CHAPTER 3 Nervous system
The nervous system has two major divisions, the central nervous system (CNS) and the peripheral nervous system (PNS). The CNS consists of the brain and spinal cord and contains the majority of neuronal cell bodies. The PNS includes all nervous tissue outside the central nervous system and is subdivided into the cranial and spinal nerves, peripheral autonomic nervous system (ANS) (including the enteric nervous system of the gut wall, ENS) and special senses (taste, olfaction, vision, hearing and balance). It is composed mainly of the axons of sensory and motor neurones that pass between the CNS and the body. However, the ENS contains as many intrinsic neurones in its ganglia as the entire spinal cord, is not connected directly to the CNS, and may be considered separately as a third division of the nervous system (Gershon 1998).
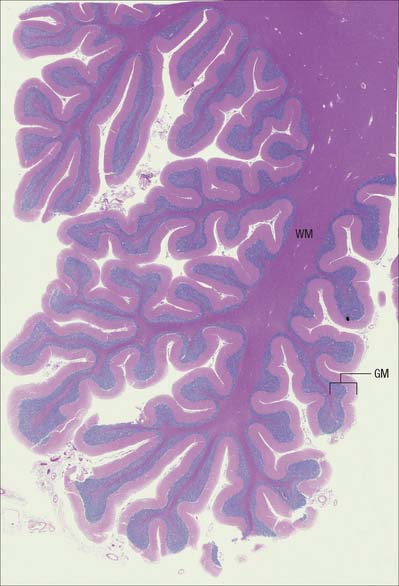
The CNS is derived from the neural tube (Ch. 24). The cell bodies of neurones are often grouped together in discrete areas termed nuclei, or they may form more extensive layers or masses of cells; collectively they constitute grey matter. Neuronal dendrites and synaptic contacts are mostly confined to areas of grey matter, and they form part of its meshwork of neuronal and glial processes termed the neuropil. Their axons join bundles of nerve fibres that tend to be grouped separately to form tracts. In the spinal cord, cerebellum, cerebral cortices (Chs 20, 22) and some other areas, concentrations of tracts constitute the white matter, so called because the axons are often ensheathed in lipid-rich sheaths of myelin which is white when fresh (Fig. 3.1).
The PNS is composed of the efferent axons (fibres) of motor neurones situated inside the CNS, and the cell bodies of sensory neurones (grouped together as ganglia) and their afferent processes. Sensory cells in dorsal root ganglia give off both centrally and peripherally directed processes: there are no synapses on their cell bodies. Also included are ganglionic neurones of the ANS, which receive synaptic contacts from the peripheral fibres of preganglionic autonomic neurones whose cell bodies lie within the CNS. Neuronal cell bodies in peripheral ganglia are all derived embryologically from cells which migrate from the neural crest (p. 201).
The nervous system contains large populations of non-neuronal cells, neuroglia or glia, which, whilst not electrically active in the same way, are responsible for creating and maintaining an appropriate environment in which neurones can operate efficiently. Indeed, it is now known that two-way communication between neurones and glial cells is essential for normal neural activity (reviewed in Fields & Stevens-Graham 2002). In the CNS, glia outnumber neurones by 10–50 times and consist of microglia and macroglia. Macroglia are further subdivided into three main types, oligodendrocytes, astrocytes and ependymal cells. The principal glial cell of the PNS is the Schwann cell. Satellite cells surround each neuronal soma in ganglia.
NEURONES
Most of the neurones in the CNS are either clustered into nuclei, columns or layers, or dispersed within grey matter. Neurones of the PNS are confined to ganglia. Irrespective of location, neurones share many general features, which are discussed here in the context of central neurones. Special characteristics of ganglionic neurones and their adjacent tissues are discussed on page 55–6.
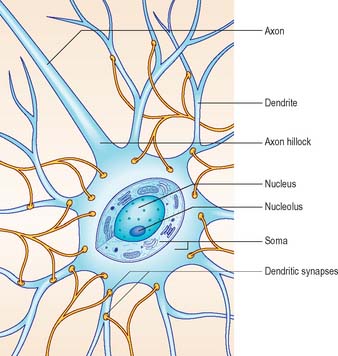
Neurones exhibit great variability in their size (cell bodies range from 5 to 100 μm diameter) and shapes. Their surface areas are extensive because most neurones display numerous narrow branched cell processes. They usually have a rounded or polygonal cell body (perikaryon or soma). This is a central mass of cytoplasm which encloses a nucleus and gives off long, branched extensions, with which most intercellular contacts are made. Typically, one of these processes, the axon, is much longer than the others, the dendrites (Fig. 3.2). Dendrites conduct electrical signals towards a soma whereas axons conduct impulses away from it.
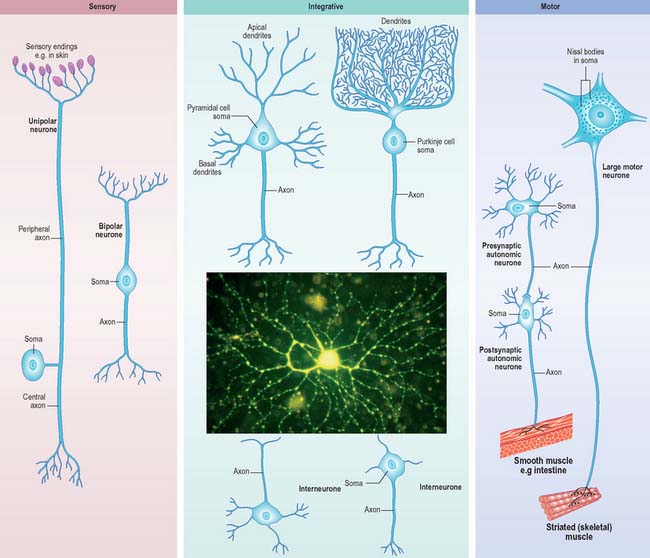
Neurones can be classified according to the number and arrangement of their processes. Multipolar neurones (Fig. 3.3) are common: they have an extensive dendritic tree which arises from either a single primary dendrite or directly from the soma, and a single axon. Bipolar neurones, which typify neurones of the special sensory systems, e.g. the retina (p. 697), have only a single dendrite which emerges from the soma opposite the axonal pole. Unipolar neurones which transmit general sensation, e.g. dorsal root ganglion neurones, have a single short process which bifurcates into peripheral and central processes (p. 55). This arrangement arises by the fusion of the proximal axonal and dendritic processes of a bipolar neurone during development: they may also, therefore, be termed pseudounipolar. Neurones are also classified according to whether their axons terminate locally on other neurones (interneurones), or transmit impulses over long distances, often to distinct territories via defined tracts (projection neurones).
Neurones are postmitotic cells and, with few exceptions, they are not replaced when lost.
SOMA
The cytoplasm of a typical soma (Fig. 3.2) is rich in rough and smooth endoplasmic reticulum and free polyribosomes, indicating a high level of protein synthetic activity. Free polyribosomes often congregate in large groups associated with the rough endoplasmic reticulum. These aggregates of RNA-rich structures are visible by light microscopy as basophilic Nissl bodies or granules. They are more obvious in large, highly active cells, such as spinal motor neurones (Fig. 3.4), which contain large stacks of rough endoplasmic reticulum and polyribosome aggregates. Maintenance and turnover of cytoplasmic and membranous components are necessary in all cells: the huge total volume of cytoplasm within the soma and processes of many neurones requires a considerable commitment of protein synthetic machinery. Neurones also synthesize other proteins (enzyme systems, etc.) involved in the production of neurotransmitters and in the reception and transduction of incoming stimuli. Various transmembrane channel proteins and enzymes are located at the surfaces of neurones where they are associated with movements of ions. The apparatus for protein synthesis (including RNA and ribosomes) occupies the soma and dendrites, but is usually absent from axons.
The neuronal cytoskeleton is a prominent feature of its cytoplasm and gives shape, strength and support to the dendrites and axons. A number of neurodegenerative diseases are characterized by abnormal aggregates of cytoskeletal proteins (reviewed in Cairns et al 2004). Neurofilaments (the intermediate filaments of neurones) and microtubules are abundant in the soma and along dendrites and axons: the proportions vary with the type of neurone and cell process. Bundles of neurofilaments constitute neurofibrils which can be seen by light microscopy in silver stained or immunolabelled sections. Neurofilaments are heteropolymers of proteins assembled from three polypeptide subunits, NF-L (68 kilodaltons [kDa]), NF-M (160 kDa) and NF-H (200 kDa). NF-M and NF-H have long C-terminal domains which project as side arms from the assembled neurofilament and bind to neighbouring filaments. They can be heavily phosphorylated, particularly in the highly stable neurofilaments of mature axons, and are thought to give axons their tensile strength. Some axons are almost filled by neurofilaments.
Microtubules are important in axonal transport, although dendrites usually have more microtubules than axons. Centrioles persist in mature postmitotic neurones, where they are concerned with the generation of microtubules rather than cell division. Centrioles are associated with cilia on the surfaces of developing neuroblasts. Their significance, other than at some sensory endings (e.g. the olfactory mucosa, p. 553), is not known.
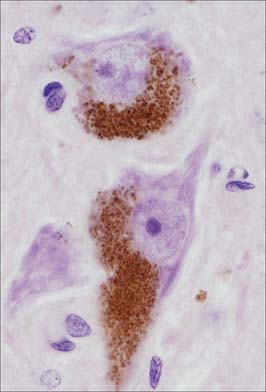
Pigment granules (Fig. 3.5) appear in certain regions, e.g. neurones of the substantia nigra contain neuromelanin, probably a waste product of catecholamine synthesis. In the locus coeruleus a similar pigment, rich in copper, gives a bluish colour to the neurones. Some neurones are unusually rich in certain metals which may form a component of enzyme systems, e.g. zinc in the hippocampus and iron in the oculomotor nucleus. Ageing neurones, especially in spinal ganglia, accumulate granules of lipofuscin (senility pigment) in residual bodies, which are lysosomes packed with partially degraded lipoprotein material (corpora amylaceae).
