Sleep and Breathing Assessment
After reading this chapter, you will be able to:
1. Describe the role of the respiratory therapist in the assessment of patients for potential sleep-disordered breathing.
2. Identify the number of Americans affected by sleep disorders.
3. Describe the expected findings in the assessment of patients with sleep-disordered breathing.
4. Explain the correlation among sleep apnea, snoring, and excessive daytime sleepiness.
5. List the criteria used with the Epworth Sleepiness Scale and the Berlin Questionnaire surveys in assessing the presence of a sleep disorder.
6. Describe the normal stages of sleep with associated physiologic changes in the cardiopulmonary system.
7. List the clinical and assessment criteria for obstructive, central, and mixed sleep apnea.
8. Describe the role of a polysomnogram in providing a differential diagnosis for sleep-disordered breathing.
9. Describe the typical physiologic parameters monitored on a polysomnogram montage.
10. List the criteria used to classify sleep apnea as mild, moderate, or severe.
11. Identify the symptoms and assessment characteristics for children with sleep apnea.
12. Understanding basic treatment options and insurance coverage criteria.
Normal Stages of Sleep
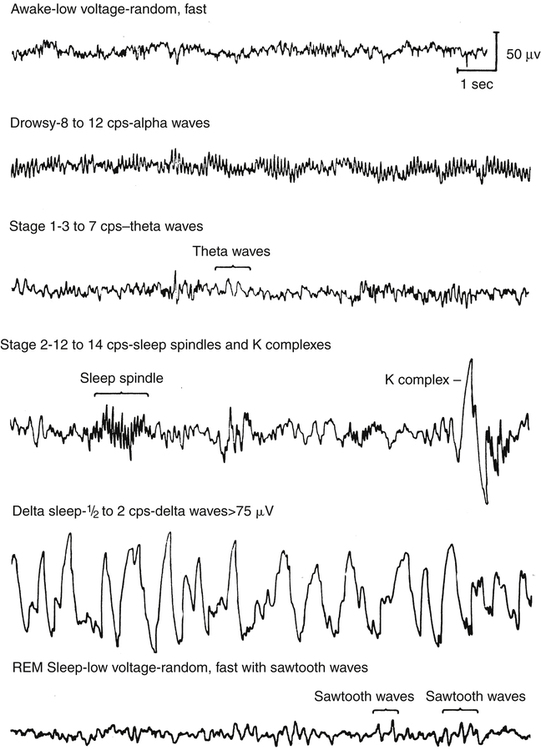
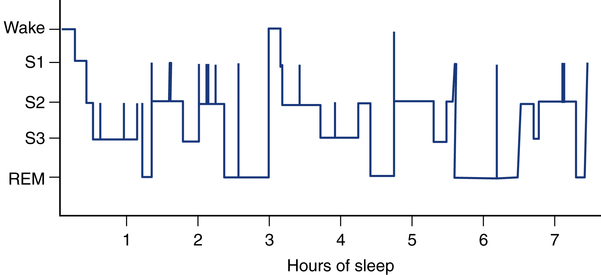
Historically, sleep has been classified in various ways; however, current nomenclature recognizes only two primary states of sleep. For adults and children, the two major states of sleep are non–rapid eye movement (NREM) and rapid eye movement (REM), named for the absence and presence, respectively, of the rapid fluttering or rolling of the eyes and the loss or retention of muscle tone during each phase (Fig. 19-1). There are special states for newborns and infants up to 6 months of age because their brain wave patterns do not yet conform to standard adult classification. They include definitions such as active, quiet, and indeterminate sleep to illustrate the ambiguity of neural activity in the very young. NREM and REM stages cycle back and forth every 60 to 90 minutes for a total of four or five cycles for adults during a normal 8-hour sleeping period. With sleep occupying one third of our life, the quality and quantity of sleep have long-range health and quality-of-life implications.
Assessment of Sleep-Disordered Breathing
Epworth Sleepiness Scale
The ESS is a simple, eight-item questionnaire that measures daytime sleepiness and is essential for initial screening of sleep disorders. The ESS was developed by Dr. Murray Johns of the Epworth Hospital in Melbourne, Australia in 1991. The survey presents eight situations, and the patient is asked to rate the chances of dozing in each situation, with the value of 0 representing no chance of dozing; 1, a slight chance of dozing; 2, a moderate chance of dozing; and 3, a high chance of dozing. The sleepiness score is totaled, and if the ESS total is 1 to 6, sleep is appropriate and no EDS is noted. If the ESS total is 7 or 8, the score is considered average. An ESS score of 9 or greater indicates that the individual should consult a sleep specialist without delay because there is evidence of EDS (Fig. 19-3). The ESS questionnaire has been shown to have a high degree of reliability and internal consistency as an evaluation instrument.
Berlin Questionnaire
The Berlin Questionnaire is an outcome of the 1996 Conference on Sleep in Primary Care held in Berlin, Germany. More than 120 American and German pulmonary and primary care physicians developed the questionnaire from the literature to identify behaviors consistent with the presence of SDB. The focus of the instrument is deliberately limited to risk factors associated with sleep apnea. The 10-item survey is divided into three categories, with category 1 addressing snoring, category 2 identifying EDS, and category 3 assessing current blood pressure and body mass index (BMI) (Fig. 19-4). To be classified as at high risk for sleep apnea, an individual must qualify for at least two categories. The Berlin Questionnaire has a high degree of sensitivity and specificity for predicting sleep apnea in individuals.