access may be needed for administration of emergency medications or fluids.
Rationale: Access may be needed for administration of emergency medications or fluids.
• Assist the patient to the supine position.  Rationale: This position prepares the patient for skin preparation, catheter insertion, and setup of the sterile field.
Rationale: This position prepares the patient for skin preparation, catheter insertion, and setup of the sterile field.
• Sedate the patient or provide prescribed analgesics as needed.  Rationale: Movement of the patient may inhibit insertion of the PA catheter.
Rationale: Movement of the patient may inhibit insertion of the PA catheter.
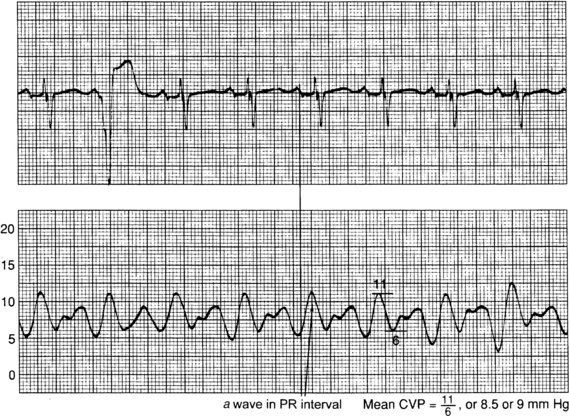
Figure 73-8 Obtaining measurements of right atrial and central venous pressures (RA/CVP). Aligning the a wave on the RA/CVP waveform with the PR interval on the electrocardiogram facilitates accurate measurement of RA/CVP at end diastole. (From Ahrens TS, Taylor LK: Hemodynamic waveform analysis, Philadelphia, 1992, Saunders.)
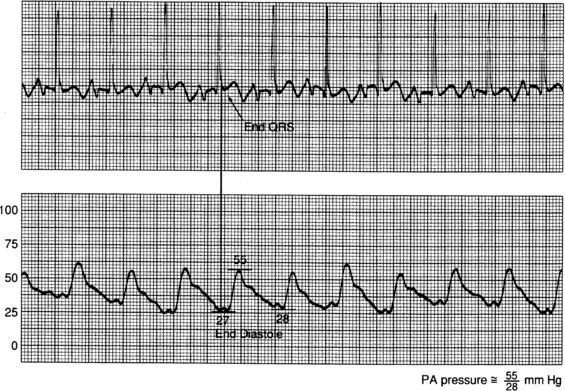
Figure 73-9 Obtaining measurements of pressure in the pulmonary artery (PA). For systolic pressure, align the peak of the systolic waveform with the QT interval on the electrocardiogram (ECG). For PA diastolic pressure, use the end of the QRS as a marker to detect the PA diastolic phase. Obtain the reading just before the upstroke of the systolic waveform. (From Ahrens TS, Taylor LK: Hemodynamic waveform analysis, Philadelphia, 1992, Saunders.)
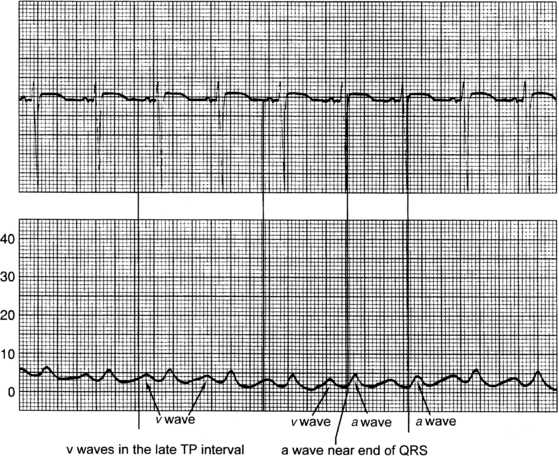
Figure 73-11 Obtaining measurement of the pulmonary artery occlusion pressure (PAOP). For accurate readings, align the a wave from the PAO waveform with the end of the QRS on the electrocardiogram (ECG) at end diastole. Pulmonary artery occlusion pressure (PAOP) is also referred to as pulmonary artery wedge pressure (PAWP). (From Ahrens TS, Taylor LK: Hemodynamic waveform analysis, Philadelphia, 1992, Saunders.)