DENDRITES
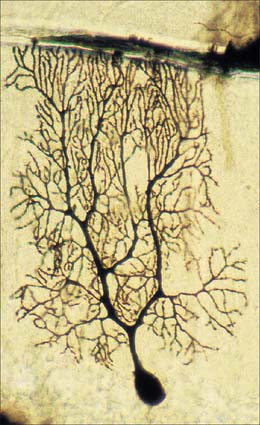
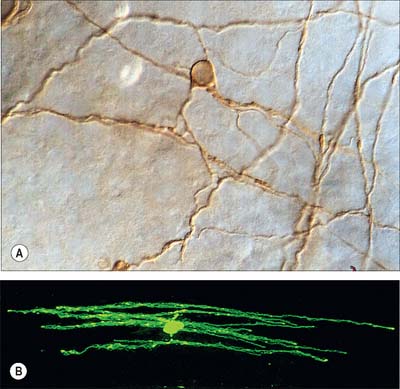
Dendrites are highly branched, usually short processes which project from the soma (Fig. 3.2). The branching patterns of many dendritic arrays are probably established by random adhesive interactions between dendritic growth cones and afferent axons that occur during development. There is an overproduction of dendrites in early development, and this is pruned in response to functional demand as the nervous system matures and information is processed through the dendritic tree. There is evidence that dendritic trees may be plastic structures throughout adult life, expanding and contracting as the traffic of synaptic activity varies through afferent axodendritic contacts (for review see Wong & Ghosh 2002). Groups of neurones with similar functions have a similar stereotypic tree structure (Fig. 3.6), suggesting that the branching patterns of dendrites are important determinants of the integration of the afferent inputs that converge on the tree.
Dendrites differ from axons in many respects. They represent the afferent rather than the efferent system of the neurone, and receive both excitatory and inhibitory axodendritic contacts. They may also make dendrodendritic and dendrosomatic connections (see Fig. 3.9), some of which are reciprocal. Synapses occur either on small projections called dendritic spines or on the smooth dendritic surface. Dendrites contain ribosomes, smooth endoplasmic reticulum, microtubules, neurofilaments, actin filaments and Golgi complexes. Their neurofilament proteins are poorly phosphorylated and their microtubules express the microtubule-associated protein (MAP)-2 almost exclusively in comparison with axons.
The shapes of dendritic spines range from simple protrusions to structures with a slender stalk and expanded distal end. Most spines are not more than 2 μm long, and have one or more terminal expansions; they can also be short and stubby, branched or bulbous. Free ribosomes and polyribosomes are concentrated at the base of the spine. Ribosomal accumulations near synaptic sites provide a mechanism for activity- dependent synaptic plasticity through the local regulation of protein synthesis.
AXONS
The axon originates either from the soma or from the proximal segment of a dendrite, at a specialized region free of Nissl granules, the axon hillock (Fig. 3.2). Action potentials are initiated here. The axonal plasma membrane (axolemma) is undercoated at the hillock by a concentration of cytoskeletal molecules, including spectrin and actin fibrils, which are important in anchoring numerous voltage-sensitive channels to the membrane. The axon hillock is unmyelinated and often participates in inhibitory axo-axonal synapses. It is unique because it contains ribosomal aggregates immediately below the postsynaptic membrane.
In the CNS, small, unmyelinated axons lie free in the neuropil, whereas in the PNS they are embedded in Schwann cell cytoplasm. Myelin, which is formed around almost all axons >2 μm diameter by oligodendrocytes in the CNS and Schwann cells in the PNS, begins at the distal end of the axon hillock. Nodes of Ranvier are specialized constricted regions of myelin-free axolemma where action potentials are generated and where an axon may branch. In both CNS and PNS, the territory of a myelinated axon between adjacent nodes is called an internode: the region close to a node, where the myelin sheath terminates, is called the paranode. Myelin thickness and internodal lengths are, in general, positively correlated with axon diameter. The density of sodium channels in the axolemma is highest at nodes of Ranvier, and very low along internodal membranes: sodium channels are spread more evenly within the axolemma of unmyelinated axons. Fast potassium channels are present in the paranodal regions of myelinated axons. Fine processes of glial cytoplasm (astrocytic in the CNS, Schwann cell in the PNS) surround the nodal axolemma.
The terminals of an axon are unmyelinated and most expand into presynaptic boutons which may form connections with axons, dendrites, neuronal somata or, in the periphery, muscle fibres, glands and lymphoid tissue. Exceptions include the free afferent sensory endings in e.g. the epidermis, which are unspecialized structurally, and the peripheral terminals of afferent sensory fibres with encapsulated endings (see Fig. 3.30). Axon terminals may themselves be contacted by other axons, forming axoaxonal presynaptic inhibitory circuits. Further details of neuronal microcircuitry are given in Kandel et al (2000).
Axoplasmic flow
Rapid flow depends on microtubules. Vesicles with side projections line up along microtubules and are transported along them by their side-arms. Two microtubule-based motor proteins with ATPase activity are involved in fast transport: kinesin family proteins are responsible for the fast component of anterograde transport, and cytoplasmic dynein is responsible for retrograde transport. Microtubule-dependent axonal transport is reviewed in Guzik and Goldstein (2004). Retrograde transport mediates the movement of neurotrophic viruses, e.g. herpes zoster, rabies and polio, from peripheral terminals, and their subsequent concentration in the neuronal soma.
SYNAPSES
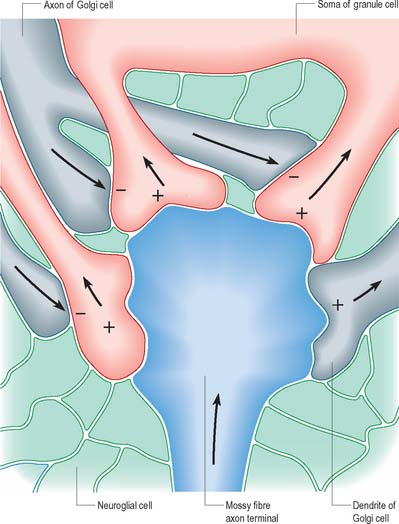
The patterns of axonal termination vary considerably. A single axon may synapse with one neurone (e.g. climbing fibres ending on cerebellar Purkinje neurones), or more often with many neurones (e.g. cerebellar parallel fibres, which provide an extreme example of this phenomenon (p. 302). In synaptic glomeruli, e.g. in the olfactory bulb and cerebellum, and synaptic cartridges, groups of synapses between two or many neurones form interactive units encapsulated by neuroglia (Fig. 3.7).
Classification of chemical synapses
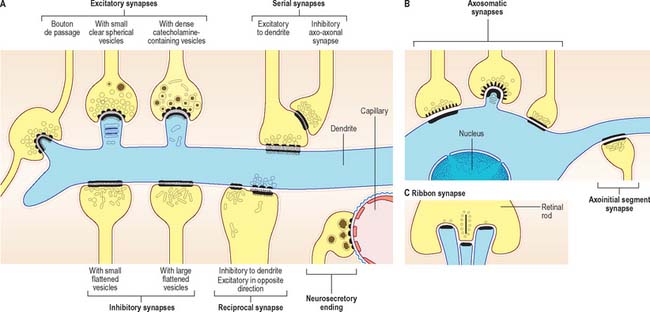
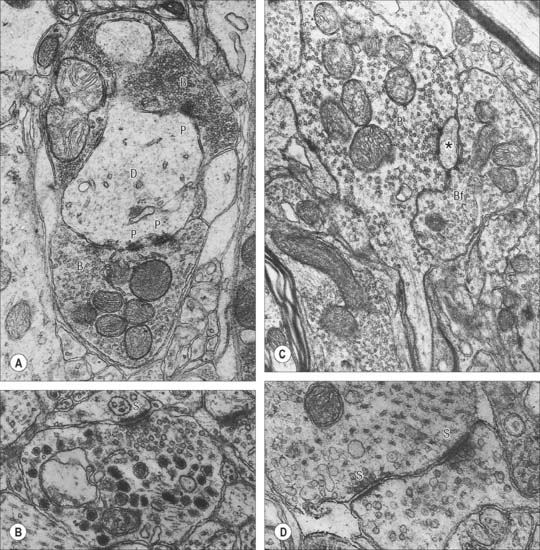
Chemical synapses have an asymmetric structural organization (Fig. 3.8 and Fig. 3.9) in keeping with the unidirectional nature of their transmission. Typical chemical synapses share a number of important features. They all display an area of presynaptic membrane apposed to a corresponding postsynaptic membrane: the two are separated by a narrow (20–30 nm) gap, the synaptic cleft. Synaptic vesicles containing neurotransmitter lie on the presynaptic side, clustered near a dense plaque on the cytoplasmic aspect of the presynaptic membrane. A corresponding region of submembrane density is present on the postsynaptic side. Together these define the active zone, the area of the synapse where neurotransmission takes place.
Chemical synapses can be classified according to a number of different parameters including the neuronal regions forming the synapse, their ultrastructural characteristics, the chemical nature of their neurotransmitter(s) and their effects on the electrical state of the postsynaptic neurone. The following classification is limited to associations between neurones. Neuromuscular junctions share many (though not all) of these parameters, and are often referred to as peripheral synapses. They are described separately on page 63.
