32 Intracranial Venous Revascularization
Introduction
Ignoring the cerebral venous system (CVS) during surgery would entail disastrous consequences; therefore, thorough knowledge of venous anatomy and physiology is of prime importance in intracranial neurosurgery.1–8
The “dangerous” venous structures
The Dural Venous Sinuses
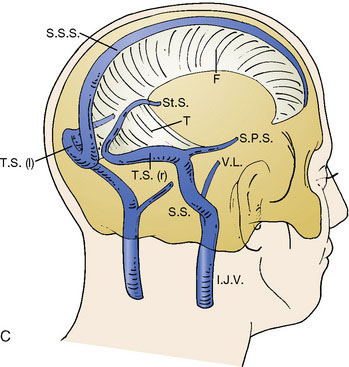
The superior sagittal sinus (SSS), a major dural sinus, has three parts. The anterior third receives the prefrontal afferent veins (Figure 32–1). It is generally admitted that its sacrifice is well tolerated. Actually, severe mental disorders, personality changes, or loss of recent memory with a general slowing of thought processes and activity, or even akinetic mutism, may occur if sacrificed. The mid-third receives the voluminous cortical veins of the central group. Interruption of this portion entails high risks of (bilateral) hemiplegia and akinesia. The posterior third, as well as the torcular Herophili, which receives the straight sinus, drains a considerable amount of blood. Interruption would inevitably provoke potentially fatal intracranial hypertension.
All of these anatomical configurations have surgical implications and must be taken into account before considering interrupting sinuses.9–11
The Deep Veins of the Brain
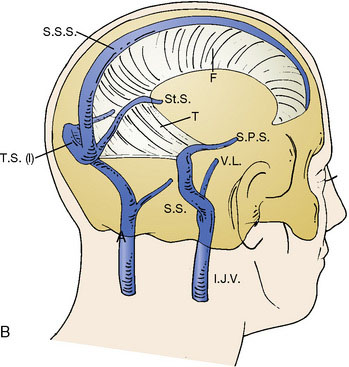
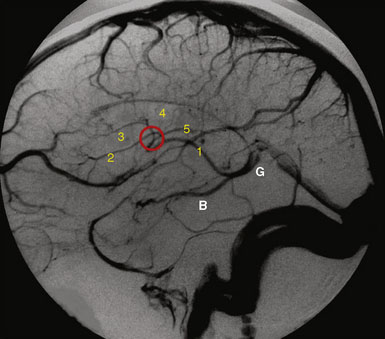
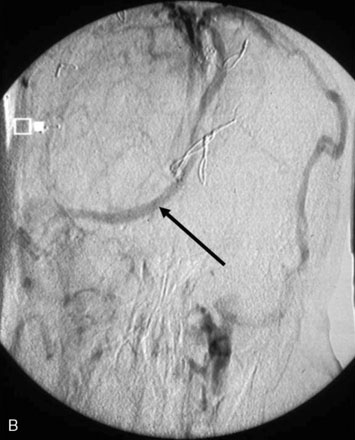
The deep veins of the brain drain toward the venous confluent of Galen (Figure 32–2). The term venous confluent is appropriate since, in addition to the two internal cerebral veins, the Galenic system receives the two basilar veins of Rosenthal, and also veins from the corpus callosum, the cerebellum (mainly the vermian precentral vein), and the occipital cortex.
