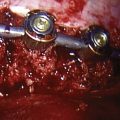
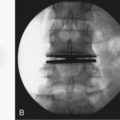
CHAPTER 60 Interspinous Process Decompressive Devices
The interspinous process space has increasingly become a target for spinal implants to address degenerative conditions of the lumbar spine. Interspinous process decompressive (IPD) devices employ a range of insertion techniques and materials but share a common goal—that distraction be maintained between the adjacent spinous processes in order to incur a clinical result. The variety of materials employed include titanium, polyether ether keytone (PEEK), silastic compounds, and allograft. Many of the implants are devised to be static in nature, while others are dynamic.1,2 The X-Stop is a titanium implant. It is the only device marketed in the United States that, at the time of publishing, has been cleared by the U.S. Food and Drug Administration (FDA) through an investigational device exemption (IDE) study. The indication for its use is spinal stenosis leading to neurogenic claudication. Other diagnoses potentially helped by this technology, but yet to be cleared by the FDA, include discogenic back pain, facet arthropathy, disc herniation, degenerative disc disease, and instability including degenerative spondylolisthesis. IPDs all share characteristics that are relatively unique among spinal implants of the lumbar spine. They can be implanted with a modest degree of destruction to the local anatomy. They do not require exposure to the neural elements, they are at least partially motion preserving, and they are relatively reversible. These features coalesce to an implant with a favorable risk profile. It is up to randomized controlled studies to prove the efficacy, indications, and ultimately role in the armamentarium available to the spine surgeon in the care of the spinal patient.
Design Rationale
The first reasonable question to pose in evaluating the design rationale of an IPD device is its net effect on implantation of the dimensions of the spinal canal. Richards and colleagues3 attempted to address this question in studying eight cadaver specimens from L2-L5 that underwent an MRI before and after implantation of an X-Stop device at the L3-4 level. Canal and foraminal dimensions were measured. The specimens were positioned, and parameters were measured in both 15 degrees of flexion and 15 degrees of extension. In extension, the canal area was increased by 18% when compared with the noninstrumented spine. Similarly the subarticular diameter was increased by 50%, the canal diameter by 10%, the foraminal area by 25%, and the foraminal width by 41%.3 In a subsequent in vivo study, Siddiqui and colleagues4,5 presented results on 12 patients with 17 instrumented levels in which positional MRIs were obtained before and after surgery in the sitting flexed, extended, neutral, and standing positions. The area for the dural sac increased from 77.8 to 93.4 mm at 6 months after surgery in the standing position. There was a similar increase in the foramina. Importantly, no change in overall lumbar lordosis was noted.4,5
The other area of study as it relates to implantation of the IPD device is the net effect on the kinematics and load sharing within the lumbar spine at both the instrumented level and the adjacent levels.6 Swanson and colleagues presented data on eight human cadaveric lumbar spines in which they tested intradiscal pressure, before and after implantation. The spines were positioned in flexion, neutral, and extension with intradiscal pressure transducers placed in the anterior and posterior aspect of the nucleus pulposus. The implants were placed at L3-L4, and the measurements of intradiscal pressure were taken at L2-L3, L3-L4, and L4-L5. The device proved to be load sharing in both extension and neutral positions. At L3-L4, which was the instrumented level, the authors measured a 63% decrease in pressure at the posterior annulus and a 41% decrease in pressure in the nucleus pulposus. In neutral position the decrease in pressure was 38% in the posterior annulus and 20% in the nucleus pulposus. The adjacent levels did not show any significant change in intradiscal pressure.7
Wiseman and colleagues presented a similar study as it relates to facet loading. Pressure film was placed in the facets at the instrument level, which was L3-L4, as well as the facets at L2-L3 and L4-L5. The film could then be measured for contact area, mean force, mean pressure, and peak pressure. At the implanted level the contact area decreased by 47%, mean force decreased by 68%, mean pressure by 39%, and peak pressure by 55%. No changes of facet pressure were seen at adjacent levels.8 These mechanical studies provide the basis for the assumption that IPD devices may be helpful in the clinical treatment of patients suffering from facet arthropathy or discogenic or degenerative disc disease-induced back pain. Yet these are only biomechanical studies. The efficacy of IPD devices has not been shown clinically in these conditions.