References
![]() 1. Blanch, L, et al. Short term effects of prone position in critically ill patients with acute respiratory distress syndrome. Intensive Care Med. 1997; 23:1033–1039.
1. Blanch, L, et al. Short term effects of prone position in critically ill patients with acute respiratory distress syndrome. Intensive Care Med. 1997; 23:1033–1039.
![]() 2. Brussel, T, et al. Mechanical ventilation in the prone position for acute respiratory failure after cardiac surgery. J Cardiothorac Vasc Anesth. 1993; 7:541–546.
2. Brussel, T, et al. Mechanical ventilation in the prone position for acute respiratory failure after cardiac surgery. J Cardiothorac Vasc Anesth. 1993; 7:541–546.
![]() 3. Cason, CL, et al. Effects of backrest elevation and position on pulmonary artery pressures. Cardiovasc Nurs. 1990; 26:1–5.
3. Cason, CL, et al. Effects of backrest elevation and position on pulmonary artery pressures. Cardiovasc Nurs. 1990; 26:1–5.
![]() 4. Chen, Y, et al. Comparison between replacement at 4 days and 7 days on the infection rate for pulmonary artery catheters in an intensive care unit. Crit Care Med. 2003; 31:1353–1358.
4. Chen, Y, et al. Comparison between replacement at 4 days and 7 days on the infection rate for pulmonary artery catheters in an intensive care unit. Crit Care Med. 2003; 31:1353–1358.
![]() 5. Chong, BH. Heparin-induced thrombocytopenia. Br J Haematol. 1995; 89:431–439.
5. Chong, BH. Heparin-induced thrombocytopenia. Br J Haematol. 1995; 89:431–439.
![]() 6. Chulay, M, Miller, T. The effect of backrest elevation on pulmonary artery and pulmonary capillary wedge pressures in patients after cardiac surgery. Heart Lung. 1984; 13:138–140.
6. Chulay, M, Miller, T. The effect of backrest elevation on pulmonary artery and pulmonary capillary wedge pressures in patients after cardiac surgery. Heart Lung. 1984; 13:138–140.
![]() 7. Clochesy, J, Hinshaw, AD, Otto, CW. Effects of change of position on pulmonary artery and pulmonary capillary wedge pressure in mechanically ventilated patients. NITA. 1984; 7:223–225.
7. Clochesy, J, Hinshaw, AD, Otto, CW. Effects of change of position on pulmonary artery and pulmonary capillary wedge pressure in mechanically ventilated patients. NITA. 1984; 7:223–225.
![]() 8. Cohen, Y, et al, The “hands-off” catheter in the prevention of systemic infections associated with pulmonary artery catheter. a prospective study. Am J Respir Crit Care Med 1998; 157:284–287.
8. Cohen, Y, et al, The “hands-off” catheter in the prevention of systemic infections associated with pulmonary artery catheter. a prospective study. Am J Respir Crit Care Med 1998; 157:284–287.
![]() 9. Daily, EK, Schroeder, JS. Techniques in bedside hemodynamic monitoring,, ed 5. St Louis: Mosby; 1994.
9. Daily, EK, Schroeder, JS. Techniques in bedside hemodynamic monitoring,, ed 5. St Louis: Mosby; 1994.
![]() 10. Dobbin, K, et al, Pulmonary artery pressure measurement in patients with elevated pressures. effect of backrest elevation and method of measurement. Am J Crit Care 1992; 1:61–69.
10. Dobbin, K, et al, Pulmonary artery pressure measurement in patients with elevated pressures. effect of backrest elevation and method of measurement. Am J Crit Care 1992; 1:61–69.
![]() 11. Eyer, S, et al, Catheter-related sepsis. prospective, randomized study of three methods of long-term catheter maintenance. Crit Care Med 1990; 18:1073–1079.
