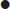Pain relief
Edited by Anthony Brown
22.1 General pain management
Daniel M Fatovich
Introduction
Pain is defined by the International Association for the Study of Pain as: ‘An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage’ [1]. Acute pain is defined as: ‘Pain of recent onset and probable limited duration. It usually has an identifiable temporal and causal relationship to injury or disease’ [2]. However, once a patient presents for medical care, severe acute pain has ceased to serve a useful purpose. Whereas in some conditions the nature and progression of the pain may be helpful in making the diagnosis of the underlying pathology, too great a reliance has been placed upon this feature, thereby allowing the patient to suffer needlessly for prolonged periods [2,3].
When severe pain is inadequately relieved it produces pathophysiological and abnormal psychological reactions that often lead to complications. This is important as acute pain is the most common presenting complaint to an emergency department (ED) [4] and its management forms part of the daily practice of emergency medicine. It should be considered poor patient care not to treat pain while attempting to arrive at a diagnosis. There can be no greater gift to one’s neighbour than to practise, teach and discover more effective methods to relieve pain and suffering [2,3]. Unfortunately, the management of acute pain is often not a specific component of medical training.
Physiology
Pain is one of the most complex aspects of an already intricate nervous system [2]. A number of theories have been developed to explain the physiology of pain, but none is proven or complete.
‘Gate Control Theory’
In 1965, the Melzack–Wall ‘Gate Control Theory’ emphasized mechanisms in the central nervous system that control the perception of a noxious stimulus and thus integrated afferent, upstream processes with downstream modulation from the brain [5]. However, this theory did not incorporate long-term changes in the central nervous system to the noxious input and to other external factors that impinge upon the individual [5].
Nociceptor function
Most pain originates when specific nerve endings (nociceptors) are stimulated, producing nerve impulses that are transmitted to the brain. Nociception is the detection of tissue damage by specialized transducers [5]. It is now recognized that nociceptor function is altered by the ‘inflammatory soup’ that characterizes a region of tissue injury [5]. The final pain experience is subject to a complex series of facilitatory and inhibitory events that precede pain awareness, such as past experience, anxiety or expectation [6].
There are two types of nociceptors [7]:
Once transduced into electrical stimuli, conduction of neuronal action potentials is dependent on voltage-gated sodium channels [2]. A number of chemicals are involved in the transmission of pain to the ascending pathways in the spinothalamic tract. These include substance P and calcitonin gene-related peptide, but many others have been identified [2,8,9]. Opioid receptors are present in the dorsal horn and it is thought that encephalins (endogenous opioid peptides) are neurotransmitters in the inhibitory interneurons [7].
Phospholipids released from damaged cell membranes trigger a cascade of reactions, culminating in the production of prostaglandins that sensitize nociceptors to other inflammatory mediators, such as histamine, serotonin and bradykinin [7].
The threshold for the perception of a painful stimulus is similar in everyone and may be lowered by certain chemicals, such as the mediators of inflammation. The discrete cognitive processes and pathways involved in the interpretation of painful stimuli remain a mystery. The cognitive and emotional reactions to a given painful stimulus are variable among individuals and may be affected by culture, personality, past experiences and underlying emotional state [2,5,10]. In addition, intense and ongoing stimuli further increase the excitability of dorsal horn neurons, leading to central sensitization [2]. With increased excitability of central nociceptive neurons, the threshold for activation is reduced and pain can occur in response to low intensity, previously non-painful stimuli known as allodynia [2]. Pain is a complex, multidimensional, subjective phenomenon [10].
Assessment of pain and pain scales
There is no truly objective measurement of pain. Doctors use a variety of methods for determining how much pain a patient feels. These include the nature of the illness or injury, the patient’s appearance and behaviour and physiological concomitants. None of these is reliable.
Pain scales have been developed because there are no accurate physiological or clinical signs to measure pain objectively. Three scales have become popular tools to quantify pain intensity [11,12]: the visual analogue scale (VAS), the numeric rating scale and the verbal rating scale.
Visual analogue scale
The VAS usually consists of a 100-mm line with one end indicating ‘no pain’ and the other end indicating the ‘worst pain imaginable’. The patient simply indicates a point on the line that best indicates the amount of pain experienced. The minimum clinically significant change in patient pain severity measured with a 100-mm visual analogue scale is 13 mm [13]. Studies of pain experience that report less than a 13 mm change in pain severity, although statistically significant, may have no clinical importance [13].
Numeric rating scale
The patient is asked in the numeric rating scale to choose a number from a range (usually 0–10) that best describes the amount of pain experienced, with zero being ‘no pain’ and 10 being the ‘worst pain imaginable’. This has been used for cardiac ischaemia pain and is also useful in illiterate patients.
Verbal rating scale
The verbal rating scale simply asks a patient to choose a phrase that best describes the pain, usually ‘mild’, ‘moderate’ or ‘severe’.
The use of pain scales has been restricted predominantly to research, where experimental pain is not associated with the strong emotional component of acute pain. In the clinical setting, anxiety, sleep disruption and illness burden are present [9]. It is difficult to use a unidimensional pain scale to measure a multidimensional process. Using pain intensity alone will often fail to capture the many other qualities of pain and the overall pain experience. The best illustration of this problem is that the same pain stimulus can be applied to two different people with dramatically different pain scores and analgesic requirements [14]. At best, the use of pain scales is an indirect reflection of ‘real’ pain, with patient self-reporting still being the most reliable indicator of the existence and intensity of pain [15].
Nevertheless, pain scales are simple and easy to use. They are now routine in many EDs, often being a standard part of the triage process, which leads to substantially faster provision of initial analgesia [4] (see Vazirani J, Knott JC in Further reading).
General principles
Patients in pain should receive timely, effective and appropriate analgesia, titrated according to response [2]. Therefore, essentially there is no role for the intramuscular route for parenteral analgesia, which simply delays the onset of analgesia. The following points should be stressed:
Specific agents
Opioids
The term ‘opioid’ refers to all naturally occurring and synthetic drugs producing morphine-like effects. Morphine is the standard opioid agonist against which others are judged [16]. These drugs are the most powerful agents available in the treatment of acute pain. A number of specific opioid receptors have been identified.
Opioid receptor effects
Opiods are responsible for a variety of effects, including analgesia, euphoria, respiratory depression and miosis (μ receptor); cough suppression and sedation (Κ); dysphoria and hallucinations (σ); nausea and vomiting, and pruritus (δ) [7]. Opioids act on injured tissue to reduce inflammation in the dorsal horn to impede transmission of nociception and supraspinally to activate inhibitory pathways that descend to the spinal segment [9].
Unfortunately, many doctors use opioids inappropriately as there are particular concerns regarding the risks of respiratory depression and inducing iatrogenic addiction. Less than 1% of patients who receive opioids for pain develop respiratory depression [17]. Tolerance to this side effect develops simultaneously with tolerance to the analgesic effect. If the opioid dose is increased so that at least half the pain is relieved, the chance of respiratory depression is small. Further, naloxone will reverse the effects of opioids. In relation to fears of addiction, large studies have shown that inducing this following opioid analgesia use is exceedingly rare [18].
Use of intravenous opiods
From a clinical practice point of view, many patients who require intravenous opioid will also require admission to hospital, as there will be ongoing opioid requirements that can only be administered in hospital. There have been occasions where patients have received opioid analgesia that has relieved their pain and they have then been discharged without a final diagnosis. This is an unacceptable practice. A patient may present with abdominal pain with vomiting and, for instance, a provisional diagnosis of gastroenteritis is made. After opioid analgesia is given the patient may feel better and be discharged. A diagnosis, such as appendicitis or bowel obstruction, has not been excluded.
It is therefore necessary for patients to have an appropriate diagnostic evaluation to confirm a benign cause and to reassess the patient after the opioid effects have waned. For patients in whom the final diagnosis is certain, such as in anterior shoulder dislocation, discharge is appropriate after a suitable period of observation until the patient is deemed clinically fit for discharge. This is a different scenario from that described previously, as it is a single system problem in which there is no doubt about the diagnosis. In summary, pain that is considered severe enough to warrant intravenous opioid analgesia requires a high index of suspicion for significant pathology.
Side effects
All potent opioid analgesics have the potential to depress the level of consciousness, protective reflexes and vital functions. It is mandatory that these are closely monitored during and after administration [7]. Specific side effects include:
 respiratory depression: rare<1%
respiratory depression: rare<1%
 nausea and vomiting: nausea occurs in approximately 40% and vomiting in 15% [7]
nausea and vomiting: nausea occurs in approximately 40% and vomiting in 15% [7]
 hypotension: opioids may provoke histamine release
hypotension: opioids may provoke histamine release
 spasm of the sphincter of Oddi, therefore patients with biliary colic may initially experience more pain. There is no good evidence to suggest that pethidine has any clinically significant advantage at equi-analgesic doses over other opioids for biliary or renal colic [16]
spasm of the sphincter of Oddi, therefore patients with biliary colic may initially experience more pain. There is no good evidence to suggest that pethidine has any clinically significant advantage at equi-analgesic doses over other opioids for biliary or renal colic [16]
Routes of administration
Opioids may be administered by many routes, including oral, subcutaneous, intramuscular, intravenous, epidural, nebulized, intrapleural, intranasal, intra-articular and transdermal. All may have a role in a specific clinical situation [4]. There is a good rationale for the use of the intravenous route in moderate-to-severe pain [4] and titration of intravenous opioids remains the standard of care for acute severe pain.
Opioid analgesics
Morphine
The standard intravenous morphine dose is 0.1–0.2 mg/kg or more with a duration of action of 2–3 h. This should be initiated as a loading dose of opioid to provide rapid initial pain relief aiming for an optimal balance between effective pain relief and minimal side effects. This means tailoring the approach to each individual patient. Thus, a young fit healthy man with renal colic may require an initial bolus of 0.1 mg/kg morphine, followed by further increments of 0.05 mg/kg. Conversely, a frail elderly patient may only tolerate 1.0–2.5 mg morphine total to begin with. There may also be considerable inter-individual variation in response to analgesia. Procedural pain may require higher-dose opioid analgesia, which has been found to be well tolerated and safe [19]. Appropriate monitoring and resuscitation equipment should be available to maximize safety.
Rapid pain relief and titration to effect are obvious advantages. Intramuscular administration results in unreliable and variable absorption and older routine practices, such as prescribing ‘5–10 mg morphine IM’, take no account of an individual’s requirements [7].
Pethidine
Pethidine should be used with caution in patients with renal failure, as there is increased risk of central nervous system toxicity due to the toxic metabolite, norpethidine. Norpethidine causes tremor, twitching, agitation and convulsions [16]. Also pethidine is contraindicated in patients receiving monoamine oxidase (MAO) inhibitors, as they interfere with pethidine metabolism, increasing the likelihood of toxicity [20]. Finally, pethidine may trigger the serotonin syndrome if used concomitantly with selective serotonin reuptake inhibitors (SSRIs).
