HIV Infection and AIDS
Perspective
The acquired immunodeficiency syndrome (AIDS) is a pandemic caused by the human immunodeficiency virus (HIV). This disease has caused a tremendous degree of human suffering and has had an immeasurable impact on demographics, cultures, economics, and politics in most societies around the globe. There are an estimated 33.3 million people living with HIV infection worldwide, and 35 million people have died of it.1,2 Significant strides have been made in areas of prevention and treatment, resulting in stabilization of the epidemic, and the annual number of new infections has steadily decreased since the 1990s. Despite this decline in incidence of HIV infection, new infections and the widespread use of highly active antiretroviral therapy (HAART), thus decreasing AIDS-related deaths, have resulted in an increase of the global prevalence of HIV/AIDS (Fig. 132-1).
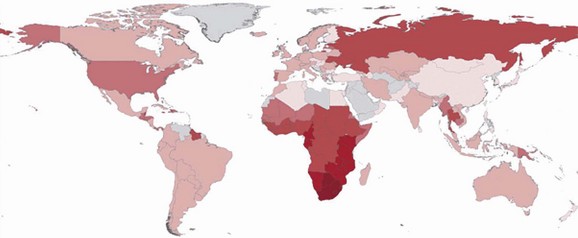
Figure 132-1 The distribution of people living with HIV infection globally. The darker shades represent higher prevalence of HIV infection. (From Joint United Nations Programme on HIV/AIDS: UNAIDS Report on the Global AIDS Epidemic 2010. Available at www.unaids.org/documents/20101123_2010_HIV_Prevalence_Map_em.pdf. Accessed November 25, 2011.)
HIV infection was first recognized in 1981 by clinicians in New York and Los Angeles in previously healthy homosexual men when they noted unusual clusters of Pneumocystis jiroveci (formerly Pneumocystis carinii) pneumonia (PCP) and Kaposi’s sarcoma. Early on, researchers were aware that these patients had a profound defect in their cell-mediated immune system. They discovered that this disease was spread through sexual activity or exposure to blood. Subsequent concerted scientific effort resulted in rapid identification of the infecting organism, prompt understanding of the natural history of the disease, and swift strides in providing therapeutic interventions.2
Means of transmission of HIV and the demographic distribution of the virus vary around the globe. Sub-Saharan Africa is the epicenter of the pandemic, with 22.5 million adults and children living with HIV infection (68% percent of the global HIV population). South Africa has the most people living with HIV infection (5.6 million), and Swaziland has the highest prevalence of HIV infection in the world (estimated to be 25.9%).1 Unprotected heterosexual intercourse with subsequent transmission of HIV to newborns and breast-fed babies (mother-to-child transmission) is the dominant mode of transmission worldwide, accounting for about 85% of all HIV infections.3 The HIV epidemic in Asia and eastern Europe is largely in people who inject drugs, sex workers and their clients, and men who have sex with men. In the Caribbean, the primary means of transmission is thought to be through unprotected sex between men and women, especially through paid sex. A large portion of the burden of disease in Central and South America is in men who have sex with men. In the Middle East and North Africa, the prevalence of HIV infection is relatively low.1 The main pattern of transmission in the higher income countries of North America and western and central Europe is in men who have sex with men. Other significant modes of transmission in these regions include injection drug use and paid unprotected sex.1
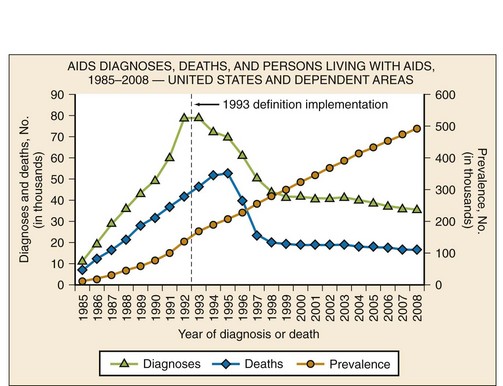
The prevalence of HIV infection in the United States, estimated to be 1,106,400 adults and adolescents in 2009, is currently the highest it has been.4 The increase in prevalence is likely to be due to a combination of factors, including more people knowing their HIV status and receiving medical therapy, identification of new infections, and fewer people dying of HIV/AIDS. Although the prevalence of HIV/AIDS in the United States continues to rise, the incidence has remained relatively stable since the late 1990s5 (Fig. 132-2). In the United States, HIV disproportionately affects men who have sex with men, African Americans, Hispanics, and people living in urban areas.4 Of the estimated 56,300 new HIV infections in the United States each year, 53% are in men who have sex with men, 31% are through high-risk heterosexual contact, 12% are in injection drug users, and 27% are in women.3,4
Principles of Disease
The mature HIV virion is a spherical structure with an outer envelope and an inner core (Fig. 132-3). The core contains two copies of the RNA genome, enzymes (reverse transcriptase and integrase), and regulatory proteins.6 Surrounding the core is the viral membrane, containing the glycoproteins gp41 and gp120, which are responsible for the attachment and entry of the virus into a CD4+ cell.7
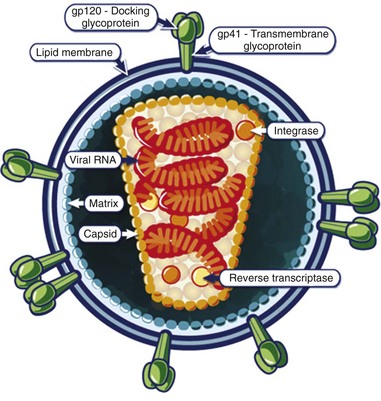
Figure 132-3 The structure of the HIV virion. (From the National Institute of Allergy and Infectious Diseases. Available at http://www.niaid.nih.gov/topics/HIVAIDS/Understanding/Biology/Pages/structure.asPx.)
