CHAPTER 20 Cerebellum
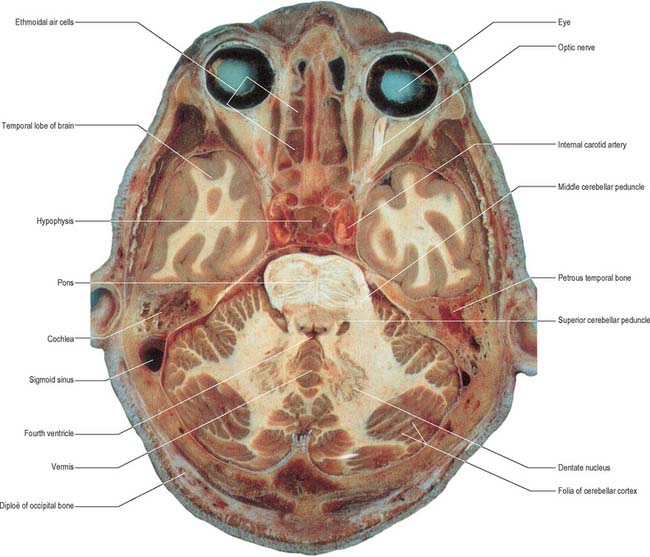
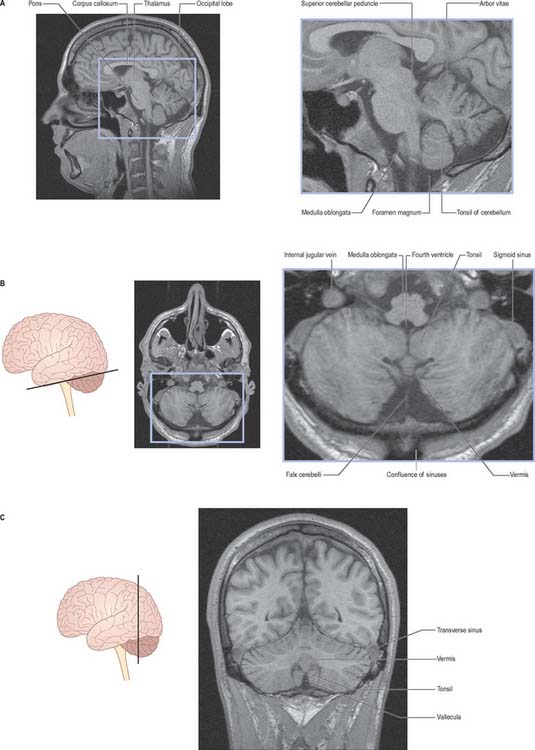
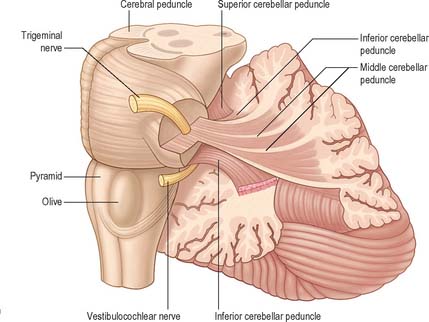
The cerebellum occupies the posterior cranial fossa, separated from the occipital lobes of the cerebral hemispheres by the tentorium cerebelli. It is the largest part of the hindbrain; in adults the weight ratio of cerebellum to cerebrum is approximately 1 : 10 and in infants 1 : 20. The cerebellum lies dorsal to the pons and medulla, from which it is separated by the fourth ventricle. It is joined to the brain stem by three bilaterally paired cerebellar peduncles, and these contain all the afferent and efferent fibres associated with the cerebellum.