11. Eyer, S, et al, Catheter-related sepsis. prospective, randomized study of three methods of long-term catheter maintenance. Crit Care Med 1990; 18:1073–1079.
![]() 12. Fridrich, P, et al. The effects of long-term prone positioning in patients with trauma-induced adult respiratory distress syndrome. Anesth Analg. 1996; 83:1206–1211.
12. Fridrich, P, et al. The effects of long-term prone positioning in patients with trauma-induced adult respiratory distress syndrome. Anesth Analg. 1996; 83:1206–1211.
![]() 13. Groom, L, Frisch, SR, Elliot, M. Reproducibility and accuracy of pulmonary artery pressure measurement in supine and lateral positions. Heart Lung. 1990; 19:147–151.
13. Groom, L, Frisch, SR, Elliot, M. Reproducibility and accuracy of pulmonary artery pressure measurement in supine and lateral positions. Heart Lung. 1990; 19:147–151.
![]() 14. Hannan, AT, Brown, M, Bigman, O. Pulmonary artery catheter induced hemorrhage. Chest. 1984; 85:128–131.
14. Hannan, AT, Brown, M, Bigman, O. Pulmonary artery catheter induced hemorrhage. Chest. 1984; 85:128–131.
15. Infusion Nurses Society. Infusion nursing standards of practice. J Infusion Nurs. 2006; 29:S1–S92.
![]() 16. Jolliet, P, Bulpa, P, Chevrolet, JC. Effects of prone position on gas exchange and hemodynamics in severe acute respiratory distress syndrome. Crit Care Med. 1998; 26:1977–1985.
16. Jolliet, P, Bulpa, P, Chevrolet, JC. Effects of prone position on gas exchange and hemodynamics in severe acute respiratory distress syndrome. Crit Care Med. 1998; 26:1977–1985.
![]() 17. Keating, D, et al. Effect of sidelying positions on pulmonary artery pressures. Heart Lung. 1986; 15:605–610.
17. Keating, D, et al. Effect of sidelying positions on pulmonary artery pressures. Heart Lung. 1986; 15:605–610.
![]() 18. Kennedy, GT, Bryant, A, Crawford, MH. The effects of lateral body positioning on measurements of pulmonary artery and pulmonary wedge pressures. Heart Lung. 1984; 13:155–158.
18. Kennedy, GT, Bryant, A, Crawford, MH. The effects of lateral body positioning on measurements of pulmonary artery and pulmonary wedge pressures. Heart Lung. 1984; 13:155–158.
![]() 19. Lambert, CW, Cason, CL, Backrest elevation and pulmonary artery pressures. research analysis. Dimens Crit Care Nurs 1990; 9:327–335.
19. Lambert, CW, Cason, CL, Backrest elevation and pulmonary artery pressures. research analysis. Dimens Crit Care Nurs 1990; 9:327–335.
![]() 20. Langer, M, et al. The prone position in ARDS patients. Chest. 1988; 94:103–107.
20. Langer, M, et al. The prone position in ARDS patients. Chest. 1988; 94:103–107.
![]() 21. Laulive, JL. Pulmonary artery pressures and position changes in the critically ill adult. Dimens Crit Care Nurs. 1982; 1:28–34.
21. Laulive, JL. Pulmonary artery pressures and position changes in the critically ill adult. Dimens Crit Care Nurs. 1982; 1:28–34.
![]() 22. Luskin, RL, et al, Extended use of disposable pressure transducers. a bacteriologic evaluation. JAMA 1986; 255:916–920.
22. Luskin, RL, et al, Extended use of disposable pressure transducers. a bacteriologic evaluation. JAMA 1986; 255:916–920.
![]() 23. O’Grady, NP, et al. Guidelines for the prevention of intravascular catheter-related infections. Am J Infect Control. 2002; 30:476–489.
23. O’Grady, NP, et al. Guidelines for the prevention of intravascular catheter-related infections. Am J Infect Control. 2002; 30:476–489.
