Chapter 115
Nonatheromatous Popliteal Artery Disease
Thomas L. Forbes
The vast majority of patients experiencing lower extremity ischemic symptoms have atherosclerotic occlusive disease. However, in the absence of significant atherosclerotic risk factors, especially in younger and more active individuals, nonatheromatous causes must be considered. Adventitial cystic disease of the popliteal artery and popliteal artery entrapment syndrome are by far the most common of these rare pathologies. Similar to the more common atherosclerosis, the symptoms associated with nonatheromatous causes of popliteal artery occlusive disease range from claudication to critical limb ischemia (CLI). Given the rarity of these symptoms and the younger age of persons affected by them, these conditions are often not diagnosed in a timely fashion, resulting in prolonged disability or occasionally progression to CLI.
Embryology
The embryologic origins of the arterial system are more completely described in Chapter 2, but it is important to review some of the key features to understand the anatomy associated with pathologies affecting the popliteal artery (Fig. 115-1).
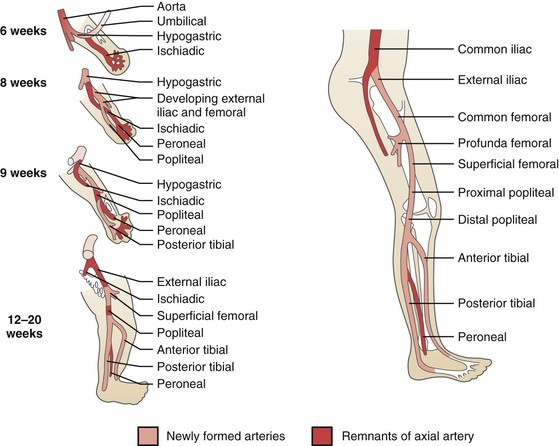
Figure 115-1 Embryologic derivation of the popliteal and other lower limb arteries. Remnants of the axial artery and arteries that develop with later differentiation are indicated. (From Levien LJ, et al: Adventitial cystic disease: a unifying hypothesis. J Vasc Surg 28:193-205, 1998.)
The lower extremity arterial system arises from two arteries—the axial and external iliac arteries—which originate from the umbilical artery. The femoral artery originates from the external iliac artery and progresses distally in the anterior compartment, whereas the axial artery elongates distally in the posterior compartment. At around 42 days of intrauterine life, the axial artery is divided into three segments, depending on its relationship to the popliteus muscle (proximal, deep, and distal); a bridging artery, the ramus communicans superius, joins the femoral artery and the proximal segment of the axial artery through the adductor hiatus. During the next week of development, the proximal component of the axial artery gives rise to a branch that runs superficial to the popliteus muscle and joins with the distal segment of the axial artery; the deep segment of the axial artery involutes. The fully developed popliteal artery results from the fusion of several arterial elements.1
Initially, both heads of the gastrocnemius muscle originate from the proximal tibia. With development, these migrate cranially along the femur to different extents. The final position of the medial head of the gastrocnemius muscle is more proximal to that of the lateral head and immediately caudal to the adductor hiatus, with the popliteal artery lying immediately lateral.1 These dynamic processes of muscle and arterial development create the potential for various anatomic variations.
Adventitial Cystic Disease
Epidemiology
Adventitial cystic disease was first reported in 1947 with the description of a case involving the external iliac artery.2 Since then approximately 400 cases have been reported, with the popliteal artery most commonly affected (85% of cases).3 In the vast majority of cases, popliteal artery involvement is unilateral; only one report of bilateral lesions has been published.4 The next most common arteries involved are the external iliac and femoral arteries,5,6 but involvement has been noted in most of the arteries lying adjacent to joint spaces (Fig. 115-2).7 Additionally, although it is primarily and most commonly a disorder of the arterial system, isolated reports exist of adventitial cystic disease involving the iliofemoral and saphenous veins.8
Adventitial cystic disease affects males predominantly, with a male-female ratio of 5 : 1. The typical age at diagnosis is the mid-30s. Some investigators have reported a slightly older age at diagnosis in women—often in their 50s.9 It must be remembered, however, that diagnosis is often delayed, given the relatively young age of these patients and the absence of atherosclerotic risk factors. As a result, the age at which the pathologic abnormality would be apparent in an asymptomatic person is uncertain. This disease has been observed primarily in western Europe, Australia, Japan, and elsewhere in Asia. Fewer cases have been reported in North America, with the majority occurring on the East Coast.10,11 The prevalence of adventitial cystic disease has been variously reported as 1 in 1200 patients with claudication, regardless of age, and 1 in 1000 diagnostic angiograms.11 These reports include predominantly symptomatic patients, so the incidence of adventitial cystic disease in the general asymptomatic population is unknown.
Pathogenesis
Etiology
The precise cause of adventitial cystic disease remains unclear and somewhat controversial, but some clarification has occurred recently. Previously, four predominant theories of etiology and pathogenesis had been described: repetitive trauma, ganglion, systemic disorder, and developmental theories.9,11 Although convincing data to support the validity of the first three theories are scarce, they are briefly described in the following paragraphs.
Repetitive Trauma Theory.
Proponents of this theory suggest that repeated flexion and extension of the knee joint lead to chronic injury of the popliteal artery characterized by cystic degeneration.7,9,12 It is thought that this repetitive distraction movement of the popliteal artery causes intramural hemorrhage between the adventitia and media, which causes cyst formation when combined with adventitial enzymatic activity. An extension of this theory suggests that subjecting the knee joint (not the popliteal artery) to this repetitive movement and stress leads to joint degeneration and changes in the surrounding connective tissue. Consequently, these connective tissue cells secrete a substance containing hydroxyproline that results in adventitial cyst formation.12
Although the repetitive trauma theory is somewhat attractive, given its simplicity and relative intuitiveness, scientific data to support it are scarce. Repetitive trauma as a causative factor does not explain cases occurring in arteries that are not subjected to such stress or in younger patients who have not been subjected to the same duration of this stimulus as have older patients. If this theory were correct, we would expect more cases of adventitial cystic disease in more active individuals or athletes, and there would be a positive correlation between age and incidence of the disease. Neither is the case.
Ganglion Theory.
Proponents of this theory of pathogenesis have been prompted by the similar content of simple ganglions and popliteal artery cysts.7,9,13 Specifically, both types of cystic structures contain high levels of hyaluronic acid. Additionally, some support is offered by periodic case reports of synovial cystic structures and Baker’s cysts directly involving adjacent vascular structures.14 Presumably, in the case of the popliteal artery, these synovial cysts enlarge and track along arterial branches, where they implant in the adventitia of the popliteal artery itself.3 Support for this theory would include evidence of histologic similarities between the lining and chemical content of the cystic fluid in the synovium and that of popliteal artery cysts. Decades ago, several researchers failed to find any such similarities, resulting in this theory’s falling into disfavor. Specifically, fluid from these cysts has a much higher hyaluronic acid content than synovial cysts.15
Systemic Disorder Theory.