Pethidine has approximately one-eighth the potency of morphine and causes the same degree of bronchospasm and increased biliary pressure as morphine [2]. Its use has now declined and it should continue to be discouraged in favour of other opioids [2].
Fentanyl
Allergic reactions are extremely rare with opioids. Fentanyl does not release histamine, making it ideal for treating patients with reactive airways disease. There are advantages in using fentanyl for brief procedures in the ED because of its short half-life. The intravenous dose of fentanyl is 1–2 μg/kg or more with a duration of action of 30–60 min. High doses of fentanyl may produce muscular rigidity, which may be so severe as to make ventilation difficult, but which responds to naloxone or muscle relaxants. Intranasal fentanyl is an effective analgesic in the ED and in the pre-hospital setting [2].
Oral opioids
Oral opioids tend to be underused in the ED, but are effective for all levels of pain and are associated with improved patient satisfaction. Their side effect profile may be better than paracetamol/codeine combinations. Oxycodone (immediate release) reaches peak levels at 45 min to 1 h but the dose should be reduced and dosing interval increased in the elderly and in those with hepatic or renal dysfunction. The main contraindication is acute respiratory depression. The initial dose is 5–10 mg. However, it is important to be aware that the increased prescribing of oral opioids is associated with increased deaths. Oxycodone/ naloxone combinations are now available.
Codeine
Codeine is the most commonly used oral opioid prodrug. Unfortunately, up to 6–10% of the Caucasian population, 2% of Asians, and 1% of Arabs have poorly functional cytochrome P450 2D6 (CYP2D6), which may render codeine largely ineffective for analgesia in these patients, although some analgesic efficacy may occur via alternate cytochrome P450 pathways.
Prescribed alone in doses as high as 120 mg, codeine has been demonstrated to be no more effective than placebo in both the adult and geriatric populations, while causing increasing gastrointestinal side effects, such as nausea, vomiting and constipation, with increasing doses [4]. It is frequently given in combination with paracetamol or aspirin.
Tramadol
Tramadol is a new opioid, with novel non-opioid properties [21]. Its efficacy lies between codeine and morphine. It has a relative lack of serious side effects, such as respiratory depression, and the potential for abuse and psychological dependence is low [21]. However, other side effects, such as nausea, vomiting, dizziness and somnolence, may be troublesome and there is a risk of seizures [21,22]. Thus, it should be avoided or used with caution in patients who are taking drugs that reduce the seizure threshold, such as tricyclic antidepressants and SSRIs. Also the concomitant administration of tramadol with monoamine oxidase inhibitors, or within 2 weeks of their withdrawal, is contraindicated [21].
The role of tramadol in emergency medicine is ill defined. One review concluded that tramadol does not offer any particular benefits over existing analgesics for the majority of emergency pain relief situations [22], with oral doses having equivalent analgesic effects in mild-to-moderate severity acute pain compared with currently available analgesics [22]. Intravenous tramadol is less effective than intravenous morphine [22].
However, tramadol may be useful in certain situations: [22]
Non-opioid analgesics
Simple analgesics
Non-steroidal anti-inflammatory drugs
Non-steroidal anti-inflammatory drugs are either non-selective cyclo-oxygenase (COX) inhibitors or selective inhibitors of COX-2 (COX-2 inhibitors). NSAIDs are effective analgesic agents for moderate pain, specifically when there is associated inflammation [4]. As with opioids, there are multiple routes of administration available. Unfortunately, their use in acute severe pain is limited by the length of onset time of 20–30 min. There is no clear superiority of one agent over another.
There is up to a 30% incidence of upper gastrointestinal bleeding when NSAIDs are used for over 1–2 weeks. The risk of bleeding in the elderly for short (3–5 days) acute therapy appears to be minimal [4]. NSAID use in pregnancy (especially late) is not recommended. Ibuprofen is considered the NSAID of choice in lactation.
NSAIDs have a spectrum of analgesic, anti-inflammatory and antipyretic effects and are effective analgesics in a variety of pain states [2]. Unfortunately, significant contraindications and adverse effects limit the use of NSAIDs, many of these being regulated by COX-1 [2]. NSAIDs are useful analgesic adjuncts and hence NSAIDs are therefore integral components of multimodal analgesia [2]. NSAID side effects are more common with long-term use. The main concerns are renal impairment, interference with platelet function, peptic ulceration and bronchospasm in individuals who have aspirin-exacerbated respiratory disease [2]. In general, the risk and severity of NSAID-associated side effects is increased in elderly people [2].
Caution is therefore needed in the elderly and in patients with renal disease, hypertension and heart failure, or with asthma. NSAIDs reduce renal cortical blood flow and may induce renal impairment, especially when used in patients already on diuretics. In patients with asthma, 2–20% are aspirin sensitive and there is a 50–100% cross-sensitivity with NSAIDs.
Ketorolac is a parenteral NSAID that is equipotent to opioids, with ketorolac and morphine equivalent in reducing pain. There is a benefit favouring ketorolac in terms of side effects when ketorolac is titrated intravenously for isolated limb injuries [23,24]. However, the utility of ketorolac in acute pain is limited due to a prolonged onset of action and a significant number of patients (25%) who exhibit little or no response [25]. There is also benefit to using ketorolac for acute renal colic [23,26]. A combination of morphine and ketorolac offered pain relief superior to either drug alone and was associated with a decreased requirement for rescue analgesia in patients with renal colic [27]. Rectal NSAIDs (e.g. indomethacin 100 mg) are an effective alternative to parenteral NSAIDs in the treatment of renal colic.
Paracetamol
Paracetamol is an effective analgesic for acute pain [2] and has useful antipyretic activity [28]. The addition of an NSAID further improves efficacy [2]. Paracetamol inhibits prostaglandin synthetase in the hypothalamus, prevents release of spinal prostaglandin and inhibits inducible nitric oxide synthesis in macrophages [28].
Indications for paracetamol include mild pain, particularly of soft tissue and musculoskeletal origin, mild procedural pain, supplementation of opioids in the management of more severe pain allowing a reduction in opioid dosage and as an alternative to aspirin [28]. Paracetamol has no gastrointestinal side effects of note and may be prescribed safely in patients with peptic ulcer disease or gastritis [4]. Aspirin has the risk of gastrointestinal side effects, such as ulceration and bleeding. It also has an antiplatelet effect which lasts for the life of the platelet.
Paracetamol is rapidly absorbed with a peak concentration reached in 30–90 min [28]. The recommended adult dose is 1 g every 4–6 h to a generally accepted maximum of 4 g per day [28]. Paracetamol has a low adverse event profile and is an excellent analgesic, especially when used in adequate dose. Parenteral paracetamol is now available and may have additional utility, e.g. in the vomiting patient. Chronic use of paracetamol alone does not seem to cause analgesic nephropathy [28]. It can be used safely in alcoholics and patients with liver metastases [28,29].
Combination drugs
Non-opioid agents, e.g. paracetamol, NSAIDs and paracetamol/codeine combinations, are all useful analgesics for mild-to-moderate pain. A systematic review found that paracetamol–codeine combinations in single dose studies produce a slightly increased analgesic effect (5%) compared with paracetamol alone [30]. However, none of the studies reviewed were based in the ED. In multidosage, paracetamol–codeine preparations have significantly increased side effects [30]. However, other reports state that the combination of paracetamol 1000 mg plus codeine 60 mg has a number needed to treat of 2.2 [2]. NSAIDs have a higher rate of serious adverse effects.
Other analgesic agents
Nitrous oxide
Nitrous oxide is an inhalational analgesic and sedative which, in a 50% mixture with oxygen (Entonox), has equivalent potency to 10 mg morphine in an adult [7]. The Entonox delivery system uses a preferential inhalational demand arrangement for self-administration, which requires an airtight fit between the mask/mouthpiece and face. As the patient holds the mask/mouthpiece, their grip will relax if drowsiness occurs, the airtight seal will be lost and the gas flow stops, thereby avoiding overdosage.
This system requires a degree of patient involvement and cooperation and is useful for patients who have difficult intravenous access or are needle-phobic. Patients who are elderly, young, confused or uncooperative will not find the technique effective. Nitrous oxide increases the volume of a pneumothorax or any other gas-filled cavity, so is contraindicated in patients with pneumothorax or pneumoperitoneum.
Ketamine
Ketamine is an N-methyl-D-aspartate (NMDA) antagonist. It is a unique anaesthetic that induces a state of dissociation between the cortical and limbic systems to produce a state of dissociative anaesthesia, with analgesia, amnesia, mild sedation and immobilization. It does not impair protective airway reflexes and random or purposeful movements are frequently observed in patients after administration. Side effects include hypersalivation, vomiting, emergence reactions, nightmares, laryngospasm, hypertension, tachycardia and increased intracranial pressure [31,32].
There are many potential contraindications to ketamine use including upper or lower respiratory infection, procedures involving the posterior pharynx, cystic fibrosis, age younger than 3 months, head injury, increased intracranial pressure, acute glaucoma or globe penetration, uncontrolled hypertension, congestive cardiac failure, arterial aneurysm, acute intermittent porphyria and thyrotoxicosis [32]. Despite this, ketamine is used increasingly in the EDs as part of procedural sedation (see Chapter 22.3). It is also an effective analgesic at sub-dissociative doses especially for opioid resistant pain, e.g. 0.2–0.3 mg/kg bolus plus infusion at 0.2 mg/kg/h.
Pain relief in pregnancy
Non-pharmacological treatment options should be considered where possible for pain management in pregnancy, because most drugs cross the placenta [2]. Use of medications for pain in pregnancy should be guided by published recommendations [2]. Paracetamol is regarded as the analgesic of choice [2]. NSAIDs are used with caution in the last trimester of pregnancy and should be avoided after the 32nd week [2]. The use of NSAIDs is associated with increased risk of miscarriage [2]. Overall, the use of opioids to treat pain in pregnancy appears safe [2].
Non-pharmacological therapies
Although pain perception involves neuroanatomical processes, the other interrelated component of pain reaction is psychophysiological. The use of non-pharmacological techniques is therefore important. These include empathy, a compassionate approach, a calm manner and reassurance. Immobilization of fractures with splinting is effective, as is the application of ice to a wound. Other techniques, such as hypnosis, transcutaneous nerve stimulation, acupuncture and manipulation, have not been widely studied in the ED setting.