The effect of the X-Stop IPD device on spinal kinematics was further measured by Lindsey and colleages.9 Seven cadaveric specimens from L2-L5 were loaded in flexion, extension, axial rotation, and lateral bending to 7.5 Nm. There was a superimposed axial load of 700 Newtons. Rigid markers were placed in each vertebral body, as well as the supporting frame to measure the relative motion. Measurements were taken both before and after implantation of an L3-L4 IPD device. There was no change in range of motion as measured in axial rotation or lateral bending. The intervertebral angle was changed by 1.9 degrees. An average of 7.6 degrees of extension at L3-L4 was reduced to 3.1 degrees after implantation of the device. Notably, the adjacent levels were not affected in flexion or extension with a device in place.9
Two studies are presented to assess the question of kyphosis of the lumbar spine. Siddiqui and colleagues, as referenced earlier, studied 12 patients with 17 implanted levels. Comparing his postoperative with preoperative MRIs, the mean intervertebral angle changed .83 degrees in extension. The over mean lumbar lordosis changed .08 degrees in extension. Therefore the change in overall lumbar lordosis was not statistically significant.4–6 The mean intervertebral angle and mean lumbar lordosis were also measured in the pivotal study trial for FDA submission. This included 41 patients with data available preoperatively and postoperatively. In those patients undergoing a single-level implant, of which there were 23, the mean lumbar lordosis changed .1 degrees in extension. In 18 patients who underwent double-level implants the mean lumbar lordosis changed 1.2 degrees in extension.10
Clinical Results
The X-Stop device is the most widely used IPD in the United States and therefore has the most abundant clinical support in the literature.11–20 The technology is new, however, and long-term support from varied centers is not present, particularly when compared with traditional surgical approaches.
Complications included incisional pain, hematoma, wound swelling, wound dehiscence, implant dislodgement, implant malposition, spinous process fracture, coronary ischemia, and respiratory distress.10
Anderson and colleagues restudied a subgroup of 75 patients in the initial cohort who carried the additional diagnosis of grade 1 degenerative spondylolisthesis. This subgroup of patients similarly followed the index group by showing statistically significant pain reduction and increase in physical function obtained by the 6-week follow-up and maintained for the 2-year study period. Additionally, there was no increase in the degree of spondylolisthesis in the study group.11
Verhoof and colleagues18 reported on 12 patients treated with the X-Stop device for spinal stenosis in the setting of degenerative spondylolisthesis. The authors also showed no progression of the deformity, but four of their initial patients showed no improvement in symptoms. Although eight patients reported complete resolution of their symptoms, three of these eventually developed recurrent symptoms for a failure rate of 58%.18
Kondrashov and colleagues presented further results of a subgroup of the initial cohort with 4 years’ follow-up. Among the patients, 77% had experienced at least a 15-point improvement in their Oswestry Disability Index (ODI) maintained at 4 years postoperation.15
Available Products
As stated previously, the X-Stop device is the only motion-preserving IPD device, at the time of writing, with FDA approval for the treatment of spinal stenosis through an IDE pathway (Table 60–1). Other products such as the Coflex, Suprion, Wallis, and Diam are in various stages of IDE investigation and currently marketed overseas. Products such as Aspen and Extensure have been approved by the FDA via a 510k pathway as fusion devices. These devices are placed in the interspinous space and apply distraction with the additional stated goal of fusion rather than motion preservation.
| Company | Product | Material |
|---|---|---|
| Sintea Biotech | Viking | PEEK |
| Globus | Flexus | Radiolucent polymer, titanium alloy, and tantalum |
| Biomech Paonan | Rocker | PEEK-OPTIMA |
| Ackermann Medical | Maxx Spine | |
| Blackstone | InSwing | PEEK and a polyester band |
| Pioneer | Bacjac | PEEK |
| Synthes | In-Space | PEEK |
| Paradign | Coflex | Titanium |
| Abbott/Zimmer | Wallis | PEEK |
| Medtronic | Aperious, Diam, and X-Stop | X-Stop—Titanium |
| Vertiflex | Superion | Titanium |
| Eden Spine | Wellex | Titanium |
| GMReis | IS Dynamic Fixation System | |
| Lanx | Aspen | Titanium fusion device |
| Nuvasive | Extensure | Allograft fusion device |
PEEK, polyetheretherkeytone.
Summary
1 Siddiqui M, Nicol M, Karadimas E, et al. The positional magnetic resonance imaging changes in the lumbar spine following insertion of a novel interspinous process distraction device. Spine. 2005;30:2677-2682.
2 Lindsey DP, Swanson KE, Fuchs P, et al. The effects of an interspinous implant on the kinematics of the instrumented and adjacent levels in the lumbar spine. Spine. 2003;28:2192-2197.