Regarding the precentral vermian vein, it is generally accepted, and we agree, that its sacrifice to approach the pineal region is without danger.1–3
The Superficial Veins
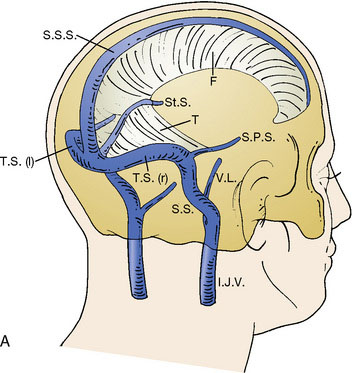
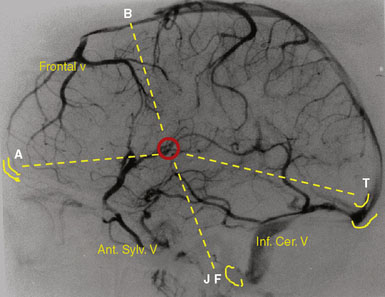
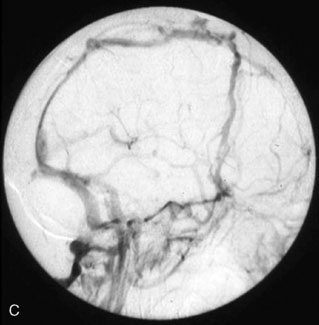
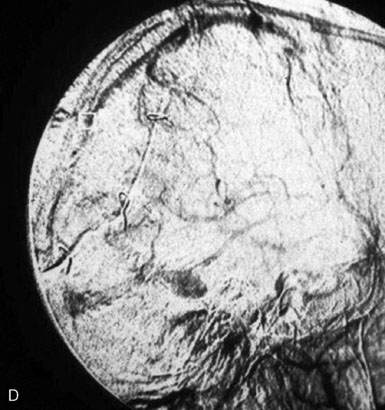
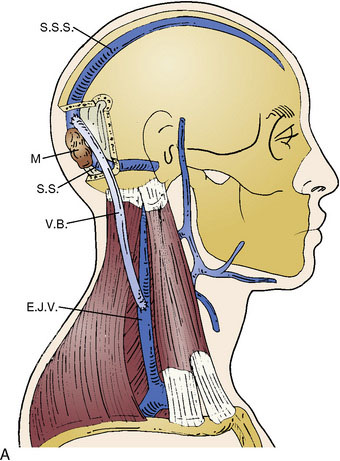
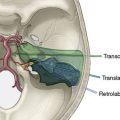
Any of the superficial veins of a certain caliber presumably have a functional role. The superficial veins belong to three “systems”: the midline afferents to the SSS, the inferior cerebral afferents to the TS, and the superficial sylvian afferents to the cavernous sinus (Figure 32–3). These three systems are strongly interconnected, but there is considerable variability among patients. The main anastomotic veins are Trolard to the SSS, Labbé to the TS, and great superficial sylvian, all of them bearing important surgical implications.
Intracranial approaches, especially skull base ones, must be prepared taking into account the organization of the superficial venous system (see Figure 32–3).7,13,14
Veins of the Posterior Fossa
Sacrifice would expose the cerebellum to swelling or infarction. The conventional statement that the superior petrosal vein, especially when voluminous, can be interrupted without danger needs to be reconsidered. Our experience is that this venous complex should be respected.1
Avoidance of venous occlusions during surgery
The role played by venous occlusions during surgery in the occurrence of postoperative hemorrhagic infarcts is undeniable.1–3,7 Retraction of the brain provokes local congestion by compressing the cortical venous network, reduction in venous flow by stretching the bridging veins, and thrombosis of veins if compression of the retractor is prolonged.15 Excessive brain retraction can be avoided by specially designed approaches and limited opening of the dura, obeying two principles, including the minimally invasive opening and bone removal associated with craniotomy at the base of the skull. Keyhole approaches or limited opening of the dura prevent excessive retraction and consequently avulsing veins.
A bridge vein acting as a limitation may occur. To be preserved, the vein has to be dissected free from the arachnoid and cortex at a length of 10 to 20 mm.16 When an important vein ruptures, its reconstruction may be considered either by resuturing or by using the silicone tubing technique.17
Wounds made in a vein wall are common. Rather than coagulating the vein, hemostasis can be attempted by simply wrapping the wall with a small piece of Surgicel (Johnson and Johnson Medical, Viroflay, France). If this is insufficient, a very localized microcoagulation with a sharp bipolar forceps or by placing a single suture with a 10-0 nylon thread is recommended. In cases with a large defect, a patching repair can be performed.18,19 In all cases, whatever the technique used, hemostasis quality has to be checked by jugular compression at the neck.