Special pain situations and non-analgesic agents
This chapter has focused on specific analgesic agents, but there are many miscellaneous agents that are effective in providing disease-specific analgesia.
 glyceryl trinitrate and β-blockers for acute cardiac ischaemia pain
glyceryl trinitrate and β-blockers for acute cardiac ischaemia pain
 antiviral agents for herpes zoster
antiviral agents for herpes zoster
 oxygen therapy for cluster headache
oxygen therapy for cluster headache
In addition, adjuvant therapy with anxiolytics, such as midazolam, contributes to pain relief. Obtaining a definitive diagnosis allows directed therapy that contributes to pain relief. If specific treatments appear to be ineffective, then the diagnosis should be reconsidered.
Acute neuropathic pain
Acute neuropathic pain is an important issue in the ED. This may be due to conditions such as sciatica and cervical radiculopathy. In addition to agents, such as the antidepressants (e.g. nortriptylline) or anticonvulsants (e.g. carbamazepine), another option includes the use of antihyperalgesic drugs, such as gabapentin 100–300 mg per dose, repeated as necessary, titrating up to a maximum of 3600 mg per day over time. The main side effects are dizziness, somnolence and ataxia. However, there have been no ED studies of gabapentin or pregabalin and there is wide variability of response.
Chronic pain
Chronic pain ‘commonly persists beyond the time of healing of an injury and frequently there may not be any clearly identifiable cause’ [2]. Patients with chronic pain attend the ED with exacerbations of their chronic pain and are often taking multimodal therapies prescribed by a pain specialist. The main difference between acute and chronic pain is that, in chronic pain, central sensitization is the main underlying pathophysiology [33]. It is important to avoid a judgemental attitude to these patients as there is a risk of overlooking serious pathology.
Antihyperalgesic drugs in the setting of chronic pain, especially ketamine, are of particular value in those with poor opioid responsiveness [2]. Other antihyperalgesics may be useful for neuropathic pain, such as gabapentin and pregabalin.
Another issue with chronic pain is to be aware of adjuvant therapies for decreasing the likelihood of chronic pain developing. For example, early management of acute zoster infection may reduce the incidence of post-herpetic neuralgia [2]. Aciclovir given within 72 h of onset of the rash accelerates the resolution of pain and reduces the risk of post-herpetic neuralgia [2]. Amitriptyline 25 mg daily in patients over 60 years for 90 days, started at the onset of acute zoster, reduces pain prevalence at 6 months post-zoster infection [34].
Acute abdomen
Traditionally, it was held that pain relief masks the clinical signs of pathology in the acute abdomen. Evidence from randomized controlled trials clearly shows that the early administration of opioids in patients with an acute abdomen does not reduce the detection rate of serious pathology and may actually facilitate the diagnosis. Thus, titrated opioid analgesia should never be withheld, certainly not pending surgical review. The effect of analgesia on physical signs cannot be used as a diagnostic test [35–37].
Likely developments over the next 5–10 years
22.2 Local anaesthesia
Anthony Brown and Tor NO Ercleve
Local anaesthesia
Local anaesthetic infiltration and nerve blocks should be used for patients presenting to the emergency department (ED) with pain, either to supplement other analgesia or for definitive pain relief. Nerve blocks are most appropriate when the pain is localized, as in certain fractures and wounds to a digit, or within a peripheral nerve distribution region. Local anaesthesia may also be used topically, particularly in children, and prior to arterial blood gas puncture and insertion of large intravenous cannulae where, contrary to popular perception, it does not increase the likelihood of failure [1,2].
Pharmacology
Local anaesthetic agents are all weak organic bases that inactivate intracellular fast sodium channels, temporarily blocking membrane depolarization and preventing nerve impulse transmission. All are vasodilators with the exception of ropivacaine and cocaine, hence the use of adrenaline to prolong their duration of activity and to improve safety by delaying absorption and/or by administering lower effective doses.
Amino ester and amino amide local anaesthetics
Local anaesthetic agents containing an ester bond between the intermediate chain and lipophilic aromatic end (amino esters) include cocaine, procaine and amethocaine. They are poorly protein bound and undergo hydrolysis by plasma pseudocholinesterase to para-amino benzoic acid. Amide-type agents containing an amide bond between the intermediate chain and aromatic end (amino amides) include lignocaine, prilocaine, bupivacaine and ropivacaine, are highly protein bound, much more stable and undergo hepatic metabolism.
Local anaesthetics are available in single or multidose vials, with or without dilute adrenaline at 1:200 000 (containing 5 μg adrenaline per millilitre) to prolong their duration of action. Antioxidants, such as sodium bisulphite or metabisulphite, are added to adrenaline-containing solutions and preservative, such as methylparaben, to multidose vials and are implicated in some apparent allergic reactions to the local anaesthetic. True allergy to local anaesthetics is extremely rare when verified by progressive challenge testing and is usually to the amino esters [3].
The duration of action of local anaesthetics is related to the degree of protein binding, vasoactivity, concentration and possibly pH, although the addition of adrenaline is the most practical way to prolong their effect. Table 22.2.1 gives standard maximum safe doses and duration of action of commonly used agents. Solutions containing adrenaline should not be injected near end arteries, such as in the fingers, toes, nose or penis, even though this well-established dogma is surprisingly not supported by the literature. Normal blood flow is restored to the digit within 60–90 min of inadvertent injection of local anaesthesia with adrenaline (epinephrine) at standard commercial dilutions, without any evidence of harm [4].
Table 22.2.1
Maximum recommended safe dose and duration of action of common local anaesthetics
| Drug | Dose (mg/kg)* | Duration (h) |
| Lignocaine | 3 | 0.5–1 |
| Lignocaine with adrenaline | 7 | 2–5 |
| Bupivacaine | 2 | 2–4 |
| Prilocaine | 6 | 0.5–1.5 |
Adverse effects
Systemic toxicity
Systemic toxicity occurs after unrecognized rapid intravenous injection or by exceeding the recommended safe maximum dose. Symptoms and signs of toxicity are related to plasma drug levels and progress from circumoral tingling, dizziness, tinnitus and visual disturbance to muscular twitching, confusion, convulsions, coma and apnoea. Cardiovascular effects are also seen with high plasma levels, including bradycardia, hypotension and cardiovascular collapse ultimately with ventricular fibrillation or asystole, which are all exacerbated by associated hypoxia. See Table 22.2.2 for the features of local anaesthetic toxicity related to increasing plasma levels.
Table 22.2.2
Features of systemic local anaesthetic toxicity (in order of increasing plasma levels)
Circumoral tingling
Dizziness
Tinnitus
Visual disturbance
Muscular twitching
Confusion
Convulsions
Coma
Apnoea
Cardiovascular collapse (highest plasma levels)
The management of systemic toxicity includes immediate cessation of the drug, summoning help, airway maintenance, supplemental oxygen and incremental doses of an intravenous benzodiazepine, such as midazolam 0.05–0.1 mg/kg, for seizures. Major reactions may require endotracheal intubation, fluids and cautious use of vasopressors and inotropes, as high doses can impede resuscitation in toxic cardiomyopathy. Refractory arrhythmias with cardiovascular collapse from local anaesthetic systemic toxicity (LAST) may respond best to intravenous 20% lipid emulsion 1.5 mL/kg bolus followed by 0.25 mL/kg/min for roughly 10 min following recovery of vital signs [5].
As adverse reactions occur immediately or within minutes after local anaesthetic use, medical expertise, resuscitation equipment and monitoring facilities must always be available.
Other reactions
Other adverse reactions to local anaesthetics involve allergy, including anaphylaxis, predominantly to the amino esters (rarely amino amides), catecholamine effects from added adrenaline, vasovagal reactions when the patient is upright, such as during a dental procedure, cytotoxic delayed wound healing, malignant hyperthermia from amino amide use and methaemoglobinaemia due to prilocaine or benzocaine (Table 22.2.3).
Table 22.2.3
Adverse reactions to local anaesthetics (other than systemic toxicity)
Allergy:
esters>>amides
additives, such as methylparaben, sodium metabisulphite
Catecholamine effects from added adrenaline
Vasovagal
Delayed wound healing
Malignant hyperthermia
Methaemoglobinaemia – prilocaine, benzocaine
Topical anaesthesia
Some agents such as EMLA (eutectic mixture of local anaesthetics including 2.5% lignocaine and 2.5% prilocaine) are used topically, particularly to decrease the pain of insertion of cannulae or for lumbar puncture and suprapubic catheter insertion in children. EMLA takes up to 1 h for maximal effect and, paradoxically, is a venoconstrictor making vessel puncture harder. A potentially superior alternative for cannula insertion is 4% amethocaine (AnGel) as this has a quicker onset and is a vasodilator, although operator experience in cannulation is likely to be of more relevance [6].
Likewise, a mixture of 1:1000 adrenaline, 4% lignocaine and 0.5% amethocaine, with the acronym ALA (or known as LET in North America standing for lidocaine, epinephrine and tetracaine) up to 0.1 mL/kg, may be used inside small wounds instead of, or to reduce the pain of, injecting local anaesthetic prior to closure, again in children or adolescents.
Specific nerve blocks
The following nerve blocks are contraindicated in uncooperative patients, those with local sepsis in the injection zone and in the rare patient with true local anaesthetic allergy. Care must be taken not to exceed the recommended maximum local anaesthetic doses (see Table 22.2.1) and monitoring facilities, resuscitation equipment and medical expertise must be available at all times.
Formal training and accreditation should occur prior to independent practice, paticularly with the more complex blocks, such as Bier’s and femoral nerve.
Digital nerve block (‘ring block’)
Indications
Wound debridement, suturing, drainage of infection, fracture or dislocation reduction around the nail, fingertip and distal finger or toe.
Contraindications
Local sepsis, Raynaud’s phenomenon and peripheral vascular disease.
Technique
Use 2% plain lignocaine. Inject 1–1.5 mL using a 25-gauge needle into the palmar aspect of the base of the finger or toe, approaching vertically from the dorsum. Withdraw the needle until subcutaneous and rotate slightly until pointing to the extensor surface of the digit and inject a further 0.5 mL (Fig. 22.2.1). Perform the same procedure on the other side of the digit. Allow at least 5 min for the block to work.
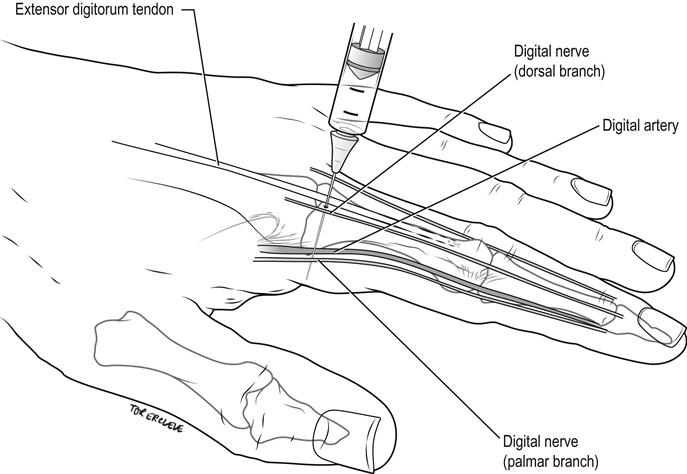
Complications
Avoid intravascular injection by aspirating prior to injection. Do not use a tourniquet or more than 4 mL total volume, to avoid impairing the circulation due to high local tissue pressures.