The third theory of pathogenesis for adventitial cystic disease, also lacking much supporting evidence, is that of a systemic disorder—specifically, the existence of a systemic mucinous or myxomatous degenerative condition. This theory was first postulated in 1967,16 but since then, little epidemiologic or pathologic supporting evidence has arisen, and the suspected systemic condition has never been discovered. In addition, reports of bilateral disease are exceedingly rare,4 as are cases of synchronous or metachronous cysts in different vascular locations.
Developmental Theory.
Currently, the developmental theory of pathogenesis has the greatest degree of acceptance and the most convincing body of evidence. Also known as the cellular inclusion theory, it proposes that adventitial cystic disease occurs when mesenchymal mucin-secreting cells are implanted in the adventitia of the vessel during development. Levien and Benn highlighted the embryologic development of these arteries in support of this proposal.7 They recognized that these nonaxial arteries form from vascular plexuses between 15 and 22 weeks of embryologic development adjacent to developing joints. During this time, mesenchymal cell rests forming these joints can be incorporated into closely adjacent vessels and may be responsible for subsequent cyst formation when these mesenchymal cells start to secrete mucoid material.
Connections between the knee joint capsule and an adjacent popliteal artery adventitial cyst have periodically been identified intraoperatively and by preoperative imaging.2,3,9,16–19 On the one hand, the presence of such a connection could lend support to the ganglion theory of development, with the connection representing a direct communication between the joint capsule and the arterial adventitial layer through which synovial cysts can migrate.3,17,19 Alternatively, proponents of the developmental theory claim that these communications represent a residuum of the embryologic process, when mesenchymal cells of the adjacent joint are included in the adventitia of the nearby developing artery.3,7 As mentioned previously, the biochemical analysis of these adventitial cysts does not support the ganglion theory because these cysts have a greater hyaluronic acid concentration than do synovial cysts.15
All four theories of development have been proposed as rational explanations for adventitial cystic disease at one time or another. However, the prevailing opinion and, more important, the current body of evidence largely support the developmental theory as the most rational explanation. In the absence of convincing supporting evidence, the other three theories can be viewed in the appropriate historical context.
Pathology
Popliteal artery adventitial cysts are filled with a gelatinous mucoid material. Microscopic examination reveals a simple cuboid cell lining in the adventitial layer, with a notable absence of any coexisting microscopic features of atherosclerotic disease (Fig. 115-3). Grossly, the popliteal artery may appear enlarged and sausage-like, with adhesions to adjacent structures (Fig. 115-4). The cyst is usually unilocular but can be multilocular. Cyst contents are usually clear or yellow but can be dark red following hemorrhage. At the time of operation, these cysts are apparent following incision of the adventitial layer.
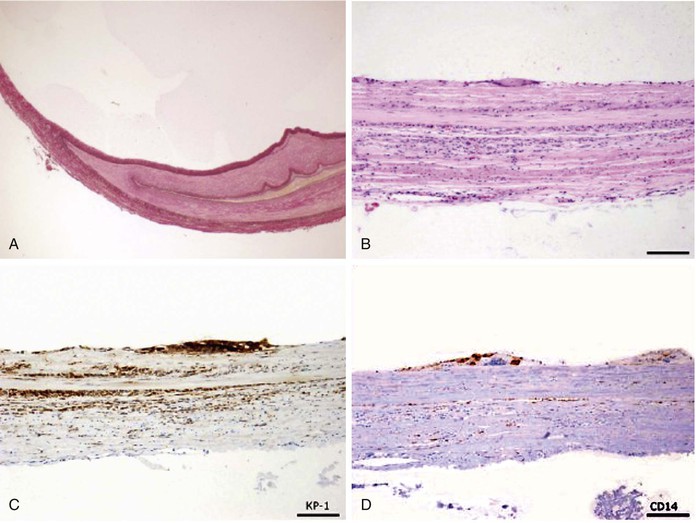
Figure 115-3 Adventitial cystic disease. A, Elastica van Gieson stain. B, Hematoxylin and eosin staining, cells lining the cysts with eosinophilic or clear cytoplasms with vacuolar or foamy structures and no basement membranes. C, CD68 stain positive cells on cyst lining. D, CD14 stain positive cells on cyst lining. (From Keiji O, et al: Rapid recurrence of cystic adventitial disease in femoral artery and an etiologic consideration for the cyst. J Vasc Surg 53:1702-1706, 2011.)
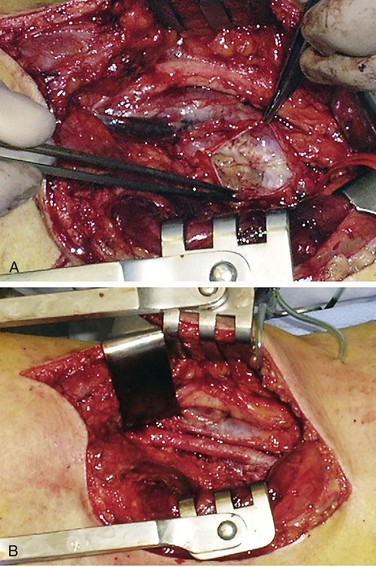
Figure 115-4 A, Adventitial cyst of popliteal artery opened and evacuated. B, Popliteal artery after cyst excision. (From van Rutte PWJ, et al: In treatment of popliteal artery cystic adventitial disease, primary bypass graft not always first choice: two case reports and a review of the literature. Eur J Vasc Endovasc Surg 42:347-454, 2011.)
Clinical Presentation
Arterial
The typical patient with adventitial cystic disease of the popliteal artery is a male in his mid-30s who complains of fairly sudden onset of short-distance calf claudication.11 The duration of symptoms is generally relatively short (weeks to a few months), and except in unusual cases, these symptoms are unilateral.4 Claudication symptoms may completely resolve for a period of time and then recur, or they may progress rapidly. Also, recovery time is often prolonged (20 minutes) compared with that of typical claudicants.20 The differential diagnosis includes popliteal artery entrapment syndrome (discussed later in this chapter), fibromuscular dysplasia, endofibrosis, and premature atherosclerosis. Given the focality of these cysts, the young age of patients, and the otherwise normal status of inflow and outflow vessels, progression to CLI is exceedingly unusual with adventitial cystic disease, although the severity of claudication can progress and become disabling. It appears that the cysts have been present and slowly enlarging for extended periods before patients enter the symptomatic phase. These enlarging cysts lead to progressive compression of the arterial lumen and can result in a “functional” occlusion of the artery without necessarily causing complete thrombosis. In fact, in cases of apparent arterial occlusion without thrombosis, evacuation of cyst contents can restore arterial patency, supporting the pathophysiology of extrinsic luminal compression with a normal intimal layer. Of course, prolonged compression of a compromised lumen can lead to popliteal artery thrombosis and a fixed occlusion. There have been anecdotal reports of spontaneous cyst resolution,21 but this appears to be extremely rare and should not be considered a feature of this disorder.