Tumors invading the major dural sinuses
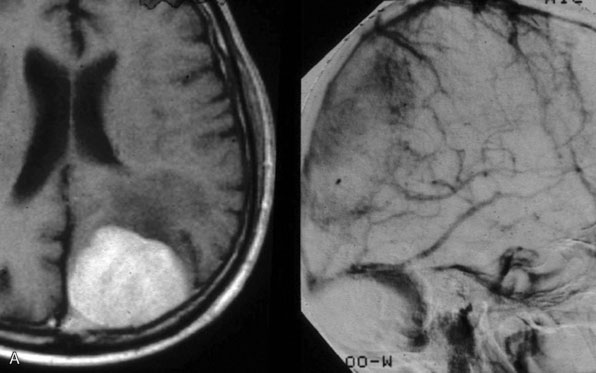
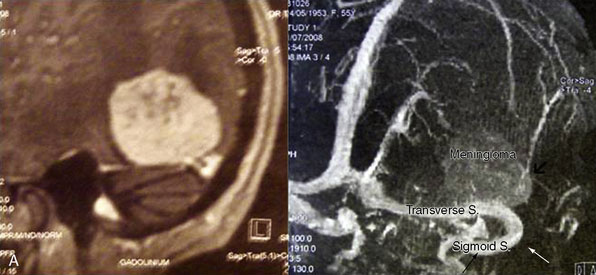
The study of long-term results in our series of 100 consecutive patients affected by meningiomas involving a major dural sinus, and in whom we attempted radical removal and venous repair,20 led us to the following conclusions. The low recurrence rate of 4% in our series, followed over a 3- to 23-year period (mean 8 years), supports resecting not only the tumor portion outside the sinus, but also the fragment invading the sinus. When radical removal is attempted, we consider venous reconstruction mandatory when the sinus is incompletely occluded, and potentially useful even in cases with complete occlusion. The goal is to restore the flow that might be compromised by impairment of the compensatory collateral channels (Figure 32–4). The traditional belief that radical removal of meningiomas with a totally occluded sinus is not dangerous must be reconsidered.21–24 In our series, the three patients who died (all three from brain swelling) had a meningioma that totally occluded the sinus and was wholly removed without any restoration of the sinus circulation.20
This is not surprising, as surgical access basically involves the destruction of some to all of the collateral pathways that naturally developed to compensate the sinus occlusion. Therefore, restoring the venous circulation at end of surgery may reduce brain swelling. Furthermore, patching or bypassing did not increase the morbidity rate in our series.20
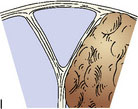
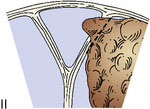
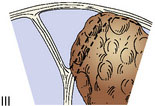
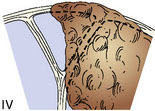
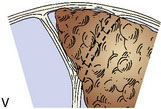
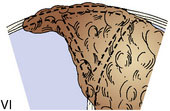
To guide in surgical decision making, we introduced a meningioma classification comprised of six types according to degree of sinus invasion1,22 (Figure 32–5). This simplified classification was developed after the ones introduced by Merrem,25 Krause,26 and Bonnal and Brotchi.27
Based on this classification, surgery should be attempted in the following ways:
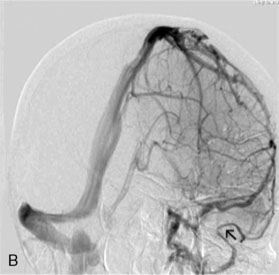
Surgery on the intracranial venous system is greatly facilitated by obeying the following technical aspects. The semi-sitting (lounging) position allows a good venous return, with a low risk of air embolism (1% in our series). Skin flap and craniotomy should extend across the midline to permit visualization of both sides of the sinus, and some 3 cm outside the margins of the occluded sinus. Approach should consider cutaneous, pericranial, and/or diploic, as well as dural venous pathways, which might be compromised during the approach. These collateral pathways can be identified on preoperative imaging (venous angio-MR and DSA with venous phases) (see Figure 32–4). Then the dura is incised in a circumferential manner around the margin of the tumor, followed by an incision along the border of the corresponding portion of the sinus. Under the microscope, attachment of the meningioma to the wall of the sinus and to the neighboring falx is deinserted by using the bipolar coagulation forceps. Then, after dural devascularization, an “intracapsular” debulking is carried out so that the meningioma can be dissected under the microscope from the underlying cortex.
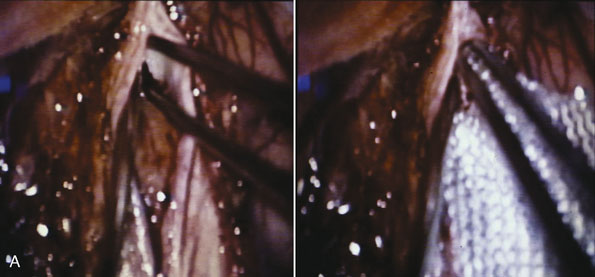
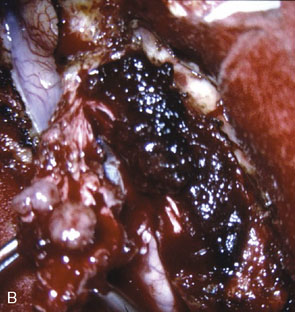
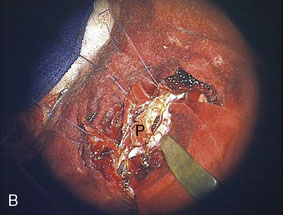
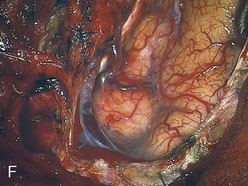
Because of frequent discrepancies between images and anatomical lesions, the sinus should be explored through a short incision to disclose any intrasinusal fragment. Control of the venous bleeding from the lumen of the sinus, can be easily obtained by plugging/packing small pledgets of Surgicel (Figure 32–6). Balloons or shunts with inflatable balloons should not be used because they do not pass easily through the sinus septa and may disrupt the sinus endothelium. Vascular clamps or aneurysm clips should be avoided because they may injure the sinus walls and afferent veins.
