Chapter 77 Pulmonary Complications of Hematopoietic Stem Cell Transplantation
Posttransplant Pulmonary Complications
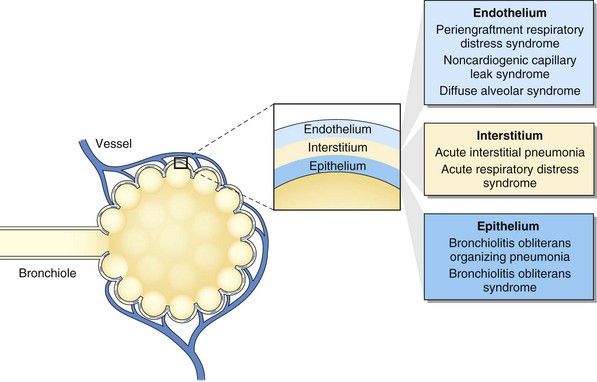
Pulmonary complications after HSCT are common, with an incidence of 40% to 60% and with up to one third of recipients requiring intensive care after transplantation. Respiratory failure is the most common cause of critical illness, and pneumonia is the leading infectious cause of death after HSCT (Figure 77-1). Pulmonary complications can occur early or late in the posttransplant course, can have infectious and noninfectious etiologies, and can present with assorted radiographic findings. The pulmonary complications of HSCT also vary depending on the indication for, type of, and preparative regimen preceding HSCT.
Risk Factors for Pulmonary Disease
Relapse status at transplant and donor-recipient HLA mismatching or nonidentity are risk factors for pulmonary complications and mortality after HSCT (Box 77-1). Active phase of malignancy, age over 21 years, and receipt of HLA-nonidentical donor marrow are risk factors for respiratory failure after HSCT.
Box 77-1
Risk Factors for Pulmonary Complications After Hematopoietic Stem Cell Transplantation (HSCT)
Infectious Complications
Supportive care in the posttransplant period has changed the microbiology of pneumonia. Prophylactic administration of trimethoprim-sulfamethoxazole (TMP-SMX), antivirals, antifungals, and fluoroquinolones has decreased the incidence of Pneumocystis jirovecii, CMV, herpes simplex, and Candida albicans infections (Box 77-2). Resistant gram-negative and gram-positive bacteria, viruses, and other fungi remain important pathogens.
Noninfectious Complications
Idiopathic Pneumonia Syndrome
The insight obtained from animal models has stimulated the classification of the entities under the term idiopathic pneumonia syndrome according to the primary anatomic site of cellular damage: interstitial tissue, vascular endothelium, or airway epithelium (see Fig. 77-1). For example, in interstitial type of injury, there are increased numbers of cytotoxic T lymphocytes and cells expressing B7 family costimulatory ligands in the alveolar and interstitial spaces. In vascular endothelium type of injury, there are injured endothelial cells, and in airway epithelium type of injury, injured type II alveolar epithelial cells. This classification reflects complex interactive mechanisms with a pathogenic role attributed to several factors, including increased soluble inflammatory mediators (e.g., tumor necrosis factor alpha), increased host flora–derived lipopolysaccharide, enhanced oxidative stress, depletion of pulmonary surfactant, donor-derived T cell effectors, host antigen-presenting cells, donor accessory cells, and leukocyte recruitment to the lung.
Afessa B, Azoulay E. Critical care of the hematopoietic stem cell transplant recipient. Crit Care Clin. 2010;26:133–150.
Afessa B, Tefferi A, Litzow MR, et al. Diffuse alveolar hemorrhage in hematopoietic stem cell transplant recipients. Am J Respir Crit Care Med. 2002;166:641–645.
Azoulay E, Mokart D, Rabbat A, et al. Diagnostic bronchoscopy in hematology and oncology patients with acute respiratory failure: prospective multicenter data. Crit Care Med. 2008;36:100–107.
Bacigalupo A, Ballen K, Rizzo D, et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant. 2009;15:1628–1633.
