Role of T Cells in Immune Responses
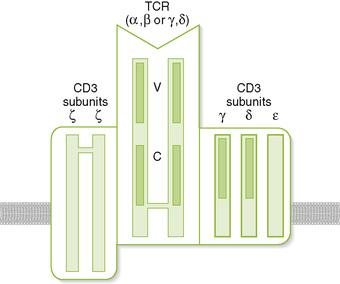
A T cell receptor (TCR) complex
• Comprises an antigen-recognizing heterodimer associated with a multimeric activation unit (CD3) (Box 2-1; Fig. 2-1)
1. All TCRs expressed by a single T cell are specific for the same antigen.
2. TCRs only recognize antigenic peptides bound to class I or II major histocompatibility complex (MHC) molecules.
1. CD4 and CD8 coreceptors define two main functional subtypes of T cells.
• CD4, present on helper T (TH) cells, binds to class II MHC molecules on the surface of antigen-presenting cells (APCs).
• CD8, present on cytolytic T (TC) and suppressor T (TS) cells, binds to class I MHC molecules on the surface of all nucleated cells.
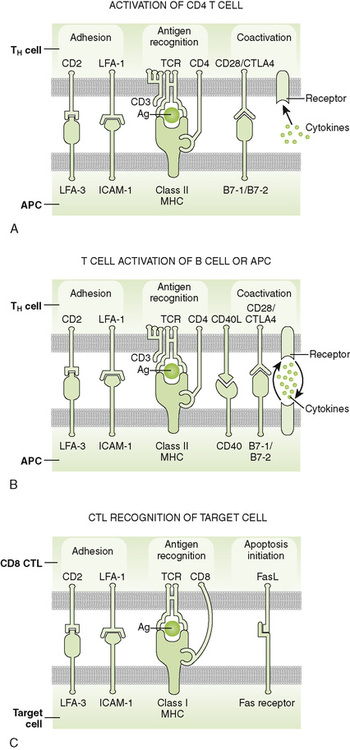
2. Adhesion molecules (e.g., CD2, LFA-1) help bind T cells to APCs and target cells or direct T cells to sites of inflammation and lymph nodes.
3. Coreceptor activating molecules (e.g., CD28 and CTLA-4) transduce signals important in regulating functional responses of T cells.
II Development and Activation of T Cells
A Antigen-independent maturation
1. Begins in bone marrow and is completed in the thymus, generates immunocompetent, MHC-restricted, naive T cells
2. Diversity of antigenic specificity of TCRs results from rearrangement of V, D, and J gene segments during maturation (similar to rearrangement of immunoglobulin genes).
• Each T cell possesses only one functional TCR gene and thus recognizes a single antigen (or a small number of related cross-reacting antigens).
3. Thymic selection eliminates developing thymocytes that react with self-antigens (including self MHC molecules).
B Antigen-dependent activation
1. Leads to proliferation and differentiation of naive T cells (clonal expansion) into effector cells and memory T cells (Fig. 2-2)
2. Effective stimulation requires primary and coactivating signals (fail-safe mechanism) that trigger intracellular signal transduction cascades, ultimately resulting in new gene expression (Fig. 2-3).
• Signal 1 (primary): specificity—dependent on antigen and MHC
a. Antigen-specific binding of TCR to antigenic peptide:MHC molecule on APC or target cell
b. Binding of CD4 or CD8 coreceptor to MHC molecule on APC or target cell
• Signal 2 (coactivating): permission—independent of antigen and MHC
3. Signal 3 (determines nature of response): direction—cytokine from dendritic cell (DC) or APC
• Determines the cytokine response and function of the T cell (TH1, TH2, TH17, regulatory T [Treg] cell)
4. Adhesion molecules: selectin (E-, L-, P-), ICAM (-1, -2, -3, LFA-3 CD2), and integrin (VLA, LFA-1, CR3)
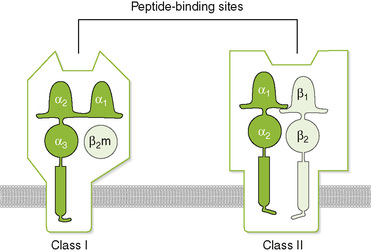
C Antigen processing and presentation by class I and II MHC molecules (Fig. 2-4)
• Different pathways are used for degradation of intracellular and internalized extracellular protein trash. Peptides resulting from digestion of nonhost (foreign) protein trash are recognized by the T cell surveillance squad, which mounts an appropriate defense (Box 2-2).