Synapses can occur between almost any surface regions of the participating neurones. The most common type occurs between an axon and either a dendrite or a soma, when the axon is expanded as a small bulb or bouton (Fig. 3.8, Fig. 3.9). This may be a terminal of an axonal branch (terminal bouton) or one of a row of bead-like endings, when the axon makes contact at several points, often with more than one neurone (bouton de passage). Boutons may synapse with dendrites, including dendritic spines or the flat surface of a dendritic shaft, a soma (usually on its flat surface, but occasionally on spines), the axon hillock and the terminal boutons of other axons.
Ultrastructurally, synaptic vesicles may be internally clear or dense, and of different size (loosely categorized as small or large) and shape (round, flat or pleomorphic, i.e. irregularly shaped). The submembranous densities may be thicker on the postsynaptic than on the presynaptic side (asymmetric synapses), or equivalent in thickness (symmetrical synapses). Synaptic ribbons are found at sites of neurotransmission in the retina and inner ear. They have a distinctive morphology, in that the synaptic vesicles are grouped around a ribbon- or rod-like density orientated perpendicular to the cell membrane (Fig. 3.9).
Synaptic boutons make obvious close contacts with postsynaptic structures, but many other terminals lack specialized contact zones. Areas of transmitter release occur in the varicosities of unmyelinated axons, where effects are sometimes diffuse, e.g. the aminergic pathways of the basal ganglia and in autonomic fibres in the periphery. In some instances, such axons may ramify widely throughout extensive areas of the brain and affect the behaviour of very large populations of neurones, e.g. the diffuse cholinergic innervation of the cerebral cortices. Pathological degeneration of these pathways can therefore cause widespread disturbances in neural function.
Neurosecretory endings found in various parts of the brain and in neuroendocrine glands and cells of the dispersed neuroendocrine system (see below; p. 31) share many features with presynaptic boutons. They all contain peptides or glycoproteins within dense-core vesicles. The latter are of characteristic size and appearance: they are often ellipsoidal or irregular in shape, and relatively large, e.g. oxytocin and vasopressin vesicles in the neurohypophysis may be up to 200 nm across.
Mechanisms of synaptic activity
Neurohormones
Neurohormones are included in the class of molecules with neurotransmitter-like activity. They are synthesized in neurones and released into the blood circulation by exocytosis at synaptic terminal-like structures. As with classic endocrine gland hormones, they may act at great distances from their site of secretion. Neurones secrete into the CSF or local interstitial fluid to affect other cells, either diffusely or at a distance. To encompass this wide range of phenomena the general term neuromediation has been used, and the chemicals involved are called neuromediators.
Neurotransmitter molecules
Monoamines
Dopamine is a neuromediator of considerable clinical importance, found mainly in neurones with cell bodies in the telencephalon, diencephalon and mesencephalon. A major dopaminergic neuronal population in the midbrain constitutes the substantia nigra, so called because its cells contain neuromelanin, a black granular byproduct of dopamine synthesis. Dopaminergic endings are particularly numerous in the corpus striatum, limbic system and cerebral cortex. Structurally, dopaminergic synapses contain numerous dense-cored vesicles that resemble those containing noradrenaline. Pathological reduction in dopaminergic activity has widespread effects on motor control, affective behaviour and other neural activities, as seen in Parkinson’s syndrome (Ch. 22).
Neuropeptides
Substance P (SP) was the first of the peptides to be characterized as a gastrointestinal neuromediator and is considered to be the prototypic neuropeptide. It is an 11-amino acid polypeptide that belongs to the tachykinin neuropeptide family, and is a major neuromediator in the brain and spinal cord. Contained within large granular synaptic vesicles, SP is found in approximately 20% of dorsal root and trigeminal ganglion cells, in particular in small nociceptive neurones, and in some fibres of the facial, glossopharyngeal and vagal nerves. Within the CNS, SP is present in several apparently unrelated major pathways, and has been described in the limbic system, basal ganglia, amygdala and hypothalamus. Its known action is prolonged postsynaptic excitation, particularly from nociceptive afferent terminals, which sustains the effects of noxious stimuli. SP is one of the main neuropeptides that trigger an inflammatory response in the skin and has also been implicated in the vomiting reflex, changes in cardiovascular tone, stimulation of salivary secretion, smooth muscle contraction, and vasodilation.
CENTRAL GLIA
Glial (neuroglial) cells vary considerably in type and number in different regions of the CNS. There are two major groups, macroglia (astrocytes and oligodendrocytes) and microglia, classified according to origin. Macroglia arise within the neural plate, in parallel with neurones, and constitute the great majority of glial cells. Their functions are diverse and are now known to extend beyond a passive supporting role (reviewed in He & Sun, 2007). Microglia are smaller cells, generally considered to be monocytic in origin, and are derived from haemopoietic tissue (see Fig. 3.21).
ASTROCYTES
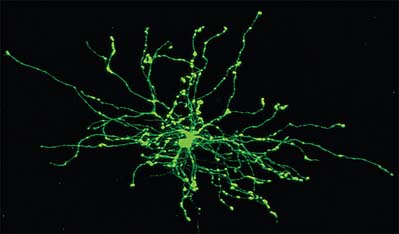
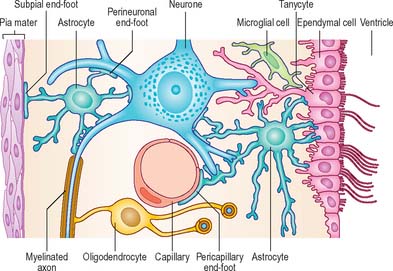
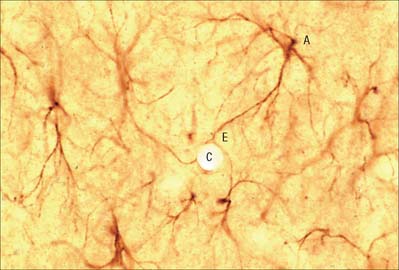
Astrocytes are star-shaped glia (Fig. 3.10). Their processes, which ramify through the entire central neuropil (Fig. 3.11), are coupled functionally at gap junctions, forming an interconnected network which ensheathes all neurones, other than at synapses and along the myelinated segments of axons. Some astrocyte processes terminate as end-feet on the basal lamina of blood vessels, and at the pial surface, where they form the glia limitans (glial limiting membrane).
Astrocytes are now thought to provide a network of communication, providing integration and regulation of function in the brain (reviewed in Fields 2004, Volterra & Meldolesi 2005). One form of communication, with other astrocytes, is via interconnecting low resistance gap junctional complexes. There is recent evidence that astrocytic (and other neural) gap junctions may be formed by a novel family of proteins, the pannexins (see p. 7). They signal to each other using intracellular calcium wave propagation, triggered by synaptically-released glutamate. Signalling presumably co-ordinates astrocyte functions that are essential for efficient neuronal activity, such as ion (particularly potassium) buffering; neurotransmitter uptake and metabolism (e.g. of excess glutamate, which is excitotoxic); membrane transport; the secretion of peptides, amino acids, trophic factors etc. The complexity of astrocytic function and dysfunction in neurological disorders is reviewed in Seifert et al (2006).
Blood–brain barrier
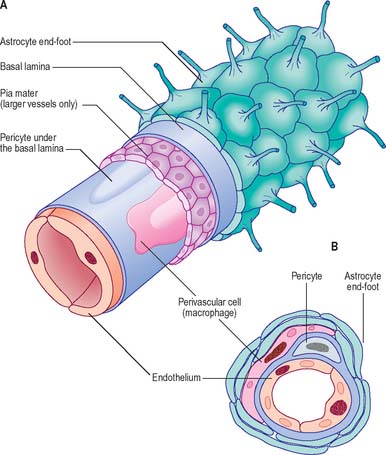
Proteins circulating in the blood enter most tissues of the body except those of the brain, spinal cord or peripheral nerves. This concept of a blood–brain or a blood–nerve barrier applies to many substances – some are actively transported across the blood–brain barrier, others are actively excluded. The blood–brain barrier is located at the capillary endothelium within the brain and is dependent upon the presence of tight junctions between endothelial cells coupled with a relative lack of transcytotic vesicular transport. The tightness of the barrier depends upon the close apposition of astrocytes, which direct the formation of endothelial tight junctions, to blood capillaries (reviewed in Abbott et al 2006) (Fig. 3.12, Fig. 3.13).
The blood–brain barrier develops during embryonic life but may not be fully completed by birth. There are certain areas of the adult brain where the endothelial cells are not linked by tight junctions, which means that a free exchange of molecules occurs between blood and adjacent brain. Most of these areas are situated close to the ventricles and are known as circumventricular organs (p. 240). Elsewhere, unrestricted diffusion through the blood–brain barrier is only possible for substances that can cross biological membranes because of their lipophilic character. Lipophilic molecules may be actively re-exported by the brain endothelium.
OLIGODENDROCYTES
Oligodendrocytes myelinate CNS axons and are most commonly seen as intrafascicular cells in myelinated tracts (Fig. 3.14, Fig. 3.15). They usually have round nuclei and their cytoplasm contains numerous mitochondria, microtubules and glycogen. They display a spectrum of morphological variation, from cells with large euchromatic nuclei and pale cytoplasm, to cells with heterochromatic nuclei and dense cytoplasm. Oligodendrocytes may enclose up to 50 axons in separate myelin sheaths, and the largest calibre axons are usually ensheathed on a 1 : 1 basis. Some oligodendrocytes are not associated with axons, and are either precursor cells or perineuronal (satellite) oligodendrocytes whose processes ramify around neuronal somata.
Oligodendrocytes originate from the ventricular neurectoderm and the subependymal layer in the fetus (p. 368), and continue to be generated from the subependymal plate postnatally. Stem cells migrate and seed into white and grey matter to form a pool of adult progenitor cells which may later differentiate to replenish lost oligodendrocytes, and possibly remyelinate pathologically demyelinated regions.