In a multistep process, the HIV virion invades the host cell and integrates its genetic material into the host’s chromosome (Fig. 132-4). The infection begins with binding of the virus to the CD4+ host cell; the interaction is mediated by a high-affinity interaction between the surface proteins gp41 and gp120 with the CD4+ antigen and coreceptors. Different coreceptors, CCR5 and CXCR4, have been identified. The virus enters the cell by fusing its envelope with the target cell membrane. After internalization, reverse transcriptase forms viral DNA from the original RNA. The viral enzyme integrase then transports the newly formed viral DNA into the nucleus, where it integrates with human chromosomal DNA. Viral polyproteins and RNA are formed and subsequently modified and spliced by the viral enzyme protease, and new infectious viral particles are created.3,8 This cycle continues with HIV infecting more CD4+ cells. Major targets of antiretroviral therapy (ART) include reverse transcriptase, protease, integrase, and the CCR5 coreceptor.
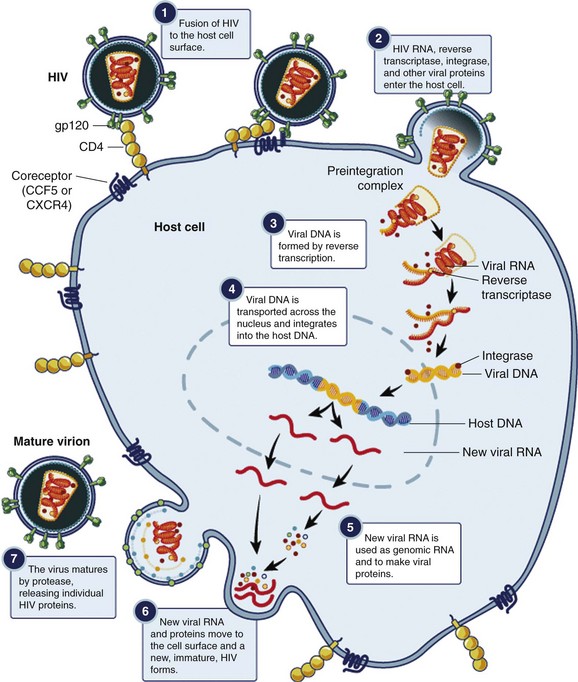
Figure 132-4 Replicative cycle of the HIV virion. (From the National Institute of Allergy and Infectious Diseases. Available at www.niaid.nih.gov/TOPICS/HIVAIDS/UNDERSTANDING/BIOLOGY/Pages/hivReplicationCycle.aspx.)
The hallmark of HIV infection is CD4+ T-cell destruction, leading to a deficient cell-mediated arm of the immune system. The opportunistic infections (PCP and infections due to Toxoplasma gondii, Mycobacterium avium complex [MAC], and cytomegalovirus [CMV]) and malignant neoplasms (including Kaposi’s sarcoma) of AIDS patients are primarily due to the lack of CD4+ activity. Although it is unclear exactly how HIV depletes CD4+ cells, increased peripheral destruction and decreased production of CD4+ cells occur with the infection. Humoral immunity is also impaired, through B-cell proliferation and production of abnormal antibodies, making HIV-infected individuals more vulnerable to infections by encapsulated bacteria.2,7
HIV infection also leads to a chronic immune activation. Ongoing viremia, along with proinflammatory cytokines, B-cell proliferation, and hypergammaglobulinemia, leads to a chronic inflammatory state that contributes to cardiovascular disease, cancer, and other chronic diseases in long-term HIV-infected individuals.7
Risk Factors for HIV Transmission
Activities that result in exposure to infectious body fluids increase the likelihood of HIV transmission, and the risk of transmission varies according to the nature of the exposure. The highest risk exposure is transfusion with HIV-positive blood. Other factors associated with increased risk of HIV transmission include exposure to serum with high viral load,9–12 lack of male circumcision,13–15 presence of ulcerative sexually transmitted disease,12 and similarity of HLA class I alleles among discordant couples.11 The risk of transmission of HIV varies by the type of sexual contact: it is 1 to 30% for receptive anal intercourse; 0.1 to 10% for receptive vaginal and insertive anal intercourse; and 0.1 to 1% for insertive vaginal intercourse.16,17
Clinical Features
Stages of HIV Infection
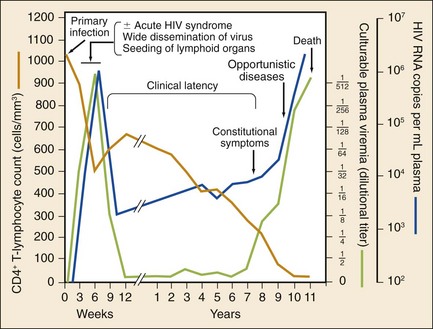
The stages of untreated HIV infection are described by the World Health Organization (Box 132-1) and the Centers for Disease Control and Prevention (CDC; Table 132-1). Figure 132-5 also illustrates the natural history of untreated HIV infection.