Nerve blocks at the wrist
These provide anaesthesia to the hand, particularly for diffuse lesions hard to infiltrate directly, such as ‘gravel rash’, or when the hand is swollen or burned.
Ulnar nerve wrist block (lateral approach)
Indications
Procedures on the medial border of the hand and medial 1.5 digits or combined with median and radial nerve blocks for hand anaesthesia.
Contraindications
Local sepsis, neuritis.
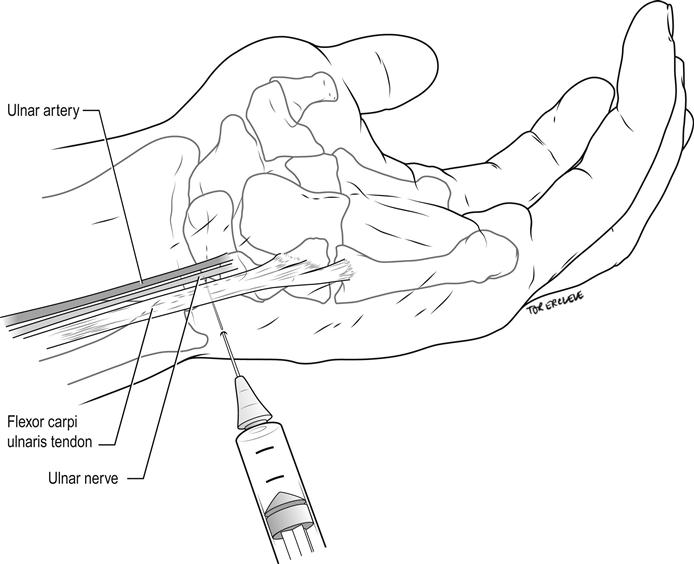
Technique
Identify the flexor carpi ulnaris tendon at the proximal palmar crease. Introduce a 25-gauge needle on the ulnar aspect of the tendon, directed horizontally and laterally for 1–1.5 cm under the tendon. Inject 4 mL of 1% lignocaine. Withdraw the needle until subcutaneous then inject 5 mL of 1% lignocaine fanwise to the dorsal midline, to block superficial cutaneous branches (Fig. 22.2.2).
Median nerve wrist block
Indications
Procedures on the lateral border of the hand in the territory supplied by the median nerve, excluding the medial 1.5 digits or combined with ulnar and radial nerve blocks for hand anaesthesia.
Contraindications
Local sepsis, carpal tunnel syndrome or neuritis.
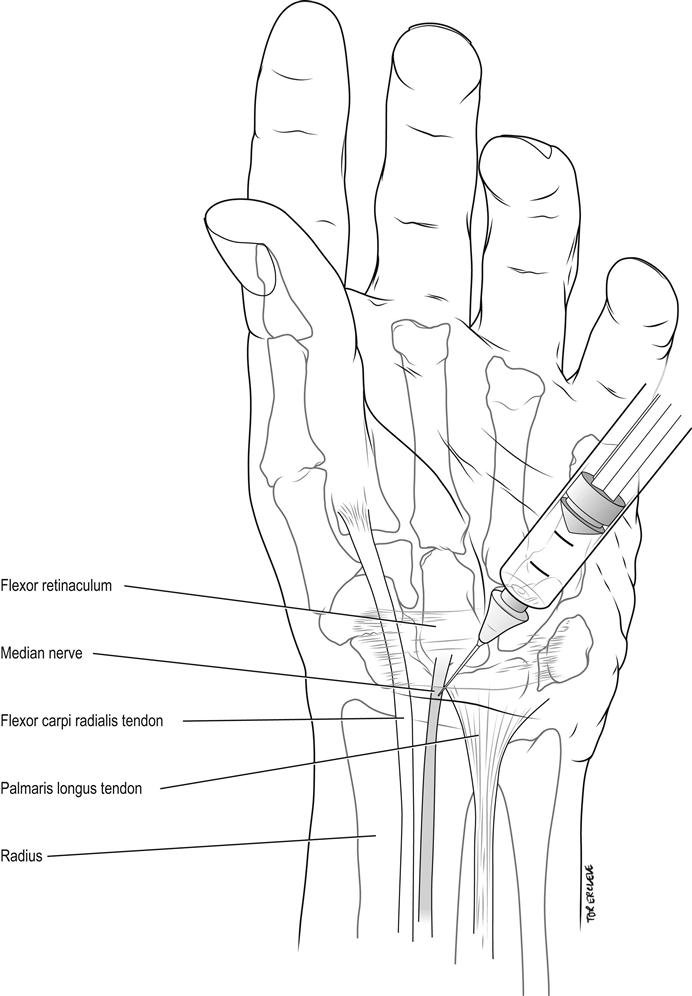
Technique
Identify the tendons of the flexor carpi radialis and palmaris longus at the proximal wrist crease. Introduce a 25-gauge needle vertically 0.5–1 cm lateral to the palmaris longus (or 0.5 cm medial to the flexor carpi radialis in the 10% of individuals lacking a palmaris longus). Inject 5 mL of 1% lignocaine when the needle gives as it penetrates the flexor retinaculum or paraesthesiae are elicited, at a depth usually of no more than 1 cm to the skin (Fig. 22.2.3). Avoid injecting into the nerve itself, as it may lie more superficial than this.
Radial nerve wrist block
Indications
Procedures on the dorsal radial aspect of the hand or combined with ulnar and median nerve blocks for hand anaesthesia.
Contraindications
Local sepsis, neuritis.
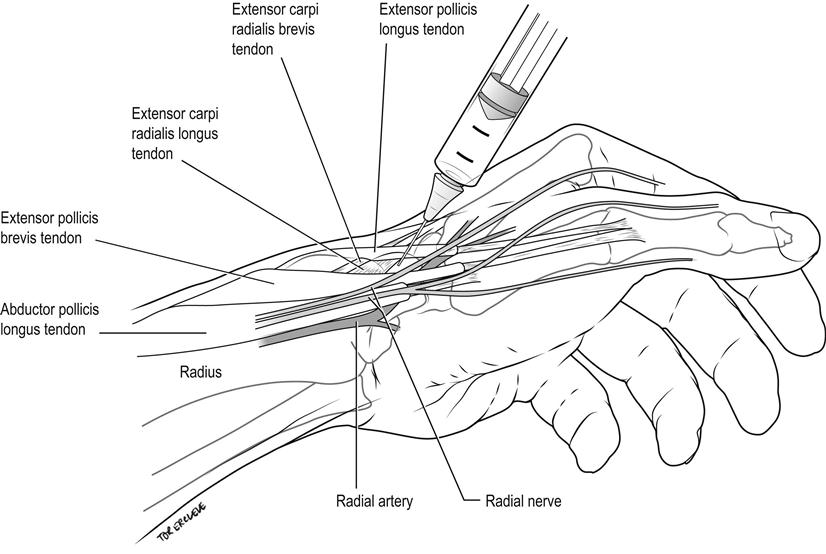
Technique
Identify the tendon of the extensor carpi radialis and infiltrate 5–10 mL of 1% lignocaine subcutaneously in a ring around the radial border of the wrist to the area overlying the radial pulse, at the level of the proximal palmar crease (Fig. 22.2.4).
Nerve blocks of the leg
Femoral nerve block
Indications
Analgesia for fractured shaft of femur, especially prior to applying dynamic splintage.
Contraindications
Local sepsis, bleeding tendency.
Technique
Palpate the femoral artery below the midpoint of the inguinal ligament, which extends from the pubic tubercle to the anterior superior iliac spine. Insert a 21-gauge needle 1 cm lateral to this point, perpendicular to the skin. Advance until paraesthesiae are elicited down the leg and withdraw slightly, aspirate to exclude intravascular placement and inject 10 mL of 0.5% bupivacaine (50 mg). Alternatively, feel for a give as the needle punctures the fascia lata, aspirate, then inject 10 mL of 0.5% bupivacaine fanwise laterally away from the artery (Fig. 22.2.5). Or preferably now use ultrasound guidance throughout. Allow up to 15–30 min for onset of maximal anaesthesia.
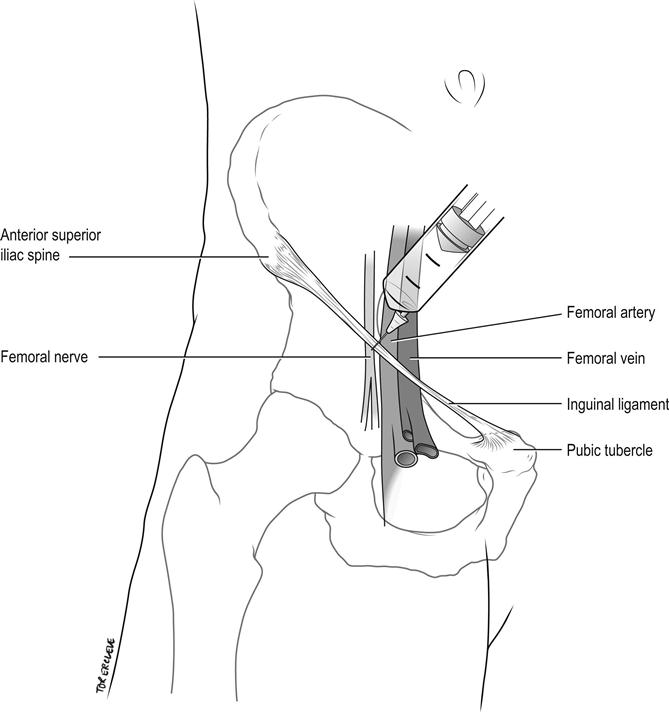
Complications
Puncture of femoral artery.
Foot blocks at the ankle
Indications
Where local anaesthetic infiltration of the foot is awkward or difficult because of thick sole skin or pain, or when excessive amounts of anaesthetic would otherwise be required.
Contraindications
Local sepsis, peripheral vascular disease.