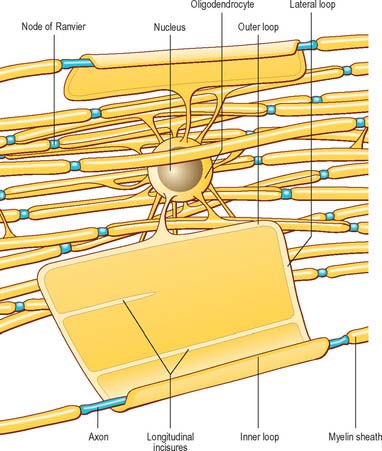
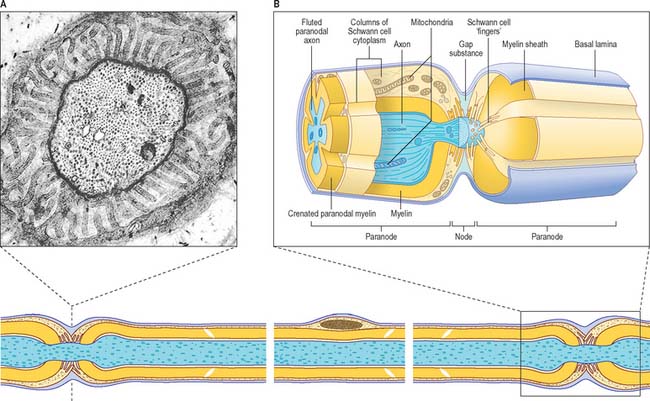
Nodes of Ranvier and incisures of Schmidt–Lanterman
The territory ensheathed by an oligodendrocyte (or Schwann cell) process defines an internode, the interval between internodes is called a node of Ranvier (Fig. 3.16) and the territory immediately adjacent to the nodal gap is a paranode, where loops of oligodendrocyte cytoplasm abut the axolemma. Nodal axolemma is contacted by fine filopodia of perinodal cells which have been shown in animal studies to have a presumptive adult oligodendrocyte progenitor phenotype: their function is unknown (reviewed in Butt et al 2005). Schmidt–Lanterman incisures are helical decompactions of internodal myelin where the major dense line of the myelin sheath splits to enclose a spiral of oligodendrocyte cytoplasm. Their structure suggests that they may play a role in the transport of molecules across the myelin sheath, but their function is not known.
MYELIN AND MYELINATION
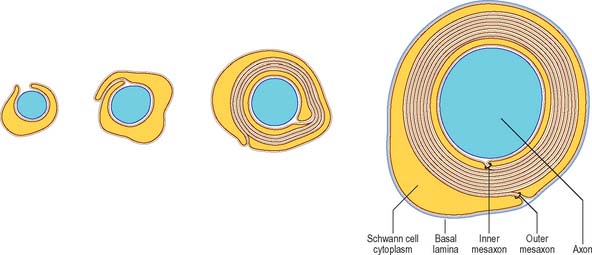
It is not known precisely how myelin is formed in either PNS or CNS, but in the CNS it has been shown recently to depend in part on expression of a protein (Wiskott–Aldrich syndrome protein family verprolin homologous; WAVE), which influences the actin cytoskeleton, oligodendrocyte lamellipodia formation and myelination (Kim et al 2006). The ultrastructural appearance of myelin is usually explained in terms of the spiral wrapping of an extensive, flat glial process (lamellipodium) around an axon, and the subsequent extrusion of cytoplasm from the sheath at all points other than incisures and paranodes. In this way, the compacted external surfaces of the plasma membrane of the ensheathing glial cell are thought to produce the minor dense lines, and the compacted inner cytoplasmic surfaces, the major dense lines, of the mature myelin sheath (Fig. 3.17). These lines, first described in early electron microscope studies of the myelin sheath, correspond to the intraperiod and period lines respectively defined in X-ray studies of myelin. The inner and outer zones of occlusion of the spiral process are continuous with the minor dense line and are called the inner and outer mesaxons.
There are significant differences between central and peripheral myelin, reflecting the fact that oligodendrocytes and Schwann cells express different proteins during myelinogenesis. The basic dimensions of the myelin membrane are different. CNS myelin has a period repeat thickness of 15.7 nm whereas PNS myelin has a period to period line thickness of 18.5 nm, and the major dense line space is approximately 1.7 nm in CNS myelin, compared with 2.5 nm in PNS myelin. Myelin membrane contains protein, lipid and water, which forms at least 20% of the wet weight. It is a relatively lipid-rich membrane and contains 70–80% lipid. All classes of lipid have been found; perhaps not surprisingly, the precise lipid composition of PNS and CNS myelin is different. The major lipid species are cholesterol (the commonest single molecule), phospholipids and glycosphingolipids. Minor lipid species include galactosylglycerides, phosphoinositides and gangliosides. The major glycolipids are galactocerebroside and its sulphate ester, sulphatide: these lipids are not unique to myelin, but they are present in characteristically high concentrations. CNS and PNS myelin also contain low concentrations of acidic glycolipids, which constitute important antigens in some inflammatory demyelinating states. Gangliosides, which are glycosphingolipids characterized by the presence of sialic acid (N-acetylneuraminic acid), account for less than 1% of the lipid in myelin.
Mutations of the major myelin structural proteins have now been recognized in a number of inherited human neurological diseases. As would be expected, these mutations produce defects in myelination, and in the stability of nodal and paranodal architecture, which are consistent with the suggested functional roles of the relevant proteins in maintaining the integrity of the myelin sheath. The molecular organization of myelinated axons is described in Scherer & Arroyo (2002).
EPENDYMA
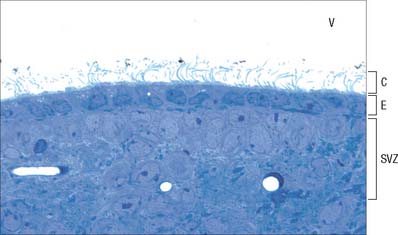
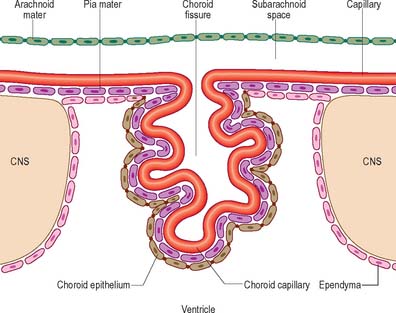
Ependymal cells line the ventricles (Fig. 3.18; see Fig. 3.11) and central canal of the spinal cord. They form a single-layered epithelium which varies from squamous to columnar in form. At the ventricular surface, cells are joined by gap junctions and occasional desmosomes. Their apical surfaces have numerous microvilli and cilia which contribute to the flow of CSF. There is considerable regional variation in the ependymal lining of the ventricles, but four major types have been described. These are: general ependyma which overlies grey matter; general ependyma which overlies white matter; specialized areas of ependyma in the third and fourth ventricles; choroidal epithelium.
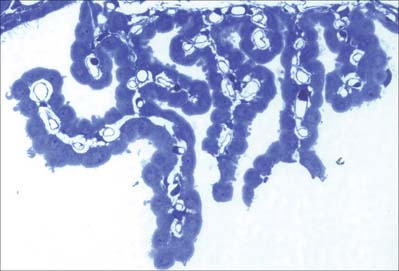
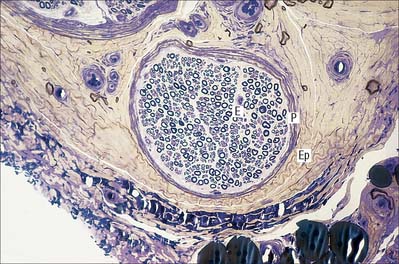
Choroid plexus
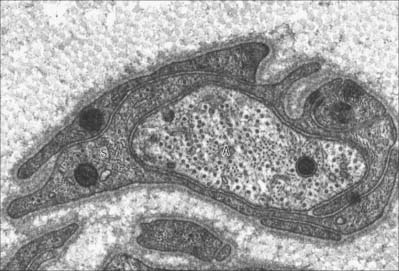
The choroid plexus forms the CSF and actively regulates the concentration of molecules in the CSF. It consists of highly vascularized masses of pia mater enclosed by pockets of ependymal cells. The ependymal cells resemble those of the circumventricular organs, except that they do not have basal processes, but form a cuboidal epithelium which rests on a basal lamina adjacent to the enclosed fold of meningeal pia mater and its capillaries (Fig. 3.19, Fig. 3.20). The cells have numerous long microvilli with only a few cilia interspersed between them. They also have many mitochondria, large Golgi complexes and basal nuclei, features that are consistent with their secretory activity: they produce most components of the CSF. They are linked by tight junctions that form a transepithelial barrier (a component of the blood–CSF barrier), and by desmosomes. Their lateral margins are highly folded.
Age-related changes occur in the choroid plexus which can be detected by neuroimaging. Calcification of the choroid plexus can be detected by X-ray or CT scan in 0.5% of individuals in the first decade of life and in 86% in the eighth decade. The incidence of calcification rises sharply, from 35% of CT scans in the fifth decade to 75% in the sixth decade. Visible calcification is usually restricted to the glomus region of the choroid plexus, i.e. the vascular bulge in the choroid plexus as it curves to follow the anterior wall of the lateral ventricle into the temporal horn.