Approximately two thirds of patients present with popliteal artery stenosis rather than occlusion. On physical examination, this may be demonstrated by normal or diminished pedal pulses and by an audible bruit in the popliteal fossa. Pedal pulses that are present at rest may disappear with flexion of the knee (Ishikawa’s sign), representing a functional stenosis that proceeds to occlusion with this physical manipulation. This is in contradistinction to popliteal artery entrapment, in which case pedal pulses disappear with gastrocnemius muscle contraction caused by active plantar flexion or passive dorsiflexion of the foot.11
Venous
Cases of lower extremity venous involvement with adventitial cystic disease have been reported sporadically.8 As with arterial involvement, these venous cysts occur predominantly in males; they most commonly involve the iliofemoral venous segments, with only occasional instances of popliteal or saphenous vein involvement. The most common presentation of this rare condition is similar to that of deep venous thrombosis. Typically, a young, healthy male presents with painless swelling of the lower extremity. Venous adventitial cystic disease should be considered when there is evidence of extrinsic compression on venous duplex imaging or a filling defect on venography. The optimal method of management of venous adventitial cystic disease is not well defined, but most authors advocate operative exploration involving a venotomy with evacuation of the cyst contents, followed by cyst wall excision in the hope of minimizing the risk of recurrence.
Diagnostic Evaluation
Noninvasive studies are consistent with an isolated arterial lesion. Ankle-brachial indices are unaffected at rest and drop following exercise. This pattern should raise the suspicion of an arterial cause of the patient’s symptoms and prompt further investigation.
As with all other arterial pathologies, we have seen a steady progression of diagnostic modalities from standard angiography and Doppler ultrasound technologies to cross-sectional imaging with computed tomography (CT) and magnetic resonance imaging (MRI). Although all these methods have advantages and disadvantages with regard to adventitial cystic disease, current recommendations advocate the use of duplex scanning followed by CT or MRI as the best diagnostic approach.11
Noninvasive Testing
Duplex ultrasound should be considered the initial diagnostic tool for this disorder.22,23 The number of cysts and their dimensions can be easily evaluated; their presence, along with elevated Doppler velocities through the affected segment, can be considered diagnostic. The boundary between the cyst and the arterial lumen is depicted by a fine bright line that pulsates. Ultrasound can also differentiate these cysts from popliteal artery aneurysms by the absence of flow signals in the cysts themselves. Following intervention, duplex scanning is a useful postoperative surveillance tool to exclude cyst recurrence and residual or recurrent stenosis.
Angiography
Traditionally, angiography was the diagnostic modality for adventitial cystic disease, but this has now been largely replaced by noninvasive methods. Complete popliteal artery occlusion is demonstrated with angiography in up to one third of cases, and the remaining studies demonstrate an eccentric compression of the popliteal artery lumen known as the “scimitar” sign (Figs. 115-5 and 115-6).22 Caution is warranted, however, because stenosis can be missed on anteroposterior views and may be evident only with lateral projections. In the absence of arterial thrombosis, this eccentric stenosis in the absence of poststenotic dilatation is a specific diagnostic sign of adventitial cystic disease. However, the diagnostic capability of conventional angiography is limited in the case of arterial occlusion. In this situation, angiography provides little accurate information regarding arterial wall pathology and the surrounding soft tissue.11
Computed Tomography and Magnetic Resonance Imaging
CT is being used more extensively in cases of popliteal artery disease (Fig. 115-7). Its main contribution to the evaluation of adventitial cystic disease is to differentiate this pathology from popliteal entrapment syndrome and aneurysmal formation, especially in cases of popliteal artery occlusion or thrombosis. CT is superior to angiography in this regard, particularly in the case of arterial occlusion; CT has the ability to demonstrate the nature of the cysts and their relationship to surrounding structures.24 It can also exclude the muscular abnormalities apparent in popliteal artery entrapment syndrome.
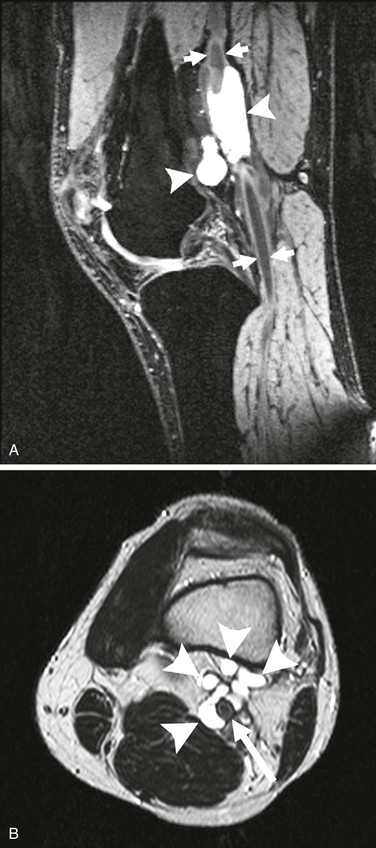
Figure 115-7 A, Cystic structures (arrowheads) in close contact with the popliteal artery (arrows). B, Cystic structures (arrowheads) in close proximity to popliteal artery (arrow). (From van Rutte PWJ, et al: In treatment of popliteal artery cystic adventitial disease, primary bypass graft not always first choice: two case reports and a review of the literature. Eur J Vasc Endovasc Surg 42:347-354, 2011.)
In the past decade, MRI has supplanted other techniques as the optimal diagnostic modality for adventitial cystic disease in the view of some investigators.25 Obvious advantages of MRI over CT include the avoidance of ionizing radiation and intravascular contrast agents. MRI clearly depicts the extent of cystic involvement, and many authors consider it essential during the planning of surgical intervention. The typical finding on MRI is an “hourglass” deformity in the popliteal artery. Technically, some authors recommend formatting MRI to include T2-weighted and gradient-echo sequences for suspected cases of adventitial cystic disease.11
Despite these strongly held beliefs, little convincing evidence supports the use of MRI over CT. However, few investigators would argue against the use of a cross-sectional imaging modality for the diagnosis of adventitial cystic disease and subsequent treatment planning. The additional information on cyst extent and anatomy provided by these techniques compared with conventional angiography can be invaluable, especially in cases of complete arterial occlusion. Duplex ultrasound remains useful as an initial diagnostic test and as an instrument of postoperative surveillance.
Treatment
Given the rarity of adventitial cystic disease, no individual center has accumulated the critical mass of patients required to offer clear guidelines about its treatment and management. Treatment recommendations are based on an evaluation of cumulative experience as opposed to randomized controlled studies. In addition, information on recurrence rates is rare. Because most case descriptions are anecdotal, so are descriptions of recurrence. Therefore data on recurrence risks are best described as being of greater or lesser risk versus other treatment options, as opposed to citing specific percentages.