Technique
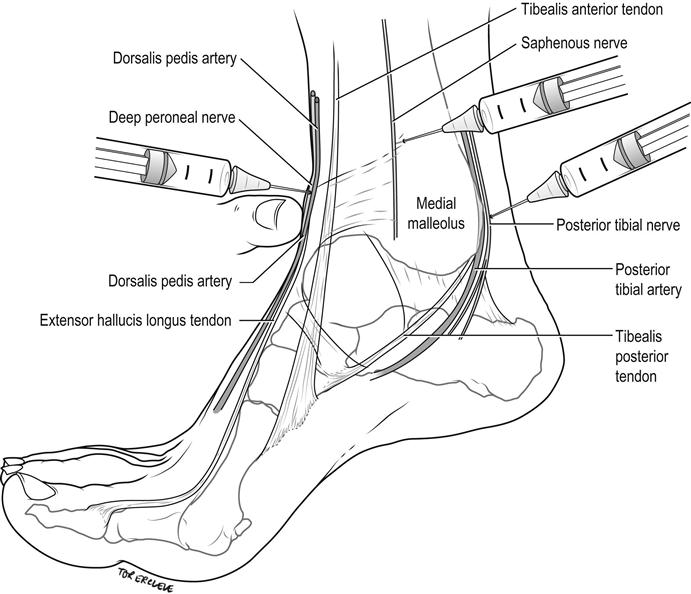
Three superficial nerves, the sural, superficial peroneal and saphenous, are blocked by subcutaneous infiltration in a band around 75% of the ankle circumference. Two deeper nerves – the posterior tibial by the posterior tibial artery and the deep peroneal (anterior tibial) nerve by the dorsalis pedis artery – are blocked, usually in combinations with the superficial ones, according to the area of anaesthesia required.
Sural nerve
The sural nerve is blocked by injecting 3–5 mL of 1% lignocaine subcutaneously in a band between the Achilles tendon and the lateral malleolus, 1 cm above and posterior to the malleolus (Fig. 22.2.6). It anaesthetizes a small strip on the lateral dorsum of the foot at the base of the little toe to the lateral malleolus and the posterolateral aspect of the ankle and heel.
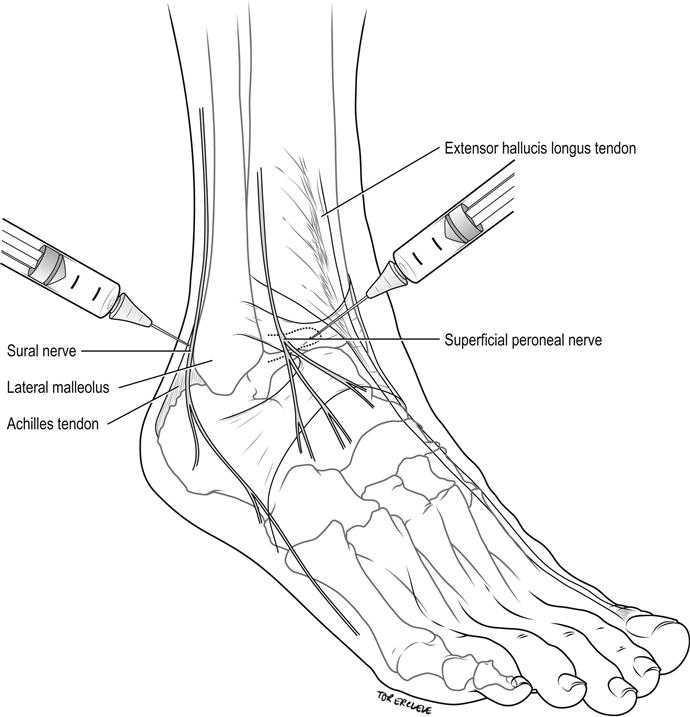
Superficial peroneal nerves
Superficial peroneal nerves are blocked by injecting 4–6 mL of 1% lignocaine subcutaneously in a band between the extensor hallucis longus tendon and the lateral malleolus, on the anterior aspect of the ankle (see Fig. 22.2.6). This block anaesthetizes the dorsum of the foot, save for the lateral aspect (see sural nerve above) and interdigital web between the hallux and second toe (see deep peroneal nerve below).
Saphenous nerve
The saphenous nerve is blocked by injecting 3–5 mL of 1% lignocaine subcutaneously above the medial malleolus, laterally until over the tibialis anterior tendon (Fig. 22.2.7). It anaesthetizes the area around the medial malleolus anteriorly and, to a lesser degree, posteriorly.
Posterior tibial nerve
The posterior tibial nerve is blocked by infiltrating 3–5 mL of 1% lignocaine immediately lateral to the posterior tibial artery as it passes behind the medial malleolus, at a depth of 0.5–1 cm to the skin (see Fig. 22.2.7). It anaesthetizes the sole of the foot, excluding the posterolateral heel (see sural nerve above), via its medial and lateral plantar branches.
Deep peroneal (anterior tibial) nerve
The deep peroneal (anterior tibial) nerve is blocked by infiltrating 1–2 mL of 1% lignocaine just above the base of the medial malleolus, lateral and behind the extensor hallucis longus by the dorsalis pedis pulse at a depth of 0.5 cm (see Fig. 22.2.7). It anaesthetizes the interdigital web between the hallux and second toe.
Complications
Exceeding a total volume of 20 mL of 1% lignocaine local anaesthetic (ie. 3 mg/kg) that risks systemic toxicity or poor peripheral perfusion due to raised tissue pressures.
Intravenous regional anaesthesia or Bier’s block
Indications
Operative procedures, such as debridement, tendon repair and foreign body removal in the forearm and hand. Reduction of fractures and dislocations, typically Colles’ fracture of the wrist.
Contraindications
Local anaesthetic sensitivity, peripheral vascular disease including Raynaud’s phenomenon, sickle cell disease, cellulitis, uncooperative patients including children, hypertension with systolic blood pressure over 200 mmHg, severe liver disease, and unstable epilepsy.
Technique
Two doctors are required, allowing one to perform the manipulation and the other, with training in the procedure and resuscitation skills, to perform the block. Explain the procedure to the patient and obtain informed consent. Assemble and check all equipment and apply standard monitoring, including ECG, non-invasive blood pressure and pulse oximetry.
Use a specifically designed and maintained single 15 cm adult cuff, placed over cotton wool padding to the upper arm.
Double-cuff tourniquets require higher inflation pressures as they are narrower. The upper cuff is inflated first, followed by the lower cuff 15 min later, after injection of the prilocaine, thereby causing less discomfort to the patient. The upper cuff is then released. The use of a double cuff does not always reduce the ischaemia pain and predisposes to accidental wrong cuff release, so requires additional expertise and understanding.
Insert a small intravenous cannula into the dorsum of the hand of the injured limb and a second cannula in the other hand or wrist as emergency access to the central circulation. Exsanguinate the injured limb by simple elevation and direct brachial artery compression for 2–3 min, carefully supporting the limb at the site of any fracture. An Esmarch bandage may be used instead, in the absence of a painful wrist fracture.
Keep the arm elevated and inflate the cuff to 100 mmHg above systolic blood pressure. The radial artery pulse should now be absent and the veins remain empty. If this is not the case, do not inject anaesthetic but repeat the exsanguination procedure and cuff inflation.
Lower the arm once the radial artery pulse is absent and the veins are empty, and inject 2.5 mg/kg (0.5 mL/kg) of 0.5% prilocaine slowly over 90 s and record the time.
Continuously monitor the cuff pressure and wait at least 5–10 min to confirm the adequacy of analgesia before removing the cannula on the injured limb. Perform the surgical procedure. Keep the tourniquet inflated for a minimum of 20 min and a maximum of 60 min.
Monitor the patient carefully for any signs of anaesthetic toxicity (see Table 22.2.2) over the next 15 min following cuff release, while organizing discharge from the monitored area.
Complications
No severe cardiac complications, deaths or methaemoglobinaemia have been reported using 0.5% prilocaine at the maximum dose of 2.5 mg/kg (0.5 mL/kg) [7]. Discomfort from the cuff is possible, but rarely significant.
22.3 Procedural sedation and analgesia
Anthony J Bell and Greg Treston
Introduction/rationale
Procedural sedation and analgesia (PSA) is a core competency for the emergency physician for the performance of brief, but painful procedures and has become standard emergency medicine practice. PSA refers to the technique of administering sedatives or dissociative agents, with or without analgesics, to induce a state that allows the patient to tolerate an unpleasant or anxiety provoking therapeutic or diagnostic procedure, while maintaining cardiorespiratory function [1,2].
Significant variation exists in the practice of PSA in Australasian emergency departments (EDs) in relation to the approach, choice and combination of agent(s) given [3]. Paediatric patients in particular represent a significant challenge to the emergency physician, as children are often frightened when in pain and their presentation to the hospital disrupts family functioning [4,5]. Medical staff underestimate and undertreat pain in children [6]. Procedures may have previously been performed with inadequate sedation or ‘oligoanalgesia’ for fear of complications, worry about prolonged recovery time or the perception that with the procedure being brief, the child will not remember it.
Painful procedures in the ED are remembered vividly by children, parents and adult patients. Denial of relief from pain that is commensurate with the expressed need for such relief must be judged as an unjustified harm and amounts to substandard and unethical medical practice [7].
Underlying principles
Guidelines
The Australasian College for Emergency Medicine (ACEM), [8] American College of Emergency Physicians (ACEP) [2] and the Canadian Association of Emergency Physicians (CAEP) [9] have all published on the underlying principles for successful procedural sedation and analgesia within the emergency department setting. These guidelines cover pre-sedation preparation and assessment, pre-sedation fasting, physician skills, staffing, equipment and setting, patient monitoring, documentation and post-sedation care. With the gradual change in the perception accorded to PSA being performed in EDs over the last decade, regulatory approval is now in place within the USA, for example, allowing the delivery of anaesthetic agents for PSA in the ED.
The ACEM guidelines require that two medical attendants, one of whom should be a specialist or advanced trainee, be present. Nursing staff are also required and the procedure must be performed in a resuscitation area. Physiological monitoring is mandated during the procedure and extending into the recovery phase. Although the Australasian guidelines were developed conjointly with ANZCA, JFICM and Faculty of Pain Medicine, there is no specific recommendation as to choice of agent, despite the potentially confounding information published by ANZCA relating to the use of intravenous anaesthetic agents [10].
Depth and duration of sedation
The optimal endpoint of any sedation episode depends on the procedure being performed and the patient’s characteristics. Classification of sedation state is now well established, ranging from minimal sedation (anxiolysis) through moderate sedation (formerly confusingly known as ‘conscious sedation’), deep sedation to general anaesthesia. Dissociative sedation is a separate state induced by ketamine [11]. The exact characteristics of respiratory and/or airway reflex depression in relation to the depth of sedation are not well defined [1].
Titration of drug and constant verbal and tactile re-assessment of the patient reduce the risk of over-sedation [2]. Some degree of responsiveness to painful stimuli should indicate preservation of airway reflexes, decreasing the risk of aspiration if vomiting occurs [12].
