CHAPTER 30 PRIMARY AUTONOMIC FAILURE
CLASSIFICATION
A convenient approach to the syndromes of autonomic failure is to distinguish those of the primary variety, in which there is no clear etiological factor or specific disease association, from those with secondary autonomic failure, in which the lesion is defined (anatomically, as in spinal cord injuries, or biochemically, as in dopamine β-hydroxylase deficiency) or is linked to specific disease processes (as in diabetes mellitus). Furthermore, drugs form a major cause of autonomic dysfunction and merit separate categorization. Moreover, another group that probably warrants a separate entity is neurally mediated syncope, in which, between episodic autonomic disturbances, usually no abnormalities can be detected.1
Primary autonomic failure syndromes can be divided into a chronic subgroup and into a rarer subgroup with acute or subacute dysautonomia (Table 30-1). Furthermore, the chronic syndromes can be subdivided into those without (i.e., pure autonomic failure [PAF]) and those with associated neurological deficits. Clinically, the latter belong to at least three categories: Parkinson’s disease associated with autonomic failure, dementia with Lewy bodies (DLB), and multiple-system atrophy (MSA). Patients with parkinsonian features may be responsive to chronic levodopa (L-dopa) therapy, probable as part of treatment for Parkinson’s disease with autonomic failure. Alternatively, there may be no or poorly (transiently) L-dopa–responsive parkinsonism, probable as part of the parkinsonian variant of MSA, i.e. MSA-P. Other patients may have cerebellar features as the predominant motor disorder and may therefore be diagnosed as the cerebellar variant of MSA, i.e., MSA-C. Some patients present initially with autonomic abnormalities, including urogenital and cardiovascular dysfunction, and only later develop the additional neurological manifestations of MSA. Eventually, some patients present with parkinsonian features accompanied by dementia within the first year of disease onset, which leads to a diagnosis of DLB.
TABLE 30-1 Classification of Primary Autonomic Failure Disorders
| Acute/subacute primary autonomic failure disorders | Pure pandysautonomia |
| Pandysautonomia with neurological features | |
| Pure cholinergic dysautonomia | |
| Acute noradrenergic autonomic neuropathy | |
| Chronic primary autonomic failure disorders | Pure autonomic failure |
| Parkinson’s disease with autonomic failure | |
| Dementia with Lewy bodies | |
| Multiple system atrophy |
Actually, it is important to classify patients or assign them to these different disease entities for a number of reasons, including prognosis. Indeed, analyses of two large series in the United Kingdom and the United States indicate that patients with PAF have a substantially better prognosis than those with additional neurological deficits.2 There are differences within this latter group as well, inasmuch as patients with L-dopa–responsive Parkinson’s disease and autonomic failure appear to live longer than do patients with MSA or DLB (personal observations).
NEUROPATHOLOGY
Degeneration of autonomic neurons with disabling dysautonomia is a prominent feature of the Lewy body syndromes and MSA. α-Synuclein is a major component of the Lewy bodies in Parkinson’s disease, DLB, and the glial and neuronal cytoplasmic inclusions of MSA. α-Synuclein also is a major component of Lewy bodies in the brain and peripheral autonomic ganglia in PAF.3 Therefore, these disorders are increasingly being referred to as “synucleinopathies.” Abnormalities in the expression or structure of α-synuclein or associated proteins may cause degeneration of catecholamine-containing neurons.4 However, the function of α-synuclein is not known, but interest in this protein derives from the finding that the gene encoding for α-synuclein is mutated in families with the autosomal-dominant form of Parkinson’s disease.5 To investigate the consequence of α-synuclein overexpression in glia, Stefanova and colleagues6 transfected U373 astrocytoma cells with vectors encoding wild-type human α-synuclein or C-terminally truncated synuclein fused to red fluorescent protein. α-Synuclein immunocytochemistry of transfected astroglial cells revealed diffuse cytoplasmic labeling associated with discrete inclusions within both cell bodies and processes. Susceptibility to oxidative stress was increased in astroglial cells overexpressing α-synuclein, particularly in the presence of cytoplasmic inclusions. However, whether the α-synuclein aggregation is induced by some other factor or factors or whether it is the primary trigger of MSA pathology is unknown. Impairment in the ability of oligodendrocytes to degrade α-synuclein, which they may normally produce at low levels, may promote abnormal subcellular aggregation in MSA.7 Alternatively, ectopic expression of oligodendroglial α-synuclein may result in glial cytoplasmic inclusions. This scenario is supported by experimental studies demonstrating glial cytoplasmic inclusion–like inclusion pathology in transgenic mice overexpressing oligodendroglial α-synuclein.8 More work is necessary to elucidate the cascade of cell death in MSA and to determine exogenous and genetic susceptibility factors, both of which are likely to drive the disease process in this disorder. It is not known what determines whether α-synuclein precipitates in neurons (Parkinson’s disease, PAF) or glial cells (MSA) or on autonomic (PAF) or striatonigral neurons (Parkinson’s disease, MSA). Anyway, there are clear distinctions between the different α-synucleinopathies and little evidence of migration from one clinical form to the other.
Lewy Body Disorders
The Lewy body syndromes are characterized by intracytoplasmic eosinophilic neuronal inclusions, so-called Lewy bodies or Lewy neurites, found in the brain, including brainstem, basal ganglia, and cortical neurons, and in the peripheral autonomic nerves of affected patients. Lewy bodies contain abnormally phosphorylated intermediate neurofilament proteins, α-synuclein, ubiquitin, and associated enzymes. There are three different but overlapping phenotypes. In PAF there is early and widespread neuronal degeneration restricted mostly to peripheral autonomic neurons; autonomic failure is the sole clinical finding.9 In fact, in patients with PAF, intracytoplasmic eosinophilic inclusion bodies with the histological appearance of Lewy bodies, similar to those found in Parkinson’s disease, are identified in neurons of the substantia nigra, locus ceruleus, thoracolumbar and sacral spinal cord, and sympathetic ganglia and in peripheral sympathetic and parasympathetic nerves.9,10 Neuropathological reports of patients with PAF showed α-synuclein–positive intraneuronal cytoplasmic inclusions (Lewy bodies) in brainstem nuclei and peripheral autonomic ganglia.3,9 In Parkinson’s disease, there is prominent degeneration of the substantia nigra (Fig. 30-1) and other brainstem nuclei, in addition to peripheral autonomic neurons; clinically, there are motor abnormalities with varying degrees of autonomic failure.11 In DLB there is extensive cortical involvement in addition to degeneration of brainstem nuclei and peripheral autonomic neurons; clinical findings are dominated by severe cognitive impairment in association with parkinsonism and autonomic dysfunction.12 It is likely that the clinical phenotype of Lewy body syndromes depends on the temporal formation and distribution of Lewy bodies and associated neurodegeneration. Individual differences in neuronal susceptibility may determine the manifesting phenotype. Patients with PAF, however, can progress to Parkinson’s disease or DLB, which suggests that phenotypes overlap and that neurodegeneration in the Lewy body syndromes may start in postganglionic autonomic neurons and later affect neurons in the central nervous system. As initially suggested by Oppenheimer,13 PAF may be a “forme fruste” of Parkinson’s disease, with early severe widespread degeneration of peripheral autonomic neurons.9,10
Multiple-System Atrophy
The second type of neurodegeneration with prominent autonomic failure is MSA. The term multiple-system atrophy was introduced in 196914; however, cases of MSA were previously reported under the rubrics of striatonigral degeneration (SND),15–17 olivopontocerebellar atrophy (OPCA),18,19 Shy-Drager syndrome,20 and idiopathic orthostatic hypotension. MSA is a sporadic neurodegenerative disorder characterized clinically by various combinations of parkinsonian, autonomic, cerebellar, or pyramidal symptoms and signs and pathologically by cell loss, gliosis, and glial cytoplasmic inclusions in several brain and spinal cord structures. Indeed, this disorder affects neurons in the basal ganglia, cortex, and spinal cord, but spares peripheral autonomic neurons. Pathologically, cytoplasmic inclusions are located in glial cells and do not form Lewy bodies.21 Clinically, two major motor presentations can be distinguished. Parkinsonian features predominate in 66% of patients (MSA-P), and cerebellar ataxia is the major motor feature in 34% of patients (MSA-C), according to a European survey.22 Severe autonomic failure is prominent in both phenotypes.23
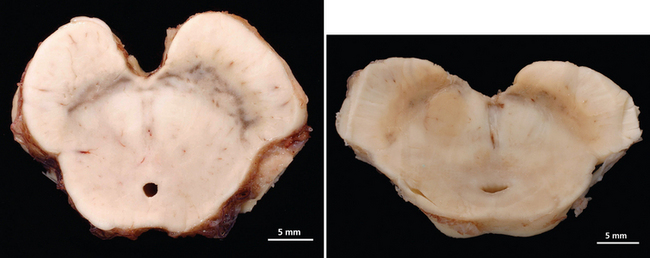
In MSA-P, the striatonigral system is the main site of pathology, but less severe degeneration can be widespread and usually includes the olivopontocerebellar system.24,25 The putamen is shrunken with gray-green discoloration. When putaminal pathology is severe, there may be a cribriform appearance. In early stages, the putaminal lesion shows a distinct topographical distribution with a predilection for the caudal and dorsolateral regions.24 Degeneration of pigmented nerve cells occurs in the substantia nigra pars compacta, whereas cells of the pars reticulata are reported as normal. The topographical patterns of neurodegeneration involving the motor neostriatum, efferent pathways, and nigral neurons reflect their anatomical relationship and suggest a common denominator or “linked” degeneration.24
The location of the α-synuclein precipitates determines not only the presence or absence of movement disorders but also the characteristics of the autonomic cardiovascular abnormality. Autonomic failure in MSA is caused by dysfunction of (1) central and preganglionic efferent autonomic activity, (2) neuronal networks in the brainstem that control cardiovascular and respiratory function, and (3) the neuroendocrine component of the autonomic regulation via the hypothalamopituitary axis. In MSA, cell loss is reported in dorsal motor nucleus of the vagus.26 Catecholaminergic neurons in the rostral (C1 group) and caudal (A1 group) ventrolateral medulla, which are involved in the control of sympathetic outflow to the cardiovascular system and reflex control of vasopressin release, are also affected, as are neurons of the arcuate nucleus that are involved in cardiorespiratory interactions.27–30 Cell loss has also been described for the Edinger-Westphal nucleus and posterior hypothalamus,20 including the tuberomammillary nucleus.31 Papp and Lantos21 demonstrated marked involvement of brainstem pontomedullary reticular formation with glial cytoplasmic inclusions, which represented a supraspinal histological counterpart for impaired visceral function. Autonomic neuronal degeneration affects the locus ceruleus, too.32 It is noteworthy that there is not always a strong correlation between nerve cell depletion or gliosis and the clinical degree of autonomic failure. It is estimated that more than 50% of cells within the intermediolateral column must decay before symptoms become evident.13 Degeneration of sympathetic preganglionic neurons in the intermediolateral column of the thoracolumbar spinal cord is considered contributory to orthostatic hypotension. On the basis of only the reports in which formal cell counts have been made, it is apparent, with very few exceptions, that all cases of MSA with predominant pathology in either the striatonigral or olivopontocerebellar system show loss of intermediolateral cells.33 Orthostatic hypotension in MSA is caused by blunted autonomic and neuroendocrine reflexes as a result of afferent and central neuronal loss; postganglionic autonomic fibers, however, are spared.34 Disordered bladder, rectal, and sexual function in MSA-P and MSA-C have been associated with cell loss in parasympathetic preganglionic nuclei of the spinal cord. These neurons are localized rostrally in Onuf’s nucleus between sacral segments S2 and S3 and more caudally in the inferior intermediolateral nucleus chiefly in the S3 to S4 segments.35 Loss of corticotrophin-releasing factor neurons in the pontine micturition area may contribute to neurogenic bladder dysfunction.30 In the peripheral component of the autonomic nervous system, Bannister and Oppenheimer36 described atrophy of the glossopharyngeal and vagus nerves. No pathology has been reported in the visceral enteric plexuses or in the innervation of glands, blood vessels, or smooth muscles. Sympathetic ganglia have not often been examined in pathological studies of autonomic failure and have seldom been described quantitatively. In MSA with autonomic failure, there are either no obvious or mild abnormalities in sympathetic ganglia. Any morphological changes reported in sympathetic ganglionic neurons in MSA have tended to be nonspecific,37 exhibiting the normal age-related range of appearances, and published micrographs and counts have indicated at least a moderate density, and sometimes quite a high density, of surviving neurons.38 Enteric and parasympathetic ganglia have been studied only in a few instances.