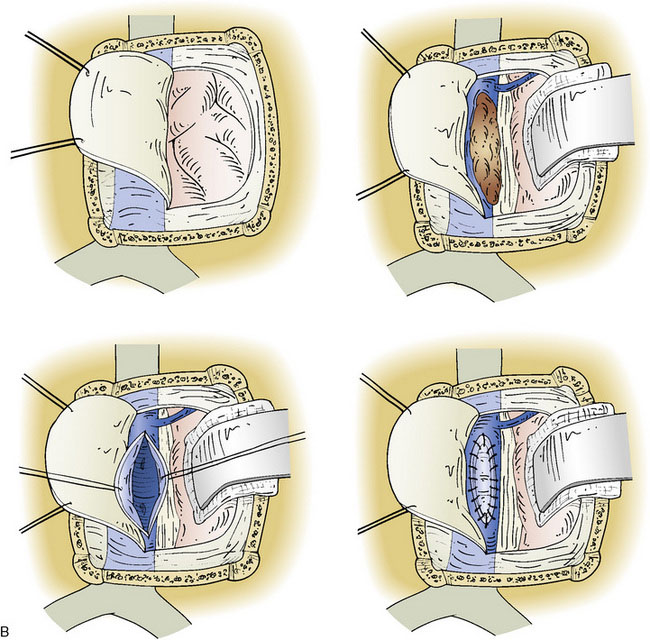
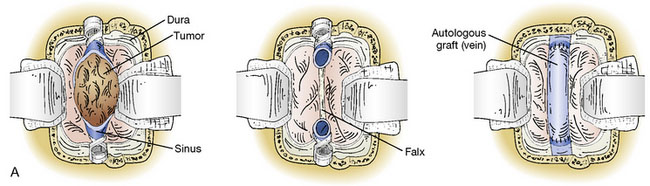
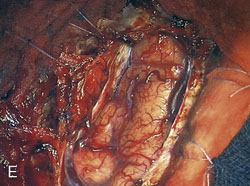
For patching, locally harvested dura mater or fascia temporalis (better than pericranium) is rigid enough to enable blood to flow inside and to use as patches. Technical steps are described in Figures 32–7 and 32–8. Also, in appropriate selective cases plasty can be performed by reflexing flap from falx.1,28
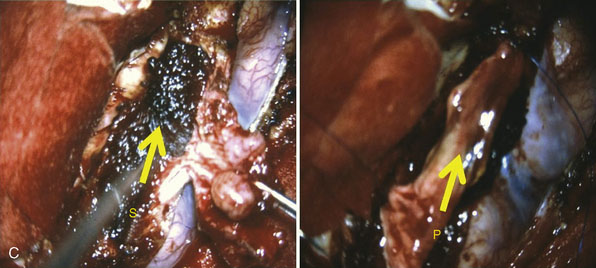
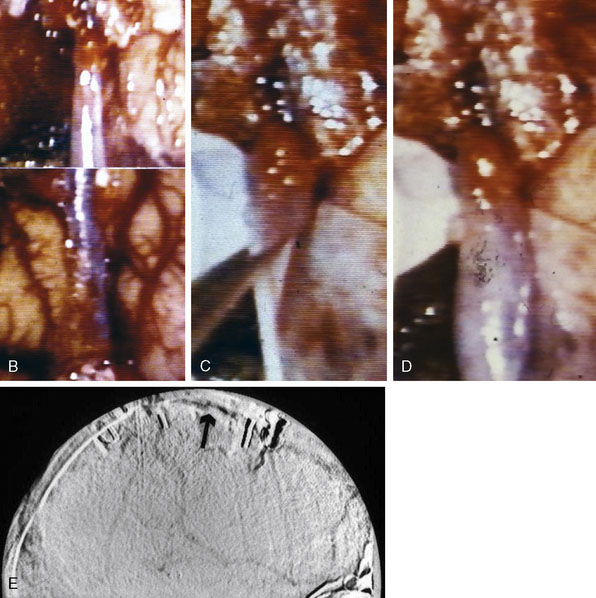
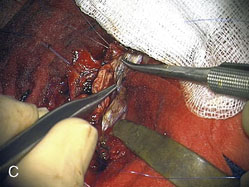
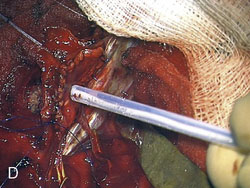
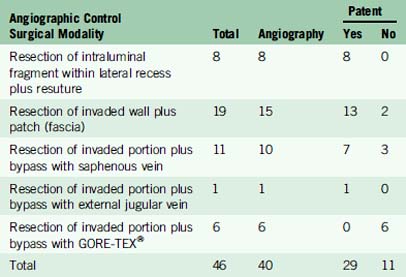
In cases with totally invaded sinus, bypassing procedures can be indicated to restore venous flow. Bypasses may be performed either immediately after removal of the occluded portion, using end-to-end anastomoses (Figure 32–9), or prior to its removal with end-to-side anastomoses (Figure 32–10). The graft should be an autologous vein: when a long graft (>10 cm) is required, the median saphenous (see Figure 32–10), or the external jugular vein when only a short graft is necessary (see Figure 32–9). GORE-TEX prosthesis should be avoided; in our six cases in whom it was used, the bypass thrombosed within the first week. Five of the thromboses were asymptomatic, but one was accompanied with an acute, but fortunately reversible, comatose state. Technical details for bypassing are provided in Figures 32–9 and 32–11. Suturing on the sinus is performed using two hemirunning sutures, with Prolene 8-0 thread (Laboratoire Ethnor, Neuilly-sur-Seine, France).