Generally, management options for adventitial cystic disease can be divided into resectional and nonresectional procedures (Table 115-1).3,11
Table 115-1
Management Options for Adventitial Cystic Disease of the Popliteal Artery, from Highest to Lowest Risk of Recurrence

Resection, with subsequent arterial reconstruction, is more commonly used in cases of complete popliteal artery occlusion secondary to thrombosis or in the presence of extensive degeneration of the arterial wall. In the majority of instances when nonocclusive stenoses are encountered, nonresectional methods are recommended.
Nonresectional Methods
Nonresectional methods of treatment include:
These treatment methods are listed in order of increasing chance of initial success and decreasing recurrence rate. Angioplasty has been discarded as a treatment option by most investigators. Its ineffectiveness is based on the normal intimal layer of these arteries and the compliant arterial segment, which can cause recoil and restenosis as early as 24 hours following balloon dilatation.26 Preservation of the secretory lining of the adventitial cysts following angioplasty almost universally leads to disease recurrence.
Cyst Aspiration.
Promising short-term outcomes have been achieved with CT- or ultrasound-guided cyst aspiration. The technique is well described, including the precise positioning of the needle tip to avoid the popliteal vein and tibial and peroneal nerves.11,27 Although the technique is relatively simple and minimally invasive, treatment failures following percutaneous cyst aspiration are not unusual in cases of multiple loculations and highly viscous cyst fluid. Symptoms recur in 10% to 30% of patients at a mean follow-up period of 15 months.20,28 Imaging-guided cyst aspiration has a place in the management of this arteriopathy, but given the risk of incomplete evacuation and recurrence, it should be limited to those patients who refuse operative intervention and agree to close imaging surveillance and probable reintervention.
Cyst Excision or Cyst Evacuation.
Operative exposure of the involved popliteal artery is best achieved via a posterior approach with the patient prone. In the case of a stenotic popliteal artery, incision into the cyst and evacuation of its contents is usually sufficient to restore arterial patency. However, recurrence rates following this form of treatment may be as high as 10%,11 causing some authors to recommend cyst excision and evacuation to limit the chance of recurrence.
Resectional Methods
In instances of popliteal artery thrombosis or extensive arterial degeneration, a resectional treatment option is preferred. The affected popliteal artery is explored through a posterior approach, and the extent of arterial resection is determined by the length of arterial involvement on preoperative cross-sectional imaging and intraoperative findings. Arterial reconstruction is performed with an autogenous venous conduit of the surgeon’s choice. Although recurrence of adventitial cystic disease after such resective therapy is unusual, there have been reports of recurrence after vein graft interposition following arterial resection.29 Presumably, this was due to incomplete resection of the involved artery at the original operation.
Recurrence
Recurrence of adventitial cystic disease has been described following all methods of therapy, although it is less likely with resection of the cyst or the involved artery. In a recent review, treatment failure or recurrence was seen in 15% of patients who underwent cyst evacuation and 6.7% of those undergoing resection.30 Choice of therapy is determined by the luminal status of the popliteal artery. In nonoccluded arteries, nonresectional methods, including imaging-guided cyst aspiration, or operative cyst evacuation and excision offer good short-term outcomes. The operative approach is most definitive and has the lowest chance of recurrence. In instances of popliteal artery thrombosis, resection is advocated, with excision of the involved artery and reconstruction with an autogenous conduit. Given the recurrence risk with all these therapies, indefinite and periodic postoperative duplex surveillance is necessary.
Popliteal Artery Entrapment Syndrome
Epidemiology
Popliteal artery entrapment syndrome should be considered in any young individual with exertional calf discomfort. As an anatomic variant, entrapment of the popliteal artery has been recognized for more than 100 years, but it was not until 40 years ago that the term popliteal artery entrapment syndrome was used to describe the clinical entity.1,31 The anatomic abnormalities were observed in 3.5% of individuals in a postmortem study,32 and up to 60% of young individuals with claudication symptoms suffer from popliteal artery entrapment syndrome.33 This makes it more common than adventitial cystic disease. Those affected tend to be active, giving this syndrome the moniker the “jogging disease.”
The vast majority (up to 80%) of reported cases have occurred in men with a reported median age of 32 years34 (range, 20.7-41 years). Cases of popliteal artery entrapment syndrome in women older than 50 years have been reported but are unusual. Of course, the anatomic variant is present since its embryologic origin in utero, but it appears that at least several decades are necessary for the development of symptoms. Popliteal artery entrapment syndrome has been described as being simultaneously symptomatic in both lower extremities in approximately 30% of cases.35 Patients with unilateral symptoms were found to have bilateral anatomic abnormalities two thirds of the time.35 Rare instances of bilateral popliteal artery occlusion secondary to popliteal artery entrapment have been reported.36
Pathogenesis
Etiology
The anatomy observed in the different types of popliteal artery entrapment is best understood by considering the embryologic development of the various structures of the popliteal fossa (see “Embryology” earlier in this chapter). Similar to the development of the lower extremity arterial system, the in utero development of the gastrocnemius muscle is a dynamic process with the potential for the occurrence of several anatomic abnormalities. Specifically, the medial head of the gastrocnemius muscle arises from the posterior fibula and lateral tibia, and during subsequent medial limb bud rotation and knee extension, it migrates across the popliteal fossa to its final attachment on the posterior aspect of the medial femoral condyle.35 As described previously, the popliteal artery forms superficial to the popliteus muscle at about the same time as this medial migration of the medial head of the gastrocnemius muscle across the popliteal fossa (Fig. 115-8).

Figure 115-8 Migration of the medial head of the gastrocnemius muscle through the popliteal fossa during formation of the popliteal artery. A, The medial head of the gastrocnemius muscle begins to migrate from the region of the fibula. At this stage, the axial distal popliteal artery lies deep to the popliteus muscle. B, The distal portion of the popliteal artery involutes as the medial head of the gastrocnemius muscle passes from lateral to medial. The proximal popliteal artery is derived from fusion with the developing femoral plexus, whereas the midportion of the popliteal artery is formed from the persistent axial artery remnant. C, A new or nonaxial distal popliteal artery now forms superficial to the popliteus muscle, after the medial head has migrated through the popliteal fossa. D, Normal definitive popliteal anatomy. (From Levien LJ, et al: Popliteal artery entrapment syndrome: more common than previously recognized. J Vasc Surg 30:587-598, 1999.)
Classification
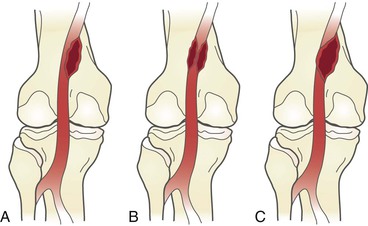
With an appreciation of the embryologic development of the popliteal fossa, a classification system based on this developmental anatomy was developed. The basis of this system was the Heidelberg classification, which originally described three types (I to III) of popliteal artery entrapment syndromes (Fig. 115-9).37 At present, the classification system describing popliteal artery entrapment syndrome consists of six variants, types I to VI.