![]() 24. O’Malley, MK, et al. Value of routine pressure monitoring system changes after 72 hours of use. Crit Care Med. 1994; 22:1424–1430.
24. O’Malley, MK, et al. Value of routine pressure monitoring system changes after 72 hours of use. Crit Care Med. 1994; 22:1424–1430.
![]() 25. Osika, C, Measurement of pulmonary artery pressures. supine verses side-lying head-elevated positions. Heart Lung 1989; 18:298–299.
25. Osika, C, Measurement of pulmonary artery pressures. supine verses side-lying head-elevated positions. Heart Lung 1989; 18:298–299.
![]() 26. Pappert, D, et al. Influence of positioning on ventilation-perfusion relationships in severe adult respiratory distress syndrome. Chest. 1994; 106:1511–1516.
26. Pappert, D, et al. Influence of positioning on ventilation-perfusion relationships in severe adult respiratory distress syndrome. Chest. 1994; 106:1511–1516.
![]() 27. Pelosi, P, et al. Effects of the prone position on respiratory mechanics and gas exchange during acute lung injury. Am J Respir Crit Care Med. 1998; 157:387–393.
27. Pelosi, P, et al. Effects of the prone position on respiratory mechanics and gas exchange during acute lung injury. Am J Respir Crit Care Med. 1998; 157:387–393.
![]() 28. Randolph, AG, et al. Benefit of heparin in central venous and pulmonary artery catheters. Chest. 1998; 113:165–171.
28. Randolph, AG, et al. Benefit of heparin in central venous and pulmonary artery catheters. Chest. 1998; 113:165–171.
![]() 29. Voggenreiter, G, et al. Intermittent prone positioning in the treatment of severe and moderate posttraumatic lung injury. Crit Care Med. 1999; 27:2375–2382.
29. Voggenreiter, G, et al. Intermittent prone positioning in the treatment of severe and moderate posttraumatic lung injury. Crit Care Med. 1999; 27:2375–2382.
![]() 30. Vollman, KM, Bander, JJ. Improved oxygenation utilizing a prone positioner in patients with acute respiratory distress syndrome. Intensive Care Med. 1996; 22:1105–1111.
30. Vollman, KM, Bander, JJ. Improved oxygenation utilizing a prone positioner in patients with acute respiratory distress syndrome. Intensive Care Med. 1996; 22:1105–1111.
![]() 31. Wild, L. Effect of lateral recumbent positions on measurement of pulmonary artery and pulmonary artery wedge pressures in critically ill adults. Heart Lung. 1984; 13:305.
31. Wild, L. Effect of lateral recumbent positions on measurement of pulmonary artery and pulmonary artery wedge pressures in critically ill adults. Heart Lung. 1984; 13:305.
![]() 32. Wilson, AE, et al. Effect of backrest position on hemodynamic and right ventricular measurements in critically ill adults. Am J Crit Care. 1996; 5:264–270.
32. Wilson, AE, et al. Effect of backrest position on hemodynamic and right ventricular measurements in critically ill adults. Am J Crit Care. 1996; 5:264–270.
![]() 33. Woods, SL, Mansfield, LW. Effect of body position upon pulmonary artery and pulmonary capillary wedge pressures in noncritically ill patients. Heart Lung. 1976; 5:83–90.
33. Woods, SL, Mansfield, LW. Effect of body position upon pulmonary artery and pulmonary capillary wedge pressures in noncritically ill patients. Heart Lung. 1976; 5:83–90.
![]() Abreu, AR, Campos, MA, Krieger, BP, Pulmonary artery rupture induced by a pulmonary artery catheter. a case report and review of the literature. J Intensive Care Med 2004; 19:291–296.
Abreu, AR, Campos, MA, Krieger, BP, Pulmonary artery rupture induced by a pulmonary artery catheter. a case report and review of the literature. J Intensive Care Med 2004; 19:291–296.
![]() Ahrens, TS, Taylor, LA. Hemodynamic waveform analysis. Philadelphia: Saunders; 1992.