Table 132-1
CDC Surveillance Case Definitions for HIV Infection
| STAGE | LABORATORY EVIDENCE |
| Stage 1 | No AIDS-defining condition and either CD4+ T-lymphocyte count of ≥500 cells/µL or CD4+ T-lymphocyte percentage of total lymphocytes of ≥29 |
| Stage 2 | No AIDS-defining condition and either CD4+ T-lymphocyte count of 200-499 cells/µL or CD4+ T-lymphocyte percentage of total lymphocytes of 14-28 |
| Stage 3 (AIDS) | CD4+ T-lymphocyte count of <200 cells/µL or CD4+ T-lymphocyte percentage of total lymphocytes of <14 or documentation of an AIDS-defining condition |
| Stage unknown | No information available on CD4+ T-lymphocyte count or percentage and no information available on AIDS-defining conditions |
CDC, Centers for Disease Control and Prevention.
From Schneider E, et al: Revised surveillance case definitions for HIV infection among adults, adolescents, and children aged <18 months and for HIV infection and AIDS among children aged 18 months to <13 years—United States, 2008. MMWR Recomm Rep 57:1-12, 2008.
Primary HIV Infection
Primary infection with HIV often causes an acute, self-limited viral infection. The most common findings are mononucleosis-like symptoms consisting of fever, pharyngitis, and lymphadenopathy. This usually occurs 2 to 6 weeks after transmission. During this time, the virus is actively replicating and antibodies to HIV have not been produced. The virus has many potential targets, and viral loads are often enormous.18 The diagnosis of acute HIV infection has significant public health benefits. Patients with acute HIV infection transmit the infection disproportionately; these patients often do not know that they are infected, and their viral load may be in the range of millions of RNA copies per milliliter.19
Diagnosis of acute HIV infection requires keeping the constellation of symptoms in mind and understanding the pitfalls of laboratory testing for early infection. The results of routine HIV antibody testing may be negative for several weeks or even months after exposure. The diagnosis of acute HIV infection is confirmed with the presence of high titers of viral RNA and a negative antibody screen. Assay reactivity is dynamic; a plasma RNA test will detect HIV infection approximately 1 week before the ability to detect the p24 antigen and 12 days before antibodies to HIV develop20 (Table 132-2). A viral load test in the absence of symptoms of acute HIV infection is not recommended; false-positive results occur, and the test is costly.21 Confirmed cases of acute HIV infection should be referred immediately to a specialist, preferably one who actively enrolls patients in clinical trials.
Table 132-2
HIV Testing by Laboratory Stage
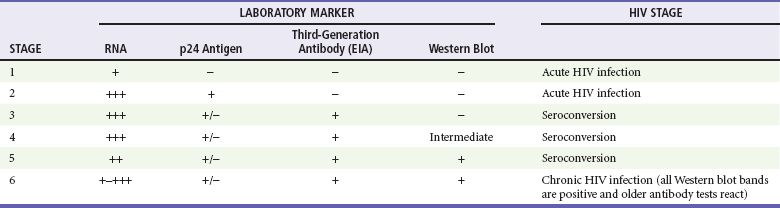
Modified from Fiebig EW, et al: Dynamics of HIV viremia and antibody seroconversion in plasma donors: Implications for diagnosis and staging of primary HIV infection. AIDS 17:1871-1879, 2003.
