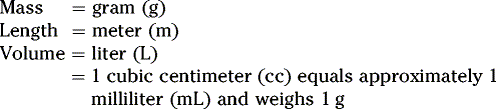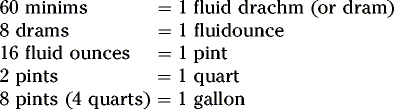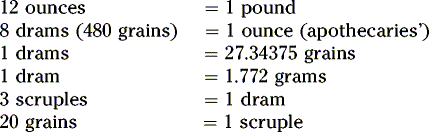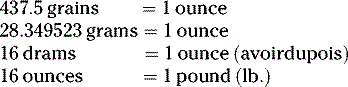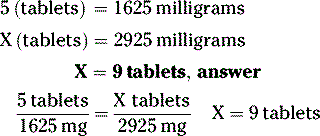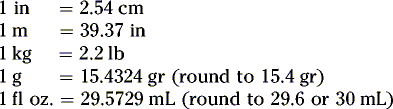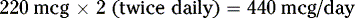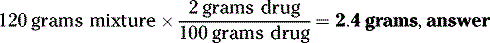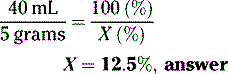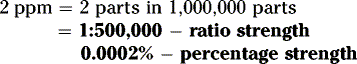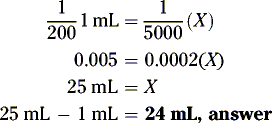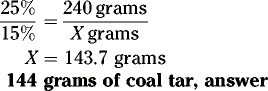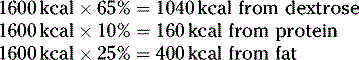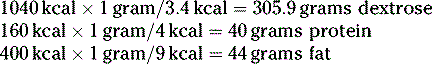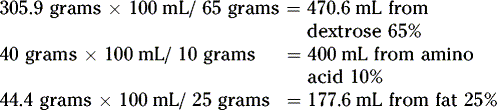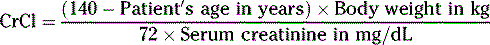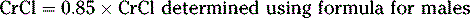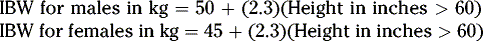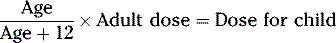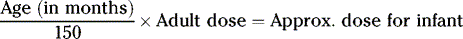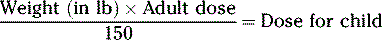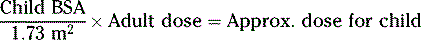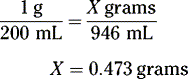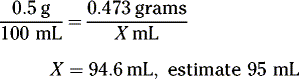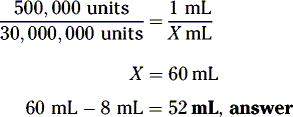CHAPTER 2 Pharmaceutical Calculations
SYSTEMS OF MEASURE
Summary of conversion between metric, apothecaries’ and avoirdupois systems:
Per the United States Pharmacopeia, 1 grain = 64.8 mg.
METRIC SYSTEM
Prefixes
| kilo- | 103 | 1 thousand (1000) times the basic unit |
| hecto- | 102 | 1 hundred (100) times the basic unit |
| deka- | 10 | 1 ten (10) times the basic unit |
| deci- | 10−1 | 1 tenth (0.1) times the basic unit |
| centi- | 10−2 | 1 hundredth (0.01) times the basic unit |
| milli- | 10−3 | 1 thousandth (0.001) times the basic unit |
| micro- | 10−6 | 1 millionth times the basic unit |
| nano- | 10−9 | 1 billionth times the basic unit |
| pico- | 10−12 | 1 trillionth times the basic unit |
INTERPRETATION OF MEDICATION ORDERS
Example 1:
A prescription for prednisone 5 mg should be taken as follows:
Example 1:
How many grams of drug should be used to prepare 120 grams of a 2% w/w solution in water?
Example 2
What is the percentage strength (w/v) of a solution of drug if 40 mL contain 5 grams?
Example
Solution
The coal tar (active ingredient) is added to a diluent (petroleum) currently containing no coal tar.
Example
First, determine how many kcal the patient needs from each component:
Next, convert these kcals into grams:
Then, calculate how many milliliters are needed from each stock solution:
For females
If the individual is obese or not within 30% of their ideal body weight, other methods of calculating creatinine clearance should be used. Ideal body weight (IBW) or adjusted body weight (ideal body weight plus 40% of obese weight) instead of actual body weight in the Cockcroft-Gault equation will provide a better estimate of creatinine clearance.
CALCULATIONS FOR PEDIATRIC DOSES
Various pediatric formulas have been used historically to calculate APPROXIMATE pediatric dosages.
Ansel H., Stoklosa M. Pharmaceutical Calculations, ed 12. Baltimore, MD: Lippincott Williams & Wilkins, 2005.
Bhatt S.H.L. Aminoglycoside Pharmacokinetics and Therapeutics. MCPHS Boston Campus, MA: White Hall, 2006.
Institute of the Certification of Pharmacy Technicians (ICPT). ExCPT Exam for the Certification of Pharmacy Technicians. Available at http://www.nationaltechexam.org/pdf/math_questions-answers070618.pdf Accessed December 24, 2008
London, Eastern and South East Specialist Pharmacy Services, Available at http://www.londonpharmacy.nhs.uk/educationandtraining/prereg/supportMaterial/calculations/download/Calculations%20WorkBook%202005.pdf Accessed December 24, 2008
Pearson J., Powers M. Systematically Initiating Insulin. The Staged Diabetes Management Approach. Diabetes Educ. 2006;32(Suppl 1):23s.
Shargel L. Applied Biopharmaceutics & Pharmacokinetics. New York: McGraw-Hill Medical Publishing Division, 2005;43-46.
Zatz J. Pharmaceutical Calculations, ed 4, Hoboken, NJ: John Wiley & Sons, Inc; 2005:30-33.
Mosteller R.D. Simplified Calculation of Body Surface Area. N Engl J Med. 1987;317:1098. (letter)
REVIEW QUESTIONS
(Answers and Rationales on page 313.)