Reducing the time a patient is sedated reduces the risk of airway compromise or respiratory depression (an adverse event in an obtunded patient). The duration of sedation is largely determined by the choice and dose of agent used and the procedure itself: whether this will be brief (e.g. shoulder reduction) or longer (e.g. compound scrub or manipulation of fractures), with most ED procedures taking less than 20 min.
Indications and patient selection
Patient selection is based on the need for sedation for a brief, painful procedure that will facilitate earlier discharge from the ED. These include, but are not limited to, fracture and dislocation reduction, incision and drainage of abscesses and cardioversion [13]. Inherently less painful but anxiety-provoking procedures in children will also be facilitated by the use of dissociative sedation, for example lumbar puncture, suturing, ocular or auditory canal foreign body (FB) removal or IV cannulation in an uncooperative and anxious child [11].
In Australasia, children have high numbers of laceration repairs or upper limb fractures reduced; young adults predominate with upper limb relocation and reductions; with older patients undergoing incision and drainage of abscesses, upper limb procedures or prosthetic hip relocations [3].
Departmental procedures and logistics
All departments that perform PSA should have written guidelines, a standardized data collection system and suitably trained staff. Internal processes should rapidly identify patients requiring PSA so that imaging, preparation for PSA and practitioner availability are ensured. Patient selection is informed by the pre-procedural risk assessment, departmental resources and ability to monitor and safely discharge the patient. In the context of time-based targets, patient flow patterns of this group need to be considered and systems put in place to optimize these. A recent Canadian paper highlights the inherent time challenges, resource intensity and foregone benefits if standardized processes are not adopted [14].
Pre-procedure risk assessment
Pre-procedural assessment is of critical importance before embarking upon PSA. Patient and pharmacological factors known to be associated with sedation-related events requiring a brief intervention include advancing age, deep sedation and pre-medication with or use of fentanyl in combination with either propofol or midazolam for PSA. Conversely, patient weight (within reason), procedure type and fasting status do not appear related to respiratory adverse events [15].
However, procedural failure is related to patient weight over 100 kg and certain procedure types, notably prosthetic hip reduction, digits and temporomandibular joint (TMJ) relocations. Ketamine is not only protective overall for respiratory adverse events, but has the highest procedural success rate [16]. Particular consideration should be given to:
Age
A young patient’s level of anxiety and cooperation will depend upon past medical experiences, anxiety of the parent(s) and the reassurance given by medical staff [11]. Elderly patients, while mostly cooperative, may have underlying impaired cardiorespiratory reserve and are at greater risk of peri-procedural cardiac events, respiratory depression or hypotension.
ASA classification
The American Society of Anesthesiologists (ASA) classification system [17] is used to classify patients (Table 22.3.1). ASA 1 and ASA 2 are the preferred candidates for procedural sedation in the ED. If an ASA 3 patient requires sedation out of necessity, such as for cardioversion, this should not preclude performance of the procedure. The management of respiratory depression becomes more relevant with increasing ASA class in all age groups.[18,19]
Table 22.3.1
American Society of Anesthesiologists (ASA) classification
| Class | |
| 1 | Healthy patient, no medical problems |
| 2 | Mild systemic disease, e.g. hypertension |
| 3 | Severe systemic disease, but is not incapacitating |
| 4 | Severe systemic disease that is a constant threat to life |
| 5 | Moribund expected to live<24 h irrespective of operation |
Airway assessment
An adverse past anaesthetic history and/or a focused airway assessment with attention to mouth opening, pharyngeal visualization using the Mallampatti score (Fig. 22.3.1), neck movement, thyromental distance and dentition help signal potential difficulty should active intervention be required. Table 22.3.2 contains a difficult airway predictor checklist.
Table 22.3.2
Airway assessment predictors of a difficult endotracheal intubation
 |
Mallampatti score III & IV |
 |
Inability to open mouth>4 cm |
 |
Thyromental distance<6 cm |
 |
Limitation of neck movement |
 |
Difficulty in protruding lower jaw |
 |
History of difficult intubation |
Past medical history
Some conditions predispose to gastro-oesophageal reflux, such as pregnancy or hiatus hernia. Unstable acute medical/neurological conditions (with the exception of arrhythmias requiring cardioversion) may carry too high a risk to proceed with PSA. Allergies to any agent in the past precludes choosing that agent, as does egg/soy allergy as regards the use of propofol in particular [20]. A history of sleep apnoea will usually be associated with some obstructed or semiobstructed airway time during procedural sedation.
Fasting status
Fasting guidelines
Emergency department patients, particularly children [3], undergoing urgent PSA are commonly not fasted on presentation, or at the time of the procedure. Furthermore, holding a patient for 6 h in an overcrowded ED to achieve a goal of fasting time is impractical if an urgent procedure needs to be performed. Fasting guidelines are consensus, not evidence-based. The ASA recommends by extrapolation at least 2 h and 6 h from last intake of fluid and food, respectively [17], despite a lack of evidence regarding these. There are no clinical data to support a consensus view regarding prolonged fasting prior to sedation [17,21]. In fact, prolonged pre-procedural fasting has been shown to increase the rate of vomiting [22,23].
Aspiration risk
The risk of aspiration is low. Fasting status is but one consideration when individualizing decisions about choice of agent, approach to dosing, desired depth of sedation or even referral for general anaesthesia (GA) instead [12,24]. PSA does not use volatile inhalational anaesthetics, which are inherently emetogenic, as may occur during GA [25]. Importantly, nor does ED PSA involve pharyngeal manipulation or instrumentation, again a potent stimulus for vomiting.
There is no association between fasting status and adverse events during procedural sedation in the ED for a range of agents that include ketamine, midazolam/fentanyl, chloral hydrate, pentobarbital [22,26,27] or nitrous oxide [20,28]. The proportion of unfasted patients in relevant studies was 53–71%. Recent data looking specifically at patients undergoing PSA with propofol with respect to fasting status showed no difference in adverse events between fasted and unfasted patients [12]. There are few reported cases of aspiration during PSA in the adult ED literature [29], with more in the paediatric literature, although the incidence is still low.
The senior ED clinician should still screen for high aspiration risk based on past history and physical characteristics, as such patients may benefit from an alternate approach or technique. The general anaesthetic literature cannot be directly extrapolated to ED PSA. Aspiration is a rare event and the decision to proceed with PSA is a pragmatic one and should be balanced against the competing harm to the patient when a procedure is unnecessarily delayed [30,31].
Post-procedure vomiting
This usually occurs well into the recovery period. Post-procedure vomiting is more common with ketamine or narcotics than it is with propofol or benzodiazepines [12,32].
A large multicentre prospective observational trial reported a vomiting incidence of 1.6% overall [15] but, as follow up occurred only during the ED phase of care, this is likely an underestimate. Oral or IV ondansetron given pre-sedation can decrease post-procedural emesis.
Procedural urgency
The endpoint of sedation in the ED should be tailored to the urgency of the procedure and availability of appropriate staff [1,9]. Procedures may thus be regarded as [1]:
Involvement of parents or carer
Parental cooperation is critical for the success of any procedural intervention in children. ED surveys show the vast majority of parents wish to be present for an invasive procedure performed on their child in the ED, with a small reduction with increasing invasiveness of the procedure, except for full resuscitation [33,34].
Despite this, more than one-third of parents were asked to leave the room in one study of children undergoing procedures [35]. The practice of requesting that parents leave the room when their child undergoes a procedure should be abandoned. Parental presence should instead be welcomed and, ultimately, their decision to stay or go should be supported. PSA in children should include ushering the parents to the bedside, not out of the room [33,36].
Informed consent
Informed consent must be obtained after explanation of specific risks of PSA and the procedure itself. In particular, when ketamine is used, warn parents to expect a ‘staring child’ with nystagmus, salivation, lacrimation, possible myoclonic jerking and vomiting in 10–15% usually after the end. This is important to parents’ overall acceptance and experience of their child’s procedure.
Likewise, although rare, the possibility of airway intervention beyond transient support should be raised with propofol. A practical point is that children will routinely (and adults occasionally) develop tears in their eyes. Family members should be reassured in advance that this is a drug effect, not a reaction to pain.
Documentation
A specific procedural sedation form or record is recommended. When designed in accordance with current best practice, they improve documentation and can act as the focus for educational initiatives and assist in audit, research and quality assurance (QA) [37–39]. As well, they can act as a de facto protocol to ensure safety during the procedure. They also increase the chance of guideline compliance and ensure essential pre-sedation checks and monitoring are performed. A specific procedural sedation form should include provision for recording adverse events, including vomiting, aspiration or respiratory depression, as well as any intervention(s) required [12].
Choice of agent
The ‘ideal’ agent for PSA in the ED should have a rapid onset, short duration of action, rapid recovery profile, minimal side effects and an amnestic effect. The different classes of drugs used in PSA include sedative hypnotics, analgesics, dissociative sedatives, inhalation agents and antagonists (flumazenil and naloxone), used alone or in combination (Table 22.3.3).
Table 22.3.3
Choice of agent and suggested IV drug dosages (adult, 70 kg, normal BMI)
| Drug | Initial bolus | Subsequent titrated IV boluses | Cumulative maximum dose |
| Morphine | 2.5 mg | 2.5 mg | 10–15 mg |
| Fentanyl | 25–50 μg | 25 μg | 150–200 μg |
| Midazolam | 2 mg | 1 mg | 10 mg |
| Diazepam | 5 mg | 2.5 mg | 10 mg |
| Propofol | 40–50 mg | 20 mg | 150 mg |
| Ketamine | 20–30 mg | 10–20 mg | 120 mg |
| Etomidate* | 5–7 mg | 2 mg | 20 mg |
| ‘Ketafol’ | 6 mL | 3 mL | 20 mL |
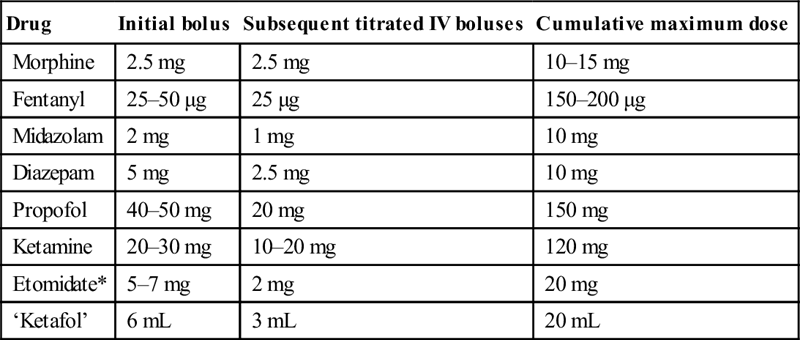
BMI: body mass index.