MICROGLIA
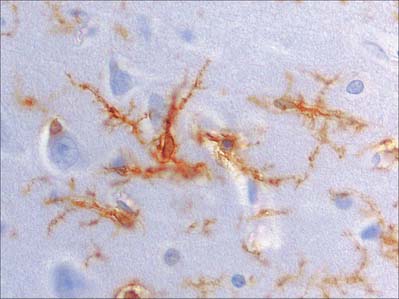
Microglia are small dendritic cells found throughout the CNS (Fig. 3.21) including the retina (p. 692). They are thought to be derived from fetal monocytes or their precursors; the haematogenous cells cross the walls of neural blood vessels and invade CNS tissue prenatally as amoeboid cells. Later they lose their motility and transform into typical microglia which bear branched processes that ramify in non-overlapping territories within the brain. All microglial domains, defined by their dendritic fields, are equivalent in size, and form a regular mosaic throughout the brain. The expression of microglia-specific antigens changes with age and many are downregulated when microglia attain the mature dendritic form.
PERIPHERAL NERVES
Peripheral nerve fibres are grouped in widely variable numbers into bundles (fasciculi). The size, number and pattern of fasciculi vary in different nerves and at different levels along their paths (Fig. 3.22). Their number increases and their size decreases some distance proximal to a point of branching. Where nerves are subjected to pressure, e.g. deep to a retinaculum, fasciculi are increased in number but reduced in size, and the amount of associated connective tissue and degree of vascularity also increase. At these points, nerves may occasionally show a pink, fusiform dilatation, sometimes termed a pseudoganglion or gangliform enlargement.
CONNECTIVE TISSUE SHEATHS
Nerve trunks, whether uni- or multifascicular, are surrounded by an epineurium. Individual fasciculi are enclosed by a multilayered perineurium, which in turn surrounds the endoneurium or intrafascicular connective tissue (Fig. 3.22).
SCHWANN CELLS
Unmyelinated axons
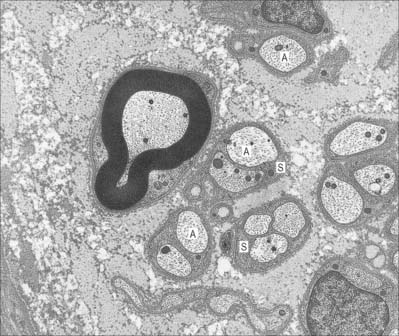
Unmyelinated axons (Fig. 3.23) are commonly 1.0 μm in diameter, although some may be 1.5 μm or even 2 μm in diameter. Groups of up to 10 small axons (0.15–2.0 μm in diameter) are enclosed within a chain of overlapping Schwann cells that is surrounded by a basal lamina. Within each Schwann cell, individual axons are usually sequestered from their neighbours by delicate processes of cytoplasm. Axons move between Schwann cell chains as they pass proximodistally along a nerve fasciculus. It seems likely, on the basis of quantitative studies in subhuman primates, that axons from adjacent cord segments may share Schwann cell columns: this phenomenon may play a role in the evolution of neuropathic pain after nerve injury. In the absence of a myelin sheath and nodes of Ranvier, conduction along unmyelinated axons is not saltatory but electrotonic, and the passage of impulses is therefore relatively slow (approximately 0.5–4.0 m/s).
Myelinated axons
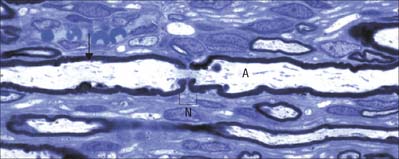
Myelinated axons (Fig. 3.24) have a 1 : 1 relationship with their ensheathing Schwann cells. The territory of an individual Schwann cell defines an internode (Fig. 3.25): internodal length varies directly with the diameter of the fibre, from 150 to 1500 μm. The interval between two internodes is a node of Ranvier. In the PNS, the myelin sheaths on either side of a node terminate in asymmetrically swollen paranodal bulbs. Schwann cell cytoplasm only forms a continuous layer in the perinuclear (mid-internodal) and paranodal regions. Between these sites, internodal Schwann cytoplasm forms a delicate network over the inner (abaxonal) surface of the myelin sheath. The outer (adaxonal) layer of Schwann cell cytoplasm is frequently discontinuous, and axons are surrounded by a narrow periaxonal space (15–20 nm) which, although nominally part of the extracellular space, is functionally isolated from it at the paranodes. For further details, see Scherer & Arroyo (2002).
Nodes of Ranvier
PNS nodes of Ranvier (Fig. 3.25) are typically approximately 0.8–1.1 μm in length. The calibre of the nodal axon is characteristically reduced relative to that of the internodal axon: this is most marked in the largest calibre axons. Node gaps are filled with an amorphous gap substance and processes of Schwann cell cytoplasm, and are surrounded by a continuous basal lamina elaborated by the ensheathing Schwann cells. In large calibre axons the surfaces of the paranodal bulbs and of the underlying axon are fluted as they approach the nodes. Fluting produces grooves in the external surface of the myelin sheath that are filled by Schwann cell cytoplasm characterized by large numbers of mitochondria. In smaller fibres this arrangement is less obvious, although the paranodal cytoplasm usually contains mitochondria.
SATELLITE CELLS
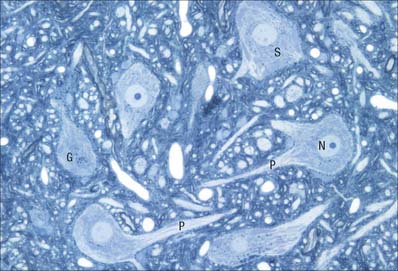
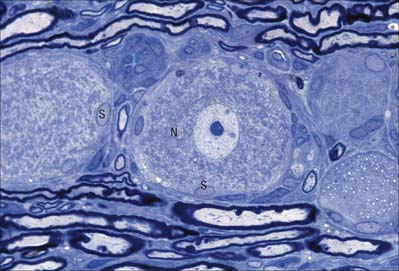
Many non-neuronal cells of the nervous system have been called satellite cells, including small round extracapsular cells in peripheral ganglia, ganglionic capsular cells, Schwann cells, any cell that is closely associated with neuronal somata, and precursor cells associated with striated muscle fibres (p. 117). Within the nervous system, the term is most commonly reserved for flat, epithelioid cells (ganglionic glial cells, capsular cells) that surround the neuronal somata of peripheral ganglia (Fig. 3.26). Their cytoplasm resembles that of Schwann cells, and their deep surfaces interdigitate with reciprocal infoldings in the membranes of the enclosed neurones. Capsular cells are succeeded by similar cells that enclose the initial part of the dendroaxonal process of unipolar sensory neurones in dorsal spinal ganglia, and these in turn are continuous with the Schwann cells that surround the peripheral and central processes of the neurones.
GANGLIA
Sensory ganglia
The sensory ganglia of dorsal spinal roots (Fig. 3.26) and the ganglia of the trigeminal, facial, glossopharyngeal and vagal cranial nerves are enclosed in periganglionic connective tissue that resembles the perineurium surrounding peripeheral nerves. Ganglionic neurones are unipolar. They have spherical or oval somata of varying size, aggregated in groups between fasciculi of myelinated and unmyelinated nerve fibres. For each neurone, the single axodendritic process bifurcates into central and peripheral processes: in myelinated fibres the junction occurs at a node of Ranvier. The peripheral process reaches a sensory ending and, since it conducts impulses towards the soma, strictly speaking it functions as an elongated dendrite. However, since it has the typical structural and functional properties of a peripheral axon, it is conventionally described as an axon.
Primary infection with the varicella zoster virus causes chickenpox. Following recovery, the virus remains dormant within dorsal root ganglia: its reactivation leads to shingles, which involves the dermatome(s) supplied by the affected sensory nerve(s). Severe pain and a rash similar to chickenpox, often confined to one of the divisions of the trigeminal nerve, or to a spinal nerve dermatome, are diagnostic. Herpes zoster involving the geniculate ganglion compresses the facial nerve and results in a lower motor neurone facial paralysis, known as Ramsay Hunt syndrome. Occasionally, if the vestibulocochlear nerve becomes involved, there is vertigo, tinnitus and some deafness.
Autonomic ganglia
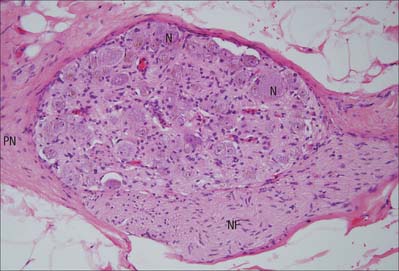
Autonomic ganglia are largely relay stations. Their neurones are multipolar and have dendritic trees on which preganglionic autonomic motor axons synapse (Fig. 3.27). The neurones are surrounded by a mixed neuropil of afferent and efferent fibres, dendrites, synapses and non-neural cells. There is considerable variation in the ratio of pre- and postganglionic fibres in different types of ganglion. Thus preganglionic sympathetic axons may synapse with many postganglionic neurones for the wide dissemination and perhaps amplification of sympathetic activity, a feature not found to the same degree in parasympathetic ganglia. Dissemination may also be achieved by connections with ganglionic interneurones or by the diffusion within the ganglion of transmitter substances produced either locally (paracrine effect) or elsewhere (endocrine effect). A small fraction of the fibres within a ganglion are either efferent fibres en route to another ganglion, or are afferent fibres from the viscera and glands: none of these fibres will synapse in the ganglion.
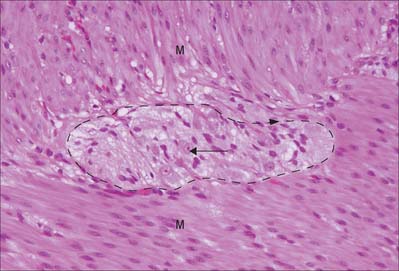
Enteric ganglia
The enteric nervous system is composed of ganglionic neurones (Fig. 3.28) and associated nerves serving different functions, including regulation of gut motility and mucosal transport (see Chs 15 and 60). Extrinsic autonomic fibres supply the gut wall and, together with intrinsic enteric ganglionic neurones, and the endocrine and cardiovascular systems, they integrate the activities of the digestive system either as a result of interaction with enteric neurones (e.g. via vagal fibres), or the direct regulation of the local blood flow (via postganglionic sympathetic fibres).