1. Endogenous antigen (class I MHC) pathway generates and presents antigenic peptides derived from intracellular viral, foreign graft, and tumor cell proteins (Fig. 2-5A).
2. Exogenous antigen (class II MHC) pathway generates and presents antigenic peptides derived from internalized microbes and extracellular proteins (Fig. 2-5B).
3. Cross-presentation pathway in DCs allows extracellular proteins (e.g., virus, tumor) to activate CD8 T cells (Fig. 2-5C).
III T Cell Effector Mechanisms
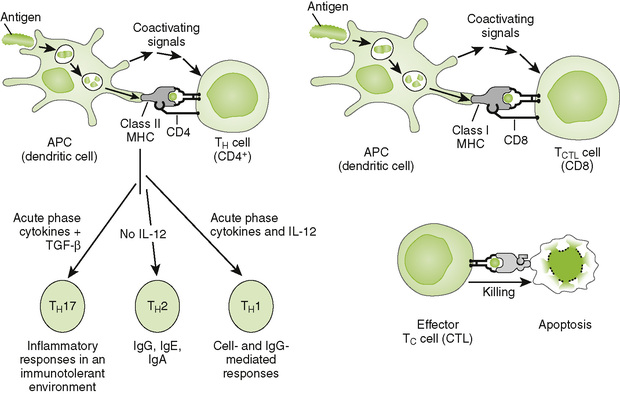
A Cytokine production by CD4 T cells
• DCs activate the naive T cells and determine the type of T cell.
• CD4 T cells differentiate into subsets of effector cells defined by the cytokines they secrete (Fig. 2-6; Table 2-1).
2. TH0 cells: presumed precursor of TH1 and TH2 subsets
3. TH1 cells: characteristic responses mediated by interferon-γ (IFN-γ), lymphotoxin (LT) (tumor necrosis factor-β [TNF-β]), and interleukin-2 (IL-2)
• IL-12 stimulates development and maintenance of TH1 responses.
• Promote cell-mediated and IgG antibody responses
• Reinforce local, innate defense by activating macrophages and stimulating lymphocyte proliferation
• Mediate type IV (delayed-type) hypersensitivity (see Chapter 4)
• Important for intracellular infections (viral, tuberculosis), fungi, and tumors
4. TH2 cells: characteristic responses mediated by IL-4, IL-5, IL-6, and IL-10
• Activate humoral (antibody) responses
• Promote allergic responses (type I hypersensitivity)
• Stimulate antiparasitic eosinophil response (immunoglobulin E [IgE])
B Cytotoxic T lymphocyte (CTL)-mediated killing of target cells
1. CD8 TC cells are activated in lymph node by DCs, which cross-present phagocytosed or internal antigen on class I MHC molecules.
• CD8 TC cells kill virus-infected cells, tumor cells, and transplanted cells expressing antigen on class I MHC molecules.
• Multiple interactions create an immune synapse between the CTL and target cell.
2. Cytotoxic substances released from granules in the CTL attack the target cell.
• Perforin pokes holes in the membrane (similar to complement component C9).
• Granzymes (serine esterases) and other toxic molecules that enter target cell through holes promote apoptosis.
3. Fas ligand on CTLs binds to Fas receptor on the target cell, stimulating apoptosis of target cell.
IV MHC and the Immune Response to Transplanted Tissue (Box 2-3)
A Clinical classification of allograft rejection
1. Hyperacute reaction is a rapid response (within hours) mediated by preexisting antibodies to transplanted alloantigens leading to complement-dependent damage to the graft.