Figure 115-9 Types of popliteal artery entrapment syndromes. (From Levien LJ, et al: Popliteal artery entrapment syndrome: more common than previously recognized. J Vasc Surg 30:587-598, 1999.)
Type I.
The popliteal artery completes its development before migration of the medial head of the gastrocnemius muscle, which then pushes the artery medially during migration. This results in the popliteal artery’s lying medial to a normally situated gastrocnemius muscle, which is associated with an anatomic and radiologic medial deviation of the artery.35
Type II.
The artery is displaced medially, but the medial head of the gastrocnemius muscle has a variable attachment on the lateral aspect of the medial femoral condyle or intercondylar area. In this case, the artery forms prematurely and partially arrests the migration of the gastrocnemius muscle, resulting in the artery’s being positioned medial to an abnormally attached muscle. In contrast to the normal position of gastrocnemius muscle insertion in type I entrapment, type II is defined by an abnormal femoral insertion site.
Type III.
An abnormal muscle slip or fibrous band that arises from either the medial or lateral femoral condyle.35 Type III entrapment occurs when embryologic remnants of the gastrocnemius muscle remain posterior to the popliteal artery or when the artery develops within this muscle mass. Occasionally, a double origin of the gastrocnemius muscle can surround and compress the popliteal artery.
Types I to III represent the same anatomic variant, the degree of which depends on the temporal relationship between popliteal artery development and migration of the medial head of the gastrocnemius muscle.
Type IV.
The mechanism resulting in type IV entrapment is fundamentally different from that causing types I to III. Type IV entrapment occurs with persistence of the axial artery as the mature distal popliteal artery. This artery remains in its embryologic position, deep to the popliteus muscle or fibrous bands.35
Type V.
Both the popliteal artery and popliteal vein are involved or entrapped via any of the previously described mechanisms. This subtype has been estimated to occur in approximately 10% to 15% of cases.38,39
Type VI.
An additional type of entrapment—previously known as functional entrapment, or type F—is now commonly referred to as type VI entrapment. This occurs in otherwise healthy individuals who describe the typical symptoms of entrapment and show appropriate compression of the artery with stress maneuvers in the absence of an explanatory anatomic abnormality.35 The anatomic abnormality in type VI entrapment has been difficult to determine, but it has been proposed that an especially lateral attachment of the medial head of the gastrocnemius muscle to the posterior aspect of the medial femoral condyle predisposes to this condition. Close to half of these asymptomatic individuals show reduction or complete occlusion of popliteal artery flow with such stressor mechanisms as plantar flexion or dorsiflexion against resistance.40 Hypertrophy of this muscle following regular exercise is thought to result in compression of the posteromedial aspect of the popliteal artery. When subjected to MRI examination, individuals with functional entrapment and popliteal artery occlusion with stressor maneuvers tend to have a more extensive midline position of the medial head of the gastrocnemius muscle. When compared with normal controls, patients with functional entrapment have more of this muscle attached toward the supracondylar femoral midline, around the lateral border of the medial femoral condyle, and within the intercondylar fossa.41
Pathology
A classification of the progression of histologic changes in popliteal artery entrapment syndrome has been described.35 These histologic changes are the result of continuing arterial compression causing the progression from fibrosis of the various arterial layers to complete occlusion or thrombosis (Fig. 115-10). In this pathologic classification scheme, the fibrosis is confined to the adventitia in stage 1; it extends into the media in stage 2, which may result in poststenotic dilatation or aneurysm formation. In stage 3, the artery may become thrombogenic, independent of the compression, as the fibrosis extends into the intimal layer.39 The degree of arterial damage does not appear to be directly related to the duration of compression or popliteal entrapment. The major clinical significance of these findings is the potential thrombogenicity of a chronically entrapped popliteal artery that might not be corrected with any surgical intervention short of arterial replacement.
Clinical Presentation
The most common presentation of an individual with popliteal artery entrapment syndrome is that of a young, healthy man who participates in sports and has symptoms of claudication. However, a range in the severity of associated symptoms has been described, and a grading scale describing these symptoms was recently proposed following a forum specifically convened to discuss popliteal artery entrapment syndrome1:
Class 1: Pain, paresthesia, and cold feet after physical training (e.g., jogging, heavy work)
Class 2: Claudication while walking (>100 m)
In a large series of patients, 46% of patients had class 1 symptoms; 35%, class 2; 14%, class 3; and 2%, class 5.1 None had class 4.
The vast majority (70% to 90%) of patients with symptomatic popliteal artery entrapment present with intermittent calf claudication. This classically resolves with rest. In some cases, the claudication symptoms may be atypical and paradoxical, in that they worsen with standing or walking but improve with more vigorous exercise, or they may occur immediately after the onset of walking rather than after walking a longer distance. Patients occasionally describe symptoms of coldness, blanching, and numbness. All types of entrapment (except type VI) can involve the tibial nerves and result in certain degrees of paresthesia. The minority of patients (10%) present with signs and symptoms of chronic CLI, and even fewer present with acute limb ischemia. Venous entrapment (type V) is suggested by calf cramping or symptoms more typical of a compartment syndrome, including swelling and a feeling of fullness.39
Typically, pedal pulses are palpable and normal at rest in these individuals, as long as compression of the popliteal artery has not progressed to occlusion. These pulses disappear with passive dorsiflexion or active plantar flexion of the foot. These maneuvers tense the gastrocnemius muscle against the entrapped artery, temporarily occluding the patent lumen. They should also be performed on the contralateral asymptomatic limb. If left untreated, chronic entrapment causes progressive fibrosis of the popliteal artery, resulting in a thrombogenic intimal surface and eventual arterial thrombosis and occlusion.35 Occlusion of the artery can be heralded by the sudden onset of more severe calf claudication with absent pedal pulses. This can occur after an episode of strenuous exercise and should result in a high degree of suspicion for popliteal artery entrapment syndrome when it occurs in a young adult with no manifestations of or risk factors for atherosclerosis.