3 Zucherman JF, Hsu KY, Hartjen CA, et al. A multicenter, prospective, randomized trial evaluating the X STOP interspinous process decompression system for the treatment of neurogenic intermittent claudication: two-year follow-up results. Spine. 2005;30:1351-1358.
1 Bono CM, Vaccaro AR. Interspinous process devices in the lumbar spine. J Spinal Disord Tech. 2007;20:255-261.
2 Wilke HJ, Drumm J, Häussler K, et al. Biomechanical effect of different lumbar interspinous implants on flexibility and intradiscal pressure. Eur Spine J. 2008;7:1049-1056.
3 Richards JC, Majumdar S, Lindsey DP, et al. The treatment mechanism of an interspinous process implant for lumbar neurogenic intermittent claudication. Spine. 2005;30:744-749.
4 Siddiqui M, Karadimas E, Nicol M, et al. Influence of X Stop on neural foramina and spinal canal area in spinal stenosis. Spine. 2006;31:2958-2962.
5 Siddiqui M, Nicol M, Karadimas E, et al. The positional magnetic resonance imaging changes in the lumbar spine following insertion of a novel interspinous process distraction device. Spine. 2005;30:2677-2682.
6 Siddiqui M, Karadimas E, Nicol M, et al. Effects of X-STOP device on sagittal lumbar spine kinematics in spinal stenosis. J Spinal Disord Tech. 2006;19:328-333.
7 Swanson KE, Lindsey DP, Hsu KY, et al. The effects of an interspinous implant on intervertebral disc pressures. Spine. 2003;28:26-32.
8 Wiseman CM, Lindsey DP, Fredrick AD, et al. The effect of an interspinous process implant on facet loading during extension. Spine. 2005;30:903-907.
9 Lindsey DP, Swanson KE, Fuchs P, et al. The effects of an interspinous implant on the kinematics of the instrumented and adjacent levels in the lumbar spine. Spine. 2003;28:2192-2197.
10 Zucherman JF, Hsu KY, Hartjen CA, et al. A multicenter, prospective, randomized trial evaluating the X STOP interspinous process decompression system for the treatment of neurogenic intermittent claudication: two-year follow-up results. Spine. 2005;30:1351-1358.
11 Anderson PA, Tribus CB, Kitchel SH. Treatment of neurogenic claudication by interspinous decompression: application of the X STOP device in patients with lumbar degenerative spondylolisthesis. J Neurosurg Spine. 2006;4:463-471.
12 Brussee P, Hauth J, Donk RD, et al. Self-rated evaluation of outcome of the implantation of interspinous process distraction (X-Stop) for neurogenic claudication. Eur Spine. 2008;17:200-203.
13 Floman Y, Millgram MA, Smorgick Y, et al. Failure of the Wallis interspinous implant to lower the incidence of recurrent lumbar disc herniations in patients undergoing primary disc excision. J Spinal Disord Tech. 2007;20:337-341.
14 Gunzburg R, Szpalski M. The conservative surgical treatment of lumbar spinal stenosis in the elderly. Eur Spine J. 2003;12(Suppl 2):S176-S180.
15 Kondrashov DG, Hannibal M, Hsu KY, et al. Interspinous process decompression with the X-STOP device for lumbar spinal stenosis: a 4-year follow-up study. J Spinal Disord Tech. 2006;19:323-327.
16 Sénégas J, Vital JM, Pointillart V, et al. Long-term actuarial survivorship analysis of an interspinous stabilization system. Eur Spine J. 2007;16:1279-1287.
17 Siddiqui M, Smith FW, Wardlaw D. One-year results of X Stop interspinous implant for the treatment of lumbar spinal stenosis. Spine. 2007;32:1345-1348.
18 Verhoof OJ, Bron JL, Wapstra FH, et al. High failure rate of the interspinous distraction device (X-Stop) for the treatment of lumbar spinal stenosis caused by degenerative spondylolisthesis. Eur Spine J. 2008;17:188-192.
19 Weiner BK. Interspinous process decompression system (X STOP) device affords superior outcomes and equal safety to nonoperative therapy. Spine. 2005;30:2846-2847.
20 Zucherman JF, Hsu KY, Hartjen CA, et al. A prospective randomized multi-center study for the treatment of lumbar spinal stenosis with the X STOP interspinous implant: 1-year results. Eur Spine J. 2004;13:22-31.