• Preexisting antibodies can arise owing to exposure to alloantigens during previous blood transfusions, transplantation, or multiple pregnancies.
2. Acute reaction, mediated primarily by T cells, begins about 10 days after transplantation.
3. Chronic reaction is marked by fibrosis and vascular injury developing months to years after transplantation.
B Graft-versus-host disease (GVHD)
1. Represents cell-mediated response mounted by lymphocytes in the graft against allogeneic host cells
2. Occurs when a graft containing many lymphocytes (e.g., bone marrow transplant) is transplanted into a host with a compromised immune system due to disease or treatment with immunosuppressive agents.
• Cytokines are low-molecular-weight proteins that induce characteristic cellular responses when they bind to specific receptors on their target cells.
A Cytokine functions and sources (Table 2-2)
TABLE 2-2
| Cytokine | Major Sources | Major Effects and Target Cells |
| IL-1 | Macrophage, dendritic cell, B cell | Acts on various nonimmune cells to initiate acute phase responses, fever Coactivates TH cells |
| IL-2 | TH1 cell | Promotes growth and activation of T and B cells |
| IL-3 | TH cell | Stimulates hematopoiesis in bone marrow |
| IL-4 | TH2 cell, mast cell | Promotes growth and differentiation of B cells Enhances IgG and IgE synthesis Stimulates TH2 response |
| IL-5 | TH2 cell | Promotes growth and differentiation of B cells Enhances IgA synthesis Stimulates growth and activation of eosinophils |
| IL-6 | TH2 cell, macrophage, dendritic cell | Promotes formation of plasma cells from B cells and antibody production Induces synthesis of acute phase proteins by liver cells |
| IL-10 | TH2 cell | Reduces TH1 response by inhibiting IL-12 production by macrophages Reduces class II MHC expression by APCs |
| IL-12 | Macrophage, dendritic cell, B cell | Stimulates formation of TH1 cells Acts with IL-2 to promote formation of CTLs, activates NK cells |
| IL-17 | TH17 cell | Promotes neutrophil activation and inflammatory responses |
| IL-23 | Dendritic cell | Promotes TH17 responses |
| IFN-γ | TH1 cell, NK cell | Enhances macrophage activity Inhibits TH2 response Mediates aspects of type IV hypersensitivity |
| TNF-α | Macrophage and other cells | Has effects similar to IL-1 Promotes cachexia associated with chronic inflammation Is cytotoxic for tumor cells |
| TNF-β (lymphotoxin) | TH1 cell, TC cell | Enhances phagocytic activity of macrophages and neutrophils Is cytotoxic for tumor cells |
| TGF-β | Macrophage, Treg cell, B cell | Generally limits inflammatory response, enhances IgA synthesis |
| CXC-type chemokines (e.g., IL-8) | Macrophage, neutrophil, endothelium, fibroblast | Attracts neutrophils and promotes their migration into tissues |
| CC-type chemokines (e.g., MIP, RANTES) | Macrophage, neutrophil, endothelium, T cell | Attracts macrophages, eosinophils, basophils, and lymphocytes |
• The diverse functions of cytokines can be grouped into several broad classes, but many cytokines exert more than one class of effect.
1. Acute phase, innate, and inflammatory responses
• Include IL-1, TNF-α, IL-6, IL-8, and chemokines
• Secreted primarily by macrophages, DCs, and other nonlymphocytes
2. Th17 antibacterial and inflammatory responses
3. Th1-related local cell-mediated and antibody immune responses
5. Treg immunosuppressive responses
6. Stimulators of inducible hematopoiesis in response to infection
1. Overproduction of IL-1, IL-6, and TNF causes a drop in blood pressure, shock, fever, and widespread blood clotting.
• Endotoxin stimulation of dendritic cells and macrophages following infection by some gram-negative bacteria → bacterial septic shock
2. Massive release of cytokines can affect many systems.
3. Inappropriate cytokine production dysregulates the immune system.