EXTERNAL FEATURES AND RELATIONS
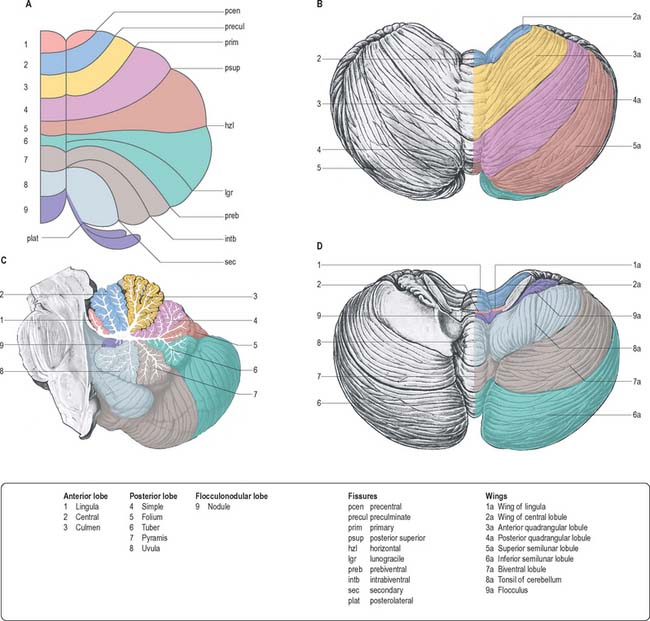
The cerebellum consists of two large, laterally located hemispheres which are united by a midline vermis (Figs 20.1, 20.2, 20.3). The superior surface of the cerebellum, which would constitute the anterior part of the unrolled cerebellar cortex, has a flat profile (Fig. 20.3). The paramedian sulci are shallow and the borders between vermis and hemispheres are indicated by kinks in the transverse fissures. The superior surface adjoins the tentorium cerebelli and projects beyond its free edge. The transverse sinus borders the cerebellum at the point where the superior and inferior surfaces meet. The inferior surface is characterized by a massive enlargement of the cerebellar hemispheres, which extend medially to overlie some of the vermis. Deep paramedian sulci demarcate the vermis from the hemispheres. Posteriorly the hemispheres are separated by a deep vallecula, which contains the dural falx cerebelli. The inferior cerebellar surface lies against the occipital squama. The shape of the surface facing the brain stem is irregular and forms the roof of the fourth ventricle and the lateral recesses on each side of it, while the cerebellar peduncles define the diamond shape of the ventricle when viewed from behind. Anterolaterally the cerebellum lies against the posterior surface of the petrous part of the temporal bone.
FUNCTIONAL DIVISIONS OF THE CEREBELLUM
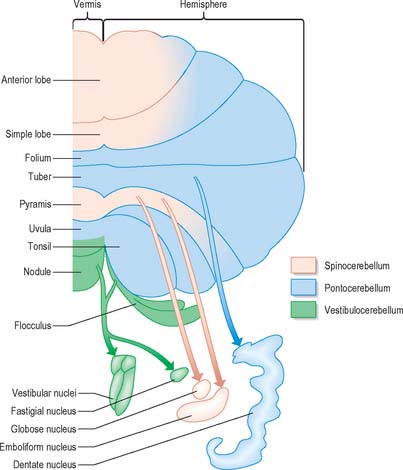
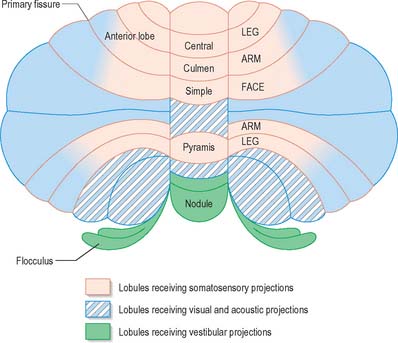
Functionally speaking, the cerebellum can be divided into a body, with inputs mainly from the spinal cord and pontine nuclei, and a flocculonodular lobe, which has strong afferent and efferent connections with the vestibular nuclei (Fig. 20.4). The body is subdivided into a series of regions dominated by their spinal or pontine inputs. The anterior lobe, simple lobule, pyramis and biventral lobules are the main recipients of spinal and trigeminal cerebellar afferents. Pontocerebellar input dominates in the folium, tuber and uvula, and throughout the entire hemisphere, including those regions that receive afferents from the spinal cord.
INTERNAL STRUCTURE
CEREBELLAR NUCLEI
Laterally, on either side within the white matter core, are four cerebellar nuclei. These are the dentate, emboliform, globose and fastigial nuclei. The dentate nucleus, which is located most laterally and is by far the largest, is the only nucleus easily visible to the naked eye (Fig. 20.1). It has the form of an irregularly folded sheet of neuronal cell bodies, with a medially directed hilum through which pass a mass of fibres mainly derived from dentate neurones and which form the bulk of the superior cerebellar peduncle. The emboliform and globose nuclei lie medial to the dentate and are equated to the nucleus interpositus (interposed nucleus) in lower species; the emboliform and globose nuclei may sometimes be referred to as the anterior and posterior interposed nuclei, respectively. Their efferent fibres join the superior cerebellar peduncle. The fastigial nucleus lies next to the midline, bordering on the roof of the fourth ventricle. A large proportion of the efferent fibres that leave this nucleus decussate within the cerebellar white matter and subsequently constitute the uncinate fasciculus, which passes dorsal to the superior cerebellar peduncle to enter the contralateral vestibular nuclei. Uncrossed fastigiobulbar fibres enter the vestibular nuclei by passing along the lateral angle of the fourth ventricle, and some fibres of the fastigial nucleus ascend in the superior cerebellar peduncle.