Boeckh M. The challenge of respiratory virus infections in hematopoietic cell transplant recipients. Br J Haematol. 2008;143:455–467.
Dickout WJ, Chan CK, Hyland RH, et al. Prevention of acute pulmonary edema after bone marrow transplantation. Chest. 1987;92:303–309.
Espinosa-Aguilar L, Green JS, Forrest GN, et al. Novel H1N1 influenza in hematopoietic stem cell transplantation recipients: two centers’ experiences. Biol Blood Marrow Transplant. 2010;17:566–573.
Gratwohl A, Baldomero H, Aljurf M, et al. Hematopoietic stem cell transplantation: a global perspective. JAMA. 2010;303:1617–1624.
Guo YL, Chen YQ, Wang K, et al. Accuracy of BAL galactomannan in diagnosing invasive aspergillosis: a bivariate metaanalysis and systematic review. Chest. 2010;138:817–824.
Herbrecht R, Denning DW, Patterson TF, et al. Voriconazole versus amphotericin B for primary therapy of invasive aspergillosis. N Engl J Med. 2002;347:408–415.
Kallianpur AR. Genomic screening and complications of hematopoietic stem cell transplantation: has the time come? Bone Marrow Transplant. 2005;35:1–16.
Maertens JA, Klont R, Masson C, et al. Optimization of the cutoff value for the Aspergillus double-sandwich enzyme immunoassay. Clin Infect Dis. 2007;44:1329–1336.
Mahony J, Chong S, Merante F, et al. Development of a respiratory virus panel test for detection of twenty human respiratory viruses by use of multiplex PCR and a fluid microbead-based assay. J Clin Microbiol. 2007;45:2965–2970.
Martino R, Porras RP, Rabella N, et al. Prospective study of the incidence, clinical features, and outcome of symptomatic upper and lower respiratory tract infections by respiratory viruses in adult recipients of hematopoietic stem cell transplants for hematologic malignancies. Biol Blood Marrow Transplant. 2005;11:781–796.
Montani D, O’Callaghan DS, Savale L. Pulmonary veno-occlusive disease: recent progress and current challenges. Respir Med. 2010;104:S23–S32.
Mullighan CG, Heatley S, Doherty K, et al. Mannose-binding lectin gene polymorphisms are associated with major infection following allogeneic hematopoietic stem cell transplantation. Blood. 2002;99:3524–3529.
Onizuka M, Kasai M, Oba T, et al. Increased frequency of the angiotensin-converting enzyme gene D-allele is associated with noninfectious pulmonary dysfunction following allogeneic stem cell transplant. Bone Marrow Transplant. 2005;36:617–620.
Pandya CM, Soubani AO. Bronchiolitis obliterans following hematopoietic stem cell transplantation: a clinical update. Clin Transplant. 2010;24:291–306.
Soubani AO, Pandya CM. The spectrum of noninfectious pulmonary complications following hematopoietic stem cell transplantation. Hematol Oncol Stem Cell Ther. 2010;3:143–157.
Sundin M, Le Blanc K, Ringdén O, et al. The role of HLA mismatch, splenectomy and recipient Epstein-Barr virus seronegativity as risk factors in post-transplant lymphoproliferative disorder following allogeneic hematopoietic stem cell transplantation. Haematologica. 2006;91:1059–1067.
Walsh TJ, Anaissie EJ, Denning DW, et al. Treatment of aspergillosis: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:327–360.
Wilczynski SW, Erasmus JJ, Petros WP, et al. Delayed pulmonary toxicity syndrome following high-dose chemotherapy and bone marrow transplantation for breast cancer. Am J Respir Crit Care Med. 1998;157:565–573.
Yanik GA, Ho VT, Levine JE, et al. The impact of soluble tumor necrosis factor receptor etanercept on the treatment of idiopathic pneumonia syndrome after allogeneic hematopoietic stem cell transplantation. Blood. 2008;112:3073–3081.