DISPERSED NEUROENDOCRINE SYSTEM
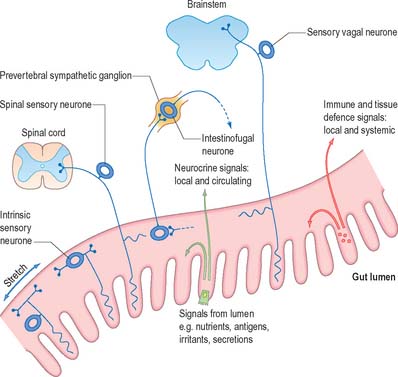
Although the nervous, neuroendocrine and endocrine systems all operate by intercellular communication, they differ in the mode, speed and degree or localization of the effects produced. The autonomic nervous system uses impulse conduction and neurotransmitter release to transmit information, and the responses induced are rapid and localized. The dispersed neuroendocrine system uses only secretion. It is slower and the induced responses are less localized, because the secretions, e.g. neuromediators, can act either on contiguous cells, or on groups of nearby cells reached by diffusion, or on distant cells via the bloodstream. Many of its effector molecules operate in both the nervous system and the neuroendocrine system. The endocrine system proper, which consists of clusters of cells and discrete, ductless, hormone-producing glands, is even slower and less localized, although its effects are specific and often prolonged. These regulatory systems overlap in function, and can be considered as a single neuroendocrine regulator of the metabolic activities and internal environment of the organism, acting to provide conditions in which it can function successfully. Neural and neuroendocrine axes appear to cooperate to modulate some forms of immunological reaction: the extensive system of vessels, circulating hormones and nerve fibres that link the brain with all viscera are thought to constitute a neuroimmune network (Fig. 3.29). For further reading see Furness (2006); Shepherd et al (2005).
Some cells can take up and decarboxylate amine precursor compounds (amine precursor uptake and decarboxylation or APUD cells. They are characterized by dense-core cytoplasmic granules (see Fig. 2.6), similar to the neurotransmitter vesicles seen in some types of neuronal terminal. The group includes cells described as chromaffin cells (phaeochromocytes), derived from neuroectoderm and innervated by preganglionic sympathetic nerve fibres. Chromaffin cells synthesize and secrete catecholamines (dopamine, noradrenaline or adrenaline). Their name refers to the finding that their cytoplasmic store of catecholamines is sufficiently concentrated to give an intense yellow-brown colouration, the positive chromaffin reaction, when they are treated with aqueous solutions of chromium salts, particularly potassium dichromate. Classic chromaffin cells include clusters of cells in the suprarenal medulla; the para-aortic bodies, which secrete noradrenaline; paraganglia (see Ch. 15); certain cells in the carotid bodies; small groups of cells irregularly dispersed among the paravertebral sympathetic ganglia, splanchnic nerves and prevertebral autonomic plexuses.
A number of descriptions and terms have been applied to cells of this system in the older literature. They include: clear cells (so named because of their poor staining properties in routine preparations); argentaffin cells (reduce silver salts); argyrophil cells (absorb silver); small intensely fluorescent cells; peptide-producing cells (particularly of the hypothalamus, hypophysis, pineal and parathyroid glands and placenta); Kulchitsky cells in the lungs; paraneurones. Many cells of the dispersed (or diffuse) neuroendocrine system, DNES, are derived embryologically from the neural crest. Some, in particular cells from the alimentary system, are now known to be endodermal in origin. For further reading, see Day and Salzet (2002).
SENSORY ENDINGS
GENERAL FEATURES OF SENSORY RECEPTORS
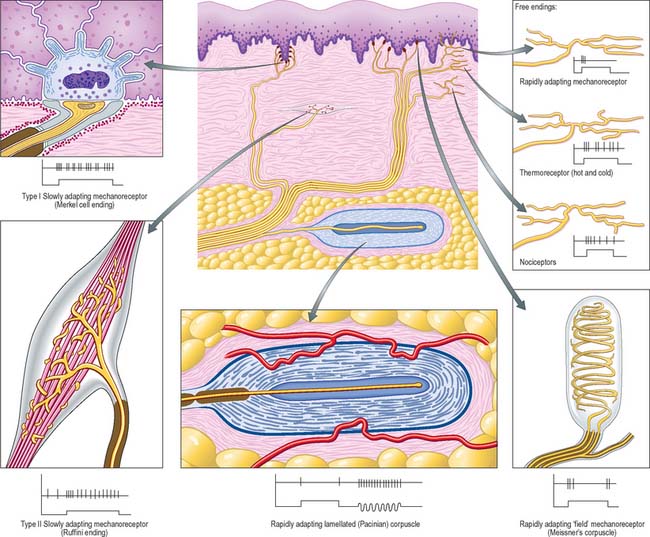
There are three major forms of sensory receptor, neuroepithelial, epithelial and neuronal (Fig. 3.30).
For further information on sensory receptors, see Nolte (2002).
FUNCTIONAL CLASSIFICATION OF RECEPTORS
Proprioceptors respond to stimuli to deeper tissues, especially of the locomotor system, and are concerned with detecting movement, mechanical stresses and position. They include Golgi tendon organs, neuromuscular spindles, Pacinian corpuscles, other endings in joints, and vestibular receptors. Proprioceptors are stimulated by the contraction of muscles, movements of joints and changes in the position of the body. They are essential for the coordination of muscles, the grading of muscular contraction, and the maintenance of equilibrium.
Interoceptors are found in the walls of the viscera, glands and vessels, where their terminations include free nerve endings, encapsulated terminals and endings associated with specialized epithelial cells. Nerve terminals are found in the layers of visceral walls and the adventitia of blood vessels, but the detailed structure and function of many of these endings are not well-established. Encapsulated (lamellated) endings occur in the heart, adventitia, and mesenteries. Free terminal arborizations occur in the endocardium, the endomysium of all muscles, and connective tissue generally. Visceral nerve terminals are not usually responsive to stimuli which act on exteroceptors, and do not respond to localized mechanical and thermal stimuli. Tension produced by excessive muscular contraction or by visceral distension often causes pain, particularly in pathological states, which is frequently poorly localized and of a deep-seated nature. Visceral pain is often referred to the corresponding dermatome (see Fig. 15.12). Polymodal nociceptors (irritant receptors) respond to a variety of stimuli such as noxious chemicals or damaging mechanical stimuli. They are mainly the free endings of fine, unmyelinated fibres that are widely distributed in the epithelia of the alimentary and respiratory tracts: they may initiate protective reflexes.
FREE NERVE ENDINGS
Sensory endings that branch to form plexuses occur in many sites (Fig. 3.30). They occur in all connective tissues, including those of the dermis, fasciae, capsules of organs, ligaments, tendons, adventitia of blood vessels, meninges, articular capsules, periosteum, perichondrium, Haversian systems in bone, parietal peritoneum, walls of viscera and the endomysium of all types of muscle. They also innervate the epithelium of the skin, cornea, buccal cavity, and the alimentary and respiratory tracts and their associated glands. Within epithelia, free sensory endings lack Schwann cell ensheathment and are enveloped instead by epithelial cells. Afferent fibres from free terminals may be myelinated or unmyelinated but are always of small diameter and low conduction velocity. When afferent axons are myelinated, their terminal arborizations are not. These terminals serve several sensory modalities. In the dermis, they may be responsive to moderate cold or heat (thermoreceptors); light mechanical touch (mechanoreceptors); damaging heat, cold or deformation (unimodal nociceptors); damaging stimuli of several kinds (polymodal nociceptors). Similar fibres in deeper tissues may also signal extreme conditions, which are experienced, as with all nociceptors, as pain. Free endings in the cornea, dentine and periosteum may be exclusively nociceptive.
Special types of free ending are associated with epidermal structures in the skin. They include terminals associated with hair follicles (peritrichial receptors) which branch from myelinated fibres in the deep dermal cutaneous plexus: the number, size and form of the endings are related to the size and type of hair follicle innervated. These endings respond mainly to movement when hair is deformed and belong to the rapidly adapting mechanoreceptor group.
ENCAPSULATED ENDINGS
Encapsulated endings are a major group of special endings that includes lamellated corpuscles of various kinds (e.g. Meissner’s, Pacinian), Golgi tendon organs, neuromuscular spindles and Ruffini endings (Fig. 3.30). They exhibit considerable variety in their size, shape and distribution, but share a common feature, in that each axon terminal is encapsulated by non-excitable cells.
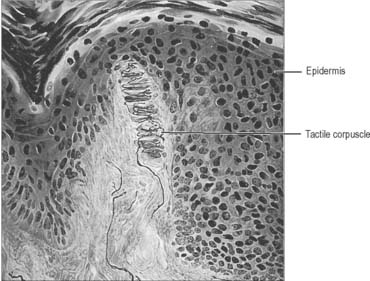
Meissner’s corpuscles
Meissner’s corpuscles are found in the dermal papillae of all parts of the hand and foot, the anterior aspect of the forearm, the lips, palpebral conjunctiva and mucous membrane of the apical part of the tongue. They are most concentrated in thick hairless skin, especially of the finger pads, where there may be up to 24 corpuscles per cm2 in young adults. Mature corpuscles are cylindrical in shape, approximately 80 μm long and 30 μm across, with their long axes perpendicular to the skin surface. Each corpuscle has a connective tissue capsule and central core composed of a stack of flat modified Schwann cells (Fig. 3.31). Meissner’s corpuscles are rapidly adapting mechanoreceptors, sensitive to shape and textural changes in exploratory and discriminatory touch: their acute sensitivity provides the neural basis for reading Braille text.