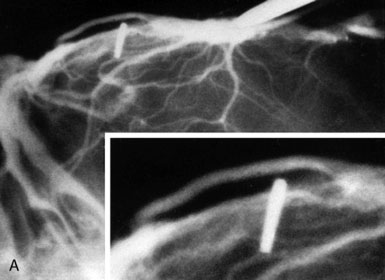
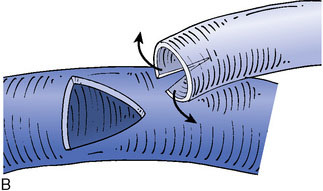
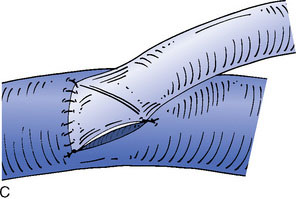
Figure 32–11 Bypass on the SSS sinus (with end-to-side anastomoses); operative technique. A, The procedure was introduced in humans in 1980,44 after experimental work in dogs in 1976.46 B, After exposure of the superficial wall (roof) of the sinus at the portion to be grafted, a sinusotomy is performed by removing an equilateral triangle of wall. Sinus hemostasis is achieved by plugging a small pledget of Surgicel into its lumen. The extremity of the graft is enlarged by a longitudinal cut. C, End-to-side anastomosis is performed as follows: triangulation with three stitches, and then running suture or interrupted stitches with an 8-0 thread on each of the three sides of the sinusotomy. Removal of the pledget of Surgicel before completing the last suture(s) must not be forgotten.
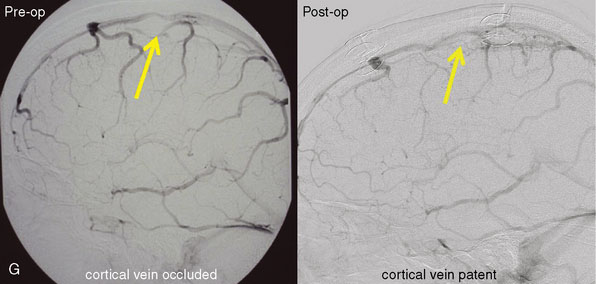
Reconstruction or reimplantation of veins may be attempted, if veins are important, such as the Labbé veins (Figure 32–12)29 or the afferent veins of the central group of the SSS (Figure 32–13).30–32
To avoid clotting, we consider postoperative anticoagulation mandatory for at least 3 months, until re-endothelization occurs. This strategy did not increase hemorrhagic complications in our series.20 Heparinotherapy is administered as soon as the morning after surgery and for the following 3 weeks, at dosages to double the coagulation time. Coumadin is then administered over the next 2 or 3 months until the (hypothetical) end of sinus, patch, graft, or endothelization.
Long-term patency of venous repair remains an unsolved problem (Table 32–1). The fact that all of the patients who had a graft that thrombosed (with the exception of one with a GORE-TEX tube) remained asymptomatic does not mean that venous reconstruction was useless. One can postulate that venous repair provides time for compensatory venous pathways to develop. Importantly, the three patients who died of brain swelling all had meningiomas with Type VI sinus invasion that had been totally resected without venous flow restoration.