AIDS
The CDC definition of AIDS is a CD4+ cell count below 200 cells/µL or the presence of an AIDS-defining condition (Box 132-2). At this level, immune dysfunction is severe, and without ART, survival is short. Those with a CD4+ cell count below 50 cells/µL have advanced AIDS and are at much higher risk for death and for development of opportunistic infections. Some infections are so common in patients with AIDS that primary prophylaxis is indicated and cost effective. Prophylaxis is started for PCP when CD4+ counts are less than 200 cells/µL; for toxoplasmosis, when CD4+ counts are less than 100 cells/µL; and for MAC disease, when CD4+ counts are less than 50 cells/µL (Table 132-3). Isoniazid is given to patients with a positive response to purified protein derivative. The risk of opportunistic infection in relation to the CD4+ cell count is progressive rather than an all-or-none phenomenon.
Table 132-3
Prophylaxis to Prevent First Episode of Selected Opportunistic Infections

ART, antiretroviral therapy; TB, tuberculosis; TMP-SMZ, trimethoprim-sulfamethoxazole.
Modified from Kaplan JE, et al: Guidelines for prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: Recommendations from CDC, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America. MMWR Recomm Rep 58:1-207, 2009. Available at www.cdc.gov/mmwr/preview/mmwrhtml/rr5804a1.htm.
Initial Evaluation
HIV-infected patients are at risk for some of the same infections and medical problems that noninfected patients have, but they are more vulnerable to opportunistic and unusual infections. Knowledge of the CD4+ count, along with the patient’s clinical presentation, is critical in the ED management of the HIV-infected patient. Before HAART, HIV-positive individuals were primarily hospitalized for opportunistic infections. Patients with HIV infection are now dying of diseases that were not traditionally considered AIDS diseases, such as heart disease, liver failure, and non–AIDS-related cancers.22
The current CD4+ count is a marker of the degree of immunosuppression and is critical background information for the interpretation of signs and symptoms. However, many patients have undiagnosed HIV infection, and they often present in later stages of disease.23 Others may not be taking ART because of issues involving access to care, difficulties with medication adherence, or viral resistance to ART. Opportunistic infections such as PCP, disseminated mycobacterial infections, cryptococcal meningitis, and CMV disease do not occur until CD4+ counts are dramatically reduced. A total lymphocyte count may provide a rough surrogate for the absolute CD4+ count; a count between 1000 and 2000 cells/µL appears to be a reasonable surrogate of significant immunosuppression. A cell count of more than 2000 cells/µL makes it less likely that a patient is profoundly immunosuppressed. Acute illness will drop peripheral lymphocyte counts and thus limits the utility of the peripheral lymphocyte count as a diagnostic aid in an acute setting. However, an ED study showed that patients with a peripheral lymphocyte count below 950 cells/µL were highly likely to have AIDS.24
Certain clinical features suggest the possibility of a new diagnosis of HIV infection. A careful look at the skin and in the mouth may be revealing; certain skin findings, such as oral hairy leukoplakia and Kaposi’s sarcoma, are found almost exclusively in HIV-infected individuals.25 Unexplained oral candidiasis is often the initial presenting symptom in someone with undiagnosed HIV infection.
Clinical Manifestations by Organ System
HIV-infected patients have an increased risk of acute coronary syndrome, although it is not fully understood whether this is the result of the HIV infection itself, a complication of ART, or a combination of both. Patients receiving ART suffer from a myriad metabolic abnormalities (hyperglycemia, hyperlipidemia, lipodystrophy) and accelerated atherosclerosis, which may increase their risk for cardiovascular disease and acute coronary syndrome.26–32 Studies have shown that the virus alone is associated with dyslipidemia,26 endothelial damage,33–35 inflammation,36,37 and hypercoagulability.38–42 Stopping of ART has been shown to result in systemic inflammation, coagulation cascade activation and an increase in biomarkers associated with endothelial activation,36,43 and increased risk of major cardiovascular events.44
Antiretroviral agents cause varying degrees of dyslipidemia. Protease inhibitors, particularly ritonavir in higher doses, can especially be a problem.30 Non-nucleoside reverse transcriptase inhibitors (NNRTIs) are associated with increases in low-density lipoprotein cholesterol and total cholesterol but also a significant increase in high-density lipoprotein cholesterol.45 The Data Collection on Adverse Events of Anti-HIV Drugs (D : A : D) study found that the nucleoside reverse transcriptase inhibitor abacavir was associated with a fourfold increased risk of myocardial infarction and a significant increase in stroke, peripheral vascular disease, and congestive heart failure.46 Despite the reality that some ART is likely to contribute to the development of coronary artery disease, the benefits of therapy outweigh the risks of accelerated atherosclerosis.
Pulmonary
Pulmonary infections are the leading cause of morbidity and mortality for people living with HIV infection.47 Noninfectious pulmonary complications are also common. The patient’s immune status (as suggested by the last CD4+ count) and the current treatment regimen determine the severity of the presenting illness. The tempo of the illness and geographic risk factors also help guide the diagnostic evaluation and initial therapy.