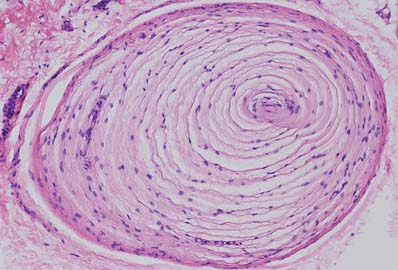
Pacinian corpuscles
Pacinian corpuscles are situated subcutaneously in the palmar and plantar aspects of the hand and foot and their digits; the external genitalia; arm; neck; nipple; periostea; interosseous membranes; near joints, and within the mesenteries. They are oval, spherical or irregularly coiled and measure up to 2 mm in length and 100–500 μm or more across: the larger ones are visible to the naked eye. Each corpuscle has a capsule, an intermediate growth zone and a central core that contains an axon terminal. The capsule is formed by approximately 30 concentrically arranged lamellae of flat cells approximately 0.2 μm thick (Fig. 3.32). Adjacent cells overlap and successive lamellae are separated by an amorphous proteoglycan matrix that contains circularly orientated collagen fibres, closely applied to the surfaces of the lamellar cells. The amount of collagen increases with age. The intermediate zone is cellular and its cells become incorporated into the capsule or core, so that it is not clearly defined in mature corpuscles. The core consists of approximately 60 bilateral, compacted lamellae lying on both sides of a central nerve terminal.
Golgi tendon organs
Golgi tendon organs are found mainly near musculotendinous junctions (Fig. 3.33), where more than 50 may occur at any one site. Each terminal is related closely to a group of muscle fibres (up to 20) as they insert into the tendon. Golgi tendon organs are approximately 500 μm long and 100 μm in diameter, and consist of small bundles of tendon fibres enclosed in a delicate capsule. The collagen bundles (intrafusal fasciculi) are less compact than elsewhere in the tendon, the collagen fibres are smaller and the fibroblasts larger and more numerous. A single, thickly myelinated 1b afferent nerve fibre enters the capsule and divides. Its branches, which may lose their ensheathing Schwann cells, terminate in leaf-like enlargements containing vesicles and mitochondria, which wrap around the tendon. A basal lamina or process of Schwann cell cytoplasm separates the nerve terminals from the collagen bundles that constitute the tendon. Golgi tendon organs are activated by passive stretch of the tendon, but are much more sensitive to active contraction of the muscle. They are important in providing proprioceptive information that complements the information coming from neuromuscular spindles. Their responses are slowly adapting and they signal maintained tension.
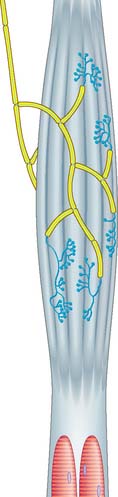
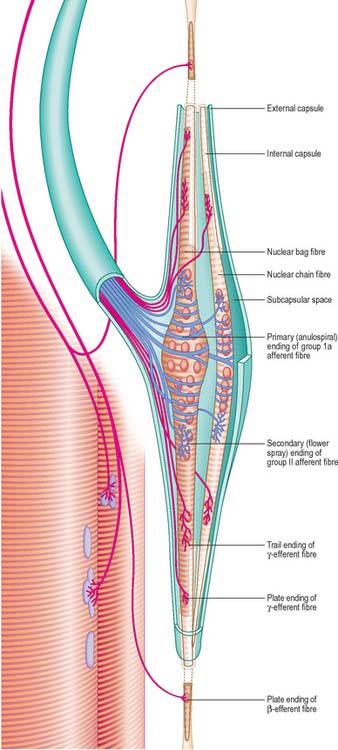
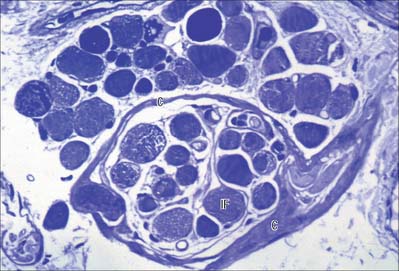
NEUROMUSCULAR SPINDLES
Neuromuscular spindles are essential for the control of muscle contraction. Each spindle contains a few small, specialized intrafusal muscle fibres, innervated by both sensory and motor nerve fibres (Fig. 3.34, Fig. 3.35). The whole is surrounded equatorially by a fusiform spindle capsule of connective tissue, consisting of an outer perineurial-like sheath of flattened fibroblasts and collagen and an inner sheath that forms delicate tubes around individual intrafusal fibres (Fig. 3.36). A gelatinous fluid rich in glycosaminoglycans fills the space between the two sheaths.
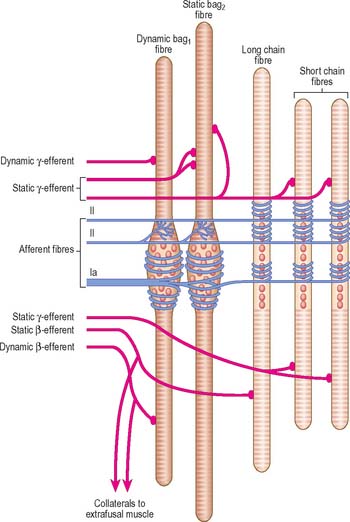
The intrafusal fibres resemble typical skeletal muscle fibres, except that the zone of myofibrils is thin around the nuclei. One subtype of nuclear bag fibre (dynamic bag 1) generally lacks M lines, possesses little sarcoplasmic reticulum and has an abundance of mitochondria and oxidative enzymes, but little glycogen. A second subtype of bag fibre (static bag 2) has distinct M lines and abundant glycogen. Nuclear chain fibres have marked M lines, sarcoplasmic reticulum and T-tubules, and abundant glycogen, but few mitochondria. These variations reflect the contractile properties of different intrafusal fibres (Boyd 1985).
There are three types of motor endings in muscle spindles. Two are from fine, myelinated, fusimotor (γ) efferents and one is from myelinated (β) efferent collaterals of axons that supply extrafusal slow twitch muscle fibres. The fusimotor efferents terminate nearer the equatorial region where their terminals either resemble the motor end-plates of extrafusal fibres (plate endings) or are more diffuse (trail endings). Stimulation of the fusimotor and β efferents causes contraction of the intrafusal fibres and consequently, activation of their sensory endings.
NEUROMUSCULAR JUNCTIONS
SKELETAL MUSCLE
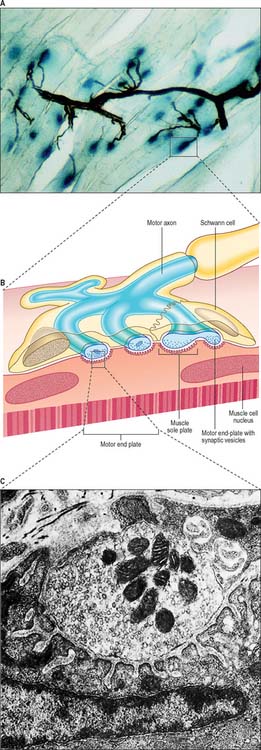
The detailed structure of a motor terminal varies with the type of muscle innervated. Two major types of ending are recognized, innervating either extrafusal muscle fibres, or the intrafusal fibres of neuromuscular spindles. In the former type, each axonal terminal usually ends midway along a muscle fibre in a discoidal motor end-plate (Fig. 3.37), and usually initiates action potentials that are rapidly conducted to all parts of the muscle fibre. In the latter type, the axon gives off numerous branches that form a cluster of small expansions extending along the muscle fibre; in the absence of propagated muscle excitation, these excite the fibre at several points. Both types of ending are associated with a specialized receptive region of the muscle fibre, the sole plate, where a number of muscle cell nuclei are grouped within the granular sarcoplasm.
The sole plate contains numerous mitochondria, endoplasmic reticulum and Golgi complexes. The terminal branches of the axon are plugged into shallow grooves in the surface of the sole plate (primary clefts), from where numerous pleats extend for a short distance into the underlying sarcoplasm (secondary clefts) (Fig. 3.37B,C). The axon terminal contains mitochondria and many clear, 60 nm spherical vesicles similar to those in presynaptic boutons, which are clustered over the zone of membrane apposition. It is ensheathed by Schwann cells whose cytoplasmic projections extend into the synaptic cleft. The plasma membranes of the axon terminal and the muscle cell are separated by a 30–50 nm gap and an interposed basal lamina which follows the surface folding of the sole plate membrane into the secondary clefts. The basal lamina contains specialized components including specific isoforms of type IV collagen and laminin and the heparan sulphate proteoglycan, agrin. Endings of fast and slow twitch muscle fibres (p. 111) differ in detail: the sarcolemmal grooves are deeper, and the presynaptic vesicles more numerous, in the fast fibres.
Junctions with skeletal muscle are cholinergic; the release of ACh changes the ionic permeability of the muscle fibre. Clustering of ACh receptors at the neuromuscular junction depends in part on the presence in the muscle basal lamina of agrin, which is secreted by the motor neurone, and is important in establishing the post-junctional molecular machinery. When the depolarization of the sarcolemma reaches a particular threshold, it initiates an all-or-none action potential in the sarcolemma, which is then propagated rapidly over the whole cell surface and also deep within the fibre via the invaginations (T-tubules) of the sarcolemma (p. 108), causing contraction. The amount of ACh released by the arrival of a single nerve impulse is sufficient to trigger an action potential. However, because ACh is very rapidly hydrolysed by the enzyme AchE, present at the sarcolemmal surface of the sole plate, a single nerve impulse only gives rise to one muscle action potential, i.e. there is a one-to-one relationship between neural and muscle action potentials. Thus the contraction of a muscle fibre is controlled by the firing frequency of its motor neurone.