CEREBELLAR PEDUNCLES
Three pairs of peduncles connect the cerebellum with the brain stem (see Ch. 19; Fig. 20.5).
The superior cerebellar peduncle contains all of the efferent fibres from the dentate, emboliform and globose nuclei and a small fascicle from the fastigial nucleus. Its fibres decussate in the caudal mesencephalon, on their way to synapse in the contralateral red nucleus and thalamus. The anterior spinocerebellar tract reaches the upper part of the pontine tegmentum before looping down within this peduncle to join the spinocerebellar fibres entering through the restiform body.
CEREBELLAR CORTEX
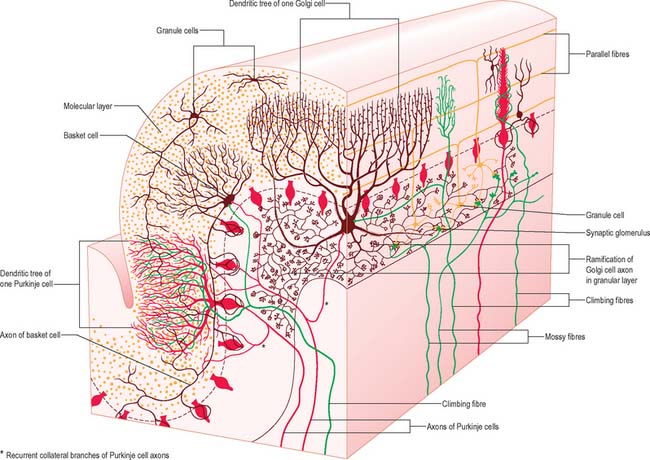
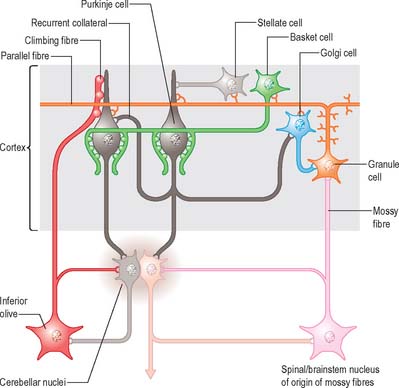
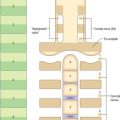
There are three main layers: molecular, Purkinje cell and granular (Fig. 20.6). The main circuit of the cerebellum involves granule cells, Purkinje cells and neurones in the cerebellar nuclei. Granule cells receive the terminals of the mossy fibre afferents (i.e. all afferent systems except the olivocerebellar fibres). The axons of the granule cells ascend to the molecular layer, where they bifurcate into parallel fibres, which are so called because they are oriented parallel to the transverse fissures and perpendicular to the dendritic trees of the Purkinje cells on which they terminate. Purkinje neurones are large and are the sole output cells of the cerebellar cortex. Their axons terminate in the cerebellar nuclei and in the vestibular nuclei. In addition to the dense array of parallel fibres, the dendritic trees of Purkinje cells receive terminals from climbing fibres which originate from neurones in the inferior olivary nucleus. The cerebellar cortex thus receives two distinct types of input: olivocerebellar climbing fibres, which synapse directly on Purkinje neurones, and mossy fibres, which connect to the Purkinje cells via granular neurones, whose axons are the parallel fibres.
The molecular layer is approximately 300–400 μm thick. It contains a sparse population of neurones, dendritic arborizations, non-myelinated axons and radial fibres of neuroglial cells. Purkinje cell dendritic trees extend towards the surface and spread out in a plane perpendicular to the long axis of the cerebellar folia. Purkinje cell dendrites are flattened. The lateral extent of the Purkinje cell dendrites is some 30 times greater in the transverse plane than it is in a plane parallel to the cerebellar folia. Parallel fibres are the axons of granule cells, the stems of which ascend into the molecular layer where they bifurcate at T-shaped branches. The two branches extend in opposite directions as parallel fibres along the axis of a folium. Parallel fibres terminate on the dendrites of the Purkinje cells and Golgi cells, which they pass on their way, and on the basket and stellate cells of the molecular layer. Dendritic trees of Golgi neurones reach towards the surface. Unlike the flattened dendritic tree of the Purkinje cell, Golgi cell dendrites span the territory of many Purkinje neurones longitudinally as well as transversely. These dendrites receive synapses from parallel fibres. Some Golgi cell dendrites enter the granular layer, where they contact mossy fibre terminals. The cell bodies of Golgi neurones lie below, in the superficial part of the granular layer. The molecular layer also contains the somata, dendrites and axons of stellate neurones (which are located superficially within the molecular layer) and of basket cells (whose somata lie deeper within the molecular layer). Climbing fibres, the terminals of olivocerebellar fibres, ascend through the granular layer to contact Purkinje dendrites in the molecular layer. Radiating branches from large epithelial (Bergmann) glial cells give off processes that surround all neuronal elements, except at the synapses. Their conical expansions join to form an external limiting membrane at the surface of the cerebellum.