These are conservative estimates for a 70-kg adult.
Dose modification is advised where appropriate in the elderly patient (lower doses) and patients with small muscle bulk (lower doses).
‘Ketafol’ is made with 1 mL of 100 mg/mL ketamine made up to 10 mL with 9 mL of saline. To this add 10 mL of 10 mg/mL propofol in the same 20 mL syringe giving 5 mg:5 mg/mL, respectively.
*Etomidate is not available in Australia.
Sedative hypnotics
Benzodiazepines
Midazolam
Midazolam is one of the most commonly used benzodiazepines with amnestic [40], anxiolytic and sedative properties. Its side effects are dose dependent. Intravenous dosing for PSA ranges from 0.025 to 0.05 mg/kg in older children and adults, up to 0.1 mg/kg in younger children. Other routes of administration include IM and intranasal, although onset of action is slower [11] and many experienced clinicians have abandoned the use of intranasal midazolam.
Midazolam/opioid combinations
Midazolam/opioid combinations have been perceived to provide more predictable response and to have a favourable safety profile compared to propofol, owing to the potential of propofol to induce dose-related deep sedation [41]. In fact, there is additive respiratory depression with opiate and midazolam-containing combinations for PSA, with prolonged recovery times when compared to propofol [42–44]. Furthermore, patients receiving midazolam have been reported to have more recall and higher pain scores [45].
Diazepam
Diazepam is less potent than midazolam, although there is little to no difference in the propensity of the two drugs to produce respiratory depression [46]. Dosing should start at 0.1–0.2 mg/kg with smaller subsequent doses. The antegrade amnestic effect of diazepam is significantly less than that of midazolam [47,48] and it causes more pain on injection but lesser early sedation [49]. The elimination half-lives of benzodiazepines do not necessarily correspond with their sedative pharmacodynamic effects and there are no clinically important sedative recovery rate differences between midazolam and diazepam [50,51].
Short-acting agents
Propofol
Propofol is a non-opioid, non-barbiturate sedative hypnotic that acts at γ-aminobutyric acid (GABA) sites within the CNS providing an amnestic effect, rapid onset<1 min and short duration (5–15 min) facilitating a rapid recovery time. It is readily titratable and has some antiemetic properties [21,52]. Hypotension is transient when propofol is titrated in a euvolaemic patient with normal cardiac function. Propofol has been shown to be safe in a wide range of settings, including PSA in the ED [2,53,54]. Propofol may cause a painful ache at the site of injection as the patient becomes sedated, which may be distressing, but is short lived.
The optimum dosing regimen for propofol in procedural sedation has yet to be defined. Options vary from single bolus [43,44,55], titration [19,56–59], bolus and infusion [42,60] or infusion alone [61–63]. Doses recommended include 1 mg/kg initial bolus and 0.5 mg/kg subsequent boluses for PSA in the ED [44,52,64,65]. In children, initial bolus doses of 2 mg/kg have been used [66,67]. An alternative is to reduce the initial dose to 0.5–1.0 mg/kg followed by 20 mg boluses [68]. Dose reduction is also essential in those over 65 years of age [60]. Higher total mg/kg doses are used in children compared with adults [68,69].
Sedation times are shorter with propofol and the reported respiratory complication rates for propofol are equivalent to midazolam alone [42], midazolam±flumazenil or etomidate, [43], and midazolam/fentanyl [44]. Discharge times are also earlier than with midazolam [70]. At excessive doses, propofol is associated with greater oxygen desaturation [55].
Respiratory depression is seen in up to 50% of ASA class 1 and 2 patients [57,71,72] and 61% in the critically ill [18]. Apnoea may occur in up to 22% of patients receiving propofol for PSA, but is transient [12,43,64]. Transient hypoxia has been observed in 6–44% of sedation episodes [18,19,42,43,55–57,59–61,69,71,72]. The combination of opiate and propofol results in higher levels of respiratory depression than propofol alone [73,74]. Supplemental oxygen was not routinely applied during PSA in some studies [56], although this practice is not supported. Supplemental oxygen should be applied in all ED sedations where practical, although with sedation of a young combative child it may be prudent to wait until the child is part-sedated before applying oxygen.
Etomidate
Etomidate is a non-barbiturate hypnotic currently unavailable in Australasia, although it is regularly used for RSI or procedural sedation in the UK and USA. Etomidate has a rapid onset<30 s with 5–15 min duration of action. The starting dose for PSA is up to 0.1 mg/kg with subsequent boluses of 0.05 mg/kg. It has a similar profile to propofol in terms of respiratory depression and duration of sedation when compared to midazolam, but is more cardiovascularly stable. Propofol is preferred as etomidate has a 20% rate of myoclonus plus the theoretical risk of adrenal suppression, emergence phenomena and higher vomiting rates [75–78].
Opiates
Opiates are the most commonly used analgesics before, during and after sedation, but up to half of all patients undergoing PSA receive no pre-procedural analgesia at all. Morphine is the preferred pre-procedure agent, but fentanyl is favoured intra-procedurally in a ratio of 4:1 [3]. Opiates provide analgesia but have no amnestic or anxiolytic properties.
Fentanyl
Fentanyl is the opiate of choice for ED PSA. It should be titrated up to 1 μg/kg IV, to avoid respiratory depression from a rapid push and should be combined with a pure sedative agent. It may also be delivered intranasally. Fentanyl compared to morphine has a rapid onset, a lack of histamine release and is cardiovascularly stable. Its duration of action is 30–45 min.
Despite its common use in sedation, fentanyl is linked to a higher respiratory depression event rate [77]. Some degree of respiratory depression should therefore be anticipated and actively managed if fentanyl is given. There is increasing evidence that ketamine is a safer analgesic than fentanyl for PSA [15,79].
Morphine
Morphine provides a longer duration of analgesia extending to hours and is useful after the procedure for ongoing analgesia. It may have been administered by ambulance officers pre-hospital.
Ultra-short-acting opiates
Ultra-short-acting opiates, such as remifentanil and alfentanil, play a role when combined with propofol to provide excellent sedation and analgesia enabling rapid recovery [80]. Like fentanyl, respiratory depression is to be expected and may even occur at lower levels of sedation. The higher recall rate in this scenario is a consideration [81,82]. As most patients will require some analgesia post-procedure, the logic for using ultra-short-acting narcotics in ED PSA appears more academic than practical.
Inhalational agents
Nitrous oxide
N2O provides anxiolysis and mild analgesia. Entonox is a 50:50 mixture of N2O and oxygen that is widely available in Australasia, delivered via a self-administered demand valve mask. It has rapid onset and offset and is safe but has little or no sedative effect. It may be useful as an anxiolytic in children and in needle-phobic individuals prior to definitive PSA.
Despite its popularly due to its convenience, N2O results in poor sedation conditions. One observational convenience sample of children aged 1–17 years [83] noted that mean sedation scores (using the 7-point Children’s Hospital of Wisconsin sedation scale; 6 being agitated, anxious, in pain and 0 being unresponsive to painful stimulus) were 4.4 with nitrous oxide 70% and 4.6 with nitrous oxide 50% (this case series utilized a nitrous oxide delivery and scavenging circuit not often found in emergency departments).
Another observational series [84] by the same authors examined the effect of high inhaled concentration N2O on intra-procedural pain scores in children undergoing painful procedures in the ED. In just over one-third of cases the recorded pain scores actually increased during the procedure and, in 15.3% of cases, pain scores during the procedure compared with baseline remained unchanged. Coupled with the rate of intra-procedural emesis associated with N2O use, this leaves N2O as ‘possibly useful’ for relatively painless procedures or as adjunct sedation for a procedure performed under a Bier’s block or similar.
Common side effects include vomiting and dizziness. Airway reflexes are preserved. It is contraindicated in patients with trapped gas, such as a pneumothorax.
Dissociative sedative
Ketamine
Ketamine is used with two distinct principal aims: as the primary sedative agent (that provides analgesia) or in a sub-dissociative dose as an analgesic adjunct with an alternative sedative. As a dissociative anaesthetic agent, ketamine has wide use in developing world and military applications [85]. In addition to excellent sedation and analgesia, it results in high levels of satisfaction and a lack of procedural recall [86]. Furthermore, ketamine is associated with the lowest procedural failure rate of any sedative in common use [16].
At sub-dissociative doses under 0.5 mg/kg, ketamine acts principally as an analgesic. With progressively larger amounts, dose-related ‘dissociative anaesthesia’, between deep sedation and general anaesthesia, occurs with doses exceeding 0.5 mg/kg. Ketamine has a rapid onset and offset with preservation of airway reflexes, although it can cause idiosyncratic laryngospasm. It is relatively contraindicated in ischaemic heart disease (IHD), due to increased sympathetic tone, or in other advanced vascular disease or uncontrolled hypertension.
Ketamine given either intravenously or intramuscularly is popular for emergency department paediatric procedural sedation. Its use is safe, with preservation of oropharyngeal reflexes and little to no respiratory depression [6,8,22,87–90]. At higher IV doses exceeding 1.5 mg/kg, subclinical respiratory depression is seen at rates similar to propofol [73], but ketamine is protective as regards the need for any actual airway intervention across the dosage spectrum [15].
There is concern about ‘emergence delirium’ [4,91], also known as ‘emergence phenomena’ [88–90,92,93]. Emergence delirium has been described as either ‘patients are agitated, restless and combative and do not seem cognizant of their surroundings. Patients refuse to be comforted, even by their parents’ [94], and ‘combative, excited and disorientated behaviour that requires transient physical restraint’ [95]. However, emergence phenomena may be as mild as an undistressing visual hallucination or a transient diplopia. Given the different definitions, the adult emergence incidence rate occurring in the recovery phase ranges from<1% to 36% [73,96–98].
Atropine as an antisialagogue with titrated IV ketamine for paediatric procedural sedation in the ED reduces hypersalivation [99] and post-procedure vomiting but, as hypersalivation rarely if ever affects the procedure, its use is optional. Hypersalivation appears less common in adults than in children.
The advantage of ketamine is its efficacy with variable routes of administration, even allowing a ‘no IV cannula’ sedation technique in the ED, particularly useful for pre-school-aged children.
Combinations with ketamine
Midazolam plus ketamine
Midazolam (or another benzodiazepine) has traditionally been used in combination with ketamine in an effort to decrease the incidence of ‘emergence delirium’, supported by studies reporting lower rates of emergence phenomena/agitation in adults who have received both ketamine and a benzodiazepine. ‘Emergence phenomena’ are fewer in children than in adults and the rate of emergence reactions in children is not lowered by adding a benzodiazepine [100–103], with unchanged rates of emergence agitation with midazolam added to ketamine [104–106].