AUTONOMIC MOTOR TERMINATIONS
Adrenergic sympathetic postganglionic terminals contain dense-cored vesicles. Cholinergic terminals, which are typical of all parasympathetic and some sympathetic endings, contain clear spherical vesicles like those in the motor end-plates of skeletal muscle. A third category of autonomic neurones has non-adrenergic, non-cholinergic endings which contain a wide variety of chemicals with transmitter properties. Conjugated purine (ATP, a nucleoside), is probably the neurotransmitter at these terminals, which are thus classed as purinergic. For further reading on purinergic signalling, see Burnstock (2006). Typically, their axons contain large, 80–200 nm, dense opaque vesicles, congregated in varicosities at intervals along axons. Such terminals are formed in many sites, including the external muscle layers and sphincters of the alimentary tract, lungs, walls of blood vessels, urogenital tract and in the CNS (see Chapter 15). In the intestinal wall, neuronal somata lie in the myenteric plexus, and their axons spread caudally for a few millimetres, mainly to innervate circular muscle. Purinergic neurones are under cholinergic control from preganglionic sympathetic neurones, via postganglionic sympathetic fibres. Their endings mainly hyperpolarize smooth muscle cells, causing relaxation, e.g. preceding peristaltic waves, opening sphincters and, probably, causing reflex distension in gastric filling.
Autonomic efferents also innervate glands, myoepithelial cells, adipose and lymphoid tissues.
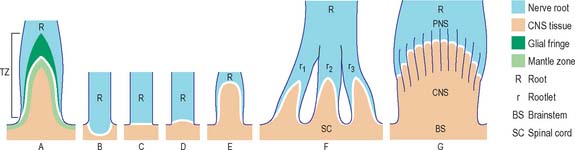
CNS–PNS TRANSITION ZONE
The transition between CNS and PNS usually occurs some distance from the point at which nerve roots emerge from the brain or the spinal cord. The segment of root that contains components of both CNS and PNS tissue is called the CNS–PNS transition zone (TZ). All axons in the PNS, other than postganglionic autonomic neurones, cross such a TZ. Macroscopically, as a nerve root is traced towards the spinal cord or the brain, it splits into several thinner rootlets which may, in turn, subdivide into minirootlets. The TZ is located within either rootlet or minirootlet (Fig. 3.38). The arrangement of roots and rootlets varies according to whether the root trunk is ventral, dorsal or cranial. Thus, in dorsal roots, the main root trunk separates into a fan of rootlets and minirootlets which enter the spinal cord in sequence along the dorsolateral sulcus. In certain cranial nerves the minirootlets come together central to the TZ and enter the brain as a stump of white matter.
A novel cell type, the boundary cap (BC) cell, has been described recently in avian and mammalian species. BCs are derived from the neural crest and form transient clusters in the presumptive dorsal root TZ of the embryonic spinal cord: they are thought to prevent cell mixing at this interface and to help dorsal root ganglion afferents navigate their path to targets in the spinal cord. It has been proposed that within dorsal root ganglia, BC-derived cells are the progenitors of neurones, mainly nociceptive afferents, satellite cells and all Schwann cell precursors that migrate into the dorsal roots (see Maro et al 2004).
CONDUCTION OF THE NERVOUS IMPULSE
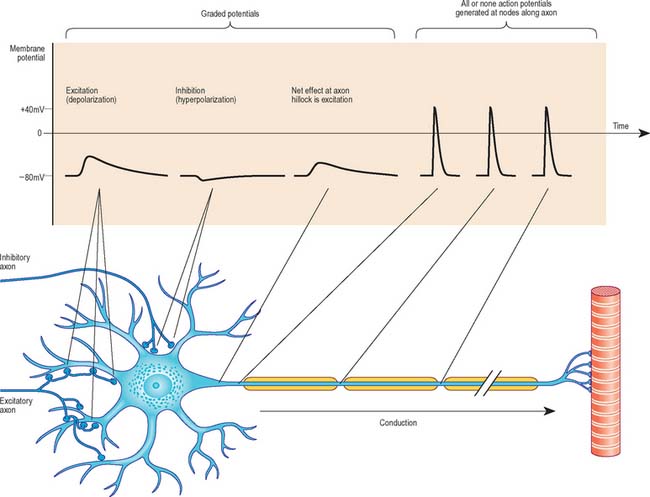
All cells generate a steady electrical potential across their plasma membranes (a membrane potential) by maintaining ionic concentrations inside the cell that differ from the extracellular fluid (Fig. 3.39). In neurones this membrane potential is known as the resting potential, and at approximately −80 mV (potential inside the cell measured relative to the outside of the cell), it is similar to that of non-excitable cells. However unlike non-excitable cells, neurones receive, conduct, and transmit information across their surfaces using minute fluctuations in membrane potential that result from changes in transmembrane ion fluxes. Sodium or, in some sites, calcium, entry causes depolarization of the cell, while chloride influx or potassium efflux results in hyperpolarization. Plasma membrane permeability to these ions is altered by the opening or closing of ion-specific transmembrane channels, triggered by chemical or electrical stimuli.
ACTION POTENTIAL
Once initiated, an action potential spreads spontaneously and at a relatively constant velocity, within the range of 4–120 m/s. Conduction velocity depends on a number of factors related to the way in which the current spreads, e.g. axonal cross-sectional area, the numbers and positioning of ion channels, and membrane capacitance (influenced particularly by the presence of myelin). In axons lacking myelin, action potential conduction is analogous to a flame moving along a fuse. Just as each segment of fuse is ignited by its upstream neighbour, each segment of axon membrane is driven to threshold by the depolarization of neighbouring membrane. Sodium channels within the newly depolarized segment open and positively charged sodium ions enter, driving the local potential inside the axon toward positive values. This inward current in turn depolarizes the neighbouring, downstream, nondepolarized membrane, and the cyclic propagation of the action potential is completed.
Axonal conduction is naturally unidirectional, from dendrites and soma to axon terminals. When an action potential reaches the axonal terminals, it causes depolarization of the presynaptic membrane and as a result, quanta of neurotransmitter (which correspond to the content of individual vesicles) are released to change the degree of excitation of the next neurone, muscle fibre or glandular cell. For further information, see Kandel et al (2000).
Abbott NJ, Rönnbäck L, Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nature Rev Neurosci. 2006;7:41-53.
Boyd IA. Muscle spindles and stretch reflexes. In: Swash M, Kennard C. Scientific Basis of Clinical Neurology. Edinburgh: Churchill Livingstone; 1985:74-97.
A detailed account of the functional aspects of neuromuscular spindles..
Burnstock G. Pathophysiology and therapeutic potential of purinergic signaling. Pharmacol Rev. 2006;58:58-86.
Butt AM, Hamilton N, Hubbard P, Pugh M, Ibrahim M. Synantocytes: the fifth element. J Anat. 2005;207:695-706.
Cairns NJ, Lee VM-Y, Trojanowski JQ. The cytoskeleton in neurodegenerative disease. J Pathol. 2004;204:438-449.
Day R, Salzet M. The neuroendocrine phenotype, cellular plasticity, and the search for genetic switches: redefining the diffuse neuroendocrine system. Neuroendocrinol Lett. 2002;23:379-384.
Fields RD. The other half of the brain. Sci Am (April). 2004:54-61.
Fields RD, Stevens-Graham B. New insights into neuron-glia communication. Science. 2002;298:552-556.
Furness JB. Novel gut afferents: Intrinsic afferent neurons and intestinofugal neurons. Auton Neurosci. 2006;125:81-85.
Gershon MD. The Second Brain. New York: HarperCollins, 1998.
Guzik BW, Goldstein LSB. Microtubule-dependent transport in neurons: steps towards an understanding of regulation, function and dysfunction. Curr Opin Cell Biol. 2004;16:443-450.
He F, Sun YE. Glial cells more than support cells? Int J Biochem Cell Biol. 2007;39:661-665.
Kandel ER, Schwartz JH, Jessel T. Principles of Neural Science, 4th edn. New York: McGraw-Hill, 2000.
Kim H-J, DiBernardo AB, Sloane JA, et al. TK WAVE1 is required for oligodendrocyte morphogenesis and normal CNS myelination. J Neurosci. 2006;26:5849-5859.
Maro GS, Vermeren M, Voiculescu O, et al. Neural cap boundary cells constitute a source of neuronal and glial cells of the PNS. Nature Neurosci. 2004;7:930-938. P
Nolte J. The Human Brain. In An Introduction to Functional Anatomy, 5th edn., Edinburgh: Mosby; 2002:197-222. Ch. 9
Scherer SS, Arroyo EJ. Recent progress on the molecular organization of myelinated axons. J Peripheral Nerv Syst. 2002;7:1-12.
Review of the molecular architecture of myelinated peripheral axons and their myelin sheaths..
Seifert G, Schilling K, Steinhäuser C. Astrocyte dysfunction in neurological disorders: a molecular perspective. Nature Rev Neurosci. 2006;7:194-206.
Shepherd AJ, Downing JEG, Miyan JA. Without nerves, immunology remains incomplete – in vivo veritas. Immunology. 2005;116:145-163.
Volterra A, Meldolesi J. Astrocytes, from brain glue to communication elements: the revolution continues. Nat Rev Neurosci. 2005;6:626-640.
Wong RO, Ghosh A. Activity-dependent regulation of dendritic growth and patterning. Nature Rev Neurosci. 2002;25:803-812.