The granular layer (Fig. 20.6) is about 100 μm thick in the fissures and 400–500 μm on foliar summits. There are approximately 2.7 million granular neurones per cubic millimetre. It has been estimated that the human cerebellum contains a total of 4.6 × 1010 granule cells, and that there are 3000 granule cells for each Purkinje cell.
Of the five cell types to be described, the first four are inhibitory, liberating γ-aminobutyric acid (GABA), and the fifth is excitatory, liberating L-glutamate. Figure 20.7 summarizes their main connections.
Purkinje cells have a specific geometry, which is conserved in all vertebrate classes (Fig. 20.6). They are arranged in a single layer between the molecular and granular layers. Individual Purkinje cells are separated by about 50 μm transversely and 50–100 μm longitudinally. Their somata measure approximately 50–70 μm vertically and 30–35 μm transversely. The subcellular structure of the Purkinje cell is similar to that of other neurones. One distinguishing feature is subsurface cisterns, often associated with mitochondria, which are present below the plasmalemma of somata and dendrites and may penetrate into the spines. The cisterns are intracellular calcium stores which are important links in the second messenger systems of the cell.
Each granule cell has a spherical nucleus, 5–8 μm in diameter, with a mere shell of cytoplasm containing a few small mitochondria, ribosomes and a diminutive Golgi complex. Granule cells give rise to 3 to 5 short dendrites, which end in claw-like terminals within the synaptic glomeruli. The fine axons of granule cells enter the molecular layer and branch at a T-junction to form parallel fibres passing in opposite directions over a distance of several millimetres. Terminals located along the parallel fibres give them a beaded appearance and are sites of synapses upon the dendrites of Purkinje, stellate, basket and Golgi cells in the molecular layer. Most numerous are the synapses with Purkinje dendritic spines. It had been estimated that about 250,000 parallel fibres cross a single Purkinje dendritic tree, although every parallel fibre may not synapse with each dendritic tree that it crosses.
Structural and functional cerebellar localization
A double, mirrored, localization exists in the anterior and posterior cerebellum (Fig. 20.8). The anterior lobe, simple lobule, pyramis and the adjoining lobules of the hemisphere of the posterior lobe all receive branches from the same mossy and climbing fibres and project to the same cerebellar nuclei. The efferent pathways of these regions monitor the activity in the corticospinal tract and in the subcortical motor systems descending from the vestibular nuclei and reticular formation. The inputs to the cerebellum and the outputs from it are organized according to the same somatotopical patterns, but the orientation of these patterns is reversed. The representation of the head is found principally in the simple lobule, and caudally in a corresponding region of the posterior lobe. The double representation of the body follows in rough somatotopic order. Vestibular connections of the cerebellum display a similar double representation in the most rostral lobules of the anterior lobe and far caudally in the vestibulocerebellum (Fig. 20.9).
The folium, tuber, uvula, tonsil and posterior biventral lobule all receive an almost pure pontine mossy fibre input. Climbing fibres from the inferior olive and mossy fibres from the basilar and tegmental pontine nuclei relay visual and acoustic information from the respective cerebral association areas and midbrain tectum to the folium and tuber thought to represent a vermal visual/acoustic area (Fig. 20.8). The efferent connections of this area travel via the fastigial nucleus to gaze centres in the pons and midbrain.
AFFERENT CONNECTIONS OF THE CEREBELLUM
Mossy fibres and climbing fibres carry the afferent connections of the cerebellum. Mossy fibre systems terminate bilaterally in transversely oriented ‘lobular’ areas; there is considerable overlap amongst the terminations. Climbing fibres from different subnuclei of the inferior olive terminate contralaterally, on discrete longitudinal strips of Purkinje cells. This longitudinal pattern closely corresponds with the zonal arrangement in the corticonuclear projection (Fig. 20.10).