In conclusion, we do not pretend that all tumors invading the major dural sinuses must be radically resected and the sinus systematically repaired. The literature discusses numerous cons33–35 and pros.36–43 Ultimately, the decision must be made after weighing benefits and risks. Before deciding to perform a radical removal with restoration of venous circulation, especially for those located in the mid-third portion of the sinus, alternatives should be discussed. For large tumors, the alternative is primary surgery to shave the tumor from the sinus and, if the tumor grows, adjuvant radiosurgery or radiotherapy. For very small meningiomas, radiosurgery can be the first option, with secondary surgery if tumor growth is not controlled.
Surgical restoration of venous flow for intracranial hypertension from venous occlusion
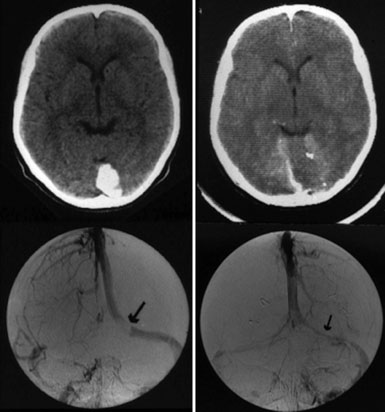
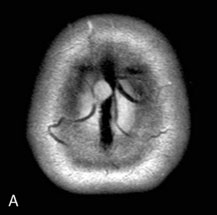
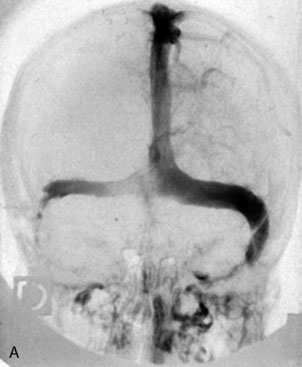
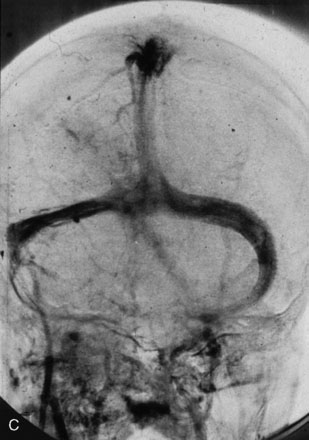
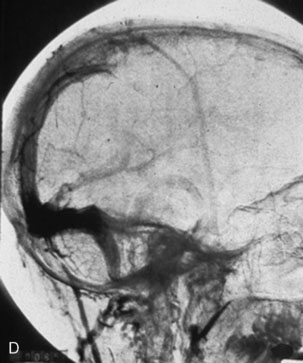
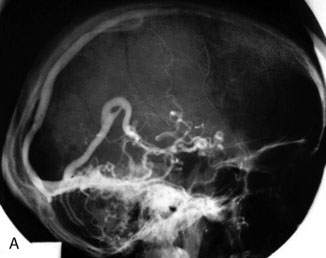
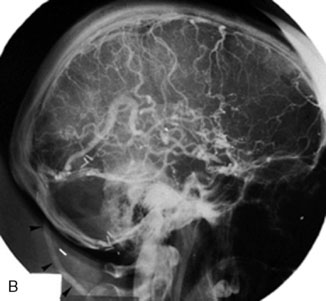
Chronic intracranial hypertension can be due to venous occlusion of the posterior third of the SSS, torcular, predominant LS, or internal jugular vein. The most frequent causes of “chronic” occlusion of dural sinuses follow: (1) fibrosis after infectious thrombophlebitis and/or cruoric thromboses44 (Figure 32–14); (2) damage or ligation after trauma or surgery; (3) tumor totally occluding the posterior third of the SSS1,20 (see Figure 32–10); and (4) dural arteriovenous fistulas accompanying (lateral) sinus thrombosis (Figure 32–15).1,45
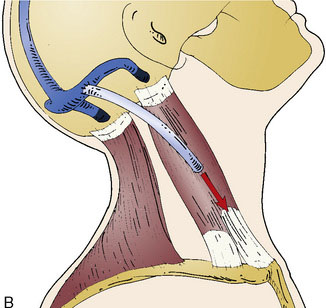
Intracranial hypertension cannot always be controlled efficiently with antiedema therapy. Lumboperitoneal CSF shunting, which appears to be a simpler technique, is not without pitfalls and complications. In the order of 20% of cases, progressive severe loss of vision and/or encephalopathy develops. Therefore, venous revascularization by sino-jugular bypass—implanted proximally to the occlusion and directed to the jugular venous system (external or internal jugular vein)—can be the solution.44 The good long-term clinical results observed in our series of five cases of sino-jugular bypasses argue in favor of this procedure as a logical treatment in these circumstances.
The original surgical technique was described in detail in 198044 and briefly more recently in 2000.1 This graft, which must generally be a long one, has to be harvested from the median saphenous vein.