Poststenotic dilatation or aneurysmal degeneration of the popliteal artery can occasionally occur and be a source of distal emboli, as can thrombus formation in a normal-caliber entrapped artery.35
Diagnostic Evaluation
The specific diagnostic and imaging protocol varies greatly among centers. Generally, the most common initial tests are ankle-brachial pressure indices before and after treadmill exercise testing and duplex scanning, combined with dynamic provocative maneuvers that cause gastrocnemius muscle contraction—namely, active plantar flexion and passive dorsiflexion of the ankle with the knee in full extension.38
Noninvasive Testing
Exercise treadmill testing is performed with the patient walking or jogging until symptoms develop. Ankle-brachial pressure measurements are performed before and following the treadmill test and should drop significantly. Subsequently, segmental pressures are taken with the Doppler probe placed on the posterior tibial artery during calf muscle contraction and relaxation. Readings are repeated several times, and care should be taken to avoid moving the probe during muscle contractions. Similarly, duplex studies are performed with visualization of the popliteal artery during calf muscle contraction and relaxation. The duplex interrogation must be repeated several times because the popliteal artery can be pushed deeper into the popliteal fossa during muscle contraction, negatively affecting visualization and volume recordings. Although a positive duplex test with provocative maneuvers should prompt further investigation, it should not serve as the only basis for operative intervention. Very high (72%) false-positive rates have been reported, resulting in an overestimation of popliteal artery compression.39
Angiography
Although cross-sectional imaging techniques have gained ground, angiography remains the mainstay of investigation at many centers with a reported median sensitivity of 97% (range 85%-100%).34
Generally, the diagnosis of popliteal artery entrapment syndrome should be considered when at least two of the following angiographic features are apparent1:
Medial deviation of the artery is most often apparent in types I, II, and III popliteal artery entrapment, but it may be subtle. Focal occlusion of the mid–popliteal artery in young patients without risk factors for or evidence of atherosclerotic disease is due to either adventitial cystic disease of the popliteal artery or popliteal artery entrapment syndrome in most cases. Poststenotic dilatation of the distal popliteal artery is apparent in an estimated 12% of angiograms of patients with popliteal artery entrapment. Angiography has the additional advantage of delineating tibial artery anatomy following emboli from a poststenotic dilatation of an entrapped popliteal artery or a thrombogenic entrapped artery.
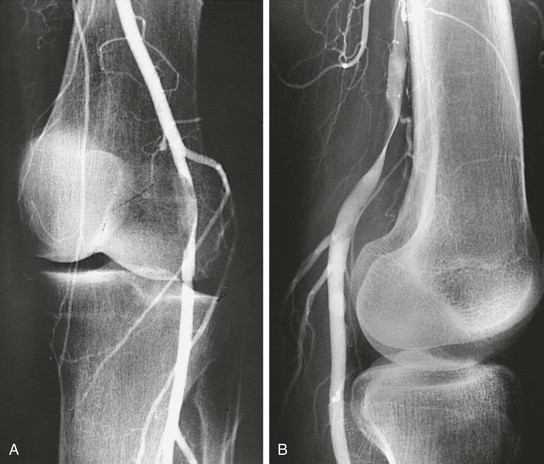
Several angiographic views should be obtained both in the neutral position and during provocative maneuvers such as passive dorsiflexion and active plantar flexion of the foot with the knee in full extension. These views increase the sensitivity of angiography in cases of popliteal artery entrapment syndrome because compression or occlusion is often not apparent in the neutral position without active contraction of the gastrocnemius muscle (Fig. 115-11). This contrasts with adventitial cystic disease of the popliteal artery; in that case, although stressor maneuvers (i.e., knee flexion) can cause complete arterial occlusion, the arterial stenosis or compression from adventitial cysts is readily apparent at rest or in the neutral position (previously described as the “scimitar” sign).
Computed Tomography and Magnetic Resonance Imaging
Less invasive imaging alternatives such as CT and MRI can be particularly useful in cases of popliteal artery entrapment syndrome when the artery is occluded. They can illustrate the particular anatomic relationships between the artery and muscle in the popliteal fossa and identify anomalous muscular insertions (Fig. 115-12). Although these claims are subjective and not backed up by extensive evidence, some investigators claim that MRI is superior to CT in this regard and should be the diagnostic test of choice in young patients suffering from intermittent claudication.39

Figure 115-12 Popliteal entrapment syndrome type I, A, medial head of gastrocnemius muscle (arrowhead) occludes popliteal artery (arrow). B, Patent artery following musculotendinous resection and popliteal artery interposition venous graft. (From Kim SY, et al: Long-term outcomes after revascularization for advanced popliteal artery entrapment syndrome with segmental arterial occlusion. J Vasc Surg 55:90-97, 2012.)
Treatment
In most cases of symptomatic popliteal artery entrapment syndrome, surgical intervention is indicated and should be offered. This is especially true for types I to V entrapment; the decision may be less clear in type VI entrapment, depending on the severity of symptoms. Generally, intervention during an earlier clinical stage results in a more limited operation—namely, myotomy alone rather than arterial reconstruction. This statement is based on knowledge of the natural history of entrapment, which is one of progressive arterial fibrosis and damage and eventual thrombosis and occlusion. As a result, most authors advocate surgical correction of types I to V popliteal artery entrapment to avoid the risk of later significant arterial degeneration.
The basic principles of intervention include release of arterial entrapment, restoration of normal anatomy, and restoration of arterial flow.39 These goals are best achieved with operative procedures because endoluminal therapies do not deal with muscular entrapment. There have been reports of small numbers of patients with occluded popliteal arteries undergoing endoluminal interventions and thrombolysis followed by myotomy several weeks later. These patients were anticoagulated for various periods.42 Anticoagulation and the preservation of a potentially thrombogenic popliteal artery are suboptimal treatment options in this young, healthy, active patient population.
A recent systematic review attempted to determine evidence-based treatment guidelines for popliteal entrapment syndrome but was unable to do so, identifying the need for prospectively obtained data and strict adherence to reporting standards.34 The specific approach for an individual patient is dictated by the anatomy, the clinical presentation, and the status of the popliteal artery (Table 115-2). These different scenarios are discussed in the following sections.
Types I to V, Normal Popliteal Artery
These symptomatic patients have normally patent popliteal arteries at rest but display evidence of occlusion during noninvasive investigations and angiography with provocative maneuvers, as previously described. Preoperative investigations often indicate whether the entrapped artery is suffering from damage or fibrosis, but it is often not until operative exposure and palpation of the artery that the degree and extent of fibrosis are apparent. In the absence of arterial fibrosis and in the presence of a normal-appearing artery, therapy limited to musculotendinous release is sufficient to restore normal anatomy.39 In one large series, three quarters of limbs were treated in this manner with myotomy of the medial head of the gastrocnemius muscle, abnormal muscle slips, or tendinous bands. When followed for up to 10 years, patients treated in such a manner were able to return to their prior sports activities, did not require any further interventions, and maintained patency of their popliteal arteries.35
Musculotendinous release can be performed through either a posterior or a medial approach. Proponents of the posterior approach highlight the operative flexibility it offers the surgeon, the wider degree of inspection possible, the greater ease of identifying and addressing the specific anatomic abnormality, and an adequate exposure to complete an arterial reconstruction if necessary.39,43 Following an S- or Z-shaped incision, flaps are raised to expose the deep fascia, which is incised longitudinally, avoiding injury to the median cutaneous sural nerve. Sacrifice of the lesser saphenous vein can facilitate exposure. As the vessels are approached, the tibial nerve is encountered and mobilized. The popliteal vein is identified, passing between the heads of the gastrocnemius muscle deep in the popliteal fossa. The popliteal artery, which is not in its normal position, is identified higher in the popliteal space and followed distally. The artery’s abnormal course can be medial to the medial head of the gastrocnemius muscle or entrapped by anomalous muscular structures or tendinous tissue.43
Through this posterior exposure, the medial head of the gastrocnemius muscle or the entrapping musculotendinous bands are completely divided. In most cases, it is not necessary to reattach the muscle because the medial head of the gastrocnemius muscle can be divided with no adverse functional sequelae, even in these young, active patients.44 In entrapment types III and IV, it is usually not necessary to divide the tendon of the medial head of the gastrocnemius muscle; mobilization of the muscular portion of the medial head off the posterior aspect of the femoral condyles usually suffices to relieve compression of the artery.