Spinocerebellar and trigeminocerebellar fibres
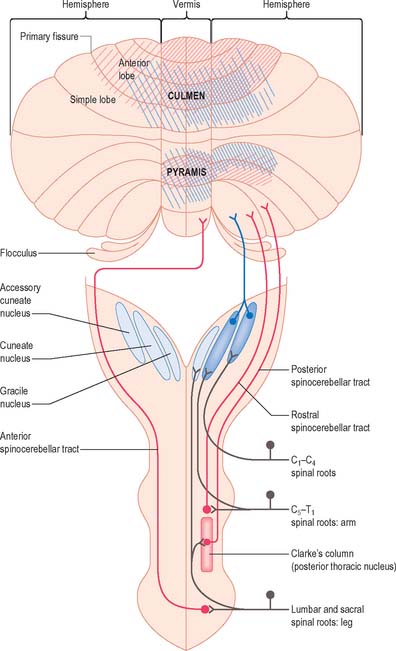
The posterior spinocerebellar tract takes its origin from the cells of Clarke’s column at the base of the dorsal horn in all thoracic segments of the spinal cord (Fig. 20.11). It enters the inferior cerebellar peduncle, gives collaterals to the cerebellar nuclei, and terminates, mainly ipsilaterally, in the vermis and adjoining regions of the anterior lobe and in the pyramis and adjoining lobules of the posterior lobe. Clarke’s column receives primary afferents of all kinds from the muscles and joints of the lower limbs, which reaches the nucleus via the fasciculus gracilis. It also receives collaterals from cutaneous sensory neurones. Accordingly, the tract transmits proprioceptive and exteroceptive information about the ipsilateral lower limb. Very fast conduction is required to keep the cerebellum informed about ongoing movements. The axons in the posterior spinocerebellar tract are the largest in the CNS, measuring 20 μm in diameter. The upper limb equivalent of the posterior spinocerebellar tract is the cuneocerebellar tract.
The anterior spinocerebellar tract is a composite pathway. It informs the cerebellum about the state of activity of spinal reflex arcs related to the lower limb and lower trunk. Its fibres originate in the intermediate grey matter of the lumbar and sacral segments of the spinal cord (Fig. 20.11). They cross near their origin, and ascend close to the surface as far as the lower midbrain before looping down in the superior cerebellar peduncle. Most fibres cross again in the cerebellar white matter. Thus their distributions to the cerebellar nuclei and cortex appear to be the same as those of the posterior tract.
The rostral spinocerebellar tract originates from cell groups of the intermediate zone and horn of the cervical enlargement. Although considered to be the upper limb and upper trunk counterpart of the anterior spinocerebellar tract, most of its fibres remain ipsilateral throughout their course. It enters the inferior cerebellar peduncle and terminates in the same cerebellar nuclei and folia as the cuneocerebellar tract.
Olivocerebellar fibres
Localization in the olivocerebellar system: zones and microzones
Climbing fibres originate exclusively from subnuclei of the contralateral inferior olivary complex and terminate on longitudinal strips of Purkinje cells in the cerebellar cortex; their collaterals end on the cerebellar or vestibular target nuclei of these Purkinje cells. A longitudinal zonal arrangement is therefore characteristic of the organization of the olivocerebellar projection. Moreover, the olivocerebellar projection zones correspond precisely to the corticonuclear projection zones already described (Fig. 20.10). Climbing fibres from the inferior olive are able to modify the cerebellar output in such a way that cells within each subnucleus of the inferior olivary complex monitor the output of a single cerebellar module.
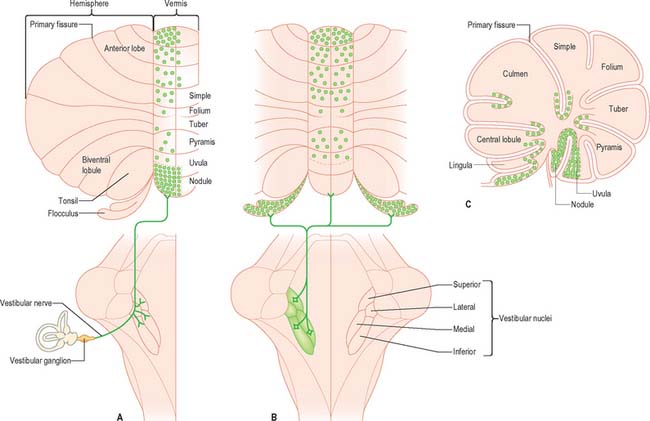
Vestibulocerebellar fibres
Primary vestibulocerebellar mossy fibres are fibres of the vestibular branch of the vestibulocochlear nerve. They enter the cerebellum with the ascending branch of the vestibular nerve, and pass through the superior vestibular nucleus and the juxtarestiform body. They terminate, mainly ipsilaterally, in the granular layer of the nodule, caudal part of the uvula, ventral part of the anterior lobe and bottom of the deep fissures of the vermis (Fig. 20.9A). Secondary vestibulocerebellar mossy fibres arise from the superior vestibular nucleus and the caudal portions of the medial and inferior vestibular nuclei. They terminate bilaterally not only in the same regions that receive primary vestibulocerebellar fibres but also in the flocculus, which lacks a primary vestibulocerebellar projection (Fig. 20.9B). Some of the mossy fibres from the medial and inferior vestibular nuclei are cholinergic.