1 Sindou M., Auque J. The intracranial venous system as a neurosurgeon’s perspective. Review. Adv Tech Stand Neurosurg. 2000;26:131-216.
2 Auque J., editor. Venous sacrifice in neurosurgery: risk, assessment and management. Neurochirurgie, 42. 1996;1-136. Suppl 1.
3 Sindou M., Auque J., Jouanneau E. Neurosurgery and the intracranial venous system. Acta Neurochir (Wien). 2005;94(Suppl):1686-1692.
4 Al-Mefty O., Krist A.F. The dangerous veins. In: Hakuba A., editor. Surgery of the Intracranial Venous System. Proceedings of First International Workshop on Surgery of the Intracranial Venous System, September 1994. Tokyo: Springer-Verlag; 1996:36-42.
5 Schmidek H.H., Auer L.M., Kapp J.P. The cerebral venous system. Review. Neurosurgery. 1985;17:663-678.
6 Hakuba A., editor. Surgery of the Intracranial Venous System. Proceedings of the First International Workshop on Surgery of the Intracranial Venous System, Osaka, September 1994. Tokyo: Springer-Verlag, 1996.
7 Malis L.I. Venous involvement in tumor resection. In: Hakuba A., editor. Surgery of the Intracranial Venous System. Proceedings of the First International Workshop on Surgery of the Intracranial Venous System, Osaka, September 1994. Tokyo: Springer-Verlag; 1996:281-288.
8 Yasargil M.G. Microneurosurgery, vol 1. Microsurgical Anatomy of the Basal Cisterns and Vessels of the Brain, Diagnostic and Studies. Stuttgart, New York: Thieme, 1984.
9 Alaywan M., Sindou M. Surgical anatomy of the lateral sinus approaches in the sigmoid region. In: Hakuba A., editor. Surgery of the Intracranial Venous System. Proceedings of the First International Workshop on Surgery of the Intracranial Venous System, Osaka, September 1994. Tokyo: Springer-Verlag; 1996:63-72.
10 Sindou M., Alaywan F., Hallacq P. Main dural sinuses surgery. Neurochirurgie Suppl. 1996;1:45-87.
11 Sindou M., Hallacq P. Venous reconstruction in surgery of meningiomas invading the sagittal and transverse sinuses. Skull Base Surg. 1998;8:57-64.
12 Kanno T., Kasama A., Shoda M., et al. pitfall in the interhemispheric translamina terminalis approach for the removal of a craniopharyngioma. Significance of preserving draining veins. Part I. Clinical study. Surg Neurol. 1989;32:111-115.
13 Samii M. Skull Base Surgery. Anatomy, Diagnosis and Treatment. Proceedings of the First International Skull Base Congress, Hannover, 1992. Basel: Karger, 1994.
14 Sindou M., Emery E., Acevedo G., et al. Respective indications for orbital rim, zygomatic arch and orbito-zygomatic osteotomies in the surgical approach to central skull base lesions. Critical, retrospective, review in 146 cases. Acta Neurochir. 2001;143:967-975.
15 Kasama A., Kanno T.A. pitfall in the interhemispheric translamina terminalis approach for the removal of a craniopharyngioma. Significance of preserving draining veins. Part II. Experimental study. Surg Neurol. 1989;32:116-120.
16 Sugita K., Kobayashi S., Yokoo A. Preservation of large bridgings veins during brain retraction. Technical note. J Neurosurg. 1982;57:856-858.
17 Sakaki T., Morimoto T., Takemura K., et al. Reconstruction of cerebral cortical veins using silicone tubing. Technical note. J Neurosurg. 1987;66:471-473.
18 Donaghy R.M., Wallman L.J., Flanagan M.J., et al. Sagittal sinus repair. Technical note. J Neurosurg. 1973;38:244-248.
19 Kapp J.P., Gielchinsky I., Deardourff S.L. Operative techniques for management of lesions involving the dural venous sinuses. Surg Neurol. 1977;7:339-342.
20 Sindou M., Alvernia J. Results of attempted radical tumor removal and venous repair in 100 consecutive meningiomas involving the major dural sinuses. J Neurosurg. 2006;105:514-525.
21 Oka K., Go Y., Kimura H., et al. Obstruction of the superior sagittal sinus caused by parasagittal meningiomas: the role of collateral venous pathways. J Neurosurg. 1994;81:520-524.
22 Sindou M. Meningiomas invading the sagittal or transverse sinuses, resection with venous reconstruction. J Clin Neurosci. Suppl, 2001;8-11.
23 Waga S., Handa H. Scalp veins as collateral pathways with parasagittal meningiomas occluding the superior sagittal sinus. Neuroradiology. 1976;11:199-204.
