51 Spinal Cord Stimulation
Placement
Position
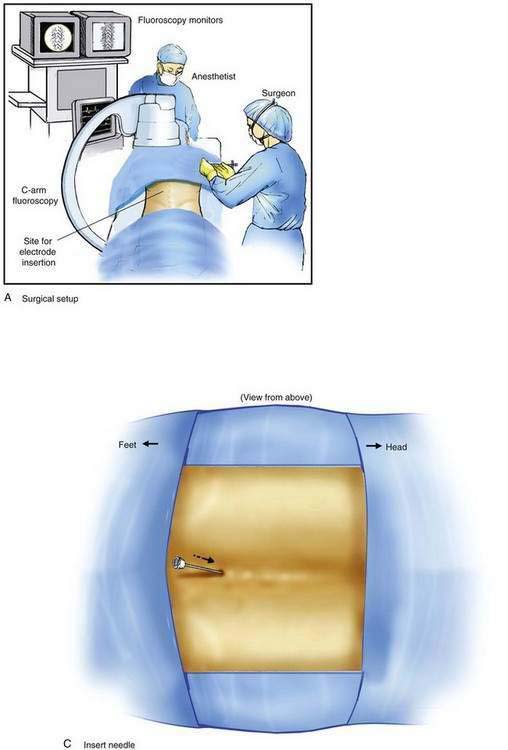
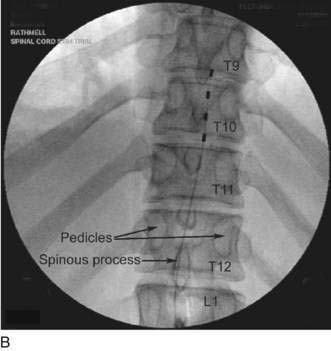
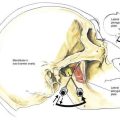
Implantation of a spinal cord stimulator lead and impulse generator is a minor surgical procedure that is carried out in the operating room using aseptic precautions, including skin preparation, sterile draping, and full surgical attire (Fig. 51-1A). The procedure must be conducted using local anesthesia and sedation light enough that the patient can report feeling the electrical stimulation during lead placement. The patient is positioned on a radiolucent table in the prone position (see Fig. 51-1A). Initial lead placement can be carried out with the patient in a lateral decubitus position, but even small degrees of rotation along the spinal axis can make positioning the lead difficult. The arms are extended upward so that they are in a position of comfort well away from the surgical field. The skin is prepared, and sterile drapes are applied. For stimulation in the low back and lower extremities, the fluoroscopic C-arm is positioned directly over the thoracolumbar junction to provide an anteroposterior view of the spine. Care must be taken to ensure that the radiographic view is not rotated by observing that the spinous processes are in the midline, halfway between the vertebral pedicles (Fig. 51-1B).
Procedure
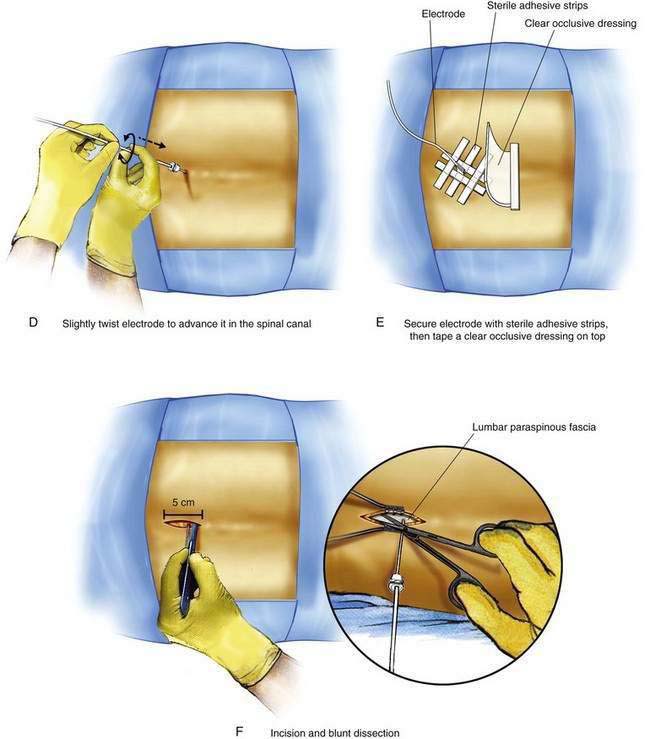
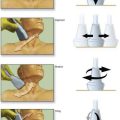
The Ll-L2 interspace is identified using fluoroscopy. The epidural needle supplied by the device manufacturer must be used to ensure that the lead can advance through the needle without damage. The needle is advanced using a paramedian approach starting 1 to 1.5 cm lateral to the spinous processes and somewhat caudad to the interspace to be entered. The needle is directed to enter the spinal space in the midline with an angle of entry no greater than 45 degrees from the plane of the epidural space (Fig. 51-1C). If the angle of attack of the needle during the initial entry into the epidural space is too great, the epidural lead is difficult to thread as it negotiates the steep angle between the needle and the plane of the epidural space. The epidural space is identified using a loss-of-resistance technique. The electrode is then advanced through the needle and directed to remain just to one side of midline as it is threaded cephalad under fluoroscopic guidance. The electrode contains a wire stylet with a slight angulation at the tip; gentle