Pontocerebellar fibres
More than 90% of fibres in the middle cerebellar peduncle belong to the cortico-ponto-cerebellar pathway. Corticopontine fibres travel in the cerebral peduncle. Fibres from the frontal lobe occupy the medial part of the peduncle, and fibres from the parietal, occipital and temporal lobes occupy the lateral part. They synapse on some 20 million neurones in corresponding regions of the basilar pons. The onward pontocerebellar mossy fibre projection is predominantly to the lateral regions of the posterior and anterior lobes (Fig. 20.4) but collaterals are given off to the dentate nucleus.
EFFERENT CONNECTIONS OF THE CEREBELLUM
The output of the cerebellum consists of the inhibitory projections of the Purkinje cells to the cerebellar and vestibular nuclei, and the efferent connections of the cerebellar nuclei to motor centres in the brain stem and, through the thalamus, the motor cortex. Their effects on movement are always indirect, because there are no direct projections from the cerebellar nuclei to motor neurones. Disynaptic connections of the Purkinje cells in the anterior vermis and vestibulocerebellum with motor neurones controlling oculogyric and proximal limb muscles are mediated by the vestibular nuclei. The vermis also influences these motor neurones bilaterally through multisynaptic pathways that involve the fastigial and vestibular nuclei and the reticular formation (Fig. 20.12). The vermis cannot be considered as a single module. Each half of the vermis is composed of several modules (each made up of a longitudinal Purkinje cell zone and a target nucleus) and their supporting climbing fibre afferent projections.
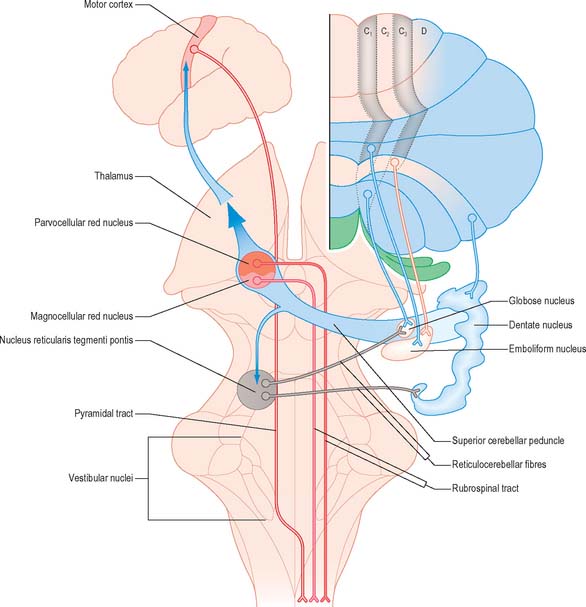
Each cerebellar hemisphere influences movements of the ipsilateral extremities by way of projections to the dentate and interposed (emboliform and globose) nuclei, which in turn project to the contralateral red nucleus, thalamus and motor cortex (Fig. 20.13).