Ahrens, TS, Taylor, LA. Hemodynamic waveform analysis. Philadelphia: Saunders; 1992.
![]() American Association of Critical-Care Nurses, AACN practice alert. pulmonary artery pressure measurement. www.aacn.org, 2004 [available at].
American Association of Critical-Care Nurses, AACN practice alert. pulmonary artery pressure measurement. www.aacn.org, 2004 [available at].
![]() Anonymous, Pulmonary artery catheter consensus conference. consensus statement. Crit Care Med 1997; 25:910–925.
Anonymous, Pulmonary artery catheter consensus conference. consensus statement. Crit Care Med 1997; 25:910–925.
![]() Bridges, EJ, Pulmonary artery pressure monitoring. when, how, and what else to use. AACN Adv Crit Care 2006; 17:286–305.
Bridges, EJ, Pulmonary artery pressure monitoring. when, how, and what else to use. AACN Adv Crit Care 2006; 17:286–305.
![]() Daily, EK. Hemodynamic waveform analysis. J Cardiovasc Nurs. 2001; 15:6–22,87-88.
Daily, EK. Hemodynamic waveform analysis. J Cardiovasc Nurs. 2001; 15:6–22,87-88.
![]() Darovic, GO, Hemodynamic monitoring. invasive and noninvasive clinical application,. ed 3. Saunders, Philadelphia, 2002.
Darovic, GO, Hemodynamic monitoring. invasive and noninvasive clinical application,. ed 3. Saunders, Philadelphia, 2002.
![]() Houghton, D, et al. Routine daily chest radiography in patients with pulmonary artery catheters. Am J Crit Care. 2002; 11:261–265.
Houghton, D, et al. Routine daily chest radiography in patients with pulmonary artery catheters. Am J Crit Care. 2002; 11:261–265.
![]() Keckeisen, M, Protocols for practice. hemodynamic monitoring seriespulmonary artery pressure monitoring,. American Association of Critical-Care Nurses, Aliso Viejo, CA, 1997.
Keckeisen, M, Protocols for practice. hemodynamic monitoring seriespulmonary artery pressure monitoring,. American Association of Critical-Care Nurses, Aliso Viejo, CA, 1997.
![]() Liu, C, Webb C, From the Food and Drug Administration pulmonary artery rupture. serious complication associated with pulmonary artery catheters. Int J Trauma Nurs 2000; 6:19–26.
Liu, C, Webb C, From the Food and Drug Administration pulmonary artery rupture. serious complication associated with pulmonary artery catheters. Int J Trauma Nurs 2000; 6:19–26.
![]() Ott, K, Johnson, K, Ahrens, T. New technologies in the assessment of hemodynamic parameters. J Cardiovasc Nurs. 2001; 15:41–55.
Ott, K, Johnson, K, Ahrens, T. New technologies in the assessment of hemodynamic parameters. J Cardiovasc Nurs. 2001; 15:41–55.
![]() Quaal, SJ. Improving the accuracy of pulmonary artery catheter measurement. J Cardiovasc Nurs. 2001; 15:71–82.
Quaal, SJ. Improving the accuracy of pulmonary artery catheter measurement. J Cardiovasc Nurs. 2001; 15:71–82.
![]() Quaal, SJ. Is it necessary to perform a square wave test routinely to test for accuracy in hemodynamic monitoring? Or is it recommended only if there is a problem. Crit Care Nurse. 1995; 15:92–93.
Quaal, SJ. Is it necessary to perform a square wave test routinely to test for accuracy in hemodynamic monitoring? Or is it recommended only if there is a problem. Crit Care Nurse. 1995; 15:92–93.
Rauen, CA, Flynn, MB, Bridges, E, Evidence-based practice habits. transforming research into bedside practice. Crit Care Nurse 2009; 29:46–59.
Rizvi, K, et al. Effect of airway pressure display on interobserver agreement in the assessment of vascular pressures in patients with acute lung injury and acute respiratory distress syndrome. Crit Care Med. 2005; 33:98–103.