rotation of the electrode as it is advanced allows the operator to direct the electrode’s path in the epidural space (Fig. 51-1D). For stimulation in the low back and lower extremities, the electrode is initially positioned 2 to 3 mm from the midline on the same side as the patient’s pain between the T8 and T10 vertebral levels (see Fig. 51-1B). Final electrode position is attained by connecting the electrode to an external impulse generator and asking the patient where the stimulation is felt. In general, cephalad advancement results in stimulation higher in the extremity, and caudad movement leads to stimulation lower in the extremity. However, if the lead is angled even slightly from medial to lateral, the pattern of stimulation may change less predictably with movement of the electrode; for example, craniad advancement can lead to stimulation lower in the extremity under these circumstances. The final electrode position should be recorded using radiography so that a permanent lead can be placed in the same position (see Fig. 51-1B). For trial stimulation, the needle is removed, the electrode secured to the back, and a sterile occlusive dressing applied (Fig. 51-1E). The patient is instructed in the use of the external pulse generator and scheduled to return in 5 to 7 days to assess his or her response and to remove the trial lead.
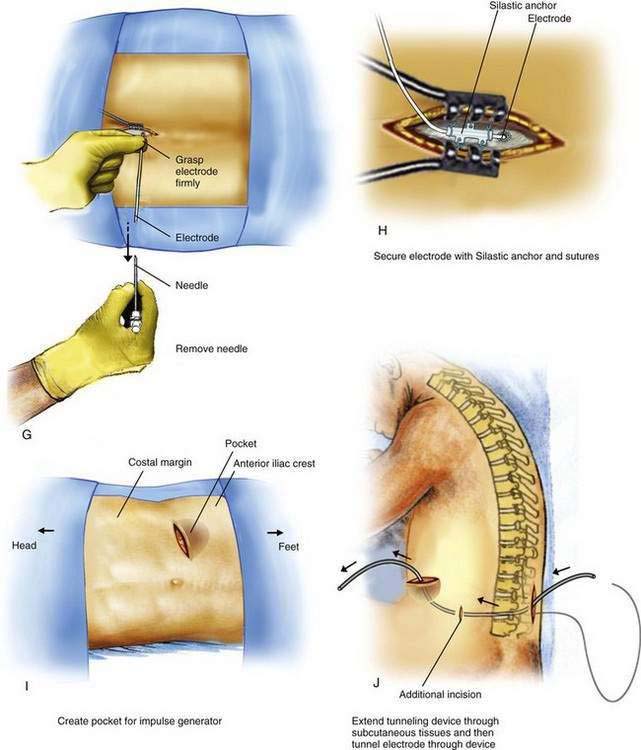
The procedure for initial lead placement for permanent implantation is identical to that for trial stimulation. Once the final lead is in position and the optimal pattern of stimulation confirmed, the lead is secured, a pocket for the impulse generator is created, and the lead is tunneled underneath the skin for connection to the impulse generator. After initial lead placement, the epidural needle is withdrawn slightly (about 1 to 2 cm) but left in place around the lead in the subcutaneous tissues to protect the lead during the subsequent incision and dissection. A 5- to 8-cm incision parallel to the axis of the spine is extended from cephalad to caudad to the needle, extending directly through the needle’s skin entry point (Fig. 51-1F). The subcutaneous tissues are divided using blunt dissection until the lumbar paraspinous fascia is visible surrounding the needle shaft. The stylet is then removed from the lead and the needle is withdrawn, using care not to dislodge the electrode (Fig. 51-1G). The lead is then secured to the paraspinous fascia using a specific anchoring device supplied by the manufacturer (Fig. 51-1H).