24 Sindou M., Hallacq P. Microsurgery of the venous system in meningiomas invading the major dural sinuses. In: Hakuba A., editor. Surgery of the Intracranial Venous System. Proceedings of the First International Workshop on Surgery of the Intracranial Venous System, Osaka, September 1994. Tokyo: Springer-Verlag; 1996:226-236.
25 Merrem G. Die parasagittalen Meningeome. Quoted in Fedor Krause memorial lecture. Acta Neurochir. 1970;23:203-216.
26 Krause F. Operative Frilegung der Vierhügel, nebst Beobachtungen über Hirndruck und Dekompression. Zentralbl Neurochir. 1926;53:2812-2819.
27 Bonnal J., Brotchi J. Surgery of the superior sagittal sinus in parasagittal meningiomas. J Neurosurg. 1978;48:935-945.
28 Masuzawa H. Superior sagittal sinus plasty using falx flap in parasagittal meningioma. Noshinkei Geka. 1977;5:707-713.
29 Guclu B., Sindou M. Reconstruction of vein of Labbé in temporo-occipital meningioma invading transverse sinus: technical report. Acta Neurochirur. 2010;152:941-945.
30 Menovsky T., De Vries J. Cortical vein end-to-end anastomosis after removal of a parasagittal meningioma. Microsurgery. 2002;22:27-29.
31 Murata J., Sawamura Y., Saito H., et al. Resection of a recurrent parasagittal meningioma with cortical vein anastomosis: technical note. Surg Neurol. 1997;48:592-597.
32 Steiger H.J., Reulen H.J., Huber P., et al. Radical resection of superior sagittal sinus meningioma with venous interpostion graft and reimplantation of the rolandic veins. Case report. Acta Neurochir (Wien). 1989;100:108-111.
33 Di Meco F., Li K.W., Casali C., et al. Meningiomas invading the superior sagittal sinus: surgical experience in 108 cases. Neurosurgery. 2004;55:1263-1274.
34 Logue V. Parasagittal meningiomas. Adv Tech Stand Neurosurg. 1975;2:171-198.
35 Brotchi J., Patay Z., Baleriaux D. Surgery of the superior sagittal sinus and neighbouring veins. In: Hakuba A., editor. Surgery of the Intracranial Venous System. Proceedings of the First International Workshop on Surgery of the Intracranial Venous System, Osaka, September 1994. Tokyo: Springer-Verlag; 1996:207-219.
36 Bederson J.B., Eisenberg M.B. Resection and replacement of the superior sagittal sinus for treatment of a parasagittal meningioma: technical case report. Neurosurgery. 1995;37:1015-1019.
37 Bonnal J. Conservative and reconstructive surgery of the superior longitudinal sinus. Neurochirurgie. 1982;52:147-172.
38 Bonnal J., Brotchi J., Stevenaert A., et al. Excision of the intrasinusal portion of rolandic parasagittal meningiomas, followed by plastic surgery of the superior longitudinal sinus. Neurochirurgie. 1971;17:341-354.
39 Bonnal J., Buduba C. Surgery of the central third of the superior sagittal sinus. Experimental study. Acta Neurochir. 1974;30:207-215.
40 Hakuba A. Reconstruction of dural sinus involved in meningiomas. In: Al-Mefty O., editor. Meningiomas. New York: Raven Press; 1991:371-382.
41 Hakuba A., Huh C.W., Tsujikawa S., et al. Total removal of a parasagittal meningioma of the posterior third of the sagittal sinus and its repair by autogenous vein graft. Case report. J Neurosurg. 1979;51:379-382.
42 Nagashima H., Kobayashi S., Takemae T., et al. Total resection of torcular herophili hemangiopericytoma with radial artery graft case report. Neurosurgery. 1995;36:1024-1027.
43 Sekhar L.N., Tzortzidis F.N., Bejjani G.K., et al. Saphenous vein graft bypass of the sigmoid sinus and jugular bulb during the removal of glomus jugular tumors. Report of two cases. J Neurosurg. 1997;86:1036-1041.
44 Sindou M., Mercier P., Bokor J., et al. Bilateral thrombosis of the transverse sinuses: microsurgical revascularization with venous bypass. Surg Neurol. 1980;13:215-220.
45 Schmid-Elsaesser R., Steiger H.J., Yousry T., et al. Radical resection of meningiomas and arteriousvenous fistulas involving critical dural sinus segments: Experience with intraoperative sinus pressure monitoring and elective sinus reconstruction in 10 patients. Neurosurgery. 1997;41:1005-1018.
46 Sindou M., Mazoyer J.F., Fischer G., et al. Experimental bypass for sagittal sinus repair. Preliminary report. J Neurosurg. 1976;44:325-330.