Corticonuclear and corticovestibular fibres
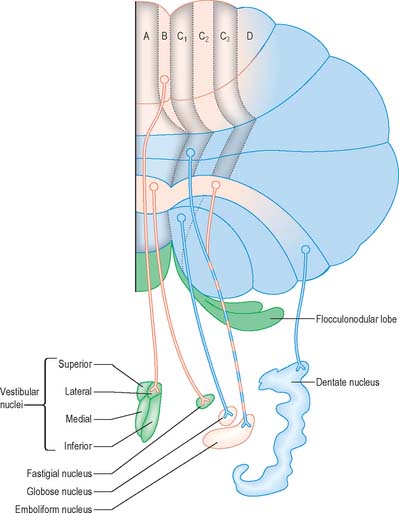
Purkinje cells of each hemivermis project to the ipsilateral fastigial and vestibular nuclei. Purkinje cells of the hemisphere project to the interposed and dentate nuclei. Although the cerebellar cortex is organized in strips of Purkinje cell zones, which project to different cerebellar and vestibular nuclei, the borders between these strips are not apparent in the structure of the cortex when it is examined histologically using conventional staining methods. The vermis of the anterior lobe and simple lobule consist of two parallel strips, A and B, of Purkinje cells (Figs 20.10, 20.12). The medial strip (A zone) projects to the rostral pole of the fastigial nucleus, and the lateral strip (B zone) projects to the lateral vestibular nucleus. The B zone does not continue beyond the simple lobule. The cortex of the entire caudal vermis, which projects to the fastigial nucleus, is included in the A zone. The folium and tuber, which represent a region of the cerebellum that receives a visual input and which are involved in the accurate calibration of saccades, project to the caudal pole of the fastigial nucleus. The pyramis, uvula and nodule can be subdivided into several Purkinje cell zones. However, the significance of their connections with the cerebellar and vestibular nuclei is not well understood. Corticovestibular projections to the superior, medial and inferior vestibular nuclei, but not to the lateral nucleus, take their origin from the nodule and the adjacent region of the uvula.
The intermediate region consists of two strips of Purkinje cells (C1 and C3 zones), which project to the emboliform (anterior interposed) nucleus. They flank a single zone (C2) that projects to the globose (posterior interposed) nucleus (Figs 20.10, 20.13). The rest of the hemisphere projects to the dentate nucleus. There are indications for a subdivision of the hemisphere into two zones that project to the caudolateral and rostromedial parts of the dentate nucleus, respectively. The neurones of the caudolateral dentate are generally smaller than those of the rostromedial portion and the convolutions are broader. The efferent connections of the flocculus are mainly with the superior, medial and inferior vestibular nuclei and resemble those from the nodule and uvula.
Cerebellovestibular and cerebelloreticular fibres
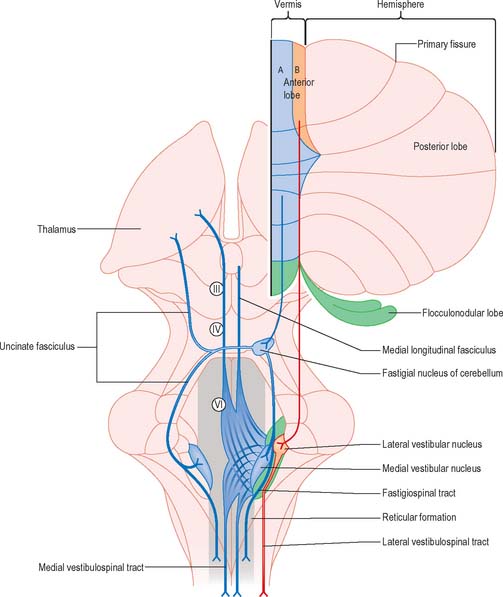
Efferent connections of the fastigial nucleus
The fastigial nucleus is connected bilaterally with the vestibular nuclei and the medullary and pontine reticular formation (Fig. 20.12). Smaller crossed connections either ascend to the midbrain and diencephalon or descend into the spinal cord. Small GABAergic neurones give rise to fastigio-olivary fibres, which terminate in the medial accessory olive. The uncinate fasciculus is the major efferent pathway of the fastigial nucleus. Its fibres cross in the rostral part of the cerebellar white matter, and pass dorsal to the superior cerebellar peduncle, to enter the vestibular nuclei from their lateral side. Uncrossed fibres enter the vestibular nuclei through the juxtarestiform body. The distribution of the fastigial projection is bilateral, but with a contralateral preponderance. Crossed and uncrossed projections end in the medial and inferior vestibular nuclei, and also traverse these nuclei to terminate in the medial reticular formation. Some crossed fibres can be traced caudally into the spinal cord. A small fascicle of crossed fibres from the fastigial nucleus ascends along the superior cerebellar peduncle and is distributed bilaterally to the dorsal tegmentum, central grey matter and deep layers of the superior colliculus and the nuclei of the posterior commissure. Fibres terminate bilaterally in the ventrolateral nucleus and the intralaminar nuclei of the thalamus.
Cerebellovestibular connections
The relationship between the cerebellum and the vestibular nuclei is complex (Figs 20.9, 20.12). In addition to the vestibulocerebellum (nodule, adjacent folia of the uvula and flocculus), the main vermis and the fastigial nucleus also project to the vestibular nuclei.