An 8- to l0-cm transverse incision is made along the previously marked line and a subcutaneous pocket is created using blunt dissection (Fig. 51-1I). The pocket should always be created caudad to the incision; if the pocket is placed cephalad to the incision, the weight of the impulse generator on the suture line is likely to cause wound dehiscence. In many patients, the blunt dissection can be accomplished using gentle but firm pressure with the fingers. It is simpler and less traumatic to use a small surgical scissors to perform the blunt dissection, using repeated opening motions rather than closing or cutting motions that are likely to cut vascular structures and provoke marked bleeding. An alternative to blunt dissection is the use of a monopolar electrocautery device in the “cut” mode, an effective means to carry out the necessary dissection without excessive tissue trauma or blood loss. After the pocket has been created, the impulse generator is placed in the pocket to ensure that the pocket is large enough. The impulse generator should fit completely in the pocket without any part of the device extending into the incision. With the device in place, the wound margins must fall into close apposition. There should be no tension on the sutures during closure of the incision, lest the wound dehisce.
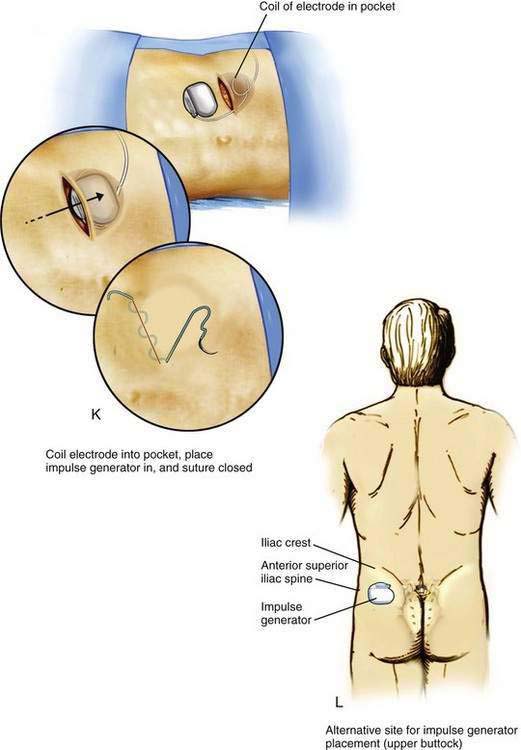
After the pocket creation is completed, a tunneling device is extended in the subcutaneous tissues between the paraspinous incision and the pocket, leaving a small tension-relief loop of lead in the subcutaneous area of the paraspinous dissection (Fig. 51-1J). The electrode is then advanced through the tunnel. Tunneling devices vary and are specific to each manufacturer. The means by which the electrode is connected to the impulse generator also varies by manufacturer; some devices use a lead extension that connects the impulse generator and the lead, and others use a one-piece lead that is connected directly to the impulse generator. After tunneling, the lead or lead extension is connected with the impulse generator. Any excess lead is coiled and placed behind the impulse generator in the pocket (Fig. 51-1K). This loop allows patient movement without placing tension on the distal electrode, causing it to be pulled from the epidural space. The skin incisions are then closed in two layers: a series of interrupted subcutaneous sutures to securely close the fascia overlying the impulse generator in the pocket and the electrode over the paraspinous fascia, followed by skin closure using sutures or staples (see Fig. 51-1K).