Midazolam use has also been associated with higher rates of airway and respiratory compromise during procedural sedation in children [17]. Thus, use of adjunctive benzodiazepines with ketamine is not recommended. The only prospective randomized controlled trial (RCT) in adults addressing this issue showed that the prophylactic use of midazolam reduced recovery agitation by 10% [107].
Midazolam as an adjunctive medication when performing ketamine PSA has repeatedly been associated with lower rates of post-procedural emesis [104,105], yet with higher rates of airway/respiratory complications [17]. Midazolam itself as a sole sedative agent has been reported to have a rate of emergence agitation of up to 42% [108–110].
Ketamine plus propofol (‘ketafol’)
International RCT studies have compared ‘ketafol’ to propofol [111,112], ‘ketafol’ to midazolam/fentanyl [113] and ketamine alone [114]. All studies were small and focused on minor outcome differences. Anecdotally, there is a growing preference for the use of ‘ketafol’ over and above that which would be expected from the reported benefits.
The rationale for the addition of ketamine to propofol is to provide intra-procedural analgesia and sedation that has been used safely in a number of non-ED settings [115]. The combination aims to use the opposing respiratory and haemodynamic effects of each drug. Additionally, the antiemetic effect of propofol may counteract the vomiting seen with ketamine and may minimize the rate of ‘emergence’, although this remains unproven. Better procedural conditions and patient comfort are also suggested with the addition of ketamine [116,117].
This combination is safe in ED studies and resulted in high staff and patient satisfaction. The putative advantages in the use of ‘ketafol’ over propofol alone include modest dose reduction [118] and reduced respiratory depression. Recovery time remains short and there are no adverse events that altered patient disposition [114,119]. As ketamine at low doses is analgesic but not dissociative, targeted depth of sedation is important when using lower doses of propofol. The addition of ketamine to propofol has also been reported to have positive mood effects, allow earlier recovery of cognition and to reduce opiate requirement post-procedure [117,120,121].
Larger trials are still needed to compare this combination with other agents alone and to find the most synergistic ratio of ketamine to propofol [76]. Ratios of propofol:ketamine combined in the same syringe include 1:1 and 4:1 [122] or they can be titrated separately. Ketamine and propofol is a stable solution and can be safely administered from the same syringe [123].
Other drugs
Dexmedetomidine
Dexmedetomidine is a selective α-2 adrenergic agonist with sedative, anxiolytic and mild analgesic properties. Chemically, it is related to clonidine, but has a greater affinity for α-2 adrenergic receptors than α-1 receptors [124]. Dexmedetomidine acts in the CNS on vasomotor centres in the medulla, where it causes decreased sympathetic tone, which results in increased inhibitory GABA activity leading to sedation and analgesia [125]. It has been used for sedation of adults in the ICU and for paediatric sedation for non-painful radiological investigations (computed tomography [CT] and magnetic resonance imaging [MRI]) with some success. However, its utility is limited by hypotension and bradycardia mediated by the sympatholytic activity. There is minimal, if any, respiratory depression.
Current ED uses could be for decreasing agitation in a child undergoing CT or MRI scan or as a sedative agent for intubated adults in the ED. Some report that its utility as a paediatric sedation agent is restricted to sedation for CT, MRI or EEG and note that it demands a longer induction and results in a longer recovery time than propofol for similar procedures.
Preparation and monitoring
Resuscitation area
PSA should always occur in a resuscitation area, with two trained physician staff; one to perform the procedure and one to be responsible for the drugs and airway, with the assistance of an ED nurse [8]. Supplemental oxygen is given for all cases of ED PSA, except if using ketamine for paediatric PSA, when the use of supplemental oxygen by mask excessively upsets the child prior to the onset of sedation.
Equipment and monitoring
Suction, oxygen, airway adjuncts and intubation equipment should be prepared and physiological monitoring set up for pulse oximetry, non-invasive blood pressure, heart rate, ECG rhythm and respiratory rate. End-tidal carbon dioxide monitoring is recommended by some [2,12]. IV access is mandatory for all cases excluding paediatric sedation using IM ketamine or N2O sedations. See Table 22.3.4 for essential equipment requirements.
Table 22.3.4
Essential equipment requirements for procedural sedation ‘SOAPMI’
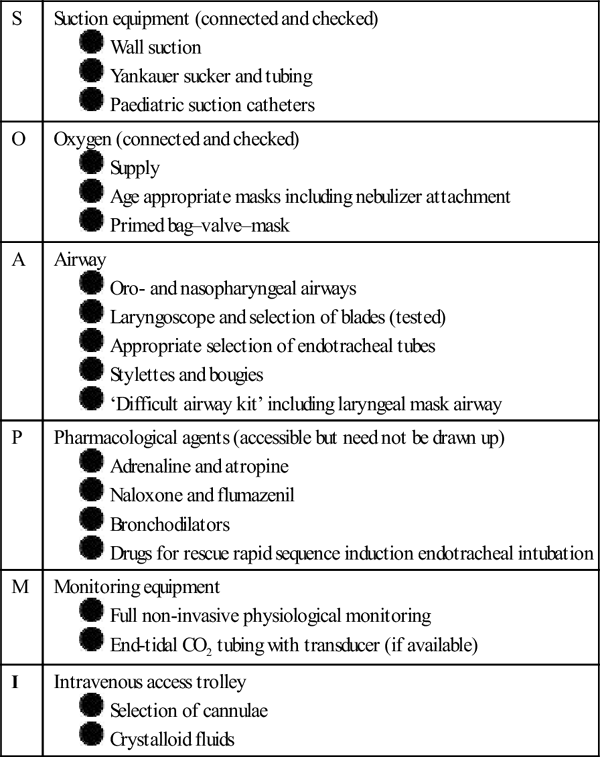
Sedation scoring
Interactive monitoring with verbal and tactile stimulation is used constantly to assess the depth of sedation. Careful dose titration and evaluation of patient responsiveness throughout the procedure is essential [41,53]. The Ramsay sedation score [126] or motor component of the observer’s assessment of alertness/sedation (OAA/S) scale [127], validated for midazolam may be used. All scales are subject to inter-observer variability and are relatively imprecise and not true objective measures of sedation. However, they require little formal training and are easily implemented (Table 22.3.5).
Table 22.3.5
| Score | Ramsay sedation scale | Sedation continuum | Score | OAA/S |
| 1 | Awake and alert | Minimal | 5 | Responds readily to name spoken in normal tone |
| 2 | Tranquil, purposeful at conversational level | Moderate | ||
| 3 | Sleepy but purposeful to verbal commands at conversational level | 4 | Responds only after name called loudly or repeatedly | |
| 4 | Sleepy and requiring louder voice or tactile stimulus to be purposeful | 3 | Responds only to mild prodding and shaking | |
| 5 | Asleep and only purposeful to loud verbal command or harder glabellar tap | Deep | 2 | Does not respond to mild prodding and shaking |
| 6 | Asleep and sluggishly purposeful only to painful stimulus | 1 | Does not respond to painful stimulus | |
| 7 | Asleep, reflex response, not purposeful | |||
| 8 | Unresponsive | Anaesthetized | 0 | Unresponsive |
OAA/S: motor component of the observer’s assessment of alertness/sedation scale.
Bispectral EEG analysis
Bispectral (BIS) EEG analysis has been studied but is not reliably predictive of conscious state in individual patients [76]. Numerical values (0–100) are assigned to a patient’s level of sedation but they are a poor measure of analgesia and ineffective when used with ketamine. Correlation with OAA/S [128,129] and Ramsay sedation scores [130,131] is poor. Lower BIS scores predict more respiratory depression (RD), but it is unclear whether BIS itself reduces the rate of RD [1,72,132]. Initial enthusiasm for BIS in ED PSA has waned as it has not proven its value over sedation scoring alone.
Capnography
Capnography can detect respiratory depression before clinical examination or oximetry, especially when on supplemental oxygen [133–136]. Capnography may even obviate the need for routine supplemental oxygen (although this is not supported by the authors of this chapter) [137]. Change in trace character or transient hypercapnoea [57,58,138] are early warning signs of hypoventilation or impending upper airway obstruction, of particular importance in children or those with reduced functional reserve [64]. Such early detection may avoid additional sedation being given and result in stimulating the patient or repositioning the airway. Only occasionally is an airway adjunct or manual (bag/valve/mask) ventilation required [12,69], and briefly ‘waiting out’ respiratory depression with propofol sedation in a pre-oxygenated patient is commonplace [65]. Thus, there may be a tendency to overintervene when capnography is used as a PSA adjunct.
One Canadian study [139] noted that capnometry (rather than capnography) offered no advantage over pulse oximetry for the detection of respiratory depression in non-pre-oxygenated adult patients undergoing procedural sedation. The capnometric changes were poorly sensitive and a late finding in patients who developed respiratory depression undergoing orthopaedic procedures. This study reinforces the benefit of pre-oxygenation and intra-procedural supplemental oxygen during sedation when sedative agents known to cause hypoventilation (propofol, narcotics, benzodiazepines) are administered.
The ‘antidote’ for apnoea due to inadvertent deep sedation is a painful stimulus, in other words, to start the painful procedure which has the benefit of stimulating respiratory effort.
Post-procedure considerations
Patients should be observed until they have returned to their baseline level of functioning [2,13,17]. The exact time will depend on the patient and drugs administered with one study in children suggesting a ‘30-min rule’ [140]. No special efforts need be made to darken the room or shield a child from the routine background visual and auditory stimuli of the busy ED.
Patients receiving propofol do not need prolonged post-procedure monitoring as re-sedation is rare [68]. Once the patient can talk, nursing staff have an ‘endpoint’ to cease physiological monitoring, confident that the patient is unlikely to develop any adverse event at this time (unless given further doses of narcotic).
The practice of making patients eat and drink prior to discharge after an ED sedation episode is without evidence and should be abandoned. This is supported by a recent Clinical Practice Advisory from the USA [13]. Nursing allocation should be tailored to the less intensive post-procedure monitoring requirements [141]. Discharge instructions, however, must be provided. See Table 22.3.6 for recommended adult discharge criteria.
Table 22.3.6
Recommended adult discharge criteria and instructions following procedural sedation
 |
Patient is alert and orientated or has returned to pre-procedure state |
 |
Patient ambulates safely or has returned to pre-procedure state |
 |
Patient is comfortable and has discharge analgesia arranged |
 |
Patient is discharged into care of a responsible adult |
 |
Driving or the like is banned for a minimum of 8 h |
 |
Alcohol or other central nervous system depressants are avoided for 12–24 h |
 |
Patients are warned about the potential for post-procedure pain, unsteadiness or dizziness. Seek medical attention if significant or disabling |
Developments in the next 5–10 years [142]