With this scenario, the medial operative approach is most suited for popliteal artery entrapment types I and II and is less appropriate for types III and IV, where it may be more difficult to delineate the arterial and muscular anatomy. Entrapment types III and IV may best be explored via the posterior approach. Type V entrapment can be explored through either route, depending on the underlying muscular abnormality. The medial approach seems to result in a quicker return to normal athletic activities in these active patients and less incision-related morbidity.35 Similar to the posterior approach, the medial head of the gastrocnemius muscle is divided when approached from its medial aspect, permitting complete arterial decompression.
Types I to V, Abnormal Popliteal Artery
Arterial bypass or replacement is indicated in cases of complete thrombosis, arterial wall degeneration from the chronic entrapment, thrombus formation on the intimal surface, fibrotic narrowing of the artery, and post-stenotic dilatation or aneurysm formation. When the popliteal artery shows evidence of chronic damage, even if the extent is only minimal fibrosis, it should be replaced or bypassed in its entirety. Allowing continued flow through the native artery leaves a residual intimal surface with unpredictable thrombogenic potential in a young, active individual and provides a setup for symptom recurrence due to arterial thrombosis.
Early reports of this syndrome described numerous instances of thromboendarterectomy with or without vein patch angioplasty. This approach produced inferior results, with a higher incidence of arterial thrombosis and reocclusion compared with arterial replacement with an autogenous conduit.39 Once a popliteal artery is occluded or thrombosed, it should be considered beyond repair and be replaced.
This arterial reconstruction can be performed through a posterior or a medial approach. The posterior approach permits use of the lesser small vein as the venous conduit, but it is less useful in cases requiring a more distal reconstruction. Several advantages exist with the medial approach, including the ease of harvesting the more proximal great saphenous vein if a conduit of larger caliber is required. Additionally, it is much easier to expose the more distal popliteal artery, or tibial arteries, through a medial exposure if a more distal revascularization is required. This may be the case with extensive poststenotic dilatation or with tibial artery occlusion secondary to thromboemboli from the entrapped popliteal artery.
In these cases, the entrapment is first relieved by dividing the muscle or tendinous segment causing the arterial compression. Resection of the thrombosed artery and a short interposition venous graft are then performed. Alternatively, a short venous bypass graft can be performed, with exclusion of the occluded artery to prevent distal thromboemboli. If poststenotic dilatation or aneurysm formation has occurred, arterial resection and venous replacement are performed. Alternatively, ligation and exclusion of the affected arterial segment may be performed, followed by bypass of the excluded segment with autogenous vein, although this type of bypass may have inferior patency rates compared with a venous interposition graft placed via the posterior approach.45
Type VI, Symptomatic
It is widely recognized that type VI functional entrapment due to hypertrophy of the medial head of the gastrocnemius muscle can lead to progressive popliteal artery damage, fibrosis, and eventual thrombosis. Most authors support an aggressive approach to surgical intervention when faced with symptomatic patients with functional entrapment.
In these cases, surgical decompression can occur through either a medial or a posterior approach. Transection and resection of the muscular portion of the medial head of the gastrocnemius muscle, with preservation of the tendon, is sufficient to relieve symptoms.39 To ensure adequate decompression, one must take care to completely transect the muscular fibers from the posterior aspect of the lateral femoral condyle and the intercondylar area. Adequacy of the extent of the myectomy can be determined with intraoperative duplex. Before resection, arterial systolic velocities are measured and compared with postmyectomy velocities in neutral, plantar, and dorsiflexion positions. Myectomy continues until no velocity changes remain.46
Type VI, Asymptomatic
As mentioned previously, up to half the normal, asymptomatic population may exhibit signs of popliteal artery compression with provocative measures such as active plantar flexion and passive dorsiflexion of the foot. This can be demonstrated by the loss of pedal pulses with these maneuvers or by angiographic investigation. When these individuals are truly asymptomatic, little evidence supports prophylactic operative intervention. These asymptomatic individuals are best followed.39
Similarly, although bilateral popliteal artery entrapment is common, often only one extremity is symptomatic (43% of cases).47 These asymptomatic contralateral extremities should be investigated, but surgical exploration is seldom recommended in the absence of symptoms or positive investigations.32
Selected Key References
Causey MW, Singh N, Miller S, Quan R, Curry T, Andesen C. Intraoperative duplex and functional popliteal entrapment syndrome: strategy for effective treatment. Ann Vasc Surg. 2010;24:556–561.
di Marzo L, Cavallaro A. Popliteal vascular entrapment. World J Surg. 2005;29:S43–S45.
Levien LJ, Veller MG. Popliteal artery entrapment syndrome: more common than previously recognized. J Vasc Surg. 1999;30:587–598.
Recent review of adventitial cystic disease, its etiology, presentation, and treatment options..
Sinha S, Houghton J, Holt PJ, Thompson MM, Loftus IM, Hinchliffe RJ. Popliteal entrapment syndrome. J Vasc Surg. 2012;55:252–262.
van Rutte PWJ, Rouwet EV, Belgers EHJ, Lim RF, Teijink JAW. In treatment of popliteal artery cystic adventitial disease, primary bypass graft not always first choice: two case reports and a review of the literature. Eur J Vasc Endovasc Surg. 2011;42:347–354.
The reference list can be found on the companion Expert Consult website at www.expertconsult.com.
References
1. di Marzo L, et al. Popliteal vascular entrapment. World J Surg. 2005;29(Suppl 1):S43–S45.
2. Atkins HJ, et al. A case of myxomatous tumour arising in the adventitia of the left external iliac artery; case report. Br J Surg. 1947;34(136):426.
3. Tsilimparis N, et al. Cystic adventitial disease of the popliteal artery: an argument for the developmental theory. J Vasc Surg. 2007;45(6):1249–1252.
4. Ortiz MW, et al. Bilateral adventitial cystic disease of the popliteal artery: a case report. Cardiovasc Intervent Radiol. 2006;29(2):306–310.