Cerebellorubral and cerebellothalamic fibres
The axons of neurones in the dentate and interposed nuclei leave the cerebellum in the superior cerebellar peduncle. The superior peduncles, including their nucleo-olivary component, decussate in the caudal midbrain (Fig. 20.13) and each then gives off a small descending branch carrying fibres that terminate in the medial reticular formation of the pons and medulla and the pontine tegmental reticular nucleus. The nucleo-olivary fibres join this descending branch and terminate in the inferior olive in a strictly ordered manner. The ascending branch is distributed to the midbrain and diencephalon, mainly to the red nucleus and thalamus. The emboliform (anterior interposed) nucleus projects to the magnocellular part of the red nucleus. In humans this projection is very small and the target neurones give rise to a relatively trivial rubrospinal tract, which crosses in the caudal midbrain and terminates on lateral medullary interneurones and a small number of motor neurones in the upper cervical spinal cord.
The thalamus receives a massive input from other major motor systems, in addition to the input it receives from the cerebellum. In particular, the output of the basal ganglia is relayed to the thalamus by a projection from the globus pallidus (Ch. 22). Available evidence suggests that these two great subcortical motor systems terminate on different regions in the ventral thalamus and project to different targets in the motor and premotor cortex.
CEREBELLAR FUNCTIONS
The vermis of the cerebellum is involved in taking anticipatory action in order to maintain an upright posture when objects are picked up. For example, when a book is taken down from a shelf, the first muscle groups to be activated are not the flexors of shoulder, elbow or fingers, but the plantar flexors of the ankle. Contraction of the ankle flexors causes the forefeet to push the lower limbs and trunk backwards at the moment the hand grasps the book. Once the lift gets under way, the erector spinae muscles correct for the combined weights of the book and the reaching arm, in order to prevent forward sway of the head and trunk. Labyrinthine receptors simultaneously inform the cerebellum of any forward movement of the head, and appropriate antigravity thrust is exerted via one or both lateral vestibulospinal tracts.
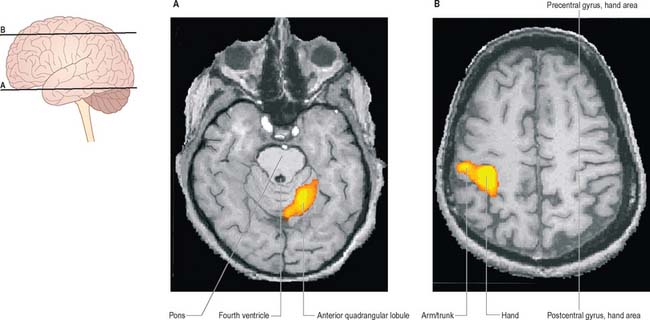
Many motor skills require precise timing, which involves an extreme degree of cooperation between prime movers and their antagonists. For example, reading a printed page requires that the scanning eyes snap back to the beginning of a line, time after time: even small errors may result in dyslexia, where slight incoordination of eye movements may cause the letters of a word to appear jumbled. Clinical testing of timing can be performed easily by checking the ability to perform rhythmic movements such as repetitive pronation/supination (Fig. 20.14).
Bastian AJ, Mugnaini E, Thach WT. Cerebellum. In: Zigmond MJ, Bloom FE, Landis SC, Roberts JL, Squire LR. Fundamental Neuroscience. San Diego: Academic Press; 1999:973-992.
Jueptner M, Kruckenberg M. Anatomic basis of functional magnetic resonance imaging. Motor system. Neuroimage Clin N Am. 2001;11:203-219.
Rae C, Harasty JA, Dzendrowskyj TE, Talcott JB, Simpson JM, Blamire AM, Dixon RM, Lee A, Thompson CH, Styles P, Richardson AJ, Stein JF. Cerebellar morphology in developmental dyslexia. Neuropsychologia. 2002;40:1285-1292.
Schmahmann JD, Sherman JC. The cerebellar cognitive affective syndrome. Brain. 1998;121:561-579.
Ivry RB, Spencer RM, Zelaznik HN, Diedrichsen J. The cerebellum and event timing. Ann NY Acad Sci. 2002;978:302-317.
Gebhart AL, Petersen SE, Thach WT. Role of the posterolateral cerebellum in language. Ann NY Acad Sci. 2002;978:318-333.
Topka H, Mescheriakov S, Boose A, et al. A cerebellar-like terminal and postural tremor induced in normal man by transcranial magnetic stimulation. Brain. 1999;122:1551-1562.