A variety of other neuronal populations are noted to show cell depletion and gliosis with considerable differences in vulnerability from case to case. Varying degrees of abnormalities in the cerebral hemisphere, including Betz cell loss, were detected in pathologically proved MSA cases.32,33 Furthermore, anterior horn cells may show some depletion, but rarely to the same extent as that occurring in motor neuron disease.39
CLINICAL PRESENTATION
Acute/Subacute Primary Autonomic Failure Disorders
Pure Pandysautonomia and Pandysautonomia with Neurological Features
There is a clinical spectrum of acute autonomic neuropathies. Acute panautonomic neuropathy (pandysautonomia), characterized by severe widespread sympathetic and parasympathetic failure, is at one extreme. Guillain-Barré syndrome is at the other end of the spectrum, in which the brunt of the disorder is borne by the somatic nervous system. Pure acute panautonomic neuropathies are relatively rare. Actually, the majority of acute autonomic neuropathies have some minor somatic features. Dysautonomia may be restricted to the cholinergic system (acute cholinergic neuropathy), the adrenergic system, or other organ systems (e.g., motility disorders).40
In medical history, a definite entity of pure pandysautonomia involving both sympathetic and parasympathetic nervous systems with a subacute onset, monophasic course, and partial recovery without significant features of somatic peripheral neuropathy was first described by Young and colleagues in 1969. Actually, there had been some earlier reports of the condition in the literature, although it was not clearly defined.47 The disorder differs from other neurological causes of autonomic dysfunction in that normal function of the central nervous system is preserved. Furthermore, there are no or only minor features of peripheral somatic nervous system involvement. Since these first descriptions, a number of other cases of acute pandysautonomia have been reported, as well as some cases of pure cholinergic dysautonomia. Some cases of acute dysautonomia with significant sensory disturbances have been described; in some, but not all, there was electrophysiological and pathological evidence of loss of small-diameter myelinated and unmyelinated fibers.41 In 1994, Suarez and colleagues42 clarified the features of acute idiopathic autonomic neuropathy. Both sexes and all ages can be affected. The onset is acute or subacute. In approximately one half of affected patients, there is an antecedent viral infection. Several cases that followed Epstein-Barr virus infection have been described, in one of which Epstein-Barr virus DNA and antibody to the virus were found in the cerebrospinal fluid.43 The most common presenting features are symptomatic orthostatic hypotension (light-headedness, dizziness, syncope) and symptoms of gastrointestinal dysfunction (nausea, vomiting, diarrhea, constipation, and postprandial bloating) or sudomotor dysfunction (failure to sweat, causing heat intolerance and flushing). Other symptoms include numbness, tingling, bladder disturbances, and impotence. Neurological examination findings are normal in about one half the patients; the remainder have depressed reflexes and distal sensory impairment. The clinical course is monophasic. Recovery tends to be gradual and frequently incomplete. The cerebrospinal fluid protein level may be mildly elevated. In rare cases, there may be evidence of sensory neuropathy with sensory symptoms of minimal intensity (mainly thermal and pain hypoesthesia in distal areas). In most cases, nerve conduction studies yield normal results. Sural nerve biopsy in some cases has demonstrated reduction of myelinated fiber density, predominantly of small fibers, and axonal degeneration. Actually, in some cases, there are minimal signs of distal denervation in electromyographic-electroneurographic studies.44 In some acute neuropathies, such as pandysautonomia, small-fiber impairment is relatively pure, but it may also appear in disorders with prominent somatic damage, such as Guillain-Barré syndrome, in which autonomic failure worsens the prognosis.45
The cause of the condition remains uncertain. Pathological features include the presence of a small inflammatory mononuclear cell infiltrate in the epineurium. It is probably a form of acute idiopathic polyneuritis restricted to autonomic nerves with an immune-mediated pathogenesis similar to that of the Guillain-Barré syndrome. Together, the acute onset, frequent antecedent viral infection, selectivity of involvement by fiber type and autonomic level, and presence of perivascular mononuclear cell infiltration suggest that the underlying mechanism is likely to be immune mediated. The following differential diagnoses have to be kept in mind: botulism, acute autonomic neuropathy associated with Guillain-Barré syndrome, porphyria, diabetes, toxic causes, systemic lupus erythematosus, and other connective tissue diseases.41
Pure Cholinergic Dysautonomia
In pure cholinergic dysautonomia, clinical and laboratory features indicate only a cholinergic failure. A number of cases of pure cholinergic dysautonomia have been described in children. Clinical features include blurred vision, impaired lacrimation, dry mouth, constipation, urinary retention and incontinence, and absence of sweating. There is no postural hypotension. Excessive salivation and sweat secretion have been described in early disease stages. Cerebrospinal fluid findings are normal.41
Chronic Primary Autonomic Failure Disorders
Primary Autonomic Failure
Bradbury and Eggleston46 were the first to describe PAF in 1925. They used the term idiopathic orthostatic hypotension. Actually, the name pure autonomic failure was introduced by Oppenheimer as one of the primary autonomic failure syndromes. It is a sporadic, adult-onset, slowly progressive, neurodegenerative disorder of the autonomic nervous system.92 Clinically, it is characterized by an isolated impairment of the autonomic nervous system with no other neurological deficits.47 PAF affects men slightly more often than women, usually in their sixth decade. Its onset is slow, and symptoms begin developing insidiously for years as minimal impairment (nonspecific weakness and orthostatic intolerance). The patient may recall that symptoms first manifested several years before he or she sought medical treatment. Common symptoms causing the patient to seek medical advice include unsteadiness, lightheadedness, or faintness on standing. Questioning often elicits descriptions of aching in the neck or occiput only when standing; lying down relieves all symptoms. In general, orthostatic symptoms are more prominent after prolonged recumbency, as in the morning hours. Moreover, postural hypotension is exacerbated after mealtimes and physical exertion. Other contributory factors are heat, alcohol ingestion, coughing, and defecation.48,49 In fact, straining during evacuation or micturition elevates intrathoracic pressure and may result in symptomatic hypotension. Mathias and colleagues50 investigated the frequency of symptoms associated with orthostatic hypotension in PAF and MSA and found that more patients with PAF had syncope (91% vs. 45%), visual disturbances (75% vs. 53%), and suboccipital/paracervical “coat hanger” neck pain (81 vs. 53%) than did the patients with MSA. The reasons for this are unclear. Patients with PAF may also develop supine hypertension. Moreover, a decreased ability to sweat may be apparent, particularly in hot climates. Men found to have PAF may have sought advice about urinary tract symptoms (hesitancy, urgency, dribbling, and occasional incontinence). Other signs of dysautonomia, including erectile and ejaculatory dysfunction, an inability to appreciate orgasm, and retrograde ejaculation may be present, too. Women may experience urinary retention or incontinence as early symptoms. In patients with neurally mediated syncope, nausea and pallor, which are prominent signs of autonomic activation, occur before loss of consciousness. In contrast, in patients with PAF, these signs are noticeably absent, and consciousness is lost with little or no warning.51
Autonomic tests are abnormal: orthostatic hypotension, cardiovagal dysfunction, and hypo- or anhidrosis of the postganglionic type (see “Laboratory Assessment” section). A definitive diagnosis of orthostatic hypotension as the cause of symptoms is made when symptoms are reproduced while declines in systolic blood pressure of at least 20 mm Hg and diastolic pressure of at least 10 mm Hg are documented, within 3 minutes of standing. The diagnosis cannot be excluded with a single measurement of upright blood pressure that does not fulfill these criteria. Several measurements of orthostatic blood pressure, preferably early in the morning or after a meal, may be necessary. Patients with PAF also have decreased sinus arrhythmia and absent blood pressure overshoot during phase IV of the Valsalva maneuver, which indicates parasympathetic and sympathetic efferent dysfunction. PAF affects mainly efferent postganglionic neurons; afferent pathways and somatic neurons are not affected. Nevertheless, there is evidence of a preganglionic disorder in 22% of patients with PAF, which suggests that such patients actually may have some central component.44
In terms of differential diagnosis, PAF should be distinguished from other forms of neurogenic orthostatic hypotension, including peripheral somatic neuropathies with autonomic involvement (e.g., diabetes and amyloid), MSA, Parkinson’s disease, and DLB. There are no symptoms or signs of sensory, cerebellar, pyramidal, or extrapyramidal dysfunction in patients with PAF. In general, this allows a clinical distinction from other forms of neurogenic orthostatic hypotension. However, it cannot be determined whether a single patient with PAF will eventually develop more widespread, nonautonomic neuronal damage that leads to a diagnosis of MSA or, in rare cases, DLB. A number of warning signs, or “red flags,” for a clinical diagnosis of MSA have been operationally defined, and their frequency has been determined in a large cohort of European patients with MSA in a natural history study conducted by the European MSA-Study Group. Some of these features that are, if present, suggestive of MSA can be attributed, at least in part, to autonomic nervous system abnormalities. Abnormal respiration occurred in 42% to 60% of patients; its manifestations included inspiratory stridor (19% to 33%), involuntary deep sighs and/or gasps (34% to 37%), sleep apnea (13% to 18%), and excessive snoring (22% to 33%). Rapid eye movement (REM) sleep behavior disorder was present in 35% to 39%. Cold hands and/or feet were noted in 26% to 34%, whereas Raynaud’s phenomenon was recorded in only 6% to 7%.52,53 Although the specificity and positive predictive value of the red flags for a diagnosis of MSA have not been determined yet, they may serve as useful “soft signs” pointing toward a diagnosis of MSA. Because of the slow disease progression in PAF, most patients probably die before central nervous system involvement can become clinically evident. Apart from dysautonomia, these patients are otherwise normal and have a relatively good prognosis. Complications are usually related to falls and associated disorders.54
Parkinson’s Disease and Autonomic Failure
In Parkinson’s disease, extrapyramidal motor problems are the presenting features. Later in the disease process, patients may also suffer severe autonomic failure, which makes the clinical distinction from MSA difficult. As in Parkinson’s disease, some patients with MSA display motor deficits before autonomic failure is apparent, which complicates the distinction further. However, dysautonomia in Parkinson’s disease is rarely as severe as that in MSA. The uncommonly encountered patients with both Parkinson’s disease and autonomic failure are usually older and are often responsive to L-dopa. Although in most cases autonomic failure occurs late, there is a subgroup of patients with Parkinson’s disease who have clinically significant autonomic failure early in the course of the disease. Orthostatic hypotension is often the key clinical feature suggestive of autonomic failure. However, there are many causes of orthostatic hypotension, including side effects of antiparkinsonian therapy (such as L-dopa or selegiline), coincidental disease causing autonomic dysfunction (e.g., diabetes mellitus), or concomitant administration of drugs for an allied condition (e.g., antihypertensives or α-adrenoceptor blockers).55 Studies on patients with Parkinson’s disease indicate that selegiline can cause orthostatic hypotension independently of autonomic failure through mechanisms that are not clearly defined.56 Together, the confounding effect of antiparkinsonian drugs that often worsens orthostatic hypotension and difficulties in the differential diagnosis (particularly between Parkinson’s disease and MSA) make it difficult to estimate accurately the prevalence of autonomic dysfunction in patients with Parkinson’s disease. Studies that mistakenly include patients with MSA-P may overestimate the frequency of autonomic dysfunction in Parkinson’s disease or underestimate it if patients with both Parkinson’s disease and autonomic dysfunction are diagnosed as MSA-P.54
In a retrospective study, almost one third of patients with Parkinson’s disease confirmed with post mortem examination had autonomic dysfunction documented in their clinical records.57 However, it has to be kept in mind that this retrospective method may underestimate the frequency of autonomic failure. Actually, bladder dysfunction (such as urgency, frequency, and incontinence) and decreased gastrointestinal motility represent the most frequent autonomic problems in Parkinson’s disease. Constipation is extremely common. Moreover, intestinal pseudo-obstruction and toxic megacolon may occur. Sexual dysfunction (loss of libido and erectile failure) is common in this disorder.54
In a study of patients whose Parkinson’s disease was diagnosed by means of clinical criteria, almost two thirds of subjects had orthostatic hypotension with symptoms of cerebral hypoperfusion, including syncope, when tested on a tilt table for 40 minutes or until symptoms developed.58 Because patients with normal responses and with orthostatic hypotension were taking similar drug regimens, antiparkinsonian medication was not the main cause of orthostatic hypotension. Senard and colleagues59 found a fall of at least 20 mm Hg of systolic blood pressure in almost 60% of patients with Parkinson’s disease. There was symptomatic orthostatic hypotension in 20% of the patients. It was related to duration and severity of the disorder, as well as with the use of higher daily L-dopa and bromocriptine dosages.59 A higher prevalence of symptomatic orthostatic hypotension (78%) was found in a retrospective study on patients with neuropathologically confirmed Parkinson’s disease.60 In an earlier study, vagal control of the heart and hemodynamic response to standing were impaired and related to duration of the clinical features of Parkinson’s disease.61
Between 20% and 40% of patients with Parkinson’s disease become demented in the course of their illness.62 Operational criteria defining the clinical boundaries between Parkinson’s disease and Parkinson’s disease with dementia (PDD) are lacking, although this distinction may have profound clinical implications for prognosis and treatment strategies.63 The criteria in Diseases and Statistical Manual of Mental Disorders (Fourth Edition, DSM-IV™) are incomplete and descriptive and do not describe several core clinical features associated with dementia in Parkinson’s disease. Peralta and colleagues showed that orthostatic hypotension is more frequent and more severe in patients with PDD than in those with Parkinson’s disease. Attentional scores during tilt testing were also more reduced in patients with PDD in comparison with those with Parkinson’s disease, which suggests that orthostatic hypotension may exacerbate cognitive dysfunction in patients with PDD.64
Dementia with Lewy Bodies
DLB is the most frequent cause of degenerative dementia after Alzheimer’s disease. Whether DLB and PDD are the same or different disorders is uncertain.65 Clinically, the central feature required for a diagnosis of DLB is progressive cognitive decline, severe enough to cause social and occupational functional impairment. Core features of DLB are fluctuating cognition, recurrent and persistent visual hallucinations, and extrapyramidal motor symptoms. Supportive features may increase diagnostic sensitivity. They include repeated falls, syncope, transient loss of consciousness, neuroleptic sensitivity, systematized delusions, and hallucinations in other modalities.
The two main differential diagnoses are Alzheimer’s disease and PDD. In order to improve the differential diagnosis of DLB, consensus criteria that establish possible and probable levels of diagnostic accuracy have been developed.12,66 In general, their sensitivity is variable and low, but their specificity is high. Current consensus is to restrict a diagnosis of DLB only to patients with parkinsonism who develop dementia within 12 months of the onset of motor symptoms. With the use of operationally defined criteria, DLB can be clinically diagnosed with an accuracy similar to that achieved for Alzheimer’s disease or Parkinson’s disease.
Autonomic failure is frequent in DLB. A retrospective analysis of autonomic symptoms in neuropathologically diagnosed DLB showed that 62% of affected patients had significant autonomic failure.67 Patients with DLB may suffer vocal cord palsy, which results in sudden death. However, autonomic function has not been well documented in patients with DLB. Some of the supportive features, including repeated falls, syncope, and transient loss of consciousness, can be attributed in part to autonomic nervous system abnormalities. Orthostasis, either asymptomatic or associated with syncope, may be observed in these patients, although symptomatic orthostatic hypotension has been found in a lower frequency (15%) than in other types of parkinsonism.60
Mean age at onset is 75 years; the age range is 50 to 80 years, with a slight male predominance.68 Although dementia is the most frequent presenting feature, psychiatric symptoms or transient alterations of consciousness are other early features. Indeed, affected patients may present with recurrent visual hallucinations even without exposure to dopaminergic antiparkinsonian agents and may have marked diurnal fluctuations in cognitive performance, which have been the most difficult feature of the disease to define but are often conspicuous in the environment. Although parkinsonism is common in DLB, occurring at some point during the course of the illness in 75% to 80% of cases,69,70 a minority of patients present with parkinsonism alone. In general, autonomic features occur later in the course of the disease, but some cases have been described in which dysautonomia was the initial and prominent feature, leading to an initial misdiagnosis of MSA.71 Fluctuating cognition, probably related to fluctuations in attention, is characteristic of DLB, occurring in 58% of cases at the time of presentation and observed during the disease course in 75%.72 The natural history of the neuropsychological changes in DLB is not well characterized, although differences with Alzheimer’s disease appear particularly pronounced in the early stages and lessen as the disease progresses. A rapidly progressive dementia, accompanied by aphasia, dyspraxia, and spatial disorientation suggestive of temporoparietal dysfunction can be seen as the disease progresses. Disability in DLB progresses at a rate similar to that in Parkinson’s disease (approximately 10% decline per year) or even at a significantly faster rate. The latency to onset of orthostatic hypotension in a postmortem series of the National Institute of Neurologic Disorders and Stroke were short in MSA patients, intermediate in patients with DLB, corticobasal degeneration, and progressive supranuclear palsy (PSP) and long in those with Parkinson’s disease.60 These data underpin the rapidly progressive nature of the disease process in DLB in comparison with that of Parkinson’s disease. As a result, mean length of survival in a series of patients with DLB confirmed with post mortem examination has been less than 10 years. It is similar to that for Alzheimer’s disease, although some patients with DLB show rapid symptom progression and die within 1 to 2 years of onset. Risk factors for increased mortality in DLB that are present at disease onset include older age, dementia, fluctuating cognition, and hallucinations.73 Strikingly, patients with DLB with neuroleptic sensitivity reactions show a twofold to threefold increase in mortality.
Multiple-System Atrophy
This disease affects both men and women, usually starts in the sixth decade, and progresses relentlessly, with a mean survival length of 6 to 9 years.74–77 There is considerable variation in disease progression, with survival lengths of more than 15 years in some instances.
Clinically, cardinal features include autonomic failure, parkinsonism, cerebellar ataxia, and pyramidal signs in various combinations. Previous studies suggest that 29% to 33% of patients with isolated late-onset cerebellar ataxia and 8% of patients with parkinsonism eventually develop MSA.78–80 Of importance, both motor presentations of MSA are associated with similar survival times.76 However, patients with MSA-P have a more rapid functional deterioration than do patients with MSA-C.74
MSA-P associated parkinsonism is characterized by progressive akinesia and rigidity. Jerky postural tremor and, less commonly, tremor at rest may be superimposed. Frequently, patients exhibit orofacial or craniocervical dystonia in association with a characteristic quivering, high-pitched dysarthria. Postural stability is compromised early on; however, recurrent falls at disease onset are unusual, in contrast to PSP. Differentiating between MSA-P and Parkinson’s disease may be exceedingly difficult in the early stages because of a number of overlapping features such as rest tremor or asymmetrical akinesia and rigidity. Furthermore, L-dopa–induced improvement of parkinsonism may be seen in 30% of MSA-P patients. However, the benefit is transient in most of these subjects, leaving 90% of the MSA-P patients unresponsive to L-dopa in the long term. L-Dopa–induced dyskinesias affecting orofacial and neck muscles occur in 50% of MSA-P patients, sometimes in the absence of motor benefit.81 In most instances, a fully developed clinical picture of MSA-P evolves within 5 years of disease onset, allowing a clinical diagnosis during follow-up.82
The cerebellar disorder of MSA-C comprises gait ataxia, limb kinetic ataxia, and scanning dysarthria, as well as cerebellar oculomotor disturbances. Patients with MSA-C usually develop additional noncerebellar symptoms and signs but, before doing so, may be indistinguishable from other patients with idiopathic late-onset cerebellar ataxia, many of whom have a disease restricted clinically to cerebellar signs and pathologically to degeneration of the cerebellum and olives.78
Dysautonomia is characteristic of both MSA motor presentations, comprising primarily urogenital and orthostatic dysfunction. During the early stages of MSA, autonomic deficits may be the sole clinical manifestation, thus resembling PAF, but after a variable period of time (sometimes 2 or 3 years, always less than 5), extrapyramidal or cerebellar deficits or both invariably develop. Early impotence (erectile dysfunction) is virtually universal in men with MSA, and urinary incontinence or retention, often early in the course or as presenting symptoms, are frequent.77 Disorders of micturition in MSA are caused by changes in the complex peripheral and central innervation of the bladder83 and generally occur more commonly, earlier, and to a more severe degree than in Parkinson’s disease. In fact, patients with MSA have early dysuria with or without chronic retention, frequently associated with a hypoactive detrusor muscle and low urethral pressure. In contrast, patients with Parkinson’s disease have urgency to void, with or without difficulty, but without chronic retention, in association with detrusor hyperreflexia and normal urethral sphincter function. Constipation occurs in equal percentages of patients in Parkinson’s disease and MSA. Symptomatic orthostatic hypotension is present in 68% of patients with clinical diagnoses of MSA, but recurrent syncope emerges in only 15%.77 L-Dopa or dopamine agonists may provoke or worsen orthostatic hypotension.
LABORATORY ASSESSMENTS
In addition to the clinical presentation, several laboratory investigations have been used to distinguish among Parkinson’s disease, PAF, and MSA (Table 30-2). Basically, most of these tests exploit the anatomopathological distinction between Lewy body syndromes, which affect postganglionic autonomic neurons, and MSA, which affects preganglionic, central autonomic neurons.
TABLE 30-2 Laboratory Investigations in Primary Autonomic Failure
| Examined Body Domain/Function | Parameters/Techniques |
|---|---|
| Cardiovascular | |
| Physiological | Head-up tilt (60 degrees); standing; Valsalva maneuver |
| Pressor stimuli (isometric exercise, cutaneous cold, mental arithmetic) | |
| Heart rate responses-deep breathing, hyperventilation, standing, head-up tilt, 30:15 R-R interval ratio | |
| Liquid meal challenge | |
| Modified exercise testing | |
| Carotid sinus massage | |
| Biochemical | Plasma noradrenaline: supine and head-up tilt or standing; urinary catecholamines; plasma renin activity, and aldosterone |
| Pharmacological | Noradrenaline: α-adrenoceptors, vascular |
| Isoprenaline: β-adrenoceptors, vascular and cardiac | |
| Tyramine: pressor and noradrenaline responses | |
| Edrophonium: noradrenaline response | |
| Atropine: parasympathetic cardiac blockade | |
| Imaging | Cardiac [123I]MIBG SPECT, 6-[18F] fluorodopamine PET |
| Brain | |
| Imaging | MRI (1.5 Tesla), diffusion-weighted imaging, voxel-based morphometry, [(123)I]β-CIT, [123I]iodobenzamide SPECT, 18F-fluorodopa PET, [11C]diprenorphine PET, 18F-fluorodeoxyglucose PET, 99mTc-hexamethylpropyleneamine oxime, [123I]FP-CIT |
| CSF studies | Neurofilament protein levels |
| Endocrine | Clonidine–α2-adrenoceptor agonist: noradrenaline suppression; growth hormone stimulation |
| Sudomotor | Thermoregulatory sweat test |
| Sweat gland response to intradermal acetylcholine, QSART, localized sweat test | |
| Sympathetic skin response | |
| Gastrointestinal | External anal sphincter EMG, video-cinefluoroscopy, barium studies, endoscopy, gastric emptying studies, transit time, lower gut studies |
| Renal function and urinary tract | Day and night urine volumes and sodium/potassium excretion measurements |
| Urodynamic studies, intravenous urography, ultrasonographic examination, sphincter electromyography | |
| Sexual | Penile plethysmography |
| Intracavernosal papaverine | |
| Respiratory | Laryngoscopy |
| Sleep studies to assess apnea and oxygen desaturation | |
| Eye and lacrimal glands | Pupillary function, pharmacological and physiological |
| Schirmer’s test | |
11C, carbon 11; β-CIT, 2β-carboxymethoxy-3β-(4-iodophenyl)tropane; CSF, cerebrospinal fluid; EMG, electromyography; 18F, fluorine 18; 123I, iodine 123; FP-CIT, 2β-carbomethoxy-3β-(4-iodophenyl)-N-(3-fluoropropyl)nortropane; MIBG, meta-iodobenzylguanedine; MRI, magnetic resonance imaging; PET, positron emission tomography; QSART, quantitative sudomotor axon reflex test; SPECT, single photon emission computed tomography.
Modified from Mathias CJ: Autonomic diseases: clinical features and laboratory evaluation. J Neurol Neurosurg Psychiatry 2003; 74(Suppl 3):iii31-iii41.
Cardiovascular Function Testing
A history of postural faintness or other evidence of orthostatic hypotension, such as neck ache on rising in the morning or posturally related changes of visual perception, should be sought in all patients in whom MSA is suspected. After a comprehensive history is documented, cardiovascular function should be tested according to consensus recommendations.47,84 A drop in systolic blood pressure of 20 mm Hg or more or in diastolic blood pressure of 10 mm Hg or more, in comparison with baseline within a standing time of 3 minutes, is defined as orthostatic hypotension and must lead to more specific assessment. This is based on continuous noninvasive measurements of blood pressure and heart rate during tilt-table testing.85–87 Although abnormal cardiovascular test results may provide evidence of sympathetic and/or parasympathetic failure, they do not differentiate autonomic failure associated with Parkinson’s disease from that associated with MSA.88
The autonomic abnormality of MSA can be distinguished biochemically from that of PAF. In MSA, during supine rest, norepinephrine (noradrenaline) levels (representing postganglionic sympathetic efferent activity) are normal,89,90 and there is no denervation hypersensitivity, which indicates a lack of increased expression of adrenergic receptors on peripheral neurons.90 In contrast to this normal or only slightly decreased plasma norepinephrine level during recumbency in MSA and varying levels in patients with Parkinson’s disease, patients with PAF have very low plasma norepinephrine levels when recumbent.90,91 On standing or tilt-table testing, patients with PAF, those with MSA, and some with Parkinson’s disease with autonomic failure do not have the expected increase in plasma norepinephrine levels, which indicates an inability to normally stimulate the release of catecholamines by baroreflex activation in all these disorders. When norepinephrine is infused into patients with PAF, there is an exaggerated increase in blood pressure. This reflects an excessive sensitivity of postsynaptic α-adrenergic receptors to exogenous catecholamines. In contrast, patients with MSA and Parkinson’s disease show only a mildly increased blood pressure response to infused norepinephrine, without leftward shift in the dose-response curve.92 Similarly, there is a greater degree of β-adrenergic receptor supersensitivity in PAF than in MSA, as shown by Baser and associates93 in a study with intravenous isoproterenol.
Sympathetic cardiac innervation is selectively affected in Parkinson’s disease and PAF but is intact in MSA. Imaging studies that measure catecholamine uptake by cardiac sympathetic neurons have confirmed that peripheral sympathetic nerves are preserved in MSA but greatly reduced in PAF.94 Visualization of sympathetic cardiac neurons through scintigraphy with norepinephrine analogue iodine 123–metaiodobenzylguanidine ([123I]MIBG) has revealed loss of binding in patients with Parkinson’s disease, regardless of disease severity, which reflects postganglionic sympathetic denervation; in comparison, cardiac binding is preserved in MSA95–100 and PSP.101 Pooled data from several studies showed that MIBG scintigraphy occurately discriminated a total of 246 cases of Parkinson’s disease from 45 of MSA with high sensitivity (90%) and specificity (95%).96 Similarly, (18F) fluorodopamine positron emission tomography (PET) is able to demonstrate cardiac sympathetic denervation in PAF and Parkinson’s disease in contrast with intact cardiac sympathetic innervation in MSA.94 6-[18F] Fluorodopamine is a catecholamine taken up by sympathetic post ganglionic neurons and handled similarly to norepinephrine. Together, these types of imaging of sympathetic cardiac neurons may turn out to be useful diagnostic tests to distinguish between Parkinson’s disease and MSA because sympathetic innervation of the heart is impaired in Parkinson’s disease and not in MSA. Moreover, in a patient with apparent PAF, finding normal sympathetic cardiac innervation should indicate a likely development of MSA.4 A caveat of this approach to be kept in mind is that published studies have compared patients with established diagnoses of MSA and PAF and, therefore, probably in later disease stages. It is not known whether these differences are apparent in patients during earlier stages of the disorder, when a diagnostic method would be more useful in the workup of patients in clinical practice.92
DLB affects peripheral postganglionic autonomic neurons (as with the other Lewy body syndromes). Actually, neuropathological studies on DLB associated with severe autonomic failure demonstrated—besides numerous Lewy bodies in the cortex and brainstem—sparse Lewy bodies in the intermediolateral columns of the spinal cord, as well as numerous Lewy bodies in autonomic ganglia and sympathetic neurons.71 These results suggests that, as in Parkinson’s disease,102 autonomic dysfunction in DLB is, at least in part, caused by degeneration of peripheral autonomic neurons. This is supported by single photon emission computed tomography (SPECT) imaging of postganglionic sympathetic cardiac innervation with 123I-MIBG, which showed that postganglionic sympathetic neurons were nonfunctional.54 Indeed, 123I-MIBG SPECT has been shown to discriminate DLB from Alzheimer’s disease with severe denervation in the former and preserved sympathetic MIBG binding in the latter disorder.103–105
Bladder Function Testing
Assessment of bladder function is a mandatory part of the diagnostic workup of parkinsonian patients and usually provides evidence of involvement of the autonomic nervous system already at an early stage of the disease (when bladder function is still normal in most patients with Parkinson’s disease). Urinary symptoms and studies of bladder function may help to distinguish between MSA-P and Parkinson’s disease. After documentation of a careful history regarding frequency of voiding, difficulties in initiating or suppressing voiding, and the presence of urinary incontinence, a standard urine analysis should be performed to rule out an infection. Postvoid residual volume needs to be determined sonographically or through catheterization to initiate intermittent self-catheterization in due course. In some patients, only cystometry can discriminate between hypocontractile detrusor function and hyperreflexic sphincter-detrusor dyssynergy. The nature of bladder dysfunction is different in MSA and Parkinson’s disease. Although frequency and urgency are common in both disorders, marked urge or stress incontinence with continuous leakage is not a feature of Parkinson’s disease, apart from very advanced cases. Urodynamic studies show a characteristic pattern of abnormality in patients with MSA.106 In the early stages, there is often detrusor hyperreflexia, often with bladder neck incompetence caused by abnormal urethral sphincter function, which results in early frequency and urgency, followed by urge incontinence. Later on, the ability to initiate a voluntary micturition reflex and the strength of the hyperreflexic detrusor contractions diminish, and the bladder may become atonic, which accounts for increasing postmicturition residual urine volumes.
Sphincter Electromyography
As a matter of fact, the striated muscle of the external anal and urethral sphincter is innervated by fibers that originate in Onuf’s nucleus (see “Neuropathology” section). This nucleus is particularly vulnerable in MSA but not in Parkinson’s disease.54 Involvement of Onuf’s nucleus is much more frequent than that of anterior horn cells in the rest of the spinal cord in MSA, although there may be some depletion of anterior horn cells.39 Interestingly, the reverse occurs in patients with amyotrophic lateral sclerosis, in whom Onuf’s nucleus is selectively spared. On electromyography (EMG) of the anal and urethral sphincter muscle, neuronal loss in Onuf’s nucleus is reflected by signs of denervation and chronic reinnervation. An abnormal finding on sphincter EMG may be found in many patients with clinically definitive MSA, including those who as yet have no urological or anorectal problems. In at least 80% of patients with MSA, EMG of the external anal sphincter reveals signs of neuronal degeneration in Onuf’s nucleus with spontaneous activity and increased polyphasia.83,107,108 Schwarz and colleagues109 suggested that abnormal spontaneous activity on sphincter EMG, although difficult to detect, may be the most useful criterion for distinguishing between Parkinson’s disease and MSA. However, the prevalence of abnormalities in early stages of MSA remains unclear. These findings do not reliably differentiate between MSA and other forms of atypical parkinsonian disorders such as PSP.110 Furthermore, neurogenic changes of external anal sphincter muscle have also been demonstrated in advanced stages of Parkinson’s disease.111 Chronic constipation, previous pelvic surgery, or vaginal deliveries can also be confounding factors that induce nonspecific abnormalities.112 However, abnormalities on anal sphincter EMG appear to distinguish MSA from Parkinson’s disease in the first 5 years after disease onset and from PAF, as well as from cerebellar ataxias, if other causes for sphincter denervation have been ruled out.113
Neuroendocrine Testing
In vivo studies in MSA, which involved testing of the endocrine component of the central autonomic nervous systems (the hypothalamopituitary axis) with a variety of challenge procedures, provided evidence of impaired humoral responses of the anterior and the posterior parts of the pituitary gland with impaired secretion of adrenocorticotropic hormone,114 growth hormone,115 and vasopressin/antidiuretic hormone.34 Although these observations can be made in virtually all patients in an advanced stage of the disease, their prevalence during the early course of MSA is unknown.
PAF selectively affects the efferent, mainly postganglionic autonomic neurons. Afferent pathways are not involved. Baroreceptor-mediated vasopressin release—a measurement of afferent baroreceptor function—is normal in patients with PAF, and presumably in those with Parkinson’s disease, but is blunted in patients suffering from MSA.34 Intravenous clonidine also tests the function of hypothalamic-pituitary pathways.92 Clonidine is a centrally active α2-adrenoceptor agonist that stimulates growth hormone secretion and lowers blood pressure predominantly by reducing central nervous system sympathetic outflow. There is an ongoing debate about the diagnostic value of the growth-hormone response to clonidine, a neuropharmacological assessment of central adrenoceptor function, in Parkinson’s disease and MSA. In an early study, there was no increase in growth hormone levels after clonidine administration in patients with MSA in comparison with those with Parkinson’s disease or PAF.116 Kimber and colleagues115 confirmed a normal increase in serum growth hormone in response to clonidine in 14 patients with Parkinson’s disease (without autonomic failure) and in 19 patients with PAF, whereas there was no growth hormone rise in 31 patients with MSA. However, these findings were challenged subsequently.117–119 After clonidine administration, growth hormone rose in patients with PSP and controls, but not in patients with MSA.120 In patients with PSP, responses to both physiological and pharmacological tests provided evidence against widespread autonomic dysfunction; this differed markedly from patients with MSA. Stimulation of growth hormone release with growth hormone–releasing hormone plus arginine rather than clonidine may differentiate MSA from idiopathic Parkinson’s disease and idiopathic late-onset cerebellar ataxia,121 but this hypothesis would need to be confirmed by further investigations. In normal humans, clonidine reduces arginine-vasopressin secretion, probably by presynaptic inhibition of noradrenergic neuron terminals in the supraoptic nucleus. A lesion of noradrenergic pathways in animals abolishes this response to clonidine. Postmortem study in MSA reveals marked loss of hypothalamic noradrenergic innervation. After clonidine administration, there was a significantly greater fall of arginine-vasopressin levels in controls than in patients with MSA, which suggests that there is an abnormal arginine-vasopressin response to clonidine in MSA, which probably represents loss of functional noradrenergic innervation of the supraoptic nucleus.122 More studies in well-defined patient cohorts are needed before clonidine challenge tests can be recommended as helpful diagnostic tests in patients with suspected MSA. Neuroendocrine responses to hypotension or centrally acting adrenergic agonists are blunted in MSA but are preserved in Parkinson’s disease and PAF, inasmuch as brainstem-hypothalamic-pituitary pathways are affected only in MSA.92
Hypothalamic dopaminergic pathways are involved in the regulation of growth hormone and prolactin release from the anterior pituitary. Neuroendocrine studies in patients with MSA, in whom there is a reported loss of hypothalamic dopamine, are few and contradictory. In patients with MSA, the growth hormone–releasing hormone and growth hormone responses to L-dopa were preserved and were similar to responses in age-matched control subjects in a study by Kimber and colleagues.123 In contrast, there was impaired dopaminergic suppression of prolactin secretion. In patients with MSA, this may represent a selective dysfunction, rather than generalized loss, of tuberoinfundibular dopaminergic neurons.
Besides orthostatic hypotension, supine hypertension, paradoxically, is present in about one half of patients with MSA or PAF.124 However, the mechanisms of supine hypertension differ between MSA and PAF. Eliminating residual sympathetic tone with the ganglionic blocker trimethaphan completely abolished hypertension in patients with MSA but not in those with PAF.125 Therefore, hypertension in MSA can be totally explained by the residual sympathetic tone unopposed by the absence of baroreflex mechanisms. However, patients with MSA are not able to engage and modulate sympathetic tone during upright posture. Therefore, they are suffering from orthostatic hypotension despite their supine hypertension. In patients with PAF, the cause of supine hypertension is not yet resolved.54
Brain Imaging
Magnetic resonance imaging (MRI) of the brain and PET of the brain and heart (see previous discussion) may be helpful in the differential diagnosis of a patient with parkinsonism and autonomic failure, distinguishing among PAF, MSA, and Parkinson’s disease. In patients with PAF, MRI results of the brain are normal.92 In Parkinson’s disease, brain MRI reveals only mild putaminal abnormalities with few brainstem or cerebellar changes. In contrast, in patients with MSA, brain MRI shows severe putaminal abnormalities, frequently accompanied by brainstem and cerebellar changes. However, MRI abnormalities in MSA frequently occur late in the time course of the disorder, and the abnormalities reviewed in the following sections have been observed in patients with advanced rather than early disease. In these early disease stages, MRI is not very sensitive but has good specificity. The diagnosis of MSA still rests on the clinical history and findings of the neurological examination. According to the Consensus Conference on the Diagnosis of MSA,23 additional investigations such as neuroimaging (and autonomic function tests or sphincter EMG, described previously) may be used to support the diagnosis or to exclude other conditions. Therefore, the Consensus Conference considered it premature to incorporate the results of laboratory investigations into the diagnostic guidelines that were established.
Structural Imaging
Routine 1.5-T MRI, including diffusion-weighted imaging, should be performed in all patients with suspected MSA, because basal ganglia and/or brainstem abnormalities suggestive of MSA may be observed even during early disease stages. These changes include an OPCA-like atrophy pattern indistinguishable from autosomal dominant cerebellar ataxia.126 MRI measures of basal ganglia pathology in MSA are less well established, and naked eye assessments are often unreliable. In advanced cases, putaminal atrophy may be detectable, and its extent may be correlated with severity of extrapyramidal symptoms.
Abnormalities on MRI may include not only OPCA126 or putaminal atrophy127 but also signal abnormalities on T2-weighted images. Signal hyperintensities within the pons and middle cerebellar peduncles are believed to reflect degeneration of pontocerebellar fibers; these changes occasionally produce an appearance resembling a hot cross bun.127 Nonspecific putaminal hypointensities in patients with atypical parkinsonism, including MSA, were first reported in 1986 by two groups who used 1.5-T T2-weighted images.128,129 This change has subsequently been confirmed by other authors in cases of pathologically proved MSA.130–132 Similar MRI abnormalities may occur in patients with classic Parkinson’s disease.133 However, Kraft and colleagues demonstrated that hypointense putaminal signal changes were more frequent in MSA than in Parkinson’s disease, by using T2*-weighted gradient echo instead of T2-weighted fast-spin echo images; this indicates that T2*-weighted gradient echo sequences are of better diagnostic value for patients with parkinsonism.134 Increased putaminal hypointensities may be associated with a slitlike hyperintense band lateral to the putamen.134,135 This finding appears to be more specific for MSA than is putaminal hypointensity127,136; however, further studies in larger cohorts of patients are needed to confirm this. The hyperintense slit signal was correlated with reactive microgliosis and astrogliosis in a case of pathologically proved MSA.132
Diffusion-weighted imaging may represent a useful diagnostic tool that can provide additional support for a diagnosis of MSA-P. Diffusion-weighted imaging is able to discriminate patients with MSA-P from both patients with Parkinson’s disease and healthy volunteers on the basis of putaminal regional apparent diffusion coefficients (rADC) values.137 The increased putaminal rADC values in MSA-P probably reflect ongoing striatal degeneration, whereas most neuropathological studies reveal intact striatum in Parkinson’s disease. However, because rADCs were also significantly increased in both putamen and globus pallidus in PSP in comparison with Parkinson’s disease,138 increased putaminal rADC values do not discriminate MSA-P from PSP.
Schulz and associates139 found significant reductions in mean striatal and brainstem volumes in patients with MSA-P, MSA-C, and PSP, whereas patients with MSA-C and MSA-P also showed a reduction in cerebellar volume. More recently, voxel-based morphometry confirmed previous region of interest–based volumetric studies139 showing basal ganglia and infratentorial volume loss in MSA-P patients.140 These data also revealed prominent cortical volume loss in MSA-P, comprising mainly the cortical targets of striatal projections such as the primary sensorimotor cortices, lateral premotor cortices, and the prefrontal cortex. MRI-based volumetry is a helpful tool to investigate the progression of cortical and subcortical atrophy patterns in MSA in comparison with other disorders; however, it cannot be applied for routine diagnostic workups of individual patients.
Structural brain imaging with MRI reveals a relative preservation of the medial temporal lobes and the hippocampus in 40% of patients with DLB, in contrast to Alzheimer’s disease.141 There is no difference from Alzheimer’s disease in terms of degree of ventricular enlargement or presence of white matter changes on MRI.142 MRI shows atrophy of the putamen in DLB but not in Alzheimer’s disease.143 Additional features such as generalized atrophy141 and rates of progression of whole-brain atrophy144 are not helpful in differential diagnosis. In volumetric studies, frontal brain atrophy was described in DLB patients, which was correlated with increasing Lewy body densities.145 Voxel-based morphometry of gray matter revealed significant atrophy of the basal forebrain in DLB, which discriminates it from Alzheimer’s disease.146
Functional Imaging
PET imaging in MSA reveals a generalized reduction in glucose utilization rate, which indicates hypometabolism, most prominently in the cerebellum, brainstem, striatum, and frontal and motor cortices. In contrast, none of these findings was present in PAF.92 In fact, the Hammersmith Cyclotron Unit, using PET, found that putaminal uptake of the presynaptic dopaminergic markers [18F]fluorodopa and S-[11C]nomifensine147,148 was similarly reduced in MSA and Parkinson’s disease; in approximately one half the patients with MSA, caudate uptake was also markedly reduced, as opposed to only moderate reduction in Parkinson’s disease. However, discriminant function analysis of striatal [18F]fluorodopa uptake distinguished patients with MSA from those with Parkinson’s disease poorly.149 Measurements of striatal dopamine D2 receptor densities with raclopride and PET failed to differentiate between Parkinson’s disease and atypical parkinsonian disorders, demonstrating a similar loss of densities in patients with advanced Parkinson’s disease, MSA, and PSP.150 PET studies with other ligands such as [11C]diprenorphine (nonselective opioid receptor antagonist)151 and [18F]fluorodeoxyglucose152–154 have proved more consistent in detecting striatal degeneration and in distinguishing patients with MSA-P from those with Parkinson’s disease, particularly when combined with a dopamine D2 receptor scan.155 Widespread functional abnormalities in MSA-C have been demonstrated through [18F]fluorodeoxyglucose and PET.156 Reduced metabolism was most marked in the brainstem and cerebellum, but other areas such as the basal ganglia and cerebral cortex were also involved, which is evidence of its nosological status as the cerebellar subtype of MSA.
Furthermore, assessing nigrostriatal dopaminergic function with 18F-fluorodopa PET may be a useful diagnostic aid in cases of DLB, inasmuch as there is a pronounced reduction of striatal dopamine uptake.157 PET examination of the cerebral glucose metabolism with 18F-fluorodeoxyglucose demonstrated that among widespread cortical regions showing glucose hypometabolism in patients with DLB, the metabolic reduction was most pronounced in the visual association cortex, in comparison with that in patients with Alzheimer’s disease.158 Therefore, among several potential antemortem biomarkers in the diagnosis of DLB, measures of the glucose metabolism in the occipital cortex may be an informative diagnostic aid to distinguish DLB from Alzheimer’s disease.158,159
SPECT evaluation of the dopamine transporter with 2β-carboxymethoxy-3β-(4-iodophenyl)tropane ([123I]β-CIT) reflects the disruption of the nigrostriatal pathway, and therefore MSA and PSP cannot be distinguished from Parkinson’s disease with this method alone.160 However, dopamine transporter SPECT may be useful in differentiating parkinsonism from controls.161 In another SPECT study, striatal [123I]β-CIT uptake was markedly reduced in both the patients with Parkinson’s disease and those with MSA,162 but patients with MSA showed a more symmetrical dopamine transporter loss, consistent with the more symmetrical clinical motor dysfunction observed in this condition.
SPECT studies using [123I]IBZM as D2 receptor ligand have revealed significant reductions of striatal IBZM binding in subjects with clinically probable MSA in comparison with patients with Parkinson’s disease or controls.163–165 However, striatal IBZM binding is also reduced in other atypical parkinsonian disorders such as PSP,164 which limits its predictive value for an early diagnosis of MSA.
IBZM SPECT imaging in patients with early parkinsonism seems to distinguish between L-dopa–responsive and L-dopa–unresponsive parkinsonism in patients not previously treated with dopaminergic drugs.166 A good response to apomorphine challenge and subsequent benefit from chronic dopaminergic therapy was observed in subjects with normal IBZM binding, whereas subjects with reduced binding failed to respond. Some of these patients developed other atypical clinical features suggestive of MSA during follow-up.167
Functional neuroimaging with technetium 99m–hexamethylpropyleneamine oxime and SPECT reveals occipital hypoperfusion in DLB, differentiating it from Alzheimer’s disease.168,169 In DLB, as well as in PDD, bilateral temporal and parietal perfusion deficits have been reported. Dopamine transporter loss in the caudate and putamen, a marker of nigrostriatal degeneration, can be detected by dopaminergic SPECT.170 [123I]-2β-carbomethoxy-3β-(4-iodophenyl)-N-(3-fluoropropyl)nortropane (FP-CIT) and SPECT reveal significant reduction in striatal uptake of a ligand for the presynaptic dopamine transporter site (FP-CIT) in DLB but not in Alzheimer’s disease, and this may prove to be a highly specific and widely applicable diagnostic test.171,172
THERAPY
Acute/Subacute Primary Autonomic Failure
Patient education is an important aspect of treatment. Sympathomimetic drugs and 9-α-fluorohydrocortisone have been of value in treating postural hypotension in cases of pandysautonomia. Correcting anemia improves orthostatic tolerance, if necessary by hemopoietin. Because the underlying mechanism is likely to be immune mediated, plasma exchange or other immunosuppressive modalities as early therapeutic intervention in patients with progressive disability may be justified. In fact, corticosteroids are frequently used, and plasmapheresis and intravenous immunoglobulin may be effective.173–175 Carbachol may be helpful for the management of urinary retention and impaired gastrointestinal motility associated with acute cholinergic neuropathy.
Chronic Primary Autonomic Failure
Because of the chronic or progressive course of primary autonomic failure syndromes, a regular review of treatment is mandatory to adjust measures according to clinical needs. Guidelines for the practical management of chronic PAF syndromes are shown in Table 30-3.
TABLE 30-3 Practical Management of Chronic Primary Autonomic Failure Syndromes
| Feature | Pharmacological Measures | Nonpharmacological Measures |
|---|---|---|
| Orthostatic hypotension | Starter drug | To be avoided |
| Fludrocortisone (0.1-0.3-0.4 mg/day) | Sudden head-up postural change (especially on waking) | |
| Sympathomimetics | Prolonged recumbency | |
| Ephedrine (15-45 mg t.i.d.) | Straining during micturition and defecation | |
| Midodrine (2.5-10 mg t.i.d.) | High environmental temperature (including hot baths) | |
| L-Threo-DOPS (300 mg b.i.d.) | “Severe” exertion | |
| Specific targeting | Large meals (especially with refined carbohydrates) | |
| For postprandial hypotension: | Alcohol | |
| Octreotide (25-50 μ g subcutaneously, 30 min before a meal) | Drugs with vasodepressor properties | |
| For nocturnal polyuria: | To be introduced | |
| Desmopressin (spray: 10-40 μ g/night or tablet: | Head-up tilt during sleep | |
| 100-400-600 μ g/night) | Small frequent meals | |
| Increasing red cell mass: | High salt intake | |
| Erythropoietin (25-50U/kg body weight subcutaneously three times a week) | Judicious exercise (including swimming; delay physical exertion until the afternoon; exercise caution on arising in the morning and immediately after meals or physical exertion) | |
| In individual cases | ||
| Vasoconstriction: | ||
| Phenylephrine, noradrenaline, clonidine, tyramine with monoamine oxidase inhibitors, yohimbine, dihydroergotamine, terlipressin | Body position and maneuvers—elevation of the legs periodically during the day | |
| To be considered | ||
| Preventing vasodilatation: | Elastic stockings or tights | |
| Indomethacin (cave: gastric ulceration and hemorrhage), flurbiprofen, metoclopramide, domperidone, propanolol, caffeine | Abdominal binders | |
| Water ingestion | ||
| Cardiac pacing | ||
| Increasing cardiac output: | ||
| Pindolol (cave: cardiac failure), xamoterol, ibopamine | ||
| Supine hypertension | Nitroglycerin (transdermal, 0.1-0.2 mg/hour) | Nocturnal snacks |
| Short-acting calcium antagonists (e.g., nifedipine, 30 mg) | Head-up tilt at night | |
| Hydralazine (50-100 mg) | ||
| Urinary difficulties | For urinary incontinence | Treatment of concurrent prostatism in men or pelvic floor muscle laxity in women |
| Oxybutynin for detrusor hyperreflexia (2.5-5 mg b.i.d-t.i.d., or 5 to 10 mg at bedtime) | ||
| Trospium chloride (15-20 mg b.i.d.-t.i.d.) | Treatment of concurrent urinary tract infection | |
| Tolterodine (1-2-4 mg b.i.d) | Urinary acidification | |
| For incomplete bladder emptying | ||
| Prazosin (1 mg t.i.d.) | ||
| Moxisylyte (10 mg t.i.d.) | Intermittent (residual volume>100 mL) or permanent urethral or suprapubic catheterization | |
| Tamsulosine (0.4 mg o.i.d.) | ||
| Alfuzosine (5 mg b.i.d) | Penile implant/prosthesis | |
| Sexual dysfunction | Erectile dysfunction | |
| Yohimbine (2.5-5 mg t.i.d.) | ||
| Sildefanil (50-100 mg) | ||
| Intracavernosal papaverine, prostaglandin E1 | ||
| Constipation | Bulk agents, laxatives, and suppositories | Dietary fiber and liquid intake |
| Macrogol 3350/electrolyte | Physical activity | |
| Inspiratory stridor | Avoid Botulinum toxin A | Continuous positive airway pressure |
| Tracheostomy | ||
| Anchoring one vocal cord in abduction/cord lateralization procedures | ||
| Laser cricoarytenoidectomy | ||
| Nasotracheal intubation | ||
| REM sleep behavior disorder | Clonazepam 0.5-1.5 mg shortly before bedtime (cave: lower initial dose in a patient older than 70 years) | |
| Sialorrhea | Anticholinergics | Tissues |
| Botulinum toxin A: | ||
| Parotid gland: Botox®, 10-40MU | ||
| Submandibular gland: Botox®, 5-15MU |
Modified from Wenning GK, Geser F, Poewe W: Therapeutic strategies in multiple system atrophy. Mov Disord 2005; 20(Suppl 12):S67-S76. DOPS, dihydroxyphenyl serine; REM, rapid eye movement.
Motor Disorder
Treatment of the motor abnormalities is fairly successful in Parkinson’s disease but remains dismal in MSA patients. These patients often do not respond to antiparkinsonian medications and fail to benefit from current surgical treatments for Parkinson’s disease. Although less effective than in Parkinson’s disease, L-dopa replacement represents the mainstay of antiparkinsonian therapy in MSA. However, a sufficiently powered double-blind controlled trial has never been performed. Results of open-label studies suggest that, in contrast to patients with Parkinson’s disease, most patients with MSA fail to benefit from treatment with L-dopa in the long run, although a transient response may occur in some cases. However, the assumption that patients with MSA are generally not responsive or poorly responsive to L-dopa is certainly misleading. L-Dopa responsiveness should be tested by administering escalating doses (with a peripheral decarboxylase inhibitor) over a 3-month period up to at least 1000 mg per day (if necessary and if tolerated).23 Reports of open-label L-dopa therapy in MSA have documented L-dopa efficacy in up to 80% of patients with clinical diagnoses.75,77,81,176–184 Data obtained from series with pathological confirmation are more variable, with rates of beneficial L-dopa response ranging between 30% and 70%.32,77,179,185–188 On occasion, a beneficial effect is evident only when seemingly unresponsive patients deteriorate after L-dopa withdrawal.179 Whatever response there is usually declines after a few years of treatment.189 The effectiveness of L-dopa on motor symptoms in DLB has not been established but is probably less than in uncomplicated Parkinson’s disease, possibly because there is additional intrinsic striatal pathology and dysfunction.190 But, although this limited L-dopa responsiveness has generally been reported in DLB, it may reflect either a failure to treat or underdosing, because of concerns about exacerbating psychotic symptoms. In fact, there are reports showing that 70% to 100% of patients with DLB do have a good response.191–193 In comparison with Parkinson’s disease, L-dopa–related motor complications appear to be less common in DLB. L-Dopa is the preferred antiparkinsonian drug in DLB because dopamine agonists may increase the occurrence of hallucinations.194 Reviews of the therapeutic values of both dopaminergic and nondopaminergic drugs for the management of the motor disorder in Parkinson’s disease, MSA, and DLB are covered in more detail elsewhere.195–198
Orthostatic Hypotension
The concept to treat symptoms of orthostatic hypotension is based on the increase of intravasal volume and the reduction of volume shift to lower body parts when a patient changes to an upright position. The selection and combination of the following options, including both nonpharmacological and pharmacological measures, depend on the severity of symptoms and their practicability in the single patient, but not on the extent of blood pressure drop during the tilt-table test. Nonpharmacological options include sufficient fluid intake, high-salt diet, more frequent but smaller meals per day to reduce postprandial hypotension by spreading the total carbohydrate intake, and as the ultima ratio custom-made elastic body garments. During the night, head-up tilt increases intravasal volume up to 1 L within a week, which is particularly helpful in improving hypotension early in the morning. This is achieved by an increased secretion of renin as a result of reduced renal perfusion pressure and by reduced atrial natriuretic hormone levels because of lower atrial filling pressure. This approach is successful, particularly in combination with the mineralocorticoid fludrocortisone, which further supports sodium retention. Indeed, medical treatment begins with attempting to increase blood volume by increasing sodium intake unless the patient is at risk for congestive heart failure or has renal insufficiency. The next group of drugs to use is the sympathomimetics. They include ephedrine (with both direct and indirect effects), which is often valuable in central autonomic failure as occurs in MSA. In fact, ephedrine can be helpful through its peripheral vasoconstrictor effects. With high doses, side effects include tremulousness, loss of appetite, and in men, urinary retention. Orthostatic hypotension is often successfully treated with midodrine,199–202 an adrenergic agonist activating α1 receptors on arterioles and veins. Midodrine increases peripheral resistance, thereby significantly reducing orthostatic hypotension. Side effects are usually mild and only rarely lead to discontinuation of treatment because of urinary retention or pruritus predominantly on the scalp. Furthermore, L-threo-3,4-dihydroxyphenyl serine has been used with some success in short clinical trials.203 It represents a precursor of norepinephrine and has been used for this indication in Japan for years. Mathias and associates showed its efficacy in an open-label, dose-finding trial in patients with MSA and PAF.204 If the above drugs do not produce the desired effect, then selective targeting is needed. The somatostatin analogue octreotide is often beneficial in postprandial hypotension,205 presumably because it inhibits release of vasodilatory gastrointestinal peptides;206 of importance, it does not enhance nocturnal hypertension.205 The vasopressin analogue desmopressin, which acts on renal tubular vasopressin-2 receptors, reduces nocturnal polyuria and improves morning postural hypotension.207 Recombinant erythropoietin, used to reverse the anemia common in MSA, increases upright blood pressure and ameliorates symptoms of orthostatic hypotension208–211 by secondarily improving cerebral oxygenation.209,211,212
Supine Hypertension
In more than one half of patients with PAF or MSA, supine hypertension is present, complicating their management. Actually, the treatment of patients with primary autonomic failure is aimed primarily at improving orthostatic hypotension, and the need to treat supine hypertension in these patients is disputed. As a matter of fact, all antihypertensive agents may worsen orthostatic hypotension and trigger symptoms of cerebral hypoperfusion. However, the finding of left ventricular hypertrophy in hypertensive patients with primary autonomic failure suggests that this question should be reconsidered.213 Effective treatment of supine hypertension can be easily accomplished simply by avoiding the supine position, at least during daytime. Sleeping in the head-up tilt position reduces nocturnal sodium loss, which improves orthostatic hypotension in the morning (see previous discussion).214,215 Although head-up tilt may reduce hypertensive cerebral perfusion pressure, it is often not sufficient to treat supine hypertension. Theoretically, a result similar to that with head-up tilt at night could be obtained pharmacologically by reducing blood pressure with antihypertensive agents. An ideal agent would decrease blood pressure and natriuresis during the night. Furthermore, it would have a short half life, in order to avoid worsening of orthostatic hypotension in the morning.54 In any case, supine hypertension does not necessitate drug treatment if systolic blood pressure is below 200 mm Hg. Patients with primary autonomic failure are particularly sensitive to transdermal nitroglycerin.124 It can be applied at bedtime and removed on arising in the morning. Short-acting calcium antagonists216 such as nifedipine given at nighttime have also been used in PAF. However, nifedipine increased the nocturnal sodium loss in a study by Jordan and colleagues,217 and nifedipine but not nitroglycerin worsened orthostatic hypotension in the morning. Oral hydralazine can also be used.54
Bladder Dysfunction
Nocturnal voiding frequency can be lessened by curtailing fluid intake after the evening meal. If this is not effective, and if there is no significant postmicturition residual, peripherally acting anticholinergics (oxybutynin, propantheline, or tolterodine) are the initial pharmacological therapeutics to be administered.54 Anticholinergic agents alleviate detrusor hyperreflexia or sphincter-detrusor dyssynergy, but commonly at the expense of inducing urinary retention.83 Muscarinic receptors of the detrusor muscle are the common target of anticholinergics, and their M1, M2, or M3 subreceptor profile seems to have little influence on their clinical efficacy. Several anticholinergics are available, but for most cases, clinical use is based on the results of open-label trials. Propantheline bromide is nonselective for muscarinic receptor subtypes and has low bioavailability. It was a first-choice agent for detrusor overactivity in the 1980s but has been largely supplanted by newer agents since. Trospium chloride is a very efficacious nonselective quaternary ammonium compound showing tissue selectivity for the bladder over the salivary glands. Oxybutynin has antimuscarinic, muscle relaxant, and local anesthetic actions. It has been demonstrated to have a higher affinity for muscarinic M1 and M3 receptors than for M2 receptor, but the clinical significance of this is unclear. Side effects are typically antimuscarinic, including dry mouth, constipation, blurred vision, and drowsiness. The administration of desmopressin at night may improve nocturia (see previous discussion). In patients with MSA and incomplete bladder emptying, clean intermittent catheterization three to four times per day is a widely accepted approach to prevent myogenic overdistension and secondary consequences from failure to micturate. In the advanced stages of MSA, a permanent urethral or transcutaneous suprapubic catheter may become necessary, when motor symptoms of MSA or mechanical obstruction in the urethra prevent uncomplicated catheterization. Pharmacological options with cholinergic or adrenergic substances are usually not successful to adequately reduce postvoid residual volume in MSA. However, α-adrenergic receptor antagonists (prazosin and moxisylyte) have been shown to improve voiding with reduction of residual volumes in patients with MSA.218 Urological surgery must be avoided in these patients because worsening of bladder control postoperatively is most likely.83
Erectile Dysfunction
The necessity of a specific treatment of sexual dysfunction needs to be evaluated individually in each MSA patient. Sildenafil is an orally active inhibitor of the type V cyclic guanosine monophosphate–specific phosphodiesterase (the predominant isoenzyme in the human corpus cavernosum) and has shown remarkable success in clinical trials.219–221 In fact, preliminary evidence in patients with Parkinson’s disease220 suggests that sildenafil citrate may also be successful in treating erectile failure in patients with MSA. A double-blind, placebo-controlled trial confirmed the efficacy of this compound in MSA but also suggested caution because of the frequent cardiovascular side effects.221 Erectile failure in MSA may also be improved by oral yohimbine, by intracavernosal injection of papaverine, or by a penile implant.83 Moreover, erectile dysfunction can be treated with intracavernosal injections or transurethral suppositories of alprostadil, a synthetic prostaglandin E1.222 Dopaminergic agents may also help with sexual dysfunction, probably by alleviating bradykinesia, as well as increasing desire. On high doses of antiparkinsonian medication, some patients may become hypersexual—even despite their inability to perform.54
Constipation
Constipation affects overall well-being and can be managed with dietary changes, adequate liquid intake, exercise, and pharmacotherapy. In fact, at least two meals per day should include high-fiber raw vegetables. Furthermore, increasing physical activity can also be helpful. Constipation can be relieved by increasing the intraluminal volume, which may be achieved with a macrogol-water solution. Indeed, it is already shown that macrogol 3350 plus electrolytes improves constipation in Parkinson’s disease and MSA.223 Stool softeners given with meals can be helpful. Lactulose may be beneficial for some patients. Bowel motility may be increased by discontinuing anticholinergic agents.
REM Sleep Behavior Disorder
In many patients, REM sleep behavior disorder is responsive to low-dose clonazepam. In patients with DLB, precipitation or aggravation of hallucinations with dopaminergic agents may occur. L-Dopa has less of a propensity to cause hallucinations and somnolence and is therefore preferred over dopamine agonists.54 Although clonazepam may be regarded as the treatment of choice for REM sleep behavior disorder, an alternative treatment is desirable for affected patients whose condition is refractory to clonazepam, who experience intolerable side effects with clonazepam, or in whom clonazepam precipitates or aggravates obstructive sleep apnea. In a series by Boeve and colleagues,224 a persistent benefit with melatonin beyond 1 year of therapy occurred in most patients, which suggests that melatonin may be considered as a possible sole or add-on therapy in selected patients with REM sleep behavior disorder.
Cognitive and Psychiatric Problems
In patients with DLB, cholinesterase inhibitor drugs are commonly used for the treatment of cognitive dysfunction. These drugs may reduce hallucinations and other neuropsychiatric symptoms of DLB, too. According to a Cochrane Database review,225 DLB patients who suffer from behavioral disturbances or psychiatric problems may benefit from rivastigmine if they tolerate it. However, the evidence is weak, and further trials with rivastigmine or other cholinesterase inhibitors in DLB are needed.54 Another important issue that has to be kept in mind is that about one third to one half of patients with DLB develop severe side effects when treated with typical or atypical neuroleptics.191,226,227 The reported side effects include increased rigidity, immobility, confusion, sedation, and postural falls.191,226–232 It is not possible to predict these neuroleptic sensitivity reactions in an individual patient before treatment starts. Severe neuroleptic sensitivity probably also occurs in about 25% of PDD patients, and caution in their use is also urged.233 Selective serotonin reuptake inhibitors have shown some effectiveness for the management of depression and for behavioral and psychological disorders in patients with dementia (particularly in Alzheimer’s disease).54
Palliative Care
Patients with PAF should be assured that their disease has a relatively benign nature. MSA patients continue to suffer from this malignant and distressing condition. Because the results of drug treatment for MSA are generally poor in the long term, other therapies are all the more important. Palliative management decisions should be based on careful clinical judgment, with the expectations of both patient and caregivers taken into account. It is most crucial that patients have maximum access to speech, occupational, and physical therapists; social workers; wheelchair clinics; and continence advisers. In fact, physiotherapy helps maintain mobility and prevent contractures. Speech therapy can improve speech and swallowing and provide communication aids. Dysphagia may necessitate feeding via a nasogastric tube or even percutaneous endoscopic gastrostomy. Continuous positive airway pressure may be helpful in some affected patients with inspiratory stridor.234,235 Tracheostomy, after all the ethical issues related to this procedure have been considered, is only rarely (about 4%) needed in mobile patients with inspiratory stridor; it should be avoided in preterminal stages of the disease. The advisability of either gastrostomy or tracheostomy should be approached on an individual basis with a realistic appraisal of the patient’s general quality of life. In advanced disease, long-term indwelling catheters become the mainstay of urinary control. Many factors, including neurological, urological, sexual, psychological, and social factors, may complicate or affect the choice of bladder management. Indeed, sometimes the preservation of quality of life for caretakers or lack of adequate nursing may dictate the use of indwelling catheters. Although urethral catheterization does provide effective bladder drainage, it is not an ideal long-term solution. Erosion of the urethra may occur in both men and women, whereas men are also prone to developing urethral stricture disease. Most urologists agree that a suprapubic catheter is the preferred route for long-term bladder drainage if an indwelling catheter is required. A reduction in maximal detrusor pressure, improvement in bladder structure, and a resolution of reflux have been reported. Complications common to both urethral and suprapubic catheterization include urine bypassing the catheter, leading to incontinence, and recurrent infections and catheter blockage. Expert uroneurological advice is invaluable. Occupational therapy helps to limit the handicap resulting from the patient’s disabilities and should include a home visit. Gait training and timely provision of a walking aide or assistive devices to prevent falling help avoid further debilitation of the patient. Provision of a wheelchair is usually dictated by the liability to falls because of postural instability and gait ataxia but not by akinesia and rigidity per se. Psychological support for patients and partners needs to be stressed. Until primary autonomic failure can be effectively treated or prevented, it will continue to present neurologists with a major challenge: that of providing the rapport, empathy, trust, and compassion necessary to support patients and family in the presence of progressive and incurable disease.
Kaufmann H, Schatz IJ. Pure autonomic failure. In: Robertson D, editor. Primer on the Autonomic Nervous System. 2nd ed. Amsterdam: Elsevier; 2004:309-311.
Mathias C, Bannister R, editors. Autonomic Failure: A Textbook of Clinical Disorders of the Autonomic Nervous System. Oxford, UK: Oxford University Press, 1999.
Mathias CJ, Polinsky RJ. Separating the primary autonomic failure syndromes, multiple system atrophy, and pure autonomic failure from Parkinson’s disease. Adv Neurol. 1999;80:353-361.
Wenning GK, Colosimo C, Geser F, et al. Multiple system atrophy. Lancet Neurol. 2004;3:203-293.
Wenning GK, Stampfer M. Dementia with Lewy Bodies. In: Robertson D, editor. Primer on the Autonomic Nervous System. 2nd ed. Amsterdam: Elsevier; 2004:293-294.
1 Mathias CJ. The classification and nomenclature of autonomic disorders—ending chaos, resolving conflict and hopefully achieving clarity. Clin Auton Res. 1995;5:307-310.
2 Bannister R, Mathias CJ, Polinsky R. Clinical features of autonomic failure. B. Autonomic failure: a comparison between UK and US experience. In: Bannister R, editor. Autonomic Failure. A Textbook of Disorders of the Autonomic Nervous System. 2nd ed. Oxford: Oxford University Press; 1988:281-288.
3 Kaufmann H, Hague K, Perl D. Accumulation of alpha-synuclein in autonomic nerves in pure autonomic failure. Neurology. 2001;56:980-981.
4 Kaufmann H. Primary autonomic failure: three clinical presentations of one disease? Ann Intern Med. 2000;133:382-384.
5 Polymeropoulos MH, Lavedan C, Leroy E, et al. Mutation in the alpha-synuclein gene identified in families with Parkinson’s disease. Science. 1997;276:2045-2047.
6 Stefanova N, Klimaschewski L, Poewe W, et al. Glial cell death induced by overexpression of alpha-synuclein. J Neurosci Res. 2001;65:432-438.
7 Tu PH, Galvin JE, Baba M, et al. Glial cytoplasmic inclusions in white matter oligodendrocytes of multiple system atrophy brains contain insoluble alpha-synuclein. Ann Neurol. 1998;44:415-422.
8 Kahle PJ, Neumann M, Ozmen L, et al. Hyperphosphorylation and insolubility of alpha-synuclein in transgenic mouse oligodendrocytes. EMBO Rep. 2002;3:583-588.
9 Hague K, Lento P, Morgello S, et al. The distribution of Lewy bodies in pure autonomic failure: autopsy findings and review of the literature. Acta Neuropathol (Berl). 1997;94:192-196.
10 van Ingelghem E, van Zandijcke M, Lammens M. Pure autonomic failure: a new case with clinical, biochemical, and necropsy data. J Neurol Neurosurg Psychiatry. 1994;57:745-747.
11 Hughes AJ, Daniel SE, Blankson S, et al. A clinicopathologic study of 100 cases of Parkinson’s disease. Arch Neurol. 1993;50:140-148.
12 McKeith IG, Galasko D, Kosaka K, et al. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology. 1996;47:1113-1124.
13 Oppenheimer DR. Lateral horn cells in progressive autonomic failure. J Neurol Sci. 1980;46:393-404.
14 Graham JG, Oppenheimer DR. Orthostatic hypotension and nicotine sensitivity in a case of multiple system atrophy. J Neurol Neurosurg Psychiatry. 1969;32:28-34.
15 van der Eecken H, Adams R, van Bogaert L. Striopallidalnigral degeneration: a hitherto undescribed lesion in paralysis agitans. J Neuropathol Exp Neurol. 1960;19:159-161.
16 Adams R, Van Bogaert L, Van der Eecken H. [Nigro-striate and cerebello-nigro-striate degeneration. Clinical uniqueness and pathological variability of presenile degeneration of the extrapyramidal rigidity type]. Psychiatr Neurol (Basel). 1961;142:219-259.
17 Adams RD, Vanbogaert L, Van der Eecken H. Striatonigral degeneration. J Neuropathol Exp Neurol. 1964;23:584-608.
18 Dejerine J, Thomas A. L’atrophie olivo-ponto-cerebelleuse. Nouv Iconogr Salpetr. 1900;13:330-370.
19 Stauffenberg W. Zur Kenntnis des extrapyramidalen motorischen Systems und Mitteilung eines Falles von sog. Atrophie olivo-pontocerbelleuse. Zeitschr Ges Neurol Psychiatrie. 1918;39:1-55.
20 Shy GM, Drager GA. A neurological syndrome associated with orthostatic hypotension: a clinical-pathologic study. Arch Neurol. 1960;2:511-527.
21 Papp MI, Lantos PL. The distribution of oligodendroglial inclusions in multiple system atrophy and its relevance to clinical symptomatology. Brain. 1994;117(Pt 2):235-243.
22 Geser F, Stampfer-Kountchev M, Seppi K, et al. The clinical presentation of multiple system atrophy (MSA) in Europe: An interim analysis of the EMSA-SG (European MSA-Study Group) Registry. Mov Disord. 2004;19(Suppl 9):347.
23 Gilman S, Low P, Quinn N, et al. Consensus statement on the diagnosis of multiple system atrophy. American Autonomic Society and American Academy of Neurology. Clin Auton Res. 1998;8:359-362.
24 Kume A, Takahashi A, Hashizume Y. Neuronal cell loss of the striatonigral system in multiple system atrophy. J Neurol Sci. 1993;117:33-40.
25 Wenning GK, Tison F, Elliott L, et al. Olivopontocerebellar pathology in multiple system atrophy. Mov Disord. 1996;11:157-162.
26 Sung JH, Mastri AR, Segal E. Pathology of Shy-Drager syndrome. J Neuropathol Exp Neurol. 1979;38:353-368.
27 Benarroch EE, Smithson IL, Low PA, et al. Depletion of catecholaminergic neurons of the rostral ventrolateral medulla in multiple systems atrophy with autonomic failure. Ann Neurol. 1998;43:156-163.
28 Benarroch EE, Schmeichel AM, Parisi JE. Depletion of cholinergic neurons of the medullary arcuate nucleus in multiple system atrophy. Auton Neurosci. 2001;87:293-299.
29 Benarroch EE, Schmeichel AM, Parisi JE. Depletion of mesopontine cholinergic and sparing of raphe neurons in multiple system atrophy. Neurology. 2002;59:944-946.
30 Benarroch EE. Brainstem in multiple system atrophy: clinicopathological correlations. Cell Mol Neurobiol. 2003;23:519-526.
31 Nakamura S, Ohnishi K, Nishimura M, et al. Large neurons in the tuberomammillary nucleus in patients with Parkinson’s disease and multiple system atrophy. Neurology. 1996;46:1693-1696.
32 Wenning GK, Tison F, Ben Shlomo Y, et al. Multiple system atrophy: a review of 203 pathologically proven cases. Mov Disord. 1997;12:133-147.
33 Daniel S. The neuropathology and neurochemistry of multiple system atrophy. In: Mathias C, Bannister R, editors. Autonomic Failure: A Textbook of Clinical Disorders of the Autonomic Nervous System. 4th ed. Oxford, UK: Oxford University Press; 1999:321-328.
34 Kaufmann H, Oribe E, Miller M, et al. Hypotension-induced vasopressin release distinguishes between pure autonomic failure and multiple system atrophy with autonomic failure. Neurology. 1992;42:590-593.
35 Konno H, Yamamoto T, Iwasaki Y, et al. Shy-Drager syndrome and amyotrophic lateral sclerosis: cytoarchitectonic and morphometric studies of sacral autonomic neurons. J Neurol Sci. 1986;73:193-204.
36 Bannister R, Oppenheimer DR. Degenerative diseases of the nervous system associated with autonomic failure. Brain. 1972;95:457-474.
37 Spokes EG, Bannister R, Oppenheimer DR. Multiple system atrophy with autonomic failure: clinical, histological and neurochemical observations on four cases. J Neurol Sci. 1979;43:59-82.
38 Matthews M. Autonomic ganglia and preganglionic neurons in autonomic failure. In: Mathias C, Bannister R, editors. Autonomic Failure: A Textbook of Clinical Disorders of the Autonomic Nervous System. 4th ed. Oxford, UK: Oxford University Press; 1999:329-339.
39 Sima AA, Caplan M, D’Amato CJ, et al. Fulminant multiple system atrophy in a young adult presenting as motor neuron disease. Neurology. 1993;43:2031-2035.
40 Low PA. Autonomic neuropathies. Curr Opin Neurol. 1994;7:402-406.
41 McLeod JG. Autonomic dysfunction in peripheral nerve disease. In: Mathias C, Bannister R, editors. Autonomic Failure: A Textbook of Clinical Disorders of the Autonomic Nervous System. 4th ed. Oxford, UK: Oxford University Press; 1999:367-377.
42 Suarez GA, Fealey RD, Camilleri M, et al. Idiopathic autonomic neuropathy: clinical, neurophysiologic, and follow-up studies on 27 patients. Neurology. 1994;44:1675-1682.
43 Bennett JL, Mahalingam R, Wellish MC, et al. Epstein-Barr virus—associated acute autonomic neuropathy. Ann Neurol. 1996;40:453-455.
44 Santiago S, Ferrer T, Espinosa ML. Neurophysiological studies of thin myelinated (A delta) and unmyelinated (C) fibers: application to peripheral neuropathies. Neurophysiol Clin. 2000;30:27-42.
45 Santiago S, Espinosa ML, Perez-Conde MC, et al. Small fiber dysfunction in peripheral neuropathies. Rev Neurol. 1999;28:543-554.
46 Bradbury S, Eggleston C. Postural hypotension: a report of three cases. Am Heart J. 1925;1:75-86.
47 Kaufmann H. Consensus statement on the definition of orthostatic hypotension, pure autonomic failure and multiple system atrophy. Clin Auton Res. 1996;6:125-126.
48 Mathias CJ. Orthostatic hypotension: causes, mechanisms, and influencing factors. Neurology. 1995;45:S6-S11.
49 Robertson D, Davis TL. Recent advances in the treatment of orthostatic hypotension. Neurology. 1995;45:S26-S32.
50 Mathias CJ, Mallipeddi R, Bleasdale-Barr K. Symptoms associated with orthostatic hypotension in pure autonomic failure and multiple system atrophy. J Neurol. 1999;246:893-898.
51 Schatz IJ. Orthostatic hypotension. I. Functional and neurogenic causes. Arch Intern Med. 1984;144:773-777.
52 Geser F, Stampfer-Kountchev M, Seppi K, et al. Warning signs (“red flags”) in multiple system atrophy (MSA): a preliminary cross-sectional analysis of 79 European MSA-P patients. Mov Disord. 2004;19(Suppl 9):346-347.
53 Geser F, Stampfer-Kountchev M, Seppi K, et al. Warning signs (“red flags”) in possible MSA: a preliminary cross-sectional analysis of the European MSA-Study Group (EMSA-SG). Funct Neurol. 2004;19:144.
54 Kaufmann H, Biaggioni I. Autonomic failure in neurodegenerative disorders. Semin Neurol. 2003;23:351-363.
55 Mathias CJ, Polinsky RJ. Separating the primary autonomic failure syndromes, multiple system atrophy, and pure autonomic failure from Parkinson’s disease. Adv Neurol. 1999;80:353-361.
56 Churchyard A, Mathias CJ, Boonkongchuen P, et al. Autonomic effects of selegiline: possible cardiovascular toxicity in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 1997;63:228-234.
57 Magalhaes M, Wenning GK, Daniel SE, et al. Autonomic dysfunction in pathologically confirmed multiple system atrophy and idiopathic Parkinson’s disease: a retrospective comparison. Acta Neurol Scand. 1995;91:98-102.
58 Kaufmann H, Nouri S, Olanow W, et al. Orthostatic intolerance in Parkinson’s disease. Neurology. 1997;48:149.
59 Senard JM, Rai S, Lapeyre-Mestre M, et al. Prevalence of orthostatic hypotension in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 1997;63:584-589.
60 Wenning GK, Scherfler C, Granata R, et al. Time course of symptomatic orthostatic hypotension and urinary incontinence in patients with postmortem confirmed parkinsonian syndromes: a clinicopathological study. J Neurol Neurosurg Psychiatry. 1999;67:620-623.
61 Orskov L, Jakobsen J, Dupont E, et al. Autonomic function in parkinsonian patients relates to duration of disease. Neurology. 1987;37:1173-1178.
62 Mayeux R, Denaro J, Hemenegildo N, et al. A population-based investigation of Parkinson’s disease with and without dementia: relationship to age and gender. Arch Neurol. 1992;49:492-497.
63 Aarsland D, Larsen JP, Karlsen K, et al. Mental symptoms in Parkinson’s disease are important contributors to caregiver distress. Int J Geriatr Psychiatry. 1999;14:866-874.
64 Peralta CM, Stampfer M, Karner E, et al. Orthostatic hypotension and attention in Lewy body disorders. Mov Disord. 2004;19(Suppl 9):331-332.
65 Geser F, Wenning GK, Poewe W, et al. How to diagnose dementia with Lewy bodies: state of the art. Mov Disord. 2005;20(Suppl 12):S11-S20.
66 McKeith IG, Perry EK, Perry RH. Report of the second dementia with Lewy body international workshop: diagnosis and treatment. Consortium on Dementia with Lewy Bodies. Neurology. 1999;53:902-905.
67 Horimoto Y, Matsumoto M, Akatsu H, et al. Autonomic dysfunctions in dementia with Lewy bodies. J Neurol. 2003;250:530-533.
68 Ransmayr G. Dementia with Lewy bodies: prevalence, clinical spectrum and natural history. J Neural Transm Suppl. 2000;60:303-314.
69 Del Ser T, McKeith I, Anand R, et al. Dementia with Lewy bodies: findings from an international multicentre study. Int J Geriatr Psychiatry. 2000;15:1034-1045.
70 Aarsland D, Ballard C, McKeith I, et al. Comparison of extrapyramidal signs in dementia with Lewy bodies and Parkinson’s disease. J Neuropsychiatry Clin Neurosci. 2001;13:374-379.
71 Pakiam AS, Bergeron C, Lang AE. Diffuse Lewy body disease presenting as multiple system atrophy. Can J Neurol Sci. 1999;26:127-131.
72 McKeith I, Burn D. Spectrum of Parkinson’s disease, Parkinson’s dementia, and Lewy body dementia. In: DeKosky S, editor. Neurologic Clinics: Dementia. Philadelphia: WB Saunders; 2000:865-883.
73 Wenning GK, Seppi K, Jellinger K, et al. Survival of patients with dementia with Lewy bodies: a meta-analysis of 236 postmortem confirmed cases. Neurology. 2000;54(Suppl 3):391-392.
74 Watanabe H, Saito Y, Terao S, et al. Progression and prognosis in multiple system atrophy: an analysis of 230 Japanese patients. Brain. 2002;125:1070-1083.
75 Testa D, Monza D, Ferrarini M, et al. Comparison of natural histories of progressive supranuclear palsy and multiple system atrophy. Neurol Sci. 2001;22:247-251.
76 Ben-Shlomo Y, Wenning GK, Tison F, et al. Survival of patients with pathologically proven multiple system atrophy: a meta-analysis. Neurology. 1997;48:384-393.
77 Wenning GK, Ben Shlomo Y, Magalhaes M, et al. Clinical features and natural history of multiple system atrophy: an analysis of 100 cases. Brain. 1994;117(Pt 4):835-845.
78 Abele M, Burk K, Schols L, et al. The aetiology of sporadic adult-onset ataxia. Brain. 2002;125:961-968.
79 Gilman S, Little R, Johanns J, et al. Evolution of sporadic olivopontocerebellar atrophy into multiple system atrophy. Neurology. 2000;55:527-532.
80 Schwarz J, Tatsch K, Gasser T, et al. 123I-IBZM binding compared with long-term clinical follow up in patients with de novo parkinsonism. Mov Disord. 1998;13:16-19.
81 Boesch SM, Wenning GK, Ransmayr G, et al. Dystonia in multiple system atrophy. J Neurol Neurosurg Psychiatry. 2002;72:300-303.
82 Wenning GK, Ben-Shlomo Y, Hughes A, et al. What clinical features are most useful to distinguish definite multiple system atrophy from Parkinson’s disease? J Neurol Neurosurg Psychiatry. 2000;68:434-440.
83 Beck RO, Betts CD, Fowler CJ. Genitourinary dysfunction in multiple system atrophy: clinical features and treatment in 62 cases. J Urol. 1994;151:1336-1341.
84 Braune S, Elam M, Baron R, et al. Assessment of blood pressure regulation. The International Federation of Clinical Neurophysiology. Electroencephalogr Clin Neurophysiol Suppl. 1999;52:287-291.
85 Bannister R, Mathias C. Investigation of autonomic disorders. In: Mathias C, Bannister R, editors. Autonomic Failure: A Textbook of Clinical Disorders of the Autonomic Nervous System. 4th ed. Oxford, UK: Oxford University Press; 1999:169-195.
86 Braune S, Auer A, Schulte-Monting J, et al. Cardiovascular parameters: sensitivity to detect autonomic dysfunction and influence of age and sex in normal subjects. Clin Auton Res. 1996;6:3-15.
87 Braune S, Schulte-Monting J, Schwerbrock S, et al. Retest variation of cardiovascular parameters in autonomic testing. J Auton Nerv Syst. 1996;60:103-107.
88 Riley DE, Chelimsky TC. Autonomic nervous system testing may not distinguish multiple system atrophy from Parkinson’s disease. J Neurol Neurosurg Psychiatry. 2003;74:56-60.
89 Ziegler MG, Lake CR, Kopin IJ. The sympathetic-nervous-system defect in primary orthostatic hypotension. N Engl J Med. 1977;296:293-297.
90 Polinsky RJ, Kopin IJ, Ebert MH, et al. Pharmacologic distinction of different orthostatic hypotension syndromes. Neurology. 1981;31:1-7.
91 Goldstein DS, Polinsky RJ, Garty M, et al. Patterns of plasma levels of catechols in neurogenic orthostatic hypotension. Ann Neurol. 1989;26:558-563.
92 Kaufmann H, Schatz IJ. Pure autonomic failure. In: Robertson D, editor. Primer on the Autonomic Nervous System. Amsterdam: Elsevier; 2004:309-311.
93 Baser SM, Brown RT, Curras MT, et al. Beta-receptor sensitivity in autonomic failure. Neurology. 1991;41:1107-1112.
94 Goldstein DS, Holmes C, Cannon RO3rd, et al. Sympathetic cardioneuropathy in dysautonomias. N Engl J Med. 1997;336:696-702.
95 Braune S, Reinhardt M, Schnitzer R, et al. Cardiac uptake of [123I]MIBG separates Parkinson’s disease from multiple system atrophy. Neurology. 1999;53:1020-1025.
96 Braune S. The role of cardiac metaiodobenzylguanidine uptake in the differential diagnosis of parkinsonian syndromes. Clin Auton Res. 2001;11:351-355.
97 Orimo S, Ozawa E, Nakade S, et al. 123I-metaiodobenzylguanidine myocardial scintigraphy in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 1999;67:189-194.
98 Takatsu H, Nagashima K, Murase M, et al. Differentiating Parkinson disease from multiple-system atrophy by measuring cardiac iodine-123 metaiodobenzylguanidine accumulation. JAMA. 2000;284:44-45.
99 Taki J, Nakajima K, Hwang EH, et al. Peripheral sympathetic dysfunction in patients with Parkinson’s disease without autonomic failure is heart selective and disease specific. Eur J Nucl Med. 2000;27:566-573.
100 Hirayama M, Hakusui S, Koike Y, et al. A scintigraphical qualitative analysis of peripheral vascular sympathetic function with meta-[123I]iodobenzylguanidine in neurological patients with autonomic failure. J Auton Nerv Syst. 1995;53:230-234.
101 Yoshita M. Differentiation of idiopathic Parkinson’s disease from striatonigral degeneration and progressive supranuclear palsy using iodine-123 metaiodobenzylguanidine myocardial scintigraphy. J Neurol Sci. 1998;155:60-67.
102 Goldstein DS, Holmes C, Li ST, et al. Cardiac sympathetic denervation in Parkinson’s disease. Ann Intern Med. 2000;133:338-347.
103 Watanabe H, Ieda T, Katayama T, et al. Cardiac 123I-metaiodobenzylguanidine (MIBG) uptake in dementia with Lewy bodies: comparison with Alzheimer’s disease. J Neurol Neurosurg Psychiatry. 2001;70:781-783.
104 Yoshita M, Taki J, Yamada M. A clinical role for [123I]MIBG myocardial scintigraphy in the distinction between dementia of the Alzheimer’s-type and dementia with Lewy bodies. J Neurol Neurosurg Psychiatry. 2001;71:583-588.
105 Oide T, Tokuda T, Momose M, et al. Usefulness of [123I]metaiodobenzylguanidine ([123I]MIBG) myocardial scintigraphy in differentiating between Alzheimer’s disease and dementia with Lewy bodies. Intern Med. 2003;42:686-690.
106 Kirby R, Fowler C, Gosling J, et al. Urethro-vesical dysfunction in progressive autonomic failure with multiple system atrophy. J Neurol Neurosurg Psychiatry. 1986;49:554-562.
107 Pramstaller PP, Wenning GK, Smith SJ, et al. Nerve conduction studies, skeletal muscle EMG, and sphincter EMG in multiple system atrophy. J Neurol Neurosurg Psychiatry. 1995;58:618-621.
108 Palace J, Chandiramani VA, Fowler CJ. Value of sphincter electromyography in the diagnosis of multiple system atrophy. Muscle Nerve. 1997;20:1396-1403.
109 Schwarz J, Kornhuber M, Bischoff C, et al. Electromyography of the external anal sphincter in patients with Parkinson’s disease and multiple system atrophy: frequency of abnormal spontaneous activity and polyphasic motor unit potentials. Muscle Nerve. 1997;20:1167-1172.
110 Valldeoriola F, Valls-Sole J, Tolosa ES, et al. Striated anal sphincter denervation in patients with progressive supranuclear palsy. Mov Disord. 1995;10:550-555.
111 Giladi N, Simon ES, Korczyn AD, et al. Anal sphincter EMG does not distinguish between multiple system atrophy and Parkinson’s disease. Muscle Nerve. 2000;23:731-734.
112 Colosimo C, Inghilleri M, Chaudhuri KR. Parkinson’s disease misdiagnosed as multiple system atrophy by sphincter electromyography. J Neurol. 2000;247:559-561.
113 Vodusek DB. Sphincter EMG and differential diagnosis of multiple system atrophy. Mov Disord. 2001;16:600-607.
114 Polinsky RJ, Brown RT, Lee GK, et al. Beta-endorphin, ACTH, and catecholamine responses in chronic autonomic failure. Ann Neurol. 1987;21:573-577.
115 Kimber JR, Watson L, Mathias CJ. Distinction of idiopathic Parkinson’s disease from multiple-system atrophy by stimulation of growth-hormone release with clonidine. Lancet. 1997;349:1877-1881.
116 Zoukos Y, Thomaides T, Pavitt DV, et al. Beta-adrenoceptor expression on circulating mononuclear cells of idiopathic Parkinson’s disease and autonomic failure patients before and after reduction of central sympathetic outflow by clonidine. Neurology. 1993;43:1181-1187.
117 Clarke CE, Ray PS, Speller JM. Failure of the clonidine growth hormone stimulation test to differentiate multiple system atrophy from early or advanced idiopathic Parkinson’s disease. Lancet. 1999;353:1329-1330.
118 Tranchant C, Guiraud-Chaumeil C, Echaniz-Laguna A, et al. Is clonidine growth hormone stimulation a good test to differentiate multiple system atrophy from idiopathic Parkinson’s disease? J Neurol. 2000;247:853-856.
119 Strijks E, van’t Hof M, Sweep F, et al. Stimulation of growth-hormone release with clonidine does not distinguish individual cases of idiopathic Parkinson’s disease from those with striatonigral degeneration. J Neurol. 2002;249:1206-1210.
120 Kimber J, Mathias CJ, Lees AJ, et al. Physiological, pharmacological and neurohormonal assessment of autonomic function in progressive supranuclear palsy. Brain. 2000;123(Pt 7):1422-1430.
121 Pellecchia MT, Salvatore E, Pivonello R, et al. Stimulation of growth hormone release in multiple system atrophy, Parkinson’s disease and idiopathic cerebellar ataxia. Neurol Sci. 2001;22:79-80.
122 Kimber J, Watson L, Mathias CJ. Abnormal suppression of arginine-vasopressin by clonidine in multiple system atrophy. Clin Auton Res. 1999;9:271-274.
123 Kimber J, Watson L, Mathias CJ. Neuroendocrine responses to levodopa in multiple system atrophy (MSA). Mov Disord. 1999;14:981-987.
124 Shannon J, Jordan J, Costa F, et al. The hypertension of autonomic failure and its treatment. Hypertension. 1997;30:1062-1067.
125 Shannon JR, Jordan J, Diedrich A, et al. Sympathetically mediated hypertension in autonomic failure. Circulation. 2000;101:2710-2715.
126 Wullner U, Klockgether T, Petersen D, et al. Magnetic resonance imaging in hereditary and idiopathic ataxia. Neurology. 1993;43:318-325.
127 Schrag A, Good CD, Miszkiel K, et al. Differentiation of atypical parkinsonian syndromes with routine MRI. Neurology. 2000;54:697-702.
128 Drayer BP, Olanow W, Burger P, et al. Parkinson plus syndrome: diagnosis using high field MR imaging of brain iron. Radiology. 1986;159:493-498.
129 Pastakia B, Polinsky R, Di Chiro G, et al. Multiple system atrophy (Shy-Drager syndrome): MR imaging. Radiology. 1986;159:499-502.
130 O’Brien C, Sung JH, McGeachie RE, et al. Striatonigral degeneration: clinical, MRI, and pathologic correlation. Neurology. 1990;40:710-711.
131 Lang AE, Curran T, Provias J, et al. Striatonigral degeneration: iron deposition in putamen correlates with the slitlike void signal of magnetic resonance imaging. Can J Neurol Sci. 1994;21:311-318.
132 Schwarz J, Weis S, Kraft E, et al. Signal changes on MRI and increases in reactive microgliosis, astrogliosis, and iron in the putamen of two patients with multiple system atrophy. J Neurol Neurosurg Psychiatry. 1996;60:98-101.
133 Schrag A, Kingsley D, Phatouros C, et al. Clinical usefulness of magnetic resonance imaging in multiple system atrophy. J Neurol Neurosurg Psychiatry. 1998;65:65-71.
134 Kraft E, Trenkwalder C, Auer DP. T2*-weighted MRI differentiates multiple system atrophy from Parkinson’s disease. Neurology. 2002;59:1265-1267.
135 Konagaya M, Konagaya Y, Iida M. Clinical and magnetic resonance imaging study of extrapyramidal symptoms in multiple system atrophy. J Neurol Neurosurg Psychiatry. 1994;57:1528-1531.
136 Kraft E, Schwarz J, Trenkwalder C, et al. The combination of hypointense and hyperintense signal changes on T2-weighted magnetic resonance imaging sequences: a specific marker of multiple system atrophy? Arch Neurol. 1999;56:225-228.
137 Schocke MF, Seppi K, Esterhammer R, et al. Diffusion-weighted MRI differentiates the Parkinson variant of multiple system atrophy from PD. Neurology. 2002;58:575-580.
138 Seppi K, Schocke MF, Esterhammer R, et al. Diffusion-weighted imaging discriminates progressive supranuclear palsy from PD, but not from the parkinson variant of multiple system atrophy. Neurology. 2003;60:922-927.
139 Schulz JB, Skalej M, Wedekind D, et al. Magnetic resonance imaging-based volumetry differentiates idiopathic Parkinson’s syndrome from multiple system atrophy and progressive supranuclear palsy. Ann Neurol. 1999;45:65-74.
140 Brenneis C, Seppi K, Schocke MF, et al. Voxel-based morphometry detects cortical atrophy in the Parkinson variant of multiple system atrophy. Mov Disord. 2003;18:1132-1138.
141 Barber R, Ballard C, McKeith IG, et al. MRI volumetric study of dementia with Lewy bodies: a comparison with AD and vascular dementia. Neurology. 2000;54:1304-1309.
142 Barber R, Scheltens P, Gholkar A, et al. White matter lesions on magnetic resonance imaging in dementia with Lewy bodies, Alzheimer’s disease, vascular dementia, and normal aging. J Neurol Neurosurg Psychiatry. 1999;67:66-72.
143 Cousins DA, Burton EJ, Burn D, et al. Atrophy of the putamen in dementia with Lewy bodies but not Alzheimer’s disease: an MRI study. Neurology. 2003;61:1191-1195.
144 O’Brien JT, Paling S, Barber R, et al. Progressive brain atrophy on serial MRI in dementia with Lewy bodies, AD, and vascular dementia. Neurology. 2001;56:1386-1388.
145 Cordato NJ, Halliday GM, Harding AJ, et al. Regional brain atrophy in progressive supranuclear palsy and Lewy body disease. Ann Neurol. 2000;47:718-728.
146 Brenneis C, Wenning GK, Egger KE, et al. Basal forebrain atrophy is a distinctive pattern in dementia with Lewy bodies. Neuroreport. 2004;15:1711-1714.
147 Brooks DJ, Ibanez V, Sawle GV, et al. Differing patterns of striatal 18F-dopa uptake in Parkinson’s disease, multiple system atrophy, and progressive supranuclear palsy. Ann Neurol. 1990;28:547-555.
148 Brooks DJ, Salmon EP, Mathias CJ, et al. The relationship between locomotor disability, autonomic dysfunction, and the integrity of the striatal dopaminergic system in patients with multiple system atrophy, pure autonomic failure, and Parkinson’s disease, studied with PET. Brain. 1990;113(Pt 5):1539-1552.
149 Burn DJ, Sawle GV, Brooks DJ. Differential diagnosis of Parkinson’s disease, multiple system atrophy, and Steele-Richardson-Olszewski syndrome: discriminant analysis of striatal 18F-dopa PET data. J Neurol Neurosurg Psychiatry. 1994;57:278-284.
150 Brooks DJ, Ibanez V, Sawle GV, et al. Striatal D2 receptor status in patients with Parkinson’s disease, striatonigral degeneration, and progressive supranuclear palsy, measured with 11C-raclopride and positron emission tomography. Ann Neurol. 1992;31:184-192.
151 Burn DJ, Rinne JO, Quinn NP, et al. Striatal opioid receptor binding in Parkinson’s disease, striatonigral degeneration and Steele-Richardson-Olszewski syndrome, a [11C]diprenorphine PET study. Brain. 1995;118(Pt 4):951-958.
152 De Volder AG, Francart J, Laterre C, et al. Decreased glucose utilization in the striatum and frontal lobe in probable striatonigral degeneration. Ann Neurol. 1989;26:239-247.
153 Perani D, Bressi S, Testa D, et al. Clinical/metabolic correlations in multiple system atrophy: a fluorodeoxyglucose F 18 positron emission tomographic study. Arch Neurol. 1995;52:179-185.
154 Antonini A, Kazumata K, Feigin A, et al. Differential diagnosis of parkinsonism with [18F]fluorodeoxyglucose and PET. Mov Disord. 1998;13:268-274.
155 Ghaemi M, Hilker R, Rudolf J, et al. Differentiating multiple system atrophy from Parkinson’s disease: contribution of striatal and midbrain MRI volumetry and multi-tracer PET imaging. J Neurol Neurosurg Psychiatry. 2002;73:517-523.
156 Gilman S, Koeppe RA, Junck L, et al. Patterns of cerebral glucose metabolism detected with positron emission tomography differ in multiple system atrophy and olivopontocerebellar atrophy. Ann Neurol. 1994;36:166-175.
157 Hu XS, Okamura N, Arai H, et al. 18F-fluorodopa PET study of striatal dopamine uptake in the diagnosis of dementia with Lewy bodies. Neurology. 2000;55:1575-1577.
158 Higuchi M, Tashiro M, Arai H, et al. Glucose hypometabolism and neuropathological correlates in brains of dementia with Lewy bodies. Exp Neurol. 2000;162:247-256.
159 Ishii K, Imamura T, Sasaki M, et al. Regional cerebral glucose metabolism in dementia with Lewy bodies and Alzheimer’s disease. Neurology. 1998;51:125-130.
160 Brucke T, Djamshidian S, Bencsits G, et al. SPECT and PET imaging of the dopaminergic system in Parkinson’s disease. J Neurol. 2000;247(Suppl 4):IV/2-IV/7.
161 Kim YJ, Ichise M, Ballinger JR, et al. Combination of dopamine transporter and D2 receptor SPECT in the diagnostic evaluation of PD, MSA, and PSP. Mov Disord. 2002;17:303-312.
162 Varrone A, Marek KL, Jennings D, et al. [123I]Beta-CIT SPECT imaging demonstrates reduced density of striatal dopamine transporters in Parkinson’s disease and multiple system atrophy. Mov Disord. 2001;16:1023-1032.
163 Schulz JB, Klockgether T, Petersen D, et al. Multiple system atrophy: natural history, MRI morphology, and dopamine receptor imaging with 123IBZM-SPECT. J Neurol Neurosurg Psychiatry. 1994;57:1047-1056.
164 van Royen E, Verhoeff NF, Speelman JD, et al. Multiple system atrophy and progressive supranuclear palsy: diminished striatal D2 dopamine receptor activity demonstrated by 123I-IBZM single photon emission computed tomography. Arch Neurol. 1993;50:513-516.
165 Brucke T, Wenger S, Asenbaum S, et al. Dopamine D2 receptor imaging and measurement with SPECT. Adv Neurol. 1993;60:494-500.
166 Schwarz J, Tatsch K, Arnold G, et al. 123I-iodobenzamide-SPECT predicts dopaminergic responsiveness in patients with de novo parkinsonism. Neurology. 1992;42:556-561.
167 Schwarz J, Tatsch K, Arnold G, et al. 123I-iodobenzamide-SPECT in 83 patients with de novo parkinsonism. Neurology. 1993;43:S17-S20.
168 Donnemiller E, Heilmann J, Wenning GK, et al. Brain perfusion scintigraphy with 99mTc-HMPAO or 99mTc-ECD and 123I-beta-CIT single-photon emission tomography in dementia of the Alzheimer-type and diffuse Lewy body disease. Eur J Nucl Med. 1997;24:320-325.
169 Lobotesis K, Fenwick JD, Phipps A, et al. Occipital hypoperfusion on SPECT in dementia with Lewy bodies but not AD. Neurology. 2001;56:643-649.
170 Ransmayr G, Seppi K, Donnemiller E, et al. Striatal dopamine transporter function in dementia with Lewy bodies and Parkinson’s disease. Eur J Nucl Med. 2001;28:1523-1528.
171 Walker Z, Costa DC, Ince P, et al. In-vivo demonstration of dopaminergic degeneration in dementia with Lewy bodies. Lancet. 1999;354:646-647.
172 Walker Z, Costa DC, Walker RW, et al. Differentiation of dementia with Lewy bodies from Alzheimer’s disease using a dopaminergic presynaptic ligand. J Neurol Neurosurg Psychiatry. 2002;73:134-140.
173 Heafield MT, Gammage MD, Nightingale S, et al. Idiopathic dysautonomia treated with intravenous gammaglobulin. Lancet. 1996;347:28-29.
174 Smit AA, Vermeulen M, Koelman JH, et al. Unusual recovery from acute panautonomic neuropathy after immunoglobulin therapy. Mayo Clin Proc. 1997;72:333-335.
175 Ramirez C, de Seze J, Stojkovic T, et al. Pure subacute pandysautonomia: an assessment of treatment with intravenous polyvalent immunoglobulins. Rev Neurol (Paris). 2004;160:939-941.
176 Goetz CG, Tanner CM, Klawans HL. The pharmacology of olivopontocerebellar atrophy. Adv Neurol. 1984;41:143-148.
177 Hughes AJ, Lees AJ, Stern GM. Apomorphine test to predict dopaminergic responsiveness in parkinsonian syndromes. Lancet. 1990;336:32-34.
178 Staal A, Meerwaldt JD, van Dongen KJ, et al. Non-familial degenerative disease and atrophy of brainstem and cerebellum: clinical and CT data in 47 patients. J Neurol Sci. 1990;95:259-269.
179 Hughes AJ, Colosimo C, Kleedorfer B, et al. The dopaminergic response in multiple system atrophy. J Neurol Neurosurg Psychiatry. 1992;55:1009-1013.
180 Parati EA, Fetoni V, Geminiani GC, et al. Response to L-DOPA in multiple system atrophy. Clin Neuropharmacol. 1993;16:139-144.
181 Albanese A, Colosimo C, Bentivoglio AR, et al. Multiple system atrophy presenting as parkinsonism: clinical features and diagnostic criteria. J Neurol Neurosurg Psychiatry. 1995;59:144-151.
182 Gouider-Khouja N, Vidailhet M, Bonnet AM, et al. “Pure” striatonigral degeneration and Parkinson’s disease: a comparative clinical study. Mov Disord. 1995;10:288-294.
183 Fetoni V, Soliveri P, Monza D, et al. Affective symptoms in multiple system atrophy and Parkinson’s disease: response to levodopa therapy. J Neurol Neurosurg Psychiatry. 1999;66:541-544.
184 Rossi P, Colosimo C, Moro E, et al. Acute challenge with apomorphine and levodopa in Parkinsonism. Eur Neurol. 2000;43:95-101.
185 Fearnley JM, Lees AJ. Striatonigral degeneration: a clinicopathological study. Brain. 1990;113(Pt 6):1823-1842.
186 Rajput AH, Rozdilsky B, Rajput A, et al. Levodopa efficacy and pathological basis of Parkinson syndrome. Clin Neuropharmacol. 1990;13:553-558.
187 Colosimo C, Albanese A, Hughes AJ, et al. Some specific clinical features differentiate multiple system atrophy (striatonigral variety) from Parkinson’s disease. Arch Neurol. 1995;52:294-298.
188 Wenning GK, Ben-Shlomo Y, Magalhaes M, et al. Clinicopathological study of 35 cases of multiple system atrophy. J Neurol Neurosurg Psychiatry. 1995;58:160-166.
189 Quinn N. Multiple system atrophy. In: Marsden CD, Fahn S, editors. Movement Disorders 3. London: Butterworth-Heinemann; 1994:262-281.
190 Duda JE, Giasson BI, Mabon ME, et al. Novel antibodies to synuclein show abundant striatal pathology in Lewy body diseases. Ann Neurol. 2002;52:205-210.
191 Gnanalingham KK, Byrne EJ, Thornton A, et al. Motor and cognitive function in Lewy body dementia: comparison with Alzheimer’s and Parkinson’s diseases. J Neurol Neurosurg Psychiatry. 1997;62:243-252.
192 Louis ED, Goldman JE, Powers JM, et al. Parkinsonian features of eight pathologically diagnosed cases of diffuse Lewy body disease. Mov Disord. 1995;10:188-194.
193 Louis ED, Klatka LA, Liu Y, et al. Comparison of extrapyramidal features in 31 pathologically confirmed cases of diffuse Lewy body disease and 34 pathologically confirmed cases of Parkinson’s disease. Neurology. 1997;48:376-380.
194 Zesiewicz TA, Baker MJ, Dunne PB, et al. Diffuse Lewy body disease. Curr Treat Options Neurol. 2001;3:507-518.
195 Poewe W, Granata R, Geser F. Pharmacologic treatment of Parkinson’s disease. In: Watts RL, Koller WC, editors. Movement Disorders: Neurological Principles and Practice. 2nd ed. New York: McGraw-Hill; 2004:247-271.
196 Lees A. The treatment of the motor disorder of multiple system atrophy. In: Mathias C, Bannister R, editors. Autonomic Failure: A Textbook of Clinical Disorders of the Autonomic Nervous System. 4th ed. Oxford, UK: Oxford: Oxford University Press; 1999:357-363.
197 Wenning GK, Geser F, Poewe W. Therapeutic strategies in multiple system atrophy (MSA). Mov Disord. 2005;20(Suppl 12):S67-S76.
198 Poewe W. Treatment of dementia with Lewy bodies and Parkinson’s disease dementia. Mov Disord. 2005;20(Suppl 12):S77-S82.
199 Kaufmann H, Brannan T, Krakoff L, et al. Treatment of orthostatic hypotension due to autonomic failure with a peripheral alpha-adrenergic agonist (midodrine). Neurology. 1988;38:951-956.
200 Wright RA, Kaufmann HC, Perera R, et al. A double-blind, dose-response study of midodrine in neurogenic orthostatic hypotension. Neurology. 1998;51:120-124.
201 Low PA, Gilden JL, Freeman R, et al. Efficacy of midodrine vs placebo in neurogenic orthostatic hypotension: a randomized, double-blind multicenter study. Midodrine Study Group. JAMA. 1997;277:1046-1051.
202 Jankovic J, Gilden JL, Hiner BC, et al. Neurogenic orthostatic hypotension: a double-blind, placebo-controlled study with midodrine. Am J Med. 1993;95:38-48.
203 Kaufmann H, Saadia D, Voustianiouk A, et al. Norepinephrine precursor therapy in neurogenic orthostatic hypotension. Circulation. 2003;108:724-728.
204 Mathias CJ, Senard JM, Braune S, et al. L-threo-dihydroxyphenylserine (L-threo-DOPS; droxidopa) in the management of neurogenic orthostatic hypotension: a multi-national, multicenter, dose-ranging study in multiple system atrophy and pure autonomic failure. Clin Auton Res. 2001;11:235-242.
205 Alam M, Smith G, Bleasdale-Barr K, et al. Effects of the peptide release inhibitor, octreotide, on daytime hypotension and on nocturnal hypertension in primary autonomic failure. J Hypertens. 1995;13:1664-1669.
206 Raimbach SJ, Cortelli P, Kooner JS, et al. Prevention of glucose-induced hypotension by the somatostatin analogue octreotide (SMS 201–995) in chronic autonomic failure: haemodynamic and hormonal changes. Clin Sci (Lond). 1989;77:623-628.
207 Mathias CJ, Fosbraey P, da Costa DF, et al. The effect of desmopressin on nocturnal polyuria, overnight weight loss, and morning postural hypotension in patients with autonomic failure. Br Med J (Clin Res Ed). 1986;293:353-354.
208 Perera R, Isola L, Kaufmann H. Erythropoietin improves orthostatic hypotension in primary autonomic failure. Neurology. 1994;44(Suppl):A363.
209 Biaggioni I, Robertson D, Krantz S, et al. The anemia of primary autonomic failure and its reversal with recombinant erythropoietin. Ann Intern Med. 1994;121:181-186.
210 Hoeldtke RD, Streeten DH. Treatment of orthostatic hypotension with erythropoietin. N Engl J Med. 1993;329:611-615.
211 Perera R, Isola L, Kaufmann H. Effect of recombinant erythropoietin on anemia and orthostatic hypotension in primary autonomic failure. Clin Auton Res. 1995;5:211-213.
212 Winkler AS, Marsden J, Parton M, et al. Erythropoietin deficiency and anaemia in multiple system atrophy. Mov Disord. 2001;16:233-239.
213 Vagaonescu TD, Saadia D, Tuhrim S, et al. Hypertensive cardiovascular damage in patients with primary autonomic failure. Lancet. 2000;355:725-726.
214 Maclean A, Allen E. Orthostatic hypotension and orthostatic tachycardia, treatment with the “head up” bed. JAMA. 1940;115:2162-2167.
215 Ten Harkel AD, Van Lieshout JJ, Wieling W. Treatment of orthostatic hypotension with sleeping in the head-up tilt position, alone and in combination with fludrocortisone. J Intern Med. 1992;232:139-145.
216 Wenning GK, Colosimo C, Geser F, et al. Multiple system atrophy. Lancet Neurol. 2004;3:93-103.
217 Jordan J, Shannon JR, Pohar B, et al. Contrasting effects of vasodilators on blood pressure and sodium balance in the hypertension of autonomic failure. J Am Soc Nephrol. 1999;10:35-42.
218 Sakakibara R, Hattori T, Uchiyama T, et al. Are alpha-blockers involved in lower urinary tract dysfunction in multiple system atrophy? A comparison of prazosin and moxisylyte. J Auton Nerv Syst. 2000;79:191-195.
219 Goldstein I, Lue TF, Padma-Nathan H, et al. Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group. N Engl J Med. 1998;338:1397-1404.
220 Zesiewicz TA, Helal M, Hauser RA. Sildenafil citrate (Viagra) for the treatment of erectile dysfunction in men with Parkinson’s disease. Mov Disord. 2000;15:305-308.
221 Hussain IF, Brady CM, Swinn MJ, et al. Treatment of erectile dysfunction with sildenafil citrate (Viagra) in parkinsonism due to Parkinson’s disease or multiple system atrophy with observations on orthostatic hypotension. J Neurol Neurosurg Psychiatry. 2001;71:371-374.
222 Padma-Nathan H, Hellstrom WJ, Kaiser FE, et al. Treatment of men with erectile dysfunction with transurethral alprostadil. Medicated Urethral System for Erection (MUSE) Study Group. N Engl J Med. 1997;336:1-7.
223 Eichhorn TE, Oertel WH. Macrogol 3350/electrolyte improves constipation in Parkinson’s disease and multiple system atrophy. Mov Disord. 2001;16:1176-1177.
224 Boeve BF, Silber MH, Ferman TJ. Melatonin for treatment of REM sleep behavior disorder in neurologic disorders: results in 14 patients. Sleep Med. 2003;4:281-284.
225 Wild R, Pettit T, Burns A. Cholinesterase inhibitors for dementia with Lewy bodies. Cochrane Database Syst Rev. (3):2003. CD003672
226 McKeith I, Fairbairn A, Perry R, et al. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. BMJ. 1992;305:673-678.
227 Ballard C, Grace J, McKeith I, et al. Neuroleptic sensitivity in dementia with Lewy bodies and Alzheimer’s disease. Lancet. 1998;351:1032-1033.
228 Sechi G, Agnetti V, Masuri R, et al. Risperidone, neuroleptic malignant syndrome and probable dementia with Lewy bodies. Prog Neuropsychopharmacol Biol Psychiatry. 2000;24:1043-1051.
229 McKeith IG, Ballard CG, Harrison RW. Neuroleptic sensitivity to risperidone in Lewy body dementia. Lancet. 1995;346:699.
230 Rich SS, Friedman JH, Ott BR. Risperidone versus clozapine in the treatment of psychosis in six patients with Parkinson’s disease and other akinetic-rigid syndromes. J Clin Psychiatry. 1995;56:556-559.
231 Burke WJ, Pfeiffer RF, McComb RD. Neuroleptic sensitivity to clozapine in dementia with Lewy bodies. J Neuropsychiatry Clin Neurosci. 1998;10:227-229.
232 Walker Z, Grace J, Overshot R, et al. Olanzapine in dementia with Lewy bodies: a clinical study. Int J Geriatr Psychiatry. 1999;14:459-466.
233 Aarsland D, Ballard C, Larsen JP, et al. Marked neuroleptic sensitivity in dementia with Lewy bodies and Parkinson’s disease. Nord J Psychiatry. 2003;57:94.
234 Iranzo A, Santamaria J, Tolosa E. Continuous positive air pressure eliminates nocturnal stridor in multiple system atrophy. Barcelona Multiple System Atrophy Study Group. Lancet. 2000;356:1329-1330.
235 Iranzo A, Santamaria J, Tolosa E, et al. Long-term effect of CPAP in the treatment of nocturnal stridor in multiple system atrophy. Neurology. 2004;63:930-932.