5. Gagnon J, et al. Adventitial cystic disease of common femoral artery. Ann Vasc Surg. 2007;21(1):84–86.
6. Oi K, et al. Rapid recurrence of cystic adventitial disease in femoral artery and an etiologic consideration for the cyst. J Vasc Surg. 2011;53(6):1702–1706.
7. Levien LJ, et al. Adventitial cystic disease: a unifying hypothesis. J Vasc Surg. 1998;28(2):193–205.
8. Dix FP, et al. Cystic adventitial disease of the femoral vein presenting as deep vein thrombosis: a case report and review of the literature. J Vasc Surg. 2006;44(4):871–874.
9. Flanigan DP, et al. Summary of cases of adventitial cystic disease of the popliteal artery. Ann Surg. 1979;189(2):165–175.
10. Ishikawa K. Cystic adventitial disease of the popliteal artery and of other stem vessels in the extremities. Jpn J Surg. 1987;17(4):221–229.
11. Tsolakis IA, et al. Cystic adventitial disease of the popliteal artery: diagnosis and treatment. Eur J Vasc Endovasc Surg. 1998;15(3):188–194.
12. Schramek A, et al. Subadventitial haematoma of the popliteal artery. J Cardiovasc Surg (Torino). 1973;14(4):447–451.
13. Vasudevan A, et al. Cystic adventitial disease: a case report and literature review. ANZ J Surg. 2005;75(12):1120–1122.
14. Schroe H, et al. Baker’s cyst connected to popliteal artery cyst. Ann Vasc Surg. 1988;2(4):385–389.
15. Jay GD, et al. Clinical and chemical characterization of an adventitial popliteal cyst. J Vasc Surg. 1989;9(3):448–451.
16. Linquette M, et al. [Cystic degeneration of the adventitia of the popliteal artery. Apropos of a further case.]. Sem Hop. 1967;43(48):3005–3013.
17. Buijsrogge MP, et al. “Intermittent claudication intermittence” as a manifestation of adventitial cystic disease communicating with the knee joint. Ann Vasc Surg. 2006;20(5):687–689.
18. Chiche L, et al. [Two cases of cystic adventitial disease of the popliteal artery. Current diagnostic approach.]. J Mal Vasc. 1994;19(1):57–61.
19. Galle C, et al. Adventitial cystic disease of the popliteal artery communicating with the knee joint. A case report. J Vasc Surg. 1998;28(4):738–741.
20. Paravastu SC, et al. A contemporary review of cystic adventitial disease. Vasc Endovascular Surg. 2012;46(1):5–14.
21. Pursell R, et al. Spontaneous and permanent resolution of cystic adventitial disease of the popliteal artery. J R Soc Med. 2004;97(2):77–78.
22. Stapff M, et al. Image-directed Doppler ultrasound findings in adventitial cystic disease of the popliteal artery. J Clin Ultrasound. 1989;17(9):689–691.
23. Vanhoenacker FM, et al. Regarding “Adventitial cystic disease: a unifying hypothesis”. J Vasc Surg. 2000;31(3):621–622.
24. Rizzo RJ, et al. Computed tomography for evaluation of arterial disease in the popliteal fossa. J Vasc Surg. 1990;11(1):112–119.
25. Crolla RM, et al. A case of cystic adventitial disease of the popliteal artery demonstrated by magnetic resonance imaging. J Vasc Surg. 1993;18(6):1052–1055.
26. Khoury M. Failed angioplasty of a popliteal artery stenosis secondary to cystic adventitial disease–a case report. Vasc Endovascular Surg. 2004;38(3):277–280.
27. Wilbur AC, et al. Adventitial cyst of the popliteal artery: CT-guided percutaneous aspiration. J Comput Assist Tomogr. 1986;10(1):161–163.
28. van Rutte PW, et al. In treatment of popliteal artery cystic adventitial disease, primary bypass graft not always first choice: two case reports and a review of the literature. Eur J Vasc Endovasc Surg. 2011;42(3):347–354.
29. Ohta T, et al. Recurrence of cystic adventitial disease in an interposed vein graft. Surgery. 1994;116(3):587–592.
30. Baxter AR, et al. Cystic adventitial disease of the popliteal artery: is there a consensus in management? Vascular. 2011;19(3):163–166.
31. Love JW, et al. Popliteal artery entrapment syndrome. Am J Surg. 1965;109:620–624.
32. Gibson MH, et al. Popliteal entrapment syndrome. Ann Surg. 1977;185(3):341–348.
33. Murray A, et al. Popliteal artery entrapment syndrome. Br J Surg. 1991;78(12):1414–1419.
34. Sinha S, et al. Popliteal entrapment syndrome. J Vasc Surg. 2012;55(1):252–262.
35. Levien LJ, et al. Popliteal artery entrapment syndrome: more common than previously recognized. J Vasc Surg. 1999;30(4):587–598.
36. McAree BJ, et al. Bilateral popliteal artery occlusion in a competitive bike rider: case report and clinical review. Vasc Endovascular Surg. 2008;42(4):380–385.
37. Hoelting T, et al. Entrapment of the popliteal artery and its surgical management in a 20-year period. Br J Surg. 1997;84(3):338–341.
38. di Marzo L, et al. Diagnosis of popliteal artery entrapment syndrome: the role of duplex scanning. J Vasc Surg. 1991;13(3):434–438.
39. Henry MF, et al. Popliteal artery entrapment syndrome. Curr Treat Options Cardiovasc Med. 2004;6(2):113–120.
40. Akkersdijk WL, et al. Colour Doppler ultrasonographic imaging in the diagnosis of popliteal artery entrapment syndrome. Br J Surg. 1995;82(1):134.
41. Pillai J, et al. Assessment of the medial head of the gastrocnemius muscle in functional compression of the popliteal artery. J Vasc Surg. 2008;48(5):1189–1196.
42. Steurer J, et al. A new therapeutic approach to popliteal artery entrapment syndrome (PAES). Eur J Vasc Endovasc Surg. 1995;10(2):243–247.
43. Beseth BD, et al. The posterior approach for repair of popliteal artery aneurysms. J Vasc Surg. 2006;43(5):940–944.
44. Lambert AW, et al. Popliteal artery entrapment syndrome. Br J Surg. 1999;86(11):1365–1370.
45. Kim SY, et al. Long-term outcomes after revascularization for advanced popliteal artery entrapment syndrome with segmental arterial occlusion. J Vasc Surg. 2012;55(1):90–97.
46. Causey MW, et al. Intraoperative duplex and functional popliteal entrapment syndrome: strategy for effective treatment. Ann Vasc Surg. 2010;24(4):556–561.
47. Rosset E, et al. Popliteal artery entrapment syndrome. Anatomic and embryologic bases, diagnostic and therapeutic considerations following a series of 15 cases with a review of the literature. Surg Radiol Anat